Abstract
Background
Citrus fruits are among the most widely cultivated fruits worldwide, and contain bioactive compounds, including flavonoids, carotenoids, limonoids, coumarins, and vitamin C, some of which are significantly bioactive. Consequently, discovering new compounds from Citrus fruits is an important objective.
Methods
The seeds of C. × junos Siebold ex Tanaka were extracted with methanol, which afforded eleven constituent compounds whose chemical structures were elucidated using NMR and MS techniques. The anti-proliferative activities of the isolated compounds were evaluated using the WST-8 assay.
Results
Junosterpene, a new limonoid derivative, was isolated from the seeds of C. × junos, along with seven known limonoids, 21,23-dihydro-23-hydroxy-21-oxodeacetylnomilin, deacetylnomilin, 21,23-dihydro-21-hydroxy-23-oxonomilin, obacunoic acid, methyldeacetylnomilinate, limonin, and ichangin, and three known furanocoumarins, (−)-heraclenol, (−)-byak-angelicin, and (+)-oxypeucedanin hydrate. None of the isolated compounds exhibited significant anti-proliferative activity against the human glioblastoma cell lines T98G and U-251 MG.
Conclusions
The newly identified limonoid derivative is possible to contribute to the diverse bioactive profile of C. × junos and provides insight into its potential physiological or pharmacological properties.
Introduction
Citrus fruits are among the most widely cultivated fruits worldwide. 1 Citrus, which is a genus in the Rutaceae family, encompasses 40 different species. 2 Mandarin-type Citrus is a heterogeneous group of East Asian Citrus with small fruit bodies. 3 Indeed, mandarins have witnessed the greatest increase in global production among Citrus fruits, 3 and contain bioactive compounds, including flavonoids, carotenoids, limonoids, coumarins, and vitamin C, 4 with some compounds exhibiting significant bioactivities, including cytotoxic, antiviral, and antimutagenic properties.5–7
Among mandarin-type Citrus fruits, C. × junos Siebold ex Tanaka is a sour orange that originated in Central China 8 and whose fruit is not only consumed fresh and as juice, but also as traditional medicine. 9 Previous studies have reported the beneficial health effects of C. × junos, including potent antidiabetic, anticancer, and anti-inflammatory properties in vitro and in vivo, 10 with flavonoids, 10 coumarins, 11 and limonoids 8 having been isolated and identified. Coumarins, such as auraptene, and limonoids, such as limonin, have been reported to have antitumor properties.12,13
In this paper, we report the isolation and structural determination of junosterpene (
Materials and Methods
General Experimental Procedures
Specific rotations were measured using a P-2200 digital polarimeter (l = 5 cm; JASCO, Tokyo, Japan). Fourier-transform infrared (FTIR) spectra were recorded on a JASCO FT/IR-4600 spectrometer. Ultraviolet–visible (UV-vis) spectra were acquired on a Shimadzu UV-1850 spectrophotometer (Shimadzu, Kyoto, Japan). High-resolution electrospray-ionization mass spectrometry (HR-ESI-MS) was performed using a JMS-T100LP AccuTOFLC-Plus 4G instrument (JEOL, Tokyo, Japan). 1H (600 MHz), 13C (150 MHz), and 2D nuclear magnetic resonance (NMR) spectra were recorded using a JEOL JNM-ECZ 600R spectrometer. Normal-phase silica-gel column chromatography was performed on Wakogel® 60N (FUJIFILM Wako Pure Chemical, Osaka, 63-212 µm). Reverse-phase silica-gel column chromatography was performed on C18-OPN (Nacalai Tesque, Kyoto, Japan, 75 µm). TLC was performed using TLC plates pre-coated with 60F254 silica gel (Merck, Darmstadt, Germany; 0.25 mm, ordinary phase) and Merck RP-18 F254S silica gel (0.25 mm, reverse phase). High-performance liquid chromatography (HPLC) was performed using a SPD-10Avp UV-vis detector (Shimadzu, Kyoto, Japan), LC-10ADvp pump (Shimadzu), SCL-10Avp system controller (Shimadzu), and LabSolutions LC software (ver. 1.25, Shimadzu)with COSMOSIL 5C18-AR-II (Nacalai Tesque, 250 × 4.6 mm i.d., 250 × 10 mm i.d.), and Luna Phenyl-Hexyl (250 × 4.6 mm i.d., 250 × 10 mm i.d.) (Phenomenex, CA, USA) columns used for analytical and preparative purposes, respectively. Solvent ratios are based on volume. IR, UV, and MS spectra were recorded using spectroscopic-grade methanol (Wako Pure Chemical, Japan).
Plant Material
C. × junos 14 fruit was supplied by a local farmer and purchased from the Japan Agricultural Cooperative (JA) farmers’ market in Yamaguchi Prefecture (Japan) in November 2022 (voucher specimen number; SOCU-2022-22). Total DNA was extracted from plant samples, and PCR amplification and sequencing were performed by Rizo Inc. (Sample number; PD0152, Tsukuba, Japan). 15 The obtained nucleotide sequences were analyzed using the BLAST tool available at the National Center for Biotechnology Information (NCBI) website (https://blast.ncbi.nlm.nih.gov/Blast.cgi). BLAST analysis was also conducted by Rizo Inc. Sequences were queried against the NCBI nucleotide collection (nr/nt) database using default parameters. The top hits with the highest sequence identities and lowest E values were used for taxonomic identification. Plant species were identified based on sequence similarity. Voucher specimens were deposited at the herbarium of the Department of Pharmacognosy, Faculty of Pharmaceutical Sciences, Sanyo-Onoda City University, Yamaguchi, Japan. The plant material was also examined and identified by Prof. Hiroyuki Tanaka (PhD) (Department of Pharmacognosy and Kampo, Faculty of Pharmaceutical Sciences, Sanyo-Onoda City University). The deposited voucher specimens, including fruit, peels, flowers, and leaves, were dried in the shade and stored.
Extraction and Isolation
Dried seeds of C. × junos were extracted three times with methanol (MeOH) under reflux for 3 h, following a modified procedure based on the method reported.
8
Solvent evaporation provided the MeOH extract. Additionally, an ethyl-acetate-soluble (EtOAc-soluble) fraction and an aqueous layer were obtained from the abovementioned MeOH extract. The aqueous layer was further extracted with n-butanol (n-BuOH) to give n-BuOH (6.5 g, 0.6%) and H2O (62.5 g, 5.5%) fractions. The EtOAc-soluble fraction was subjected to normal-phase silica-gel column chromatography [1.0 kg, n-hexane:CHCl3 (5:1→ 1:1→ 1:5) → CHCl3→ CHCl3:MeOH (1:0→ 200:1→ 100:1→ 50:1→ 10:1→ 7:1→ 5:1→ 1:1)]. C. × junos EtOAc-soluble fraction number five (CJEA5) (4.5 g) was further separated via reverse-phase silica-gel column chromatography [225.0 g, MeOH:H2O (4:6→ 5:5→ 6:4→ 7:3→ 8:2→ 9:1→ 1:0)] to give eight fractions (CJEA5-1-5-8). Compounds
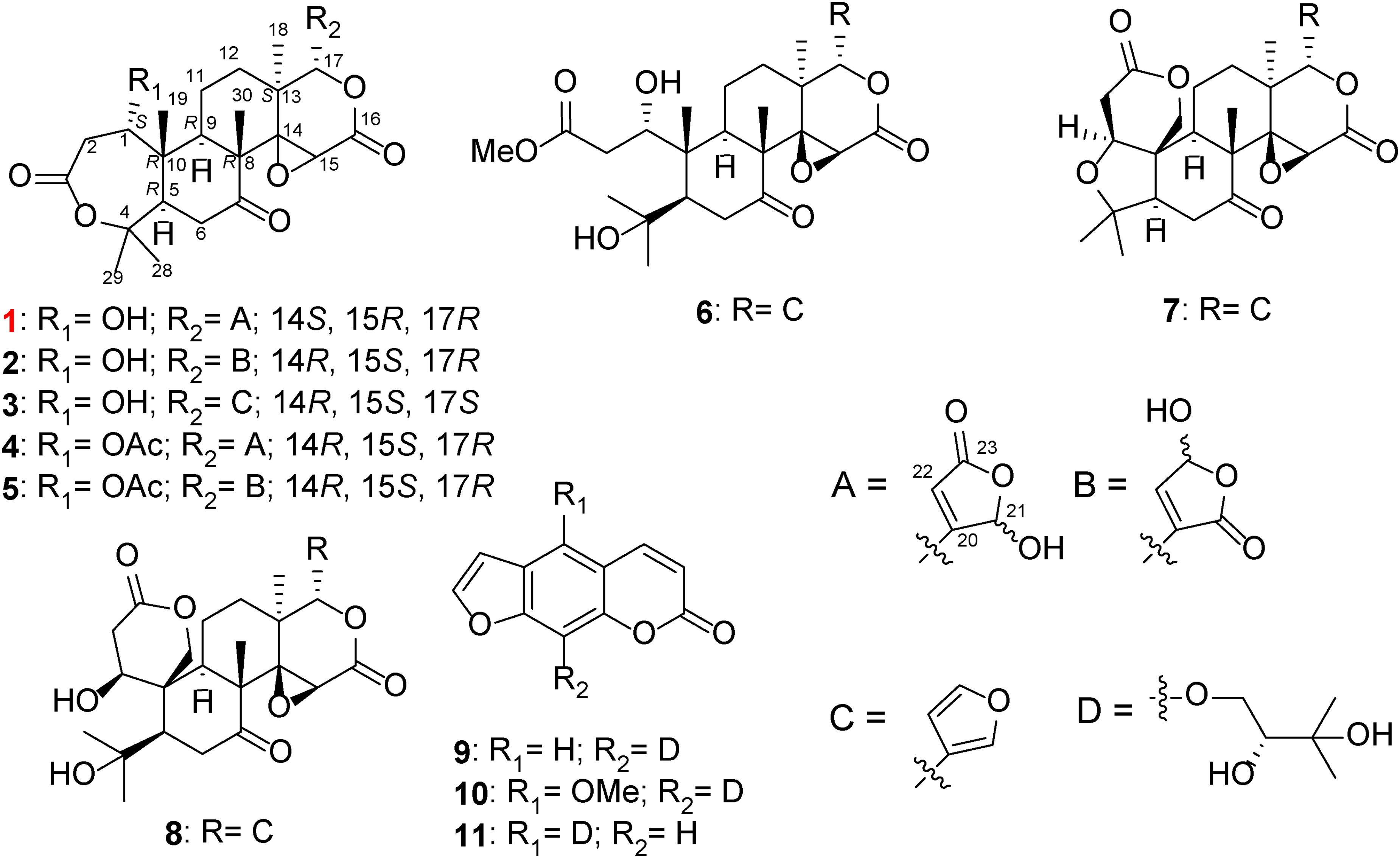
Skeletal (shorthand) structures of compounds isolated from C.
Junosterpene (1 )
White amorphous solid; [α]D25 −28.8 (c 0.1, MeOH); FTIR (ATR) vmax 1655, 2833, 2949, and 3307 cm−1; UV (MeOH) λmax (logε) 204.5 nm (3.47); 1H NMR (acetic acid-d4, 600 MHz) and 13C NMR (acetic acid-d4, 150 MHz) (Table 1); HR-ESI-MS m/z: 527.1908 (Calcd. for C26H32O10Na [M + Na]+ m/z: 527.1888).
13C (150 MHz) and 1H (600 MHz) NMR Data for Junosterpene (

*in acetic acid-d4, ** in methanol-d3, aOverlapping signals.
Cell Culture
U-251 MG (IFO50288, Japanese Collection of Research Bioresources Cell Bank, Osaka, Japan) and T98G (RCB1954, RIKEN BRC through the National Bio-Resource Project of the MEXT, Ibaraki, Japan) cells were cultured in Dulbecco's modified Eagle's medium (DMEM) with low glucose (FUJIFILM Wako Pure Chemical Industries, Osaka, Japan) supplemented with 10% fetal bovine serum (FBS: FUJIFILM Wako Pure Chemical Industries) and 5% penicillin-streptomycin solution (FUJIFILM Wako Pure Chemical Industries) under 5% CO2 at 37 °C. 16
WST-8 Assay
Cell proliferation was determined using a cell counting kit 8 (CCK-8: Wako Pure Chemical Industries) according to modifying the manufacturer's instructions and the method described previously. 17 Cells were seeded at a density of 1.0 × 104 cells/100 µL per well in 96-well cell culture plates (Coster 3596; Corning, NY, USA). After approximately 24 h, the cells were treated with adriamycin (Wako Pure Chemical Industries) or the isolated compounds (30 µM) for 24 h. CCK-8 solution containing WST-8 [2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4disulfophenyl)-2H-tetrazolium, monosodium salt] (10 µL) was added to the plates and incubated in a CO2 incubator for 2 h. 17 Absorbances was measured at 450 and 620 nm using a microplate reader (Multiskan FC; Thermo Fisher Scientific, MA, USA).
Statistical Analysis
Statistical analyses were performed with GraphPad Prism 8.43 software using the Dunnett's test to analyze differences between treatment groups. Differences were considered significant at *P < 0.01 when compared against DMSO-treated cells.
Results
Isolating Limonoids from Citrus × junos
The MeOH extract (136.5 g, 12.0%) of the seeds of C.
Structure of Junosterpene (1 )
Junosterpene (
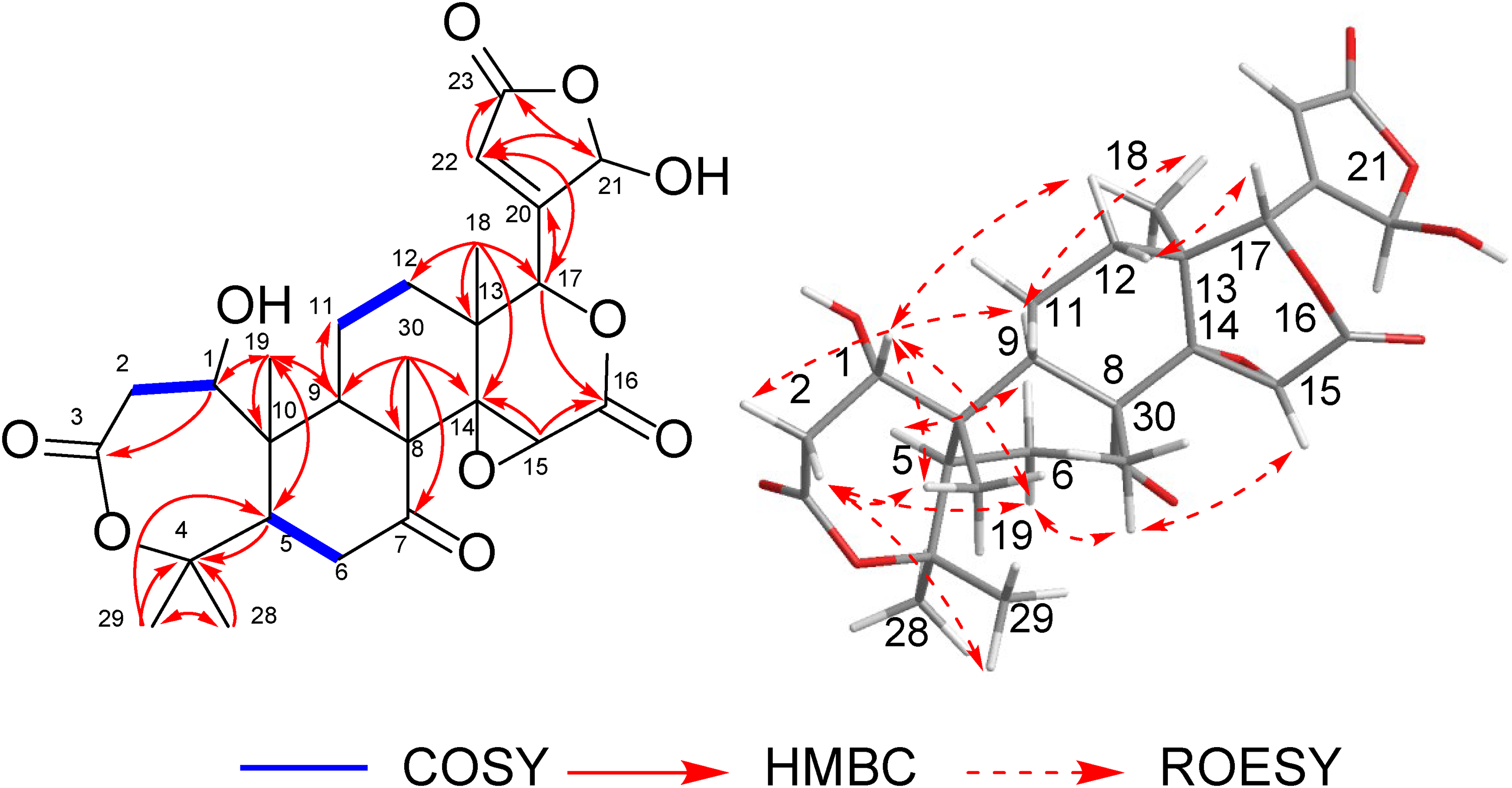
Important 2D NMR and ROESY correlations for junosterpene (
Evaluating Anti-Proliferative Effects
The anti-proliferative activities of the isolated compounds (
Discussion
Citrus fruits are rich sources of bioactive compounds, such as flavonoids, carotenoids, limonoids, sesquiterpenoids, and coumarins. 4 While hundreds of limonoids have been isolated from various plants, their occurrence is largely limited to plant families within the order Rutales, predominantly Meliaceae and Rutaceae. 13 Limonoids are unusual triterpenes owing to their extensively rearranged biosynthetic scaffolds. 29 At least two main scaffold modifications are proposed to be conserved in both plant families: a C-30 methyl shift of the protolimonoid scaffold (apo-rearrangement), and the conversion of the hemiacetal ring of melianol into a mature furan ring with the concomitant loss of a C4 carbon side-chain 30 ; additional modifications that are specific to Rutaceae hen yield nomilin-type intermediates. 30
However, the constituent components of the fruit of C. × junos have been less studied, although some studies on the bioactivities of its extracts have been reported.10,31 The primary objective of this study was to isolate novel limonoids, which led to the isolation of junosterpene (
Conclusion
Junosterpene (
Supplemental Material
sj-doc-1-npx-10.1177_1934578X251401118 - Supplemental material for Limonoids and Furanocoumarins from a Cultivated Citrus × junos Siebold ex Tanaka
Supplemental material, sj-doc-1-npx-10.1177_1934578X251401118 for Limonoids and Furanocoumarins from a Cultivated Citrus × junos Siebold ex Tanaka by Daisuke Imahori, Takuya Muraoka, Miharu Kubota and Hiroyuki Tanaka in Natural Product Communications
Footnotes
Acknowledgements
Ethical Approval
Not applicable.
Consent for Publication
Not applicable.
CRediT Authorship Contribution Statement
Daisuke Imahori: Writing–original draft, Project administration, Conceptualization. Takuya Muraoka: Data curation, Formal analysis. Miharu Kubota: Data curation. Hiroyuki Tanaka: Writing – review & editing, Supervision, Conceptualization.
Author Agreement Statement
We declare that this manuscript is original, has not been published previously, and is not currently under consideration for publication elsewhere.
We confirm that the manuscript has been read and approved by all named authors, and that there are no other persons who satisfy the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all authors.
We understand that the Corresponding Author is the sole contact for the Editorial process. He/she is responsible for communicating with the other authors about progress, submissions of revisions and final approval of proofs.
Funding
This work was supported by the Japan Society for the Promotion of Science (JSPS): KAKENHI Grant Numbers 22K20720 and 23K16308.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statements and Declarations
There are no human subjects in this study, therefore informed consent is not required.
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
