Abstract
Atherosclerosis is no longer a disease attributed mainly to high cholesterol content in the body; it has come to be regarded as a chronic inflammatory disease with an autoimmune component. The purpose of this study was to investigate the effect of the prunin fraction (PF) isolated from the ethanolic extract of Bauhinia variegata against the release of various proinflammatory mediators in rats fed an atherogenic diet. The diet was administered orally to Sprague Dawley rats for 60 days to induce atherosclerosis. The blood serum of the rats was used to estimate the levels of tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), thiobarbituric acid reactive substance, catalase, total cholesterol, triglyceride, low-density lipoprotein, and high-density lipoprotein using assay kits. Other physical parameters, such as body weight, feed intake, and systolic blood pressure, were also determined during the study. The results showed a significant protective effect of the PF against diet-induced atherosclerosis by decreasing the levels of proinflammatory mediators such as TNF-α and IL-6. Rats treated with PF (20 and 40 mg/kg) showed a change in systolic blood pressure and a reduction in oxidative stress induced by the atherogenic diet. Reduction in body weight and modulation of food intake were observed in PF-treated rats, which indicated atheroprotective, hypolipidemic, and antioxidant effects. The study concludes that the atheroprotective properties of PF are due to effects on the initial phase of plaque formation to thrombus formation. This study may help researchers to find a better alternative for selecting optimal therapies and preventing plaque formation.
Atherosclerosis is a multifocal, immune-inflammatory disease of medium and large arteries characterized by cholesterol accumulation, connective tissue components, macrophage infiltration, smooth muscle cell (SMC) proliferation, and thrombus formation. 1,2 The atherosclerotic process is initiated when low-density lipoprotein (LDL) cholesterol (“bad” cholesterol) accumulates in the artery. 3 This LDL is subjected to various types of modifications, including oxidation. In response, cells of the immune system are recruited to the artery to absorb the modified LDL, which leads to local inflammation. As this process continues, atherosclerotic plaques form. A ruptured plaque triggers the formation of a blood clot inside the artery, and a heart attack occurs if the blood clot blocks the artery. 4 A combination of cholesterol clefts and cell disappearance was interpreted as showing the origin of the atherosclerotic core. 5
Advancements in pharmacotherapy are limited to a few targeted factors, which stimulate the release of lipoproteins. Recent research has shown the major causative factors of atherosclerosis, such as nuclear factor kappa-light-chain-enhancer of activated B cells, endotoxins, oxidative stress, and impaired calcium signaling.
6
Despite modern drugs, those obtained from other resources, such as plants, have been found to be prophylactic against atherosclerosis. These traditionally used drugs are taken from a wide population. Some of the traditionally used plants are Quercus robur, Q. infectoria, Medicago sativa, Gundelia tournefortii, Pulicaria gnaphalodes, Valeriana officinalis, Eremurus persicus, and Sesamum indicus
7
. Bauhinia variegata (Fabaceae) is found throughout India, at altitudes of up to 1300 m in the Himalayas.
8
The constituents of B. variegata root include β-sitosterol, lupeol, kaempferol-3-glucoside, 5,7-dehydroxy and 5,7-dimethoxyflavanone-4-O-α-
Materials and Methods
Collection and Authentication of Plant Material
Roots of B. variegata L., collected from the herbal garden of Shandong, China, in July 2017, were identified and authenticated by Sr. scientist Dr Qi Lin, Herbarium (PE), Institute of Botany, Chinese Academy of Sciences, 20 Nanxincun Xiangshan Beijing 100093, China. A voucher specimen was lodged in the Herbarium (V. no.475494).
Preparation of Ethanolic Plant Extracts
The freshly collected root of B. variegata was dried in the shade and pulverized into a coarse powder. The root powder (1200 g) was extracted in a Soxhlet apparatus using hot ethanol for 8 complete cycles over 48 hours. The filtrate was evaporated to dryness under reduced pressure in a rotary vacuum evaporator. The yield of ethanolic extract was 8.83% (w/w).
Estimation of Total Flavonoids
The aluminum chloride colorimetric technique was used for the estimation of flavonoids. 9 The amount of flavonoid was calculated from a standard quercetin graph. 10
Thin-Layer Chromatography
All extracts were examined by thin-layer chromatography (TLC). Identification of compounds was based on the use of appropriate standards and previous methods described by Harborne. 7 The mobile phase was nitromethane:methanol (5:2 v/v), and the compounds were identified by spraying with 1% methanolic aluminum chloride solution and observing under ultraviolet light.
Isolation of Prunin From Bauhinia variegata
Prunin was isolated from B. variegata roots according to the method described by Choi et al 23 and identified by direct comparison with a standard sample (1H and 13C nuclear magnetic resonance [NMR] spectra). 20 The melting point was found to be −220°C to 223°C. The molar concentration of prunin was determined by comparing naringenin released by complete hydrolysis of the prunin fraction (PF) with a standard curve of naringenin. 24
Experimental Animals
The experimental protocols used in this study were approved by the Institutional Animal Ethical Committee. Sprague Dawley rats (6-8 weeks) weighing about 180-200 g were used. The animals were maintained in controlled temperature and humidity, with a standard diet and water ad libitum.
Experimental Methodology
Atherogenic diet-induced aAtherosclerosis
Animals were weighed, numbered, and divided randomly into 5 groups of 6 animals each. All groups except for the control group were given an atherogenic diet (Table 1). Group II received an atherogenic diet for 60 days. Group III received atorvastatin along with an atherogenic diet for 60 days. Group IV received PF (20 mg/kg, orally) and Group V PF (40 mg/kg, orally), along with an atherogenic diet for 60 days (Table 2). The atorvastatin and PF were administered orally and suspended in distilled water using carboxymethylcellulose (CMC, 2%) as a suspending agent before administration.
Composition of Atherogenic Diet.

Animals Grouping of Atherogenic Diet-Induced Atherosclerotic Model.

Estimation of total cholesterol (CHOD-PAP method)
Total cholesterol was estimated by the cholesterol oxidase/peroxidase (CHOD-PAP) method described by Allain, 25 using a commercially available kit.
Estimation of triglycerides (GO-PAP method)
Serum triglyceride was estimated by the glycerophosphate oxidase peroxidase (GO-PAP) method 20 using a commercially available kit.
Estimation of HDL-cholesterol (phosphotungstate method)
High-density lipoprotein (HDL) was estimated by a phosphotungstic acid method 21 using a commercially available kit.
Estimation of LDL-cholesterol
LDL-cholesterol was determined using the Friedewald formula. 22

Estimation of very LDL-cholesterol
Very LDL (VLDL)-cholesterol was estimated using the Friedewald formula. 23

Atherosclerotic index and LDL/HDL ratio
This was measured by the following equation.
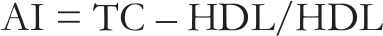

In vivo pharmacological evaluation parameters
The body weight of healthy animals in each group was recorded from day 1 and at weekly intervals throughout the study period using a digital balance.
Feed intake of each group was measured on daily consumption by each rat for 60 days using a digital balance.
Measurement of systolic blood pressure (SBP): This was measured using a noninvasive tail-cuff method (AD Instrument PowerLab Data Acquisition System, Sydney, Australia) on the first and last days of the experiment (days 1 and 60) in conscious rats. The animals were restrained in heated chambers at 38 ± 1 °C for 10 minutes. Rats were trained with a blood pressure measuring equipment for 1 week before the initiation of the experiment. Blood pressure was recorded for each rat three times and averaged to obtain the mean SBP.
Estimation of oxidative stress parameters
Estimation of thiobarbituric acid reactive substance (TBARS) in rat heart: malonaldehyde (MDA), a decomposition product of lipid hydroperoxides, is used as an indicator of oxidative damage to cells and tissues. In this method, the sample was heated with thiobarbituric acid (TBA) under acidic conditions. A small amount of MDA, produced during the lipid peroxidation of polyunsaturated fatty acids in the plasma membrane, reacts with TBA to generate a colored product. In acid solution, the product, which absorbs light at 532 nm, is readily extractable into organic solvents, such as n-butanol. 24
Estimation of catalase (CAT) activity in rat heart: the samples were incubated with hydrogen peroxide (H2O2) at pH 7; catalase breaks down part of the H2O2, and the remaining H2O2 is determined by potassium dichromate reagent. H2O2 reacts with potassium dichromate in the presence of glacial acetic acid at high temperatures to produce chromic acetate, which was measured using a colorimeter at 560-630 nm. Dichromate itself does not show any absorbance in this region. 26
Estimation of proinflammatory cytokines: tumor necrosis factor-alpha (TNF-α) and interleukin-1β (IL-1β) were estimated using an enzyme-linked immunosorbent assay kit. 25
Statistical Analysis
The data were analyzed using Graph Pad Prism 7.0. The results are expressed as mean ± SEM (n = 6), followed by one-way analysis of variance with Tukey’s multiple comparison tests, with significance set at P < 0.001.
Results
Results of the Atherogenic Diet-Induced Model of Atherosclerosis
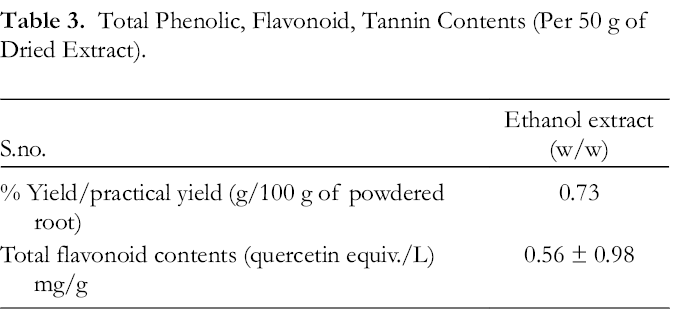
The total flavonoid content of the B. variegata root extract was found to be 0.56 ± 0.98 quercetin equiv./L/mg/g of extract (Table 3). Prunin content in fractions was identified by direct comparison with authentic samples (1H and 13 CNMR). The prunin content in the PF was found to be 0.016 ± 0.56 naringenin equiv./L, where naringenin was used as a standard for prunin content determination. Colorless crystals of PF were observed, and the structure of prunin is shown in Figure 1.
Total Phenolic, Flavonoid, Tannin Contents (Per 50 g of Dried Extract).

Structure of prunin.
Effect of PF on body weight
There was no significant difference in the treatment groups at the beginning of the study. Group II animals fed on an atherogenic diet exhibited a significant (P < 0.001) increase in body weight between weeks 1 and 8 compared with the control group. Treatment with PF (40 mg/kg, orally) produced a significant (P < 0.001) decrease in body weight as compared with the atherogenic diet-treated Group II (Figure 2).

Effect of PF on body weight. Body weight of rats fed a high-fat diet and treated with PF extract for 8 weeks; all values are expressed as mean ± SEM (n = 6) and the significant difference compared with that in the atherogenic diet group: *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.
Effect of PF on food intake
There was an increase in food intake in the atherogenic diet-induced rats (Group II) as compared with those in Group I. Similar food intake was observed in the first 4 weeks of the study in the remaining 4 groups, suggesting that treatment with PF at doses of 20 and 40 mg/kg of extract in groups IV and V significantly decreased the food intake compared with that in the atherogenic Group II. The results are shown in Figure 3.

Effect of PF on food intake: rats fed a high-fat diet and treated with PF extract for 8 weeks; all values are expressed as mean ± SEM (n = 6). Significant difference compared with that in the atherogenic diet group; *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.
Effect of PF on lipid profile
The levels of plasma lipids, including total cholesterol, triglycerides, LDL, and VLDL, was significantly (P < 0.001) higher, while the HDL level was significantly (P < 0.001) decreased, in the atherosclerotic rats of Group II compared with those in Group I. Treatment with 20 and 40 mg PF produced a significant (P < 0.001) decrease in total cholesterol, triglycerides, LDL, and VLDL compared with the atherogenic diet control Group II. However, the HDL level was increased in the 20 and 40 mg PF groups compared with that in Group II. The results are shown in Figures 4 -8.

Effect of PF on total cholesterol content; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.

Effect of PF on total triglyceride content. All values are expressed as mean ± SEM (n = 6); significant difference compared with that in the diet group; *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.

Effect of PF on HDL content. All values are expressed as mean ± SEM, (n = 6); significant difference compared with that in the atherogenic diet group; *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). HDL, high-density lipoprotein; PF, prunin fraction.

Effect of PF on LDL content; all values are expressed as mean ± SEM (n= 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). LDL, low-density lipoprotein; PF, prunin fraction.

Effect of PF on VLDL content; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group; *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction; VLDL, very low-density lipoprotein.
Effect of PF on the atherogenic index and LDL/HDL levels
The levels of LDL/HDL and the atherogenic index were significantly increased in the atherogenic diet-treated rats (Group II) than in the normal rats. Treatment with 20 and 40 mg PF significantly decreased the atherogenic index and LDL/HDL levels compared with those in atherosclerotic rats (Figures 9–10).

Effect of PF on atherogenic index; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group,*** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.
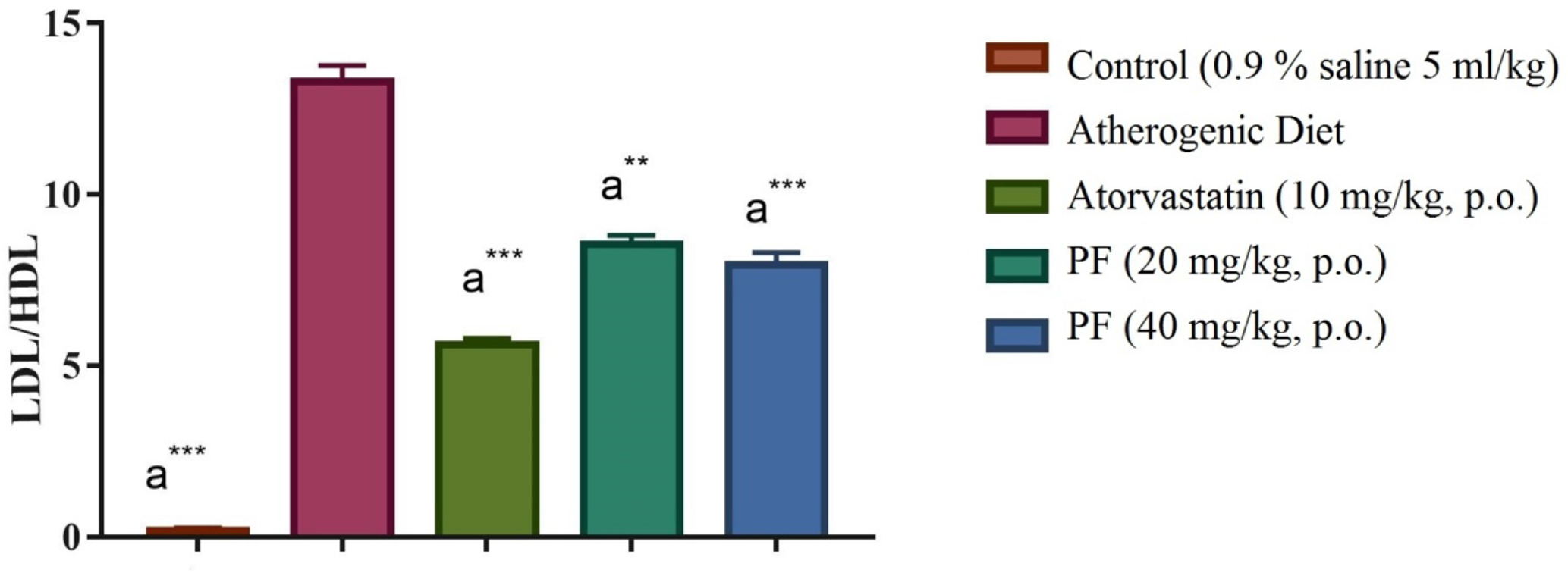
Effect of PF on LDL/HDL ratio; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). HDL, high-density lipoprotein; LDL, low-density lipoprotein; PF, prunin fraction.
Effect of PF on TBARS level
The TBARS level in Group II was significantly (P < 0.001) higher than that in Group I. However, treatment with 20 and 40 mg PF showed significantly (P < 0.001) decreased TBARS levels in the heart when compared with the atherogenic diet group (Group II). The PF in Group VI also resulted in a significant (P < 0.001) decrease in TBARS levels compared with those in Group I. The results are shown in Figure 11.

Effect of PF on TBARS level; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction; TBARS, thiobarbituric acid reactive substance.
Effect of PF on catalase levels
The catalase levels in Group II were significantly (P < 0.001) lower than those in the normal control rats in Group I. However, Groups IV and V showed a significantly (P < 0.001) increased level compared with those in Group II. The results are shown in Figure 12.
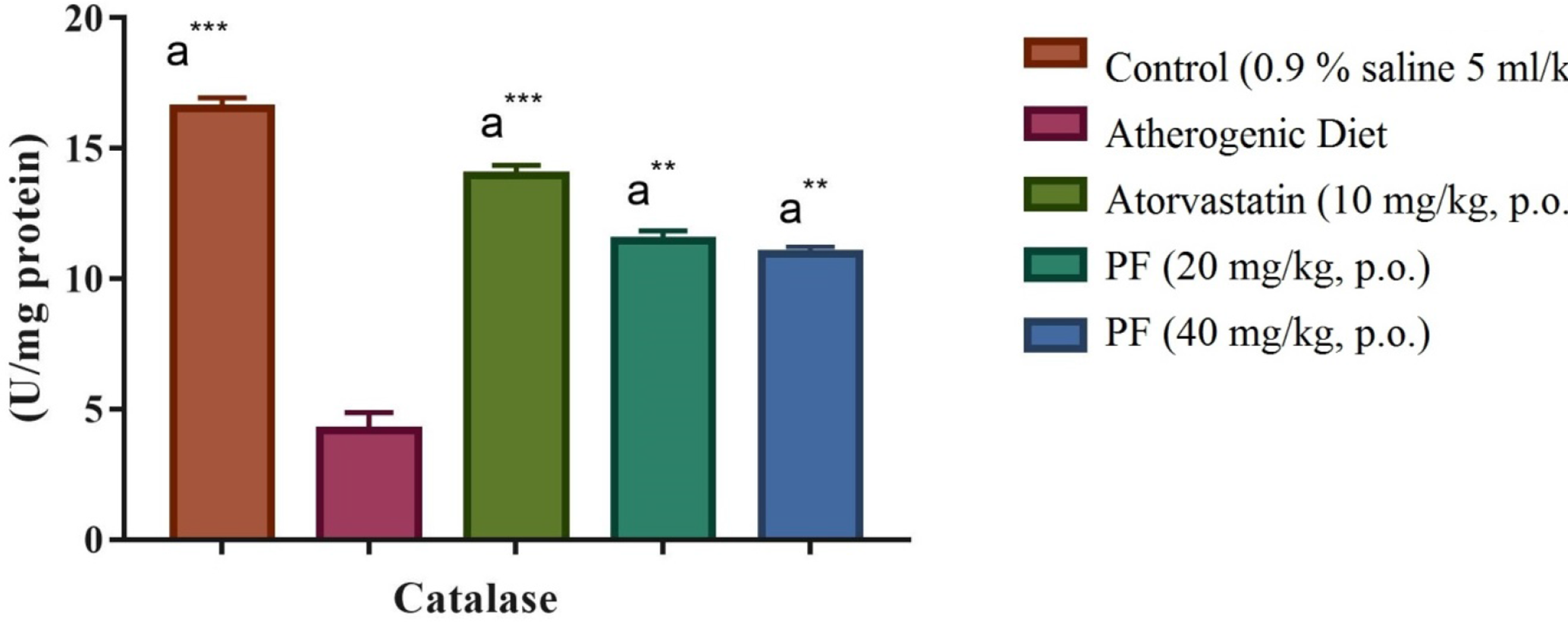
Effect of PF on catalase level; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction.
Effect of PF on SBP of rats
The initial SBP was determined and found to be normal. Later, the animals treated with an atherogenic diet showed significantly increased blood pressure owing to elevated levels of fatty acids. The rats treated with atorvastatin showed reduced blood pressure to near the normal range. Those treated with PF extract produced a significant reduction in the elevated blood pressure compared with the negative control group. The effects observed in PF extract-treated mice showed a significant reduction in the elevated level of SBP, as compared with those in the negative control group (Figure 13).

Change in systolic blood pressure from day 1 and on the 60th day of the experiment in rats. All values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, P < 0.001 (one-way analysis of variance, n = 6 per group). PF, prunin fraction.
Effect of PF on proinflammatory cytokines
All of the treated animals showed increased TNF-α levels in the serum. PF treatment produced a significant reduction compared with that in the control (Figure 14). The data shown in Figure 15 reveals that the treatment of rats with PF at all doses significantly reduced the serum content of IL-1 compared with that in the negative control group.

Effect of PF on TNF-α level; all values are expressed as mean ± SEM (n = 6); significant difference compared with atherogenic diet group; *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). PF, prunin fraction; TNF-α, tumor necrosis factor-alpha.

Effect of PF on IL-1β level; all values are expressed as mean ± SEM (n = 6); significant difference compared with that in the atherogenic diet group, *** P < 0.001, ** P < 0.01, * P < 0.05 (one-way analysis of variance, followed by Dunnett’s test, n = 6 per group). IL, interleukin; PF, prunin fraction.
Histopathological Observations of the Atherogenic Diet-Induced Model
The aorta of the atherogenic diet-fed rats developed plaques and thus thickening of the intima, migration of smooth muscle cells into the intima, adhesion, and infiltration of macrophages, which resulted in the loss of elastic lamellae under the endothelium, as shown in Figure 16. Standard and test drugs showed normal intimal thickening without any inflammatory infiltration.

Demonstrated no obsessive changes (ordinary intima) in rats of the control group (100×). Rats sustained on an atherogenic diet created typical plaques (shown by arrow), described by thickening of the intima, movement of smooth muscle cells to the intima, attachment and penetration of macrophages, appearance of foam cells, and loss of normal arrangement of elastic lamellae under the endothelium (100×). Standard and test compound-treated rats demonstrated typical intimal average thickening without provocative cell penetration comparable with that of control (100×).
Discussion
Atherosclerosis and its thrombotic complications remain the most common cause of morbidity and mortality in developed countries. Atherosclerosis is an oxidative, inflammatory, and thrombotic disease of the arterial wall that is precipitated by elevated levels of LDL cholesterol in the blood. 27 Hyperlipidemia is one of the important factors associated with atherosclerosis other than hypertension, smoking, and diabetes mellitus. Diet is an important tool for the prevention of cardiovascular diseases. 28
In dietary hyperlipidemia, on one hand, increased accumulation of cholesterol in the serum and tissue increases free radical production. 29,30 On the other hand, the free radicals produced are capable of enhancing β-hydroxy β-methylglutaryl-CoA (HMG Co-A) reductase activity, resulting in the increased endogenous production of cholesterol. 29 The combined effects of cholesterol reduction and mitigating oxidative stress may improve endothelial function in hyperlipidemic individuals. 31 Our study supported the previous findings in reducing oxidative stress levels at cellular sites. A cholesterol-enriched diet resulted in a significant increase in the total cholesterol, triacylglycerol, and phospholipid levels in plasma, which is accompanied by an increased serum LDL-C level with decreased circulating HDL-C, thus providing a model for dietary hyperlipidemia. 32 The increase in lipid parameters is a strong risk factor for coronary heart disease in many populations. The high cholesterol level in tissue and serum may be due to the increased uptake of exogenous cholesterol and subsequent deposition and decreased cholesterol catabolism, as evidenced by a reduction in bile acid production and the turnover of bile acids. The metabolism of free and ester cholesterol is impaired in the liver, spleen, and thymus tissues, and the rate of turnover was specifically decreased in all tissues of hyperlipidemic rats. 33 A high-fat diet or hypercholesterolemia also induces oxidative stress, leading to free radical generation that promotes lipid peroxidation and enhances foam cell formation. 34 In hypercholesterolemia, there are high levels of lipids and phospholipids. Therefore, there is increased production of arachidonic acid and prostaglandins (PGs) with the help of phospholipase A2 and cyclooxygenase enzymes. 35 Free radicals are produced during the production of PGs. 33 Leukocytes and monocytes are the main sources of free radical generation. 36 Elevated levels of serum glutamic pyruvic transaminase have also been implicated in increased cardiovascular disease risk, via underlying nonalcoholic fatty liver disease (which is closely related to 3 important pathogenic risk factors for diabetes and cardiovascular disease: (1) ectopic fat deposition; (2) insulin resistance; and (3) adverse profiles in adipokine levels), endothelial dysfunction and vessel wall damage, inflammation, oxidative stress, and impaired homeostasis. The excess cardiovascular risk associated with alkaline phosphatase has been suggested to be via mechanisms related to vascular calcification through increased bone metabolism, impaired vascular homeostasis, proinflammatory activities, and subclinical liver dysfunction. 36
Atorvastatin is a lipid-lowering agent that comes from a class of HMG-CoA reductase inhibitors. It competitively inhibits the enzyme HMG-CoA reductase in the liver that converts HMG-CoA to mevalonate, an early precursor for cholesterol synthesis. 37 Lipid deposition is a major clinical complication of hyperlipidemia. 38 High serum lipid levels, especially elevated levels of LDL, are strongly related to the development of atherosclerosis. It is generally accepted that atherosclerotic lesions are initiated via the enhancement of LDL uptake by monocytes and macrophages. 39 In this study, atherogenic diet-induced rats resulted in increased total cholesterol, triglycerides, LDL, and VLDL levels and decreased HDL levels. However, treatment with PF 20 and 40 mg significantly (P < 0.001) decreased levels of total cholesterol, triglycerides, LDL, and VLDL and increased levels of HDL. 40 -45
Cardiovascular diseases, such as atherosclerosis and heart failure, are associated with obesity and insulin resistance. There is controversy about the cardiovascular benefits of yo-yo dieting, and an animal model is needed to explore the contributions of major diet and body weight changes on the heart and vascular functions. 46 In this study, there was a significant difference between the body weights of the atherogenic and normal diet groups. This result indicates that an increase in body weight is independent of the amount of food consumed by animals. The results of this study established that an atherogenic diet causes an elevation in body weight and feed intake. However, supplementation with PF reverses all of the parameters, thus suggesting its weight-reducing potential. 47
Reactive oxygen species can impair endothelial function and increase systemic and intrarenal proinflammatory and fibrogenic factors, possibly triggering a complex sequence of mechanisms involved in atherosclerosis. 48 -52 In our study, TBARS levels were significantly increased in the atherogenic diet and Triton-induced rats, while the catalase level was decreased in both groups. However, treatment with PF showed decreased levels of TBARS and increased levels of catalase compared with those in Group II for both models.
Histopathological evaluation of the aorta of the atherogenic diet-fed rats 53 showed developed plaques and thus thickening of the intima, migration of smooth muscle cells into the intima, adhesion, and infiltration of macrophages, which resulted in the loss of elastic lamellae under the endothelium. Standard and test drugs showed normal intimal thickening without any inflammatory infiltration. 54 -58
Our findings from this study reveal that PF may act as an antiatherosclerotic, antioxidant, anti-inflammatory, and hypolipidemic agent. Thus, we can say that treatment with PF may have an antihyperlipidemic effect. 59,60 Since the findings are novel and preliminary, further studies need to be conducted to confirm the safety profile of PF, such as side effects and drug interactions, and provide new alternatives for clinical use.
This study evaluated a diet-based, cost-effective, and time-efficient experimental model, which can be used to study important markers of cardiovascular disease. This model can be further developed to explore new therapeutic moieties to ameliorate atherosclerosis-like disease and reduce the length of the treatment period.
Conclusion
The findings of this study suggest that prunin protects against plaque formation and thrombus formation around arterial walls. It also protects against vascular damage caused by oxidative stress and high lipid content in the serum of rats. These results pave the path for conducting a clinical study for future investigation.
Footnotes
Acknowledgments
The authors thank all affiliated institutes for providing the necessary research facilities.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the institutional animal care review board guidelines of Taizhou Third People’s Hospital, China, and approved by the Administration Committee of Experimental Animals Jiangsu, Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
