Abstract
Eight limonoids (
Limonoids are highly oxygenated triterpenes, which have lost the terminal 4 carbon atoms of the side chain, with the remaining side chain cyclized into a furan ring. Limonoids are widely found in leaves, bark, fruits, seeds, and kernels throughout the Meliaceae and Rutaceae. 1 This type of compound revealed various interesting biological effects such as antifeedant, 1,2 cytotoxic, 3,4 antibacterial, anti-human immunodeficiency virus, and human carboxylesterase 2 inhibitory 5 activities.
As a part of our ongoing investigations on medicinal plants growing in Lamdong province, Vietnam,
6
-8
the methanol (MeOH) extract of Choerospondias axillaris branches and leaves wre found to exhibit cytotoxicity against hepatocellular carcinoma (Hep-G2) and rhabdomyosarcoma (RD) human cancer cell lines with half-maximal inhibitory concentration (IC50) values of 8.02 and 10.08 µg/mL, respectively. In Vietnam, this plant is found in glades in semideciduous forests in Son La, Lao Cai, Bac Giang, Quang Ninh, Nghe An, Ha Tinh, Kon Tum, Dak Lak, and Lam Dong provinces. The stem bark and fruit of this plant have been used in folk medicine for their antibacterial, detox, and anti-inflammatory effects.
9
Recent investigations of this plant reported the main chemical constituents as bridged-ring ketones,
10
proanthocyanidins,
11
and flavonoids,
12
with some exhibiting cytotoxic,
10,12
antioxidant, antiangiogenic,
11
antihypoxia, and antibacterial
12
activities. In this article, we address the isolation and structure elucidation of 8 limonoids (
Results and Discussion
The dry MeOH extract of C. axillaris branches and leaves was suspended in water (H2O) and partitioned in turn with n-hexane, dichloromethane (CH2Cl2), and ethyl acetate (EtOAc). Subsequently, 8 limonoids (

The structures of compounds 1-8.
Axillariol A (

Key correlation spectroscopy (▬) and heteronuclear multiple bond correlation ( ) correlations of 1.
) correlations of 1.
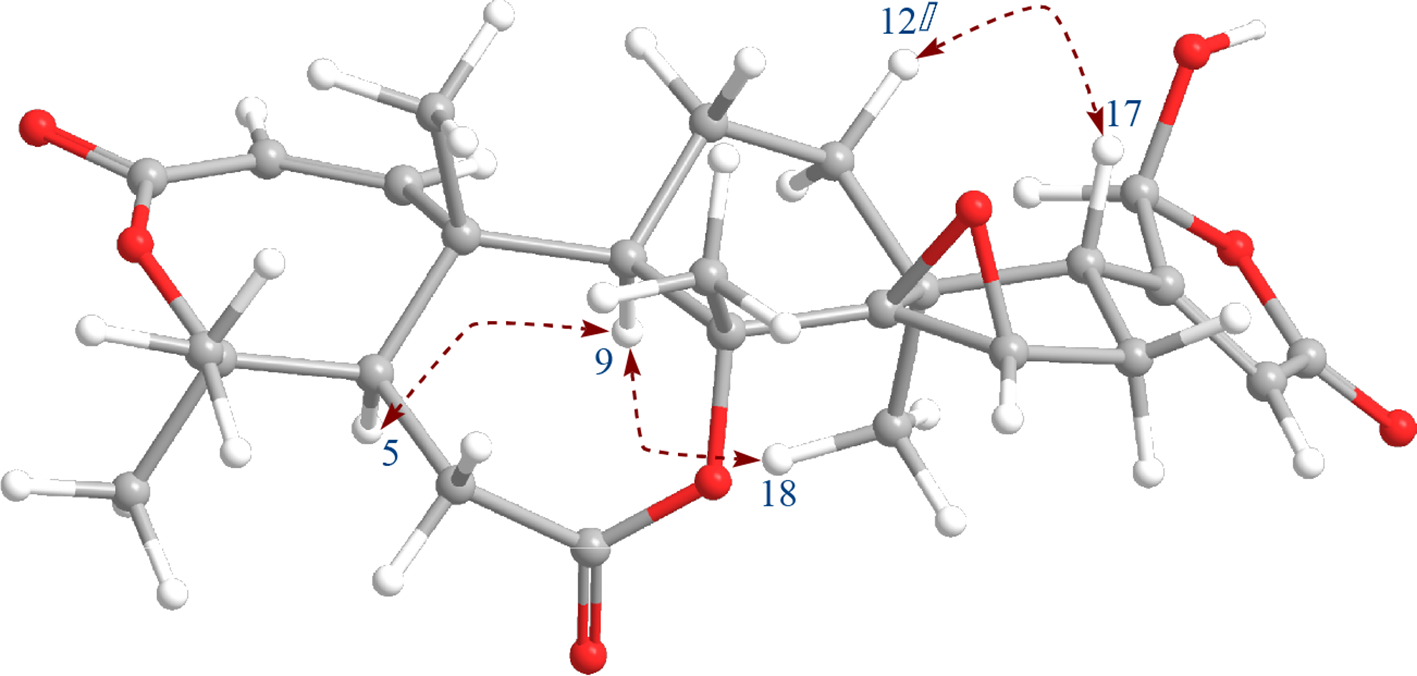
Key rotating frame Overhauser effect spectroscopy correlations and computer-generated 3-dimensional model using MM2 force field calculations of 1.
1H (500 MHz, DMSO-d 6) and 13C NMR (125 MHz, DMSO-d 6) Spectroscopic Data of 1.

Abbreviation: DMSO, dimethyl sulfoxide.
All assignments were based on heteronuclear single quantum correlation, correlation spectroscopy, heteronuclear multiple bond correlation, and rotating frame Overhauser effect spectroscopy experiments.
Since the MeOH extract of C. axillaris was found to exhibit cytotoxicity to Hep-G2 (hepatocellular carcinoma) and RD (rhabdomyosarcoma) cancer cell lines, isolated compounds
Cytotoxic Activity of Compounds 1-

Abbreviation: IC50, half-maximal inhibitory concentration.
aEllipticine was used as positive control; (−): IC50 >10 µM; results are the average means of triplicate experiments.
Experimental
General
Optical rotations were determined on a JASCO P-2000 polarimeter (Tokyo, Japan), HR-MS on a MicroQ-TOF III mass spectrometer (Bruker Daltonics, Germany), and 1H NMR (500 MHz) and 13C NMR (125 MHz) spectra on an AVANCE III HD 500 FT-NMR spectrometer (Bruker, Germany), with trimethylsilane used as an internal standard. Medium pressure liquid chromatography (MPLC) was carried out on a Biotage-Isolera One system (SE-751 03 Uppsala, Sweden), and column chromatography (CC) on silica gel (Kieselgel 60, 70-230 mesh and 230-400 mesh, Merck, Darmstadt, Germany), reversed-phase silica gel (ODS-A, 12 nm S-150 mm, YMC Co., Ltd.), and Sephadex LH-20 (Sigma) resins. Thin-layer chromatography (TLC) used precoated silica gel 60 F254 (1.05554.0001, Merck) and RP-18 F254S plates (1.15685.0001, Merck), and compounds were visualized by spraying with aqueous 10% sulfuric acid, followed by heating for 3-5 minutes.
Plant Material
Choerospondias axillaris (Roxb.) Burtt et Hill (Anacardiaceae) branches and leaves were collected in Lamdong province, Vietnam, in June 2018. The voucher specimen (No: TN3/201) was identified by Dr Nong Van Duy and deposited at the Tay Nguyen Institute of Scientific Research, VAST, Vietnam.
Extraction and Isolation
Air-dried powder of C. axillaris branches and leaves (3 kg) was extracted with MeOH (5 L) under ultrasonic condition (3 times, 1 hour each). The resulting solutions were filtered, combined, and concentrated in vacuum to give the MeOH residue (M, 250 g). This was suspended in H2O (2 L) and partitioned in turn with n-hexane (H; 3 × 2 L), CH2Cl2 (D; 3 × 2 L), and EtOAc (E; 3 × 2 L) to obtain the corresponding n-hexane (40.0 g), CH2Cl2 (14.5 g), and EtOAc (15.5 g) extracts, and H2O residue. Extract D (14.5 g) was separated into 4 fractions, D1-D4, by using silica gel MPLC with step-wise elution using n-hexane-acetone (10:1, 5:1, 3:1, and 1:1, v/v). Fraction D2 (3.0 g) was further separated by silica gel CC with n-hexane-CH2Cl2-EtOAc (1:2:0.2, v/v) to obtain 6 subfractions, D2A-D2F. Purification of subfraction D2B (740 mg) on ODS-A CC with acetone-H2O (2:1, v/v) furnished compound
Axillariol A (1)
White amorphous powder.
[α]D 25 + 35 (c 0.05, MeOH);
1H (dimethyl sulfoxide [DMSO-d 6], 500 MHz) and 13C NMR (DMSO-d 6, 125 MHz) data are given in Table 1.
HR ESI QTOF MS m/z 495.2021 [M + Na]+ (calcd. for C26H32NaO8 +, 495.1995).
Cytotoxic Assays
Cytotoxic evaluations were performed by SRB colorimetric assay 20,21 employing 4 human cancer cell lines: Hep-G2, Lu, RD, and RD. The assay was adapted for the quantitative measurement of cell growth and viability. Briefly, cancer cells were seeded at 5 × 103 cells per well in 96-well microtiter plates and incubated at 37 °C (48 hours, 5% carbon dioxide). Cultures were fixed with trichloroacetic acid and stained for 30 minutes with 0.4% (w/v) SRB. The protein-bound dye was extracted with 10 mM Tris base for determination of optical density (OD) at a single wavelength of 564 nm in a 96-well plate reader. The percentages of cell survival of test samples were calculated using the following formula: % cell survival = 100% × [OD(sample) – OD(day 0)]/[OD(DMSO) – OD(day 0)]. IC50 values of test samples were calculated by interpolation by linear regression analysis.
Conclusions
From C. axillaris branches and leaves, 8 limonoids, including 1 new compound named axillariol A (
Supplemental Material
Figure S1 - Supplemental material for Limonoids From Choerospondias axillaris
Supplemental material, Figure S1, for Limonoids From Choerospondias axillaris by Nguyen Huu Toan Phan, Nguyen Thi Dieu Thuan, Nguyen Thi Thu Hien, Pham Van Huyen, Tran Thi Hong Hanh, Le Thi Vien, Tran Hong Quang, Nguyen Xuan Cuong, Nguyen Hoai Nam, Phan Van Kiem and Chau Van Minh in Natural Product Communications
Footnotes
Acknowledgments
The authors are grateful to Dr Tran Thi Nhu Hang, Institute of Natural Products Chemistry, VAST for the cytotoxic evaluation, and the Institute of Chemistry, VAST for measurement of the NMR spectra.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by Vietnam national project of the Tay Nguyen Program (TN18/C09).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
