Abstract
Oligosaccharides have important alimental and medical applications. Dextranase has been used to produce isomalto-oligosaccharides (IMOs). In this study, we isolated dextranase-producing bacteria from sugarcane-cultivated soil. Identification of the isolate based on its phenotypical, physiological, and biochemical characteristics, as well as 16S ribosomal deoxyribonucleic acid gene sequencing yielded Shewanella sp. strain GZ-7. The molecular weight of the dextranase produced by this strain was 100-135 kDa. The optimum temperature and pH for dextranase production were 40 °C and 7.5, respectively. The enzyme was found to be stable at the pH range of 6.0-8.0 and the temperature range of 20 °C-40 °C. Thin-layer chromatography and high-performance liquid chromatography of the enzymolysis products of the substrate confirmed the enzyme to be endodextranase. Under the optimal conditions, the ratio of IMOs could reach 91.8% of the hydrolyzate. The final products were found to efficiently scavenge the 2,2-diphenyl-1-picrylhydrazyl, hydroxyl, and superoxide anion radicals. In general, dextranase and hydrolyzates have high potential prospects for application in the future.
Nondigestible oligosaccharides have gained importance as functional foods in recent years.
1
Isomalto-oligosaccharides (IMOs) have a low glycemic index and are prebiotic in nature.
2
IMOs are added to or used as nutritional supplements, foods, and beverages. The production technology of IMOs and their special characteristics are in demand for a variety of purposes.
3
Dextran is fermented by microorganisms to form the main chain via linkage of
Ingelman et al were the first to report Cellvibrio fulva dextranase in the 1940s. 8 Since then, various dextranase-producing microorganisms have been reported, including bacteria and fungi. 9,10 The currently reported dextranase-producing bacteria include some species of Streptococcus, 11 Flavobacterium, 12 Bacillus, 13 Thermoanaerobacter, 14 and Arthrobacter. 15 Considering the diversity of its features and applications, the search for new dextranases with special characteristics are always in demand.
In this study, we screened dextranase-producing bacteria for some special properties of dextranase from different soil samples collected from sugarcane plants. In addition, we focused on dextranases that can be used to mass produce IMOs. We identified a new strain, GZ-7, that could produce endodextranase with a novel property. In the hydrolyzates, the IMOs could be accumulated in greater quantity. Moreover, the antioxidant activity of the hydrolyzates was found to be significant.
A total of 9 strains were screened. Of which, GZ-7 demonstrated the highest activity of dextranase, with Figure 1(A) depicting the hyaline halos on the screening medium formed by this strain. The morphological, physiological, and biochemical characteristics of the isolated strain GZ-7 are listed under online supplementary file 1. The strain GZ-7 grew under aerobic conditions and demonstrated a dextranase activity of 0.89 U/mL in the culture supernatant. After incubation on Luria-Bertani (LB) plates at 30 °C, the strain formed grayish-white colored colonies and appeared as gram-negative, rod-shaped organisms under the scanning electron microscope (size: 0.8-1 μm × 1.5-3 μm; Figure 1(B)). The 16S ribosomal ribonucleic acid (rRNA) gene of the strain GZ-7 was sequenced.

The clear zone around the colony of strain GZ-7 on the plate (A) and scanning electron micrograph (B) of strain GZ-7. The phylogenetic tree of GZ-7 based on 16s ribosomal ribonucleic acid gene sequencing (C). The evolutionary distances were calculated using MEGA 7, with 0.002 substitutions per nucleotide.
The 16S rRNA gene of strain GZ-7 was sequenced and compared with the other 16S rRNA sequences of the GenBank. The similarity levels between GZ-7 and Shewanella sp. are summarized in Figure 1(C), with GZ-7 sharing 99.7% similarity with Shewanella baltica (NR 025267.1). A phylogenetic tree was constructed to compare the 16S rRNA gene sequences, which indicated that the strain GZ-7 belonged to the genus Shewanella. Based on the morphological and biochemical characterization, the selected strain was identified to be Shewanella sp. and denoted as “Shewanella sp. GZ-7”. Until date, to the best of our knowledge, this is the first report of dextranase produced by a Shewanella species.
Presently, most strains reported to produce dextranase include molds, bacteria, and yeast, such as Chaetomium, 10 Penicillium, 9,16,17 Aspergillus subolivaceus, 18 Sporothrix schenckii, 19 Bacillus sp., 13 Lipomyces starkeyi, 20 and L. starkeyi NCYC1436. 21 Dextranases produced by molds have a longer fermentation time and higher energy consumption when compared with bacteria. 22 In addition, the properties of dextranases obtained from various sources are discrepant. Therefore, screening for prokaryotic microorganisms that produce dextranase is meaningful.
Until date, the number of known bacterial dextranases is lesser than that of fungal dextranases. For instance, Wynter et al reported an anaerobic thermophilic bacterium; 23 Igarashi et al reported Streptococcus rattus 8 ; Ruanhong et al reported Catenovulum sp. DP03, 24 and Purushe et al reported Streptomyces sp. NK458. 6 The strain Shewanella GZ-7 reported herein is a new bacterium screened for dextranase production.
The optimal culture conditions for strain GZ-7 to induce dextranase production were investigated. The evaluation of different carbon and nitrogen sources revealed dextrin and tryptone extract (Figure 2(A) and (B)) to be the most suitable ones, respectively. Strain GZ-7 could grow at 0 °C, but its optimal growth temperature was 30 °C (Figure 2(C)). As shown in Figure 2(D), the strain GZ-7 grew well at the pH range 6.5-8.5, but its optimum growth pH was at 7.5.

Effects of carbon (A) and nitrogen (B) sources, temperature (C), and pH (D) on the growth of strain GZ-7. OD, optical density.
Oxygen is critical for the growth of the strain GZ-7. The higher the rotational speed of the growth medium, the more is the availability of oxygen, and hence better is the growth of the strain. At the incubational rotation speed of 180 rpm, the number of cells grown was the maximum (Figure 3(A)). The inoculation concentration of 2.5% was found to be optimal (Figure 3(B)). When the liquid medium volume was 20%, the growth of the strain was the maximum (Figure 3(C)). The GZ-7 had a short lag phase (less than 2 hours) (Figure 3(D)), but it entered a stable phase after 10 hours of incubation.
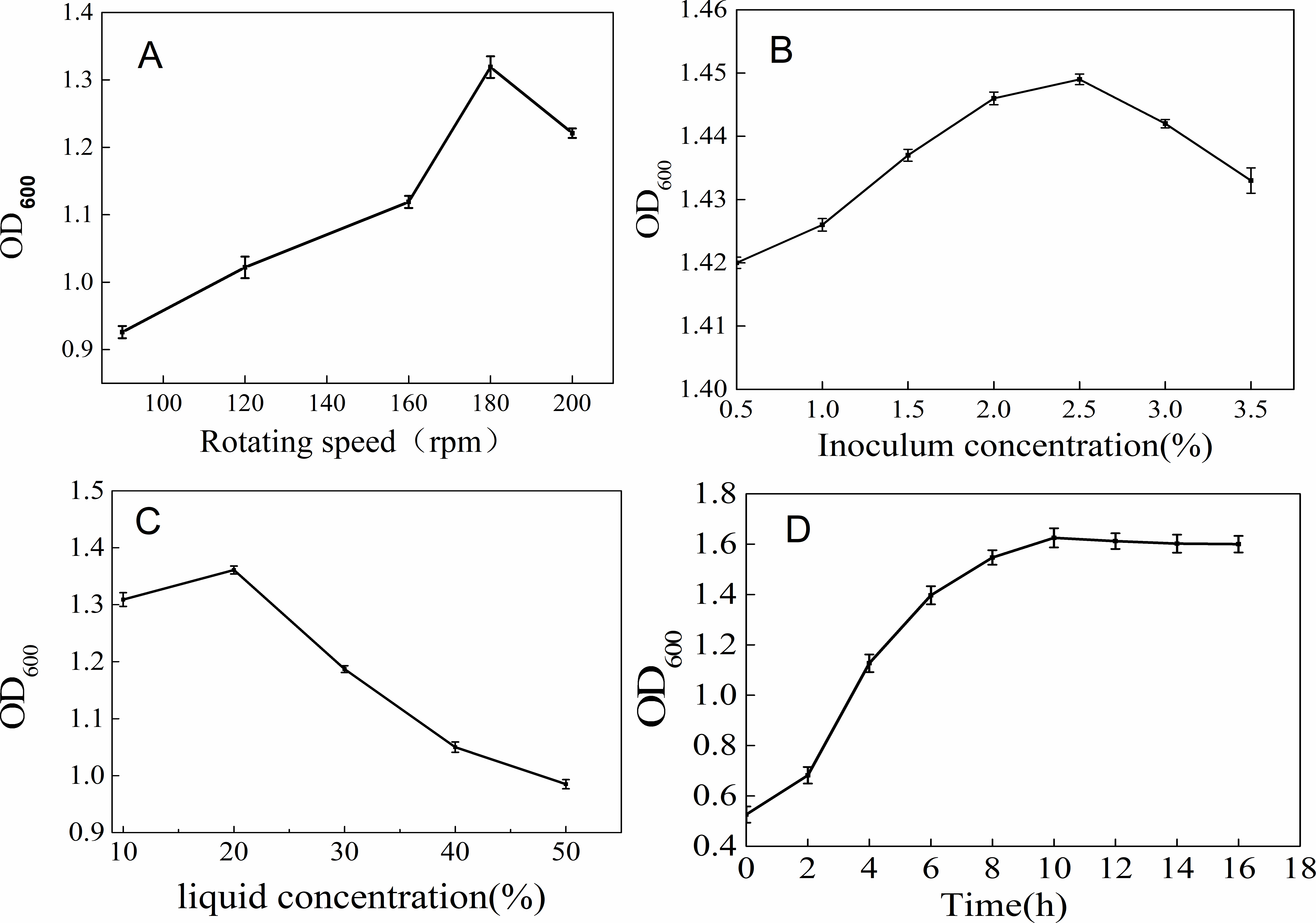
Effects of rotating speed (A), inoculum concentration (B), liquid concentration (C), and culture time (D) on the growth of strain GZ-7. OD, optical density.
According to the literature, species related to Shewanella include S. baltica NCTC 10735 T, 25 S. denitrificans OS217T, 26 S. frigidimarina ACAM591T, S. oneidensis ATCC 700550T, S. putrefaciens ATCC 8071T, 27 S. japonica KMM 3299T, 28 S. livingstonensis LMG 19866T, 29 and S. olleyana ACEM9T. 30 All these strains produced different enzymes, except for dextranase. The optimum growth temperature and growth pH of the Shewanella sp. GZ-7 were 30 °C and 7.5, respectively. Its growth characteristics were the same as for the above-mentioned Shewanella species.
The optimum temperature for the dextranase activity was found to be 40 °C, and the catalytic efficiency was maintained at the temperature range of 35 °C-45 °C (Figure 4(A)). The enzyme was denatured at 60 °C. The relative enzyme activity was >80% after the dextranase was incubated for 5 hours at 20 °C-40 °C. The thermal stability of the dextranase in this study was significant, being relatively stable in comparison with that of the dextranases reported elsewhere. 5,31 After incubation for 2 hours at 50 °C, the relative enzyme activity remained >50% (Figure 4(B)).

Effect of temperature on dextranase (A), stability (B), the effect of pH (C), and stability (D).
The optimum pH for dextranase activity was 7.5, and the enzyme activity was 70% at the pH range of 5-9 (Figure 4(C)). For the stability of dextranase, 80% activity was maintained (Figure 4(D)) at the pH range 6-8.
The optimal pH of dextranases reported in the literature was generally 4.5-5.0, 17,19,31,32 and they could function under acidic conditions as well. On the contrary, the optimal pH for dextranase activity produced by the GZ-7 strain was 7.5, which could be maintained at the pH range 6-8. This feature of the enzyme extends its range of applications quite widely.
As shown in Table 1, Co2+ and Li+ have a promoting effect on the dextranase activity, while the remaining tested metal ions showed an inhibitory effect.
Effects of Metal Ions on Dextranase.

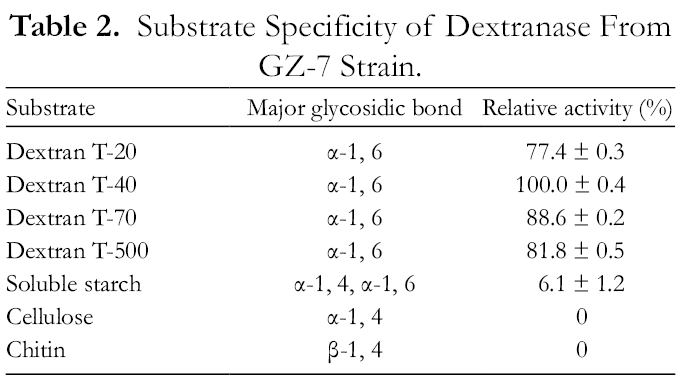
For the selected substrate, soluble starch contained α-1,4 and α-1,6 glycosidic bonds, cellulose contained α-1,4 glycosidic bonds, chitin contained β-1,4 glycosidic bonds, and dextran mainly contained α-1,6 glycosidic bonds. As shown in Table 2, dextranase could only hydrolyze the α-1,6 glycosidic bonds, but not the α-1,4 bond. Dextran T40 was found to be the most-favored substrate of the dextranase.
Substrate Specificity of Dextranase From GZ-7 Strain.
As shown in Figure 5A and A, a single dextranase activity band was noted, which indicated the presence of the enzyme. In line 5, the band was located at 100-135 kDa. The blue dextran-sodium dodecyl sulfate-polyacrylamide gel electrophoresis (BD-SDS-PAGE) was found to be an effective method for determining the molecular weight of dextranase. 13,33,34

(A) The activity staining of dextranase. Lane M: marker; lane 1: crude enzyme; lane 2: the crude enzyme was filtered through a 10 K filter; lane 3: the crude enzyme was filtered through a 50 K filter; lane 4: the crude enzyme was filtered through a 100 K filter; and lane 5: blue dextran-sodium dodecyl sulfate-polyacrylamide gel electrophoresis. (B) Thin-layer chromatographic analysis of hydrolyzates. 1: standards 2: the products of hydrolysis for 1 hour. 3: the products of hydrolysis for 3 hours. 4: the products of hydrolysis for 5 hours. 5: dextran. 6: the supernatant of broth. (C) High-performance liquid chromatographic (HPLC) analysis of standard. (D) HPLC analysis of hydrolyzates.
The molecular weight of dextranase is distributed over 40-100 kDa. For example, dextranase produced by Penicillium minioluteum is approximately 67 kDa, 35 while that by Trichoderma harzianum is 62 kDa. 31 Arnold et al reported that the molecular weight of dextranase produced by S. schenckii is 79 kDa, 19 by Arthrobacter oxydans KQ11 is 66.2 kDa 22 , and that by S. rattus was 100 kDa 8 . The lowest molecular weight (26.5 kDa) of dextranase is produced by Penicillium lilacinum, 36 while the highest one (158 kDa) is produced by KIBGE-IB25 (Bacillus licheniformis). 37 The molecular weight of dextranase produced by the GZ-7 strain is 100-135 kDa. For future research on dextranase, it would be meaningful to investigate the structure and hydrolysis mechanism of glycosidase.
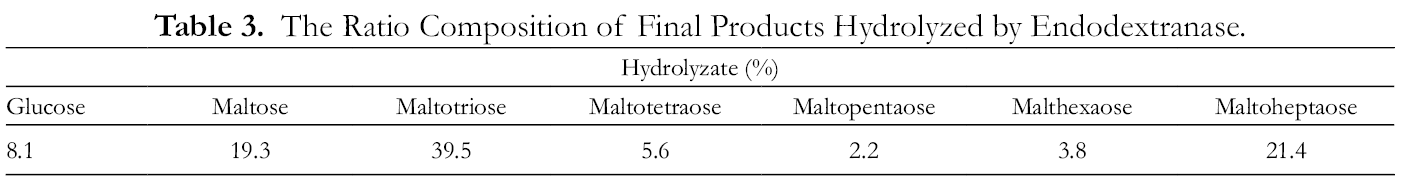
The thin-layer chromatography (TLC) results revealed that the dextranase exhibited endo-type activity and produced various oligosaccharides. HPLC analyses demonstrated that the hydrolyzate obtained was mainly composed of glucose, maltotriose, maltotetraose, maltopentaose, maltohexaose, and maltoheptaose, with a total of 91.8% content of IMOs (Table 3). The content of isomaltotriose was the highest, while that of isomaltopentose was the lowest. Functional oligosaccharides can thus promote the growth of intestinal probiotics and improve the immune system and intestinal environment.
The Ratio Composition of Final Products Hydrolyzed by Endodextranase.
Oligosaccharides possess antioxidant activities, although the different components of oligosaccharides have their own special features. 3 The hydrolyzates possess strong 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical and hydroxyl radical scavenging activities (Figure 6), but the superoxide anion radical scavenging activity occurs at a lower level. The reducing power was enhanced by the increasing concentration of final products.

The antioxidant activities of the hydrolyzates. (A) 2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical scavenging, (B) superoxide anion radical scavenging, (C) hydroxyl radical scavenging, and (D) determination of reducing power.
Functional oligosaccharides prepared by endodextranases possess the prebiotic properties that can be applied as antioxidants, 38,39 with far-reaching significance toward health development.
In summary, Shewanella sp. Gz-7 was isolated from the soil, identified, and then used for endodextranase production. Based on the experimental evaluation, the following optimal culture conditions were determined to be as follows: 30 °C, 180 rpm, pH 7.5, and 8 hours of culturing. The optimum reaction temperature and pH were 40 °C and 7.5, respectively. The dextranase maintained 80% of its activity after 5 hours of incubation at 40 °C, and the activity was found to be stable at pH 6-8. The molecular weight of the enzyme was approximately 100-135 kDa. HPLC identified that the products were mainly oligosaccharides. More than 91.8% of IMOs were produced, including maltose, maltotriose, maltotetraose, maltopentaose, and maltopeptaose. The tested hydrolyzates exhibited moderate antioxidant activity. Thus, dextranase and the corresponding hydrolyzates can be produced for wide applications in the near future.
Experimental
Samples and Materials
The samples were collected from the sugarcane-cultivated soil in the Guangdong province. Dextran T20 and blue dextran 2000 were obtained from GE Healthcare (Uppsala, Sweden). All other reagents were purchased from Sinopharm Chemical Reagent Company (Shanghai, China), and they were of analytical grade purity.
Screening for Dextranase-Producing Strains
Microorganisms possessing the ability of degrading dextran were screened and isolated from soil samples. Briefly, the soil suspension was serially diluted and tested by the spread plate method on a screening medium composed of 2 g blue dextran, 5 g yeast extract, 10 g peptone, 10 g sodium chloride, 10 g dextran T20, 20 g agar powder, and 1 L distilled water (pH 7.4). The plates were incubated at 30 °C. The strains that produced dextranases hydrolyzed the blue dextran in the medium to form transparent zones around their colonies. 24
Identification of the Strains
The morphological, physiological, and biochemical characteristics, as well as the 16S rRNA sequencing of the strain were performed with reference to Bergey’s Manual of Systematic Bacteriology (eighth edition), and the selected strain was subjected to various physiological and biochemical reactions for identification purpose. The 16S rRNA sequence of the strain GZ-7 was amplified by polymerase chain reaction with the bacterial universal primers 27F (5′-AGAGTTTGATCCTGGCTCAG-3′) and 1492R (5′-GGTTA CCTTGTTACGACTT-3′) (Shanghai Shenggong Bioengineering Co., Ltd.). The amplified products were sequenced by Shanghai Shenggong Bioengineering Co., Ltd. A similarity search was performed by using the GenBank database (hhit://www.ncbi.nih.gov), and the phylogenetic tree was constructed by the MEGA7.0 using the Neighbor-Joining method. 40
The Strain Growth Characteristic
Seed broth was prepared by adding 10 g/L tryptone, 5 g/L yeast extract, 10 g/L dextran T20, and 10 g/L sodium chloride to 50 mL distilled water at pH 7.4. The broth was cultured by incubation for 16 hours at 30 °C at 180 rpm shaking condition. Next, seed broth (2%) was inoculated into 50 mL fermentation broth (in 250 mL Erlenmeyer flask), incubated at 30 °C for 18 hours under 180 rpm shaking condition. The density of the cultivated bacteria was measured with an ultraviolet/visible spectrophotometer (OD600nm). All experiments were performed in triplicate.
Effect of Culture Conditions
Dextrin, potato starch, mung bean starch, pea starch, corn starch, tapioca starch, soluble starch, lactose, maltose,
Dextranase Assay
The cultured liquid medium was centrifuged (for 10 minutes at 10 000×g under 4 °C condition), and the supernatant was collected and concentrated 10 times via ultrafiltration through a hollow fiber membrane of molecular weight 10 kDa. The reducing sugar content was determined by the 3,5-dinitrosalicylic acid method. Briefly, 50 µL of the enzyme solution was mixed with 150 µL of 3% dextran 20 000 (50 mM; pH 5.5 sodium acetate buffer) and kept in a water bath at 40 °C for 15 minutes. 41 One unit of the enzyme activity was defined as the amount of enzyme required to release 1 μmol of maltose in a minute under the above-mentioned reaction conditions. 42
Molecular Weight of Dextranase
Protein samples and unstained protein markers were separated on 10% (w/v) SDS-PAGE and 0.5% BD-SDS-PAGE. The gel was treated in a BIO-RAD electrophoresis apparatus at 80 volts (V), and the gel portion was separated at 120 V. Next, the gel was split into 2 halves. One half was subjected to denaturing dyeing and the other to renaturation. The denatured gel was incubated in 20 mM sodium phosphate buffer (PB) containing 2.5% Triton X-100 at room temperature for 30 minutes. Next, the gel was incubated in 20 mM sodium PB (pH 7.5) at 37 °C for 3 hours or until a clear band appeared.
Characteristics of Dextranase
The effect of temperature on the dextranase produced was evaluated by incubating the enzyme with the substrate at 20 °C-60 °C, followed by measurement of the residual enzyme activity. The thermal stability of dextranase was evaluated by incubating the enzyme in 50 mM PB at pH 7.5 for 1 hour at different temperatures, followed by the measurement of the residual enzyme activity.
The dextranase activity was detected under different pH from 4.0 to 9.0 (sodium acetate buffer, pH 4.0-6.0; PB, pH 6.0-7.5; Tris-hydrochloric acid, pH 7.5-9.0). The relative activities were measured. The pH stability of the dextranase was evaluated by incubating the enzyme under different pHs from 4.0 to 9.0 at 25 °C for 1 hour. The residual enzyme activity was measured, and the relative activity was calculated.
To study the effect of metal ions on dextranase, the enzyme was mixed with various metal ions (Mg2+, Ca2+, K+, Na+, Sr2+, Co2+, Li+, Cd2+, and Ba2+), the final concentrations of metal ions were 1 and 10 mM, respectively. Then, the enzyme was incubated at 30 °C for 1 hour, followed by the measurement of the enzyme activity.
To investigate the most-favored substrate of dextranase, dextran T-20, T-40, T-70, T-500, soluble starch, cellulose, and chitin were tested as substrates, followed by the measurement of the enzyme activity.
HPLC and TLC Analyses of the Final Products
HPLC was performed on the Agilent 1260 Infinity Analytical SFC System with the standards of glucose, maltotriose, maltotetraose, maltopentaose, malthexaose, and maltoheptaose (glycarbo#GY series products). Methanol used was chromatographically pure. The system was equipped with the Waters Suger-PakI (6.5 × 300 mm) column, with deionized water used as the mobile phase. The flow rate was maintained at 0.4 mL/min, and the column temperature was 75 °C. The standard oligosaccharides were dissolved in 1 mL of deionized water and sonicated. The final products of hydrolysis were collected under the optimal reaction conditions of dextranase, and the reaction solution was further filtered through a micropore filter for HPLC analysis.
The hydrolyzates were analyzed by TLC using silica gel plates. The same standards mentioned above were used. The running solution was composed of 10 mL N-butyl alcohol, 6 mL ethanol, and 4 mL water. The TLC plate was dipped into the solution for 40 minutes, and the oligosaccharides were visualized by heating the plate at 120 °C for 10 minutes.
Antioxidant Activity of the Final Products
The DPPH radical, superoxide anion radical, hydroxyl radical, and reducing power scavenging activities were determined as suggested by Zhang et al. 43
Supplemental Material
online supplementary file 1 - Supplemental material for Isomalto-Oligosaccharides Produced by Endodextranase Shewanella sp. GZ-7 From Sugarcane Plants
Supplemental material, online supplementary file 1, for Isomalto-Oligosaccharides Produced by Endodextranase Shewanella sp. GZ-7 From Sugarcane Plants by Xin Liu, Tian Deng, Xueqin Liu, Xiaohua Lai, Yanli Feng, Mingsheng Lyu and Shujun Wang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the National Key R&D Program of China 317 (2018YFC0311106), the Priority Academic Program Development of Jiangsu higher 318 Education Institutions (PAPD), and the Postgraduate Research and Practice Innovation 319 Program of Jiangsu (SJCX18-0949, KYCX18-2586).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
