Abstract
The pathogenesis of Alzheimer’s disease (AD) is complex as various mechanisms interact with each other and, therefore, intervention from a single target is often ineffective. Many studies have shown that herbal medicines, such as curcumin, fisetin, icariin, and ginsenosides, have significant intervention effects on AD with different treatment mechanisms. Therefore, we have designed this study to know whether the combination of these herbal medicines can have an intervention effect on AD through multiple targets. Amyloid precursor protein/presenilin 1(APP/PS1) double transgenic AD mice were used to study the protective effects of a combination of curcumin, piperine, icariin, and ginsenosides, as well as a combination of fisetin, piperine, icariin, and ginsenosides, which were separately mixed into the feed. These herbal medicinal compounds (HMCs) lowered the serum lipid levels, reduced the Aβ oligomers, decreased the pS404-tau protein, as well as neurofibrillary tangles, and restored the reduction of synaptic protein levels and neuronal death of AD mice without causing toxicity to liver and kidneys. In this study, we found that HMCs have significant intervention against AD through multiple targets, providing a novel therapeutic idea for the prevention of AD.

Alzheimer’s disease (AD), the most common form of dementia, currently afflicts approximately 50 million people, and is considered as one of the worst global health and social crises of the 21st century, becoming the world’s seventh leading cause of death. 1 According to “World Alzheimer Report 2018,” 1 there will be 1 new dementia patient every 3 seconds. It is predicted that by 2030, the number of patients will increase to 82 million, and 152 million by 2050. The global socioeconomic cost of dementia was around $1 trillion in 2018 and will rise to $2 trillion by 2030, bringing huge economic and human burden to society. However, there is still a lack of effective clinical drugs to either cure or control the progression of AD. Since 1998, more than 100 attempts have been made to develop effective drugs for the disease, but most of them have ended in failure. Therefore, in-depth study of the pathogenesis of AD, exploration of reliable early diagnostic indicators, and further development of AD drugs are still main research directions.
AD is an extremely complex pathogenic process mediated by many factors and mechanisms with multiple genes and targets. 2 The amyloid cascade hypothesis, abnormal phosphorylation and aggregation of tau protein, and the hypothesis of neuronal loss currently occupy the main position in clinical and scientific research. Aβ, the core component of plaque, is generated from amyloid precursor protein (APP) progressively cleaved by β-secretase (BACE1) and γ -secretase. Many studies have shown that Aβ is a common pathway to induce AD, and a key factor in its occurrence and development. 3 The neurotoxic mechanism of Aβ is complex, including triggering oxidative stress, inducing apoptosis, and causing inflammation. 4,5 Tau is a microtubule-associated protein mainly in neurons. Its primary function is to bind to tubules, facilitate the assembly of microtubules, and stabilize the microtubule structure. 6 Tau plays an important role in the assembly of microtubules, and this process is regulated by its phosphorylation state. In AD patients, tau becomes abnormally phosphorylated and the hyperphosphorylated tau loses its ability to facilitate microtubule assembly, leading to axoplasmic transport impairment and the death of neurons. 7 -10 Hyperphosphorylated tau protein dissociates from the microtubule structure to form paired helical filaments, which further accumulate to form neurofibrillary tangless (NFT). 11 The formation of NFT can also cause cytotoxicity, resulting in neuronal abnormalities. Both Aβ and tau pathology are highly correlated with synaptic loss, neuronal dysfunction, and memory decline in AD. 12 -14
At present, the treatment of AD using modern medicines is mainly to improve clinical symptoms, which cannot stop the disease process. Herbal medicines have the advantages of overall regulation of the occurrence and development of diseases. 15 At the same time, it can maintain the homeostasis of important organelles, such as mitochondria and endoplasmic reticulum. 16 Therefore, the development of drugs from plants to treat and delay the pathological process of AD has attracted much attention in recent years. 17 -19 Curcumin is a component of turmeric with anticancer, anti-Alzheimer’s disease activity, and antioxidative effects. 20 -23 However, the efficacy of curcumin is limited by its quite low bioavailability. However, studies have shown that piperine can improve the bioavailability of curcumin. 24 Fisetin is one of the most antioxidant potent flavonoids, a family of antioxidants found in vegetables, fruits, red wine, and tea, 25 which has a range of pharmacological activities, such as antioxidant, anti-inflammatory, and anticancer effects. 26 -29 Icariin, the main pharmacologically active component of herba Epimedium, has been traditionally used as an antirheumatic remedy and aphrodisiac. Our previous studies have shown that icariin significantly improved the spatial learning and memory of AD model mice. 30 Ginsenosides, with a wide range of pharmacological activities, including anticancer, antioxidation, anti-inflammation, antifatigue, antiaging, and other biological activities, are the major constituents of Panax species (Araliaceae), which are traditional, valuable, medicinal herbs. 31 -33 Many studies have shown that curcumin, fisetin, icariin, and ginsenosides have significant intervention effects on AD, with different treatment mechanisms. 27,34 -37
AD is a complex disease caused by multiple pathogenic factors. Thus, the current “One molecule-one-target” (OMOT) strategy cannot solve the multiple pathological stages of AD. In contrast, the “Combination-drugs-multi-targets” strategy, which is an inevitable trend in future AD treatment, is more effective in addressing multiple pathological processes in AD. This may overcome the shortcomings of poor efficacy of OMOT. Thus, we have designed this study to explore tentatively the effects of the combination of curcumin, piperine, icariin, and ginsenosides (CPIG), and the combination of fisetin, piperine, icariin, and ginsenosides (FPIG), on AD pathology.
Materials and Methods
Materials and Reagents
Curcumin, piperine, fisetin, icariin, and ginsenosides were all purchased from Shanxi Senlang Biochemical Co., Ltd. Ginsenosides (80.6%) used in the experiment were a mixture of a variety of ginsenoside analogs, consisting of Rg1 (9%), Re (22.6%), Rb1 (4.7%), Rc (3.1%), Rb2 (5.1%), Rd (9.8%), and other types of ginsenoside analogs (26.3%). Biochemical reagents were obtained from Shenzhen iCubio Biomedical Technology Co., Ltd. The primary antibody of 6E10 was obtained from Biolegend. Antibodies of pS404-tau, post-synaptic density protein 95 (PSD95), Synapsin I (Syn1), and Synaptophysin (Syna) were acquired from Abcam. Secondary antibodies were purchased from Abmart. Glycine silver staining kit and Nissl staining kit were obtained from Wuhan Servicebio Technology Co., Ltd and Solarbio Life Sciences, respectively. All the other reagents were of analytical grade.
Animals and Treatment
The APP/PS1 transgenic mice (B6C3-Tg [APPswe, PSEN1dE9] 85Dbo/Mmjax) were purchased from Guangdong Medical Lab Animal Center. All mice were kept in a 12-hour light/12-hour dark cycle and given access to food and water ad libitum. Fifty male mice (4 months old) were randomly divided into 5 groups: high dose of CPIG (CH), low dose of CPIG (CL), high dose of FPIG (FH), low dose of FPIG (FL), and AD control mice. Ten wild-type (WT) mice (C57/BL6) were used as the normal control group. Detail of the composition and dose of the experimental diet is shown in Table 1.
Composition and Dose of Experimental Standard Chow Diet.

aH1 stands for high dose. L2 stands for the low dose.
After 4 months of drug treatment, all the mice were tested by the open-field test to assess their cognitive functions. After the behavioral test, the mice were euthanized, and eyeballs blood and brain tissues rapidly collected. The left hemisphere was fixed for frozen sections, which were used for immunofluorescence analysis, glycine silver staining, and Nissl staining. The right hemisphere was immediately frozen at −80 °C for western blot analysis.
All the procedures and experiments were approved by the Animal Ethical and Welfare Committee of Shenzhen University with permit number AEWC-20140615-002. Every effort was made to reduce mice suffering.
Open-Field Test
The apparatus is an open field made of gray plastic, measuring 40 × 40 × 40 cm. The floor is made up of hard plastic and divided into 25 equal-sized grids. Mice were put in the corner of the field and moved freely for 3 minutes to monitor the spontaneous locomotor activity of the mice in the CH, CL, FH, FL, AD, and WT groups. Between trials, the arena was cleaned with 75% ethanol to reduce olfactory cues. Each mouse was introduced into the field and the number of grids entered with all paws and rearing, ie, the animal stood on its hind legs, were recorded.
Biochemical Analysis
Eyeballs blood was collected and serum was obtained after centrifugation at 12 000 rpm for 30 minutes. Serum biochemical indexes were measured using a biochemical analyzer (iMagic-M7, China) according to the manufacturer’s protocol.
Western Blot Assay
The hippocampal tissue of mice was fully lysed in lysis buffer with the addition of phosphatase inhibitors, protease inhibitors, and phenylmethanesulfonyl fluoride. The protein supernatant was obtained after centrifugation at 12 000 rpm for 30 minutes and the protein concentration was determined using the bicinchoninic acid method. After heating at 100 °C for 3-5 minutes with the addition of loading buffer, 12 µg of each protein extract was separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto polyvinylidene fluoride membranes. The membranes were blocked with 5% fat-free milk and 6E10, pS-404 tau, PSD95, Syn1, and Syna were detected by corresponding primary antibodies, followed by a combination of horseradish peroxidase-conjugated secondary antibody. The protein blots were detected by enhanced chemiluminescence reagents and imaged via Image Station (Tanon-5200, China). Relative band intensities were quantified by Quantity One software.
Immunofluorescence Analysis
For immunofluorescence, frozen sections were washed with phosphate-buffered saline (PBS) for 5 minutes, repaired by antigen repair solution for 5 minutes, permeabilized with 2% Triton X-100 in PBS (PBST) for 10 minutes, blocked with 3% bovine serum in PBST for 1.5 hours, and incubated with primary anti-6E10 antibody (1:1000, mouse monoclonal, Biolegend, CA, USA) diluted in blocking solution overnight at 4 °C. These were followed by 3 washes with PBS. Secondary antibody conjugated to Dylight-488 (1:1000, Abcam, Cambridgeshire, England) was then added and incubated at room temperature for 2 hours. After 3 times of washing with PBS, frozen sections were incubated with DAPI (1:500, Beyotime, Shanghai, China) for 5 minutes, washed 3 times with PBS again, and then visualized using a confocal microscope (ZEISS, Jena, Germany).
Glycine Silver Staining
Glycine silver staining was performed according to the instructions of the manufacturer. Briefly, the frozen sections were stained with acid formaldehyde for 5 minutes and washed 3 times with double deionized water. The sections were then treated with silver glycine solution preheated at 37 °C for 4 minutes, and subsequently put into reduction solution preheated at 45 °C for several seconds followed again by 3 times washing with double deionized water, and finally visualized using an upright fluorescence microscope (OLYMPUS BX51).
Nissl Staining
Nissl staining was performed according to the instructions of the kit manufacturer. Briefly, the frozen sections were stained with methyl violet stain solution for 15 minutes and washed with double deionized water. The sections were then washed with Nissl differentiation for seconds until most of the stain was eliminated, and subsequently dehydrated with anhydrous ethanol and xylene, and visualized using an upright fluorescence microscope (OLYMPUS BX51).
Statistical Analysis
The data were analyzed with GraphPad Prism 7.0 software and expressed as mean ± standard error of the mean. The degree of statistical significance is shown as follows: ****P < .0001; ***P < .001; **P < .01; *P < .05.
Results
HMCs Significantly Improved Locomotor and Exploring Ability of APP/PS1 Transgenic Mice
The open-field test was used to assess locomotor and exploring activity behavior indicated by the numbers of crossed grids and rearing of the mice in C57, AD, CH, CL, FH, and FL groups. As shown in Figure 1, compared with the C57 group, the mice in AD group showed a significant decrease in the number of crossed grids and rearing within 180 seconds (Figure 1(A) and (B)), indicating the decreased exploratory and locomotor activity of AD mice. Furthermore, the mice in FH group demonstrated a significant increase in the number of crossed grids (Figure 1); CH as well as CL mice reared more frequently in comparison with the AD mice (Figure 1(B)), indicating a notable increase in locomotor and exploring ability, respectively. However, there was no significant difference in either the crossed grids or rearing between FL and AD mice.

HMCs administration improved locomotor and exploring ability of APP/PS1 transgenic mice by open-field test. (A) The numbers of crossed grids; (B) frequencies of rearing (***P < .001, *P < .05; n = 10). AD, Alzheimer’s disease; CH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; FH, high dose of FPIG; FL; low dose of FPIG; FPIG, combination of fisetin, piperine, icariin, and ginsenosides; HMC, herbal medicinal compound.
Effect of HMCs on Serum Lipids and Toxicity to Liver and Kidneys
To explore the toxicity of herbal medicinal compounds (HMCs) to liver and kidneys of mice, several biochemical indicators in the serum of mice were analyzed. As shown in Figure 2, there was no difference in the protein levels of either aspartate transferase or glutamine transferase, the 2 biochemical indexes of liver cell injury, among the mice from all groups (Figure 2(A)). Similarly, the levels of uric acid and creatinine that are the main biochemical indicators reflecting the metabolic capacity of the kidney in HMCs-treated groups and C57 mice did not differ either (Figure 2(B)), suggesting that HMCs had no toxicity to liver and kidneys of mice. Interestingly, triglycerides of all the CH, CL, FH, and FL groups were remarkably lower than that of AD mice, indicating the serum lipid reduction ability of HMCs (Figure 2(C)).
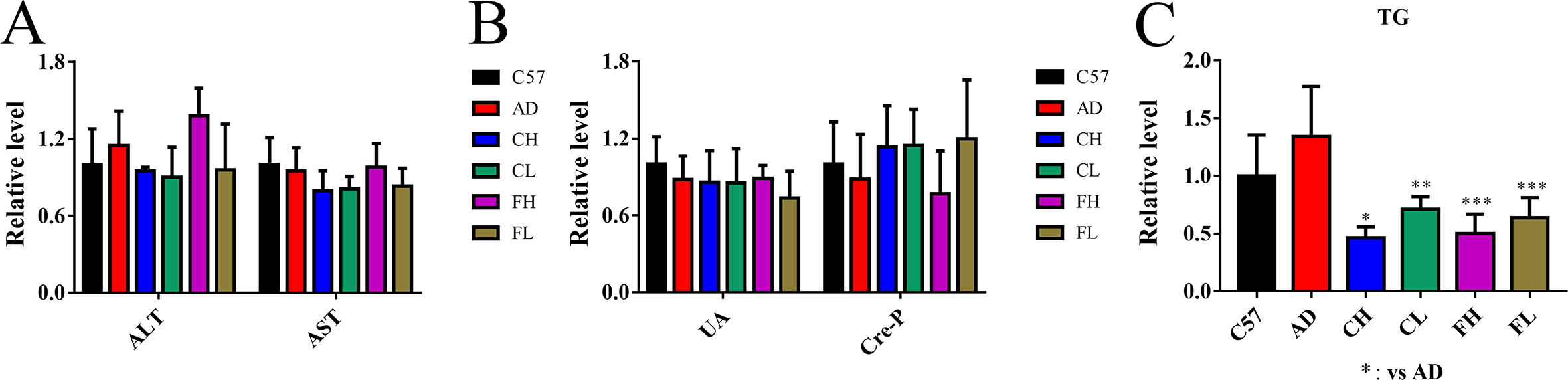
Effect of herbal medicinal compounds on biochemical indexes of AD mice: (A) Relative level of liver-related biochemical indicators involving AST and ALT. (B) Relative level of kidney-related biochemical indicators involving UA and Cre-P. (C) Relative level of serum lipid-related biochemical indicator, TG. (***P < .001; **P < .01; *P < .05; n = 10). AD, Alzheimer’s disease; ALT, glutamine transferase; AST, aspartate transferase; CH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; Cre-P, creatinine; FH, high dose of FPIG; FL; low dose of FPIG; FPIG, combination of fisetin, piperine, icariin, and ginsenosides; TG, triglyceride; UA, uric acid.
HMCs Inhibited Aβ Generation and Aggregation in the Brains of APP/PS1 TranCH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; sgenic Mice
Amyloid plaques formed by Aβ aggregation are one of the hallmarks of AD. Thus, we examined the effects of HMCs on Aβ generation and deposition in the brains of AD mice. As shown in Figure 3, the levels of Aβ oligomers (14× and 6×) in the hippocampus of AD mice were much higher than those of C57 mice by western blot analysis (Figure 3(A) and (B)), while HMC treatments significantly reduced the levels of Aβ oligomers (Figure 3(A) and (B)). In agreement with this result, immunofluorescence analysis with anti-Aβ antibody (6E10) has shown that Aβ immunoreactivity in CAI, CA3, DG, and cortex of AD mice were much higher than that in the C57 group, and HMCs inhibited the deposition of Aβ in these regions of AD mice (Figure 3(C)-(F)).

The level and distribution of Aβ oligomers in brain tissues (A) Aβ oligomer (14× and 6×) blots of hippocampal tissues. (B) Gray value analysis of western blotting. (C)–(F) Confocal microscopy images showing the expression patterns of Aβ oligomers.(scale bar, 100 µm) (****P < .0001; ***P < .001; **P < .01; n = 4). AD, Alzheimer’s disease; CH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; FH, high dose of FPIG; FL; low dose of FPIG; FPIG, combination of fisetin, piperine, icariin, and ginsenosides; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
HMCs Attenuated Tau Hyperphosphorylation and NFT Formation in APP/PS1 Transgenic Mice
NFT formed by hyperphosphorylated tau protein are another important feature of AD, which induce toxicity in the nervous system. Herein, we determined the level of tau phosphorylated at serine 404 (pS404-tau) in hippocampus of AD mice by western blots. As shown in Figure 4, the expression level of pS404-tau protein in hippocampus of AD mice is higher than that of C57 mice (Figure 4(A) and (B)), while CH and CL decreased the pS404-tau protein level. Moreover, FH and FL significantly reduced the pS404-tau protein level in hippocampus of AD mice (Figure 4(A) and (B)). Meanwhile, we performed glycine silver staining of brain frozen slices using a glycine silver staining kit to directly observe the levels and distributions of NFT. The results showed that the contents of NFT in CAI, CA3, DG, and cortex of HMCs-treated AD mice were significantly decreased as compared with untreated AD mice (Figure 4(C) and (D)).
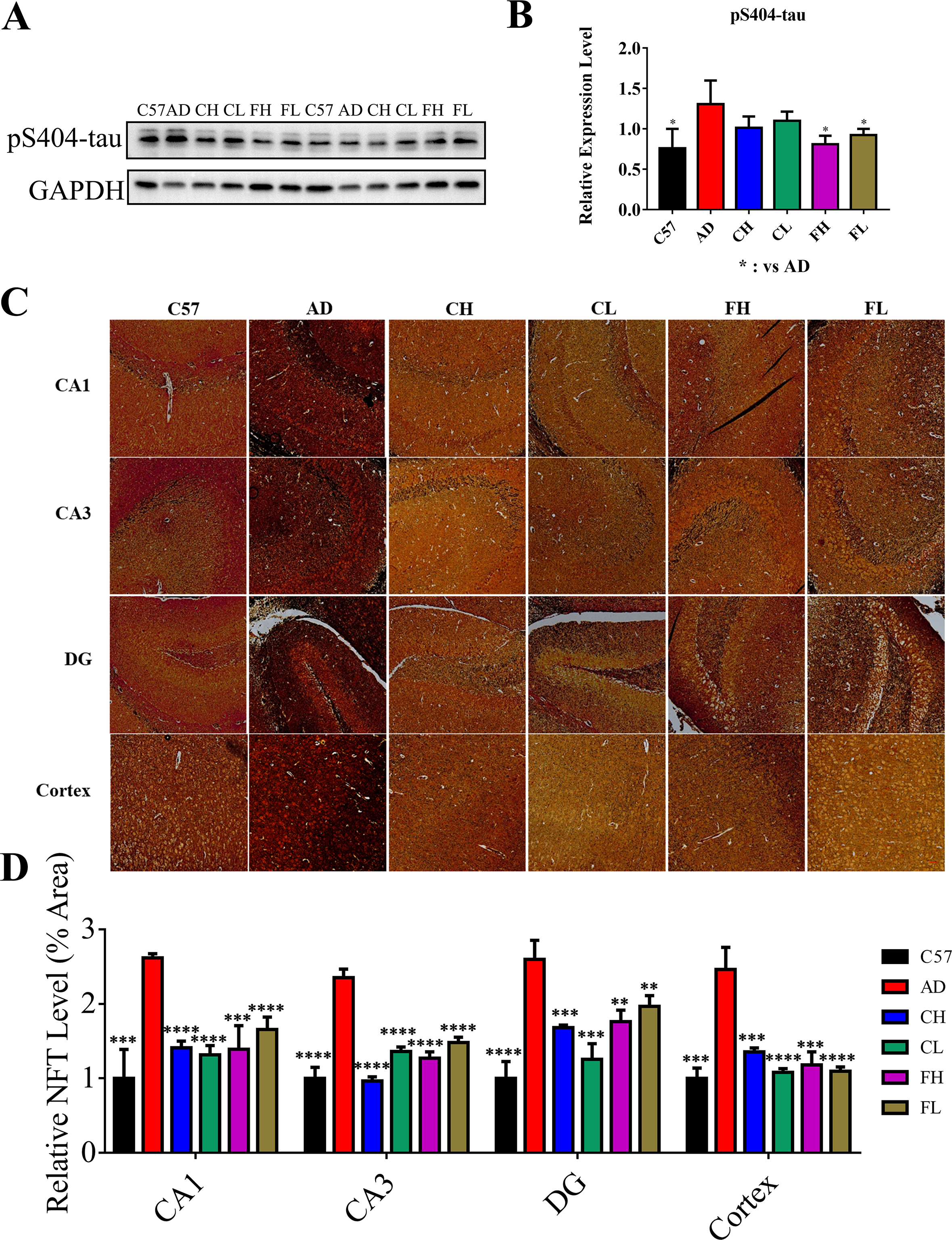
Herbal medicinal compounds attenuated tau hyperphosphorylation and NFT formation in APP/PS1 transgenic mice. (A) pS404-tau blots of hippocampal tissues. (B) Gray value analysis of western blotting. (C) Glycine silver staining images showing the levels and distributions of NFT (scale bar, 50 µm). (D) Quantitative analysis of glycine silver staining (****P < .0001; ***P < .001; **P < .01; *P < .05; n = 4). AD, Alzheimer’s disease; CH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; FH, high dose of FPIG; FL; low dose of FPIG; FPIG, combination of fisetin, piperine, icariin, and ginsenosides; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; NFT, neurofibrillary tangles.
HMCs Restored the Reduction of Synaptic Protein Levels and Neuronal Death in APP/PS1 Transgenic Mice
Synapses are the sites of functional communication between neurons and the key sites for information transmission. Numerous studies have demonstrated that synaptic damage occurred in the brain of APP/PS1 transgenic mice. We, therefore, have investigated the expression of synaptic proteins including PSD95, Syn1, and Syna of hippocampal tissues, which were all decreased in AD mice and reversed in HMCs-treated mice (Figure 5(A) and (B)). This reveals the recovery of synaptic deficits in AD by HMCs. The loss and death of neurons is an important cause of synaptic dysfunction, which is closely related to the development of AD. Here, Nissl staining was conducted to detect the density of neurons in AD mice treated with or without HMCs. As shown in Figure 5, the number and density of Nissl bodies in CA1, CA3, DG, and cortex regions of AD mice were significantly decreased in comparison with C57 mice (Figure 5(C) and (D)). This reflects that the neurons of AD mice were damaged to some extent. As expected, the amount and density of Nissl bodies in these regions of HMCs-treated AD mice were restored (Figure 5(C) and (D)). Our results illustrated that HMCs administration not only protected the synaptic proteins but also alleviated neuronal death and impairment in AD mice.

Herbal medicinal compounds restored the reduction of synaptic protein levels and neuronal death in APP/PS1 mice. (A) PSD95, Syn1, and Syna blots of hippocampal tissues. (B) Gray value analysis of western blotting. (C) Nissl staining images showing the levels and distributions of Nissl bodies (scale bar, 100 µm). (D) Quantitative analysis of Nissl staining (***P < .001; **P < .01; *P < .05; n = 4). AD, Alzheimer’s disease; CH, high dose of CPIG; CL, low dose of CPIG; CPIG, combination of curcumin, piperine, icariin, and ginsenosides; FH, high dose of FPIG; FL; low dose of FPIG; FPIG, combination of fisetin, piperine, icariin, and ginsenosides; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; PSD95, post-synaptic density protein 95; Syn1, Synapsin I; Syna, Synaptophysin.
Discussion
The pathogenesis of AD is very complex and involves many factors and organs. At present, the main drugs used to treat the disease are single-target drugs, which can only improve or alleviate the symptoms to a certain extent and cannot reverse or even delay the development of AD. In addition, the patients may have adverse reactions after drug administration. 38 Herbal medicines have long been used to prevent and treat cognitive impairment and have the advantages of little side effect, multiple targets, and overall regulatory effect, focusing not only on the remission of symptoms, but also on restoring and maintaining homeostasis. This is like the principle of modern multitarget therapy for complex diseases such as AD and is more appropriate for long-term use. 39,40
Curcumin, fisetin, icariin, and ginsenosides were reported having significant intervention effects on AD. Curcumin showed significant protection, especially in inhibiting the formation of amyloid beta plaques and promoting their disaggregation. It has been reported that curcumin stimulated phagocytosis, altered expression of inflammatory cytokines by decreasing CD33, and increased TREM2 expression to emulate Aβ immunotherapy and control signaling and phagocytic clearance of amyloid. 34 In addition, curcumin can attenuate the hyperphosphorylation of tau and enhance its clearance, lower cholesterol, bind copper, inhibit acetylcholinesterase, modify microglial activity, and mediate the insulin signaling pathway. 22 Fisetin has been reported to maintain the cognitive function in AD transgenic mice by reducing the levels of the cyclin-dependent kinase 5 activator p35 cleavage product, p25, which is closely related to neuroinflammation and neurodegeneration. 27 Moreover, fisetin stimulated autophagy degradation of phosphorylated tau by activating TFEB and Nrf2 transcription factors. 35 Icariin reduced Aβ contents, amyloid plaques, and APP and BACE-1 levels. 36 Furthermore, icariin reduced neuronal apoptosis, as well as suppressing endoplasmic reticulum stress, and protected the structure and function of mitochondria in AD neurons. 41,42 Ginsenoside could inhibit tau phosphorylation by reducing expression of glycogen synthase kinase 3beta, the most important kinase involved in tau phosphorylation, and enhanced the activity of protein phosphatase 2A, a key phosphatase involved in tau dephosphorylation. 37 Ginsenosides could also increase γ-aminobutyric acid, acetylcholine, dopamine, glycine, and serotonin levels, decrease Aβ, glutamate, and aspartic acid levels, and repair the damage of the hippocampus. 43 Moreover, all these compounds have functions of antioxidation, anti-inflammation, and antiaging, which have been implicated in the intervention of AD. Therefore, we have made an attempt to explore the multitarget treatment of AD by 4 groups of CH, CL, FH, and FL consisting of either curcumin, piperine, icariin, and ginsenosides or of fisetin, piperine, icariin, and ginsenosides.
Several studies have shown that curcumin inhibits the formation of Aβ oligomers and fibrils, and reduces amyloid levels and plaque burden. 44 -46 Many researchers have also reported that curcumin ameliorates cognitive decline and improves synaptic functions in mouse models of AD. 45,47 Similarly, we found that CH and CL including curcumin have reduced the level of Aβ oligomers and increased the levels of synapse-associated proteins PSD95, Syn1, and Syna, which can indicate synaptic functions. Piperine was added to increase the bioavailability of curcumin in the CH and CL group, and as a control in the FH and FL group. Fisetin is a small, orally active molecule, which can act on many of the target pathways involved in AD. Recent research revealed that fisetin could reduce cognitive deficits, restore multiple markers associated with impaired synaptic function, and decrease levels of phosphorylated tau. 27,35,48,49 All these results coincide with our findings that FH and FL have decreased the expression of pS404-tau protein, as well as the contents of NFT, and restored the reduction of synaptic protein levels and neuronal death of AD mice. Icariin, a main constituent of Epimedii Herba and a well-known tonic crude drug, has been found to have a remarkable effect on AD in the past 10 years. Researchers found that icariin could reduce the Aβ burden and amyloid plaque deposition in the hippocampus of AD transgenic mice, 36,50,51 and preserve the expression of synaptic functional proteins and improve synaptic function. 42 Ginsenoside could attenuate pathological tau phosphorylation 37 and ameliorate hippocampal Aβ deposition in AD mice. 52 In this study, CH, CL, FH, and FL that all contained icariin and ginsenoside exhibited all of these similar effects.
The current study has thought-provoking results, but still has certain limitations. We have only used the combinations of these natural drugs that can improve locomotor and exploring activity as well as ameliorating AD-related neuropathologies. However, it is not clear whether one of the herbal medicines has exerted the effect or whether these 4 drugs have played a synergistic role. In addition, we do not know the specific receptor/enzyme or specific cellular pathway on which the combinations work during neuroprotection. Thus, our future study will focus on the comparison of drug combinations and individual drugs on AD, and the specific targets and pathways that the drug combinations work on. This may underline the merits of combinations of natural products acting through a polytherapeutic approach over a monotherapy option of AD therapy.
Conclusions
In the present study, the effects of CH, CL, FH, and FL on AD mice were investigated and the results showed that all these combinations lowered the level of serum lipids and improved the locomotor and exploring ability of APP/PS1 transgenic mice by mitigating Aβ pathology, reducing tau pathology, and reversing synaptic deficits without inducing toxicity to the liver and kidneys. The overall goal of the study was to test whether these herbal medicines could provide protective effects against AD when used in combination, and, if positive, could be useful for future AD drug development.
Patent
Chinese patent no. 201910613202.6 has resulted from the work reported in this manuscript.
Footnotes
Acknowledgment
The authors would like to acknowledge Dr Xin Liu of the drug dosage recommendation and experimental design.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: XD, XL, and JN are the inventors and owners of Chinese patent no. 201910613202.6.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (21771126), the Shenzhen Bureau of Science, Technology and Information (JCYJ20180305124000597, JCYJ20170818142241972), and Shenzhen-Hong Kong Institute of Brain Science-Shenzhen Fundamental Research Institutions (2019SHIBS0003).
