Abstract
Pepper is one of the most popular spices over the world and is called the King of Spices. Its essential oils (EOs) could alleviate neuronal ailments due to the inhibitory effect against acetylcholinesterase (AChE). In this study, the chemical compositions of 26 EOs prepared from white and black pepper collecting from 6 different cultivars were analyzed by gas chromatography-mass spectrometry (GC-MS). A total of 133 compounds were identified in the white and black pepper EOs. Monoterpenes and sesquiterpenes were found to be riched in these EOs, of which α-pinene, β-pinene, sabinene, 3-carene, limonene, and (E)-β-caryophyllene were the major constituents. Most of pepper EOs showed potential AChE inhibitory activity with half-maximal inhibitory concentration (IC50) values in the range of 0.5-182.5 µg/mL. Comparison of chemical constitutes of pepper EOs from different cultivars suggested that α-pinene, β-pinene, and 3-carene with an IC50 value of 3.2, 53.3, and 2.9 µg/mL, respectively, might be used as Quality-marker (Q-marker) of pepper oil in inhibiting AChE.
Pepper is the most commonly used spice and is called “the King of Spices,” which is mainly made of fruit or seed of Piper nigrum (Piperaceae). 1 They are also used as medicine and natural food preservative. 1,2 Four different forms of pepper products, including green, red, black, and white peppers are available in the market, which is prepared by different process methods. 3 The most commonly known ones are black and white peppers, which are made of ripe fruit and ripen fruit without the peel, respectively. Essential oils (EOs) are one of the main chemical compositions of pepper, of which monoterpenes (60%-80%) and sesquiterpenes (20%-30%) are predominant compounds. 4 Among them, the oxygenated terpenes mainly contributed to the characteristic odor of pepper oil, although they are comprised of less than 4%. Pepper EOs are widely used for making perfumes. Aromatherapy also used pepper EOs to quitting nicotine, 5 improving neck pain, 6 reducing the symptoms of lymphoedema, 7 and so on. It is reported that pepper EOs showed antimicrobial, 8 antioxidant, 9 antihyperuricemic, 9 herbicidal, 9 insecticidal, 10 antipoliferative, 11 and neuroprotective 12 activity as well.
Pepper is originally native to India. Now there are many cultivars of peppers over the world, for example, Malaysia, Vietnam, Madagascar, Cambodia, China, and so on. 13 Previous publications reported that the four major cultivars of black pepper EO from Kerala, viz Kottanadan, Ottaplackal, Kuthiravally, and Cheriakaniakadanin, India, contained different main components. 13 We reported that black pepper EO had more diverse chemical constituents than that of white pepper. 14 It is noted that the yields and chemical compositions of the pepper EO have been found to vary significantly in the samples depending on cultivating locations and process methods. Dr. M. McCarron et al suggested that the ratio of the total content of sabinene, β-pinene, and 3-carene to the content of caryophyllene should be suitable pepper oil characteristics. 15 Wang et al reported that some stereospecific compositions of pepper oil, for example, β-pinene, sabinene, limonene, and terpinen-4-ol, can be used as chiral signatures of black pepper oils. 16 However, comparative research on AChE inhibitory activity of different pepper EOs was unavailable. Herein, 26 pepper EOs from 6 major cultivars were analyzed by gas chromatography-mass spectrometry (GC-MS), and their acetylcholinesterase (AChE) inhibitory activity was tested. The results suggested that pepper EOs from different locations showed different AChE inhibitory activities. α-pinene, β-pinene, and 3-carene as one of the major active chemical constituents were used as Q-markers 15 of pepper oil in inhibiting AChE.
Materials and Methods
Chemicals
Acetyl thiocholine iodide (ATCI) and 5,5′-dithiobis (2-nitrobenzoic acid) (DTNB) were purchased from Adamas Reagent Co., Ltd. Fast Blue B salt, 1-naphthyl acetate, and AChE (from electric eel [EC 3:1.1.7], Sigma C 2888; 1000U) were purchased from Sigma. Huperzine A was purchased from Shanghai Mindray Chemical Technology Co., Ltd. purity >98% by high-performance liquid chromatography. The deionized water was used in the bioassay.
General Procedures
The AChE inhibitory assay was performed in 96-well microplates, and absorbances were measured by using an Emax precision microplate reader.
Plant Materials
The white pepper (SX61) and the black pepper (SX73) were collected from Wenchang county, Hainan province, China. The white pepper (SX63) and the black pepper (SX75) were collected from Wanning county, Hainan province, China. The white pepper (SX62, SX132) and the black pepper (SX133, SX134) were collected from Baoshan county, Yunnan Province, China. The white pepper (SX97) and the black pepper (SX104) were collected from Malaysia. The white pepper (SX99) and the black pepper (SX107) were collected from Cambodia. The white pepper (SX100-SX103) and the black pepper (SX106) were collected from Indonesia. The white pepper (SX128) and the black pepper (SX129) were collected from Lvchun county, Yunnan province, China. The white pepper (SX130) and the black pepper (SX131) were collected from Yingjiang County, Yunnan Province, China. The white pepper (SX98) and the black pepper (SX82) were collected from Hainan province, China. The black pepper (SX105) was collected from India. The black pepper (SX108-SX111) was collected from Vietnam, identified by Dr Shao-Bin Shen at Yunnan Academy of Agricultural Sciences, Baoshan, Chinese Academy of Science. Voucher specimens were deposited at the College of Life Science and Technology, Kunming University of Technology, Kunming, Yunnan, China. The detailed information of all the aforementioned samples is shown in Supplemental Table S1.
Extraction of EOs
The dried ground pepper material (100 g) and distilled water (1500 mL) was added in a round bottom flask. After standing alone overnight, the mixture was refluxed for 6 hours by the essential oil extractor (keeping micro-boiling). The refluxing liquid was collected and extracted 3 times with ether, then, dried with anhydrous sodium sulfate, and evaporated to dryness at room temperature.
GC-MS Analysis of EOs
GC-MS was used to analyze the chemical ingredients of pepper EOs. Chromatographic separation was accomplished on an HP-5 MS column (30 m × 0.25 mm i.d.; film thickness: 0.25 µm; Agilent, USA). Helium was used as carrier gas at a flow rate of 1.5 mL/min. Each essential oil was diluted with n-hexane (analytically pure) to 1 mg/mL and filtrated using a 0.45-µm filter. A measure of 0.2 µL sample was injected into the GC-MS (Agilent US15173042) and unsplit stream sampling. The oven temperature was programmed as follows: 50 °C held for 2 minutes, then increased to 220 °C at 5 °C/min, held isothermal for 10 minutes. The injector temperature was set at 250 °C. The mass-selective detector was operated in an electron-impact ionization (EI) source (70 eV) was used at full scan mode with a mass scan range from m/z 50 to 550. Ingredients of these 26 EOs were analyzed by GC-MS 5977A and identified by the National Institute of Standards and Technology 14 (IST, USA) with a similarity of more than 80%. Data were deconvolution processed with Masshunter Qualitative Analysis B.07.00. Using saturated n-alkane mixture (C7-C30), the retention index (RI) value for each ingredient was calculated.
AChE Inhibition Assay
A modified Ellman’s method was applied to measure the AChE inhibitory activity. 14
Statistical Analysis
All experiments were carried out in triplicate. Data were expressed as means ± SD. The concentration giving a half-maximal inhibitory concentration (IC50) was calculated by nonlinear regression using Excel 2016 for Windows (Microsoft office Software, 2016, USA).
Results and Discussion
The Yields of the Pepper EOs
The yields of 26 pepper EOs, including 12 white ones, 13 black ones, and 1 peel were shown in Figure 1 and Table S1 of Supplementary material. The data suggested that the yield of white pepper EOs and black ones are 2.43% and 2.53%, respectively. Among them, the yield of black peppers EOs was higher than that of white ones if they were collected from the same location. For example, the yield of the black one (SX-129; 4.12%) was higher than that of the white one (SX-128; 3.01%). Both of them were collected from Lvchun, Yunnan province, China. In addition, the yields of white pepper EO from Indonesia and black ones from Vietnam were similar, approximately 2.0%. However, the yield of white and black pepper EOs from China was diverse, which is approximately 3 times from different locations of China. Among them, the highest yield of the black pepper EO (4.12%) was from the cultivar (SX-129) in Lvchun country of China, while the highest yield of white pepper EO (3.68%) was from the cultivar (SX-134) in Baoshan county of China. It is noted that the yield of EO from pepper peel was very low, approximately 0.01%.
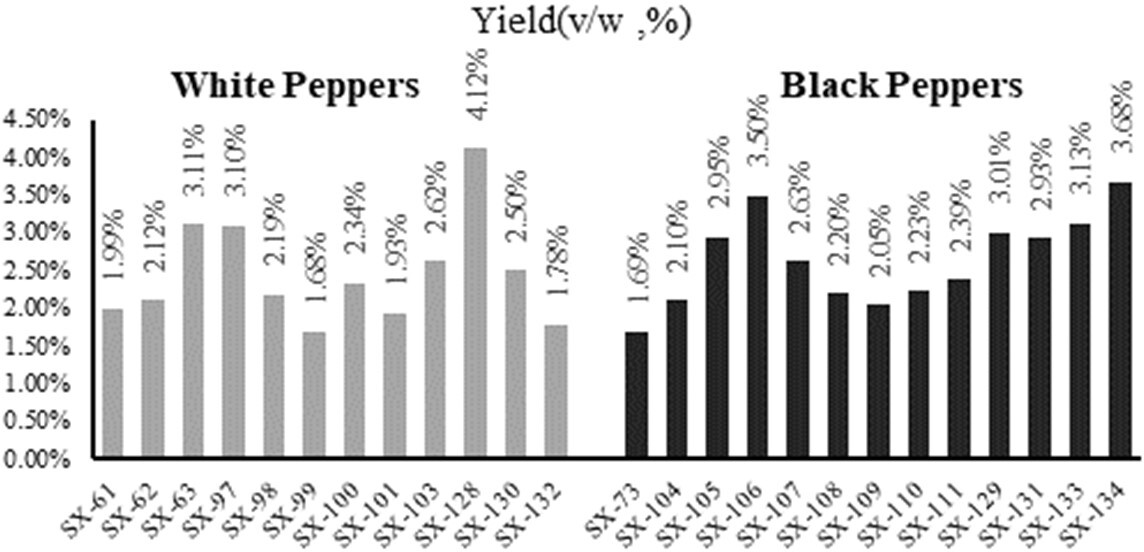
The yields of white and black pepper essential oils from different cultivars.
The Chemical Compositions of the EOs From the Pepper Samples
The data (Figure 2 and Tables S2 and S3 of Supplementary material) showed the chemical compositions of 26 pepper EOs collected in 6 main producing areas, including Hainan and Yunnan province of China, Malaysia, Cambodia, India, Indonesia, and Vietnam. Herein, 58 volatile compounds were identified from 12 white pepper EOs, including 12 monoterpene hydrocarbons, 10 oxygenated monoterpenes, 35 sesquiterpenoids, and 1 aromatic compound. Almost 75 volatile compounds were identified from the 13 black pepper EOs, including 16 monoterpene hydrocarbons, 10 oxygenated monoterpenes, 41 sesquiterpene hydrocarbons, 7 oxygenated sesquiterpenes, and 1 aromatic. Thirty-seven volatile compounds were identified from the pepper peel EO collected from the Hainan province of China, including 26 sesquiterpenes, 9 monoterpenes, 1 aromatic, and 1 aliphatic compound. The data suggested that the total contents of monoterpenes and sesquiterpenes of peppers from different areas were similar, approximately 60% and 30%, respectively. Among them, 11 common compounds were detected in the black pepper EOs, including 5 monoterpenoids (α-pinene, α-phellandrene, limonene, δ-terpinene, and β-geraniolene) and 6 sesquiterpenes (δ-elemene, β-elemene, (E)-β-caryophyllene, α-cubebene, α-selinene, and α-gurjunene). Nine common chemical constituents were detected in white pepper EOs, including 5 monoterpenes (α-pinene, α-phellandrene, limonene, δ-terpinene, and β-pinene) and 4 sesquiterpenes (δ-elemene, (E)-β-caryophyllene, germacrene D, and isospathulenol). It is noted that 4 major chemical constituents, for example, α-pinene, limonene, δ-terpinene, and (E)-β-caryophyllene, were detected in both black and white peppers EOs. (E)-β-caryophyllene was the major sesquiterpenes with more than 30% of contents in the white pepper EOs from the cultivar in Lvchun county, Yunnan, China. The content of (E)-β-caryophyllene with almost 27% in black pepper produced in Lvchun county, Yunnan province, China, was highest in all the black samples. (E)-β-caryophyllene was approved by United States Food and Drug Administration and European agencies as a food additive, taste enhancer, and flavoring agent and termed as a phytocannabinoid. 15 Furthermore, a previous publication reported that sabinene and terpinen-4-ol seemed to be the most important components in the contribution of volatile substrates to the characteristic odor of black pepper oil. 17 However, the 2 major odor constituents, sabinene and terpinen-4-ol, were not detected in EOs of black pepper from China and Vietnam in this study.

The main chemical families of essential oils of peppers.
Currently, the quality of black pepper oil is regulated under the International Standard Organization (ISO) “ISO 3061:2008 Oil of black pepper (P. nigrum L.)”, in which the content of some major chemical constituents from 4 countries, including India, Sri Lanka, Indonesia, and Madagascar, was limited. 16 For example, it suggested that the content of β-pinene of the black pepper EOs produced in India should be 5%-12%. The content of β-selinene was 3%-6% in the EOs of black pepper produced in Indonesia. However, in this study, neither the β-pinene was detectable in Indian black pepper nor the β-selinene was detectable in Indonesian black pepper. Furthermore, the international standard of white pepper was unavailable, as well as that of the black pepper from other areas such as China, Vietnam, and Cambodia. As we all know, the diverse components from different locations may be due to the geographical factors, post-crop processing, and different nutritional status of the plants. We suggested that the ISO 3061:2008 oil of black pepper (P. nigrum L.) should be revised considering the absence of β-pinene and β-selinene constituent in black pepper EOs from China, Vietnam, and Cambodia. Also, the ISO of white pepper EOs should be established since it is one of the popular spices in the world.
AChE Inhibitory Activity of the EOs
The AChE inhibitory bioassay was utilized to identify cholinergic compounds for the potential treatment of Alzheimer’s disease (AD). Twenty-six pepper EOs and their major active chemical constituents, α-pinene, β-pinene, and 3-carene, were tested for AChE inhibitory activity by a modified Ellman’s method. It is noted that some white pepper EOs (SX-99, SX-63, SX-103, SX-100, SX-130, and SX-97) showed potential activity with IC50 values of 0.7, 11.8, 12.5, 14.3, 14.3, and 14.6 µg/mL, respectively, as well as some black pepper EOs (SX-105, SX-106, SX-107, SX-110, SX-111, and SX-109) with IC50 values of 0.5, 3.3, 6.5, 6.9, 13.1, and 13.6 µg/mL, respectively (Table 1). Comparison of chemical constitutes of pepper EOs from different cultivars led to speculate that α-pinene, β-pinene, and 3-carene might be major active compounds in both white and black pepper EOs, and α-pinene should be the major component contributing to AChE inhibitory activity. Most of the white pepper EOs contained a higher concentration of α-pinene than that of black pepper EOs, and most of the white pepper EOs showed more potential activity than those of black pepper. Experimental verification of AChE inhibitory activity showed α-pinene, β-pinene, and 3-carene with an IC50 value of 3.2, 53.3, and 2.9 µg/mL, respectively. And total contents of α-pinene, β-pinene, and 3-carene were consistent with the order of AChE inhibitory activity of pepper EOs. We suggest that pepper EOs, especially white ones, are an important resource for exploring AChE inhibitors for AD therapy, and α-pinene, β-pinene, and 3-carene could be used as Q-markers (Figure 3).

Selective structures of chemical constituents of pepper (active compounds marked blue and odor compounds marked red).
Acetylcholinesterase Inhibitory Activity of the Pepper Essential Oils.

Abbreviation: IC50, half-maximal inhibitory concentration.
Data are expressed as mean ± SD (n = 3).
Conclusion
Monoterpenes and sesquiterpenes were found to be rich in the EOs from different cultivars, of which (E)-caryophyllene, α-pinene, β-pinene, sabinene, 3-carene, and limonene were the major constituents. Sabinene was the typical chemical constituent of black pepper EOs which gives them the odor. However, due to geographical factors, post-crop processing, and different nutritional status, the pepper EOs showed different chemical constituents and AChE inhibitory activity. Among them, white pepper produced in Cambodia and black pepper from India showed potential AChE inhibitory activity with IC50 values of 0.7 and 0.5 µg/mL, respectively. Comparison of chemical constitutes of pepper EOs from different cultivars suggested that α-pinene, β-pinene, and 3-carene with an IC50 value of 3.2, 53.3, and 2.9 µg/mL, respectively, should be used as Q-markers of pepper oil in AChE inhibitory activity. The quality of black pepper from India and white pepper from Cambodia and Indonesia was better than those from other cultivars due to their potential AChE inhibitory activity and sweet smell. In summary, herein, it is the first time to compare AChE inhibitory activity of the pepper EOs from different cultivars, which led to the suggestion of α-pinene, β-pinene, and 3-carene being Q-markers of pepper oil as a potential AChE inhibitor. However, it is important to collect more pepper samples to establish the authenticity and quality of white and black pepper from different cultivar by Q-markers. 16
Supplemental Material
Supplementary Material 1 - Supplemental material for Comparison of Chemical Compositions of the Pepper EOs From Different Cultivars and Their AChE Inhibitory Activity
Supplemental material, Supplementary Material 1, for Comparison of Chemical Compositions of the Pepper EOs From Different Cultivars and Their AChE Inhibitory Activity by Shu-Xia Chen, Kong Yang, Jia-Yao Xiang, Osafo Raymond Kwaku, Jia-Xin Han, Xian-Ai Zhu, Ya-Ting Huang, Le-Jiang Liu, Shao-Bin Shen, Hai-Zhou Li, Xing-Cong Li, Yang Feng and Min Xu in Natural Product Communications
Footnotes
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Kunming University of Science and Technology, China, and approved by the Administration Committee of Experimental Animals, Yunnan Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSFC 31960093, Program for Innovative Research Team (in Science and Technology) in University of Yunnan Province and Ronald J Quinn AM Academician Workstation (2019IC003).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
