Abstract
Naringin possesses strong antioxidative activity and can protect against some respiratory diseases. Oxidative stress is thought to be a major factor in the development of many tobacco-caused diseases. The nuclear factor erythroid 2-related factor 2 (Nrf2) signaling pathway plays a critical role in the regulation of oxidative stress. The dynamic changes in the antioxidant system in the lung that are induced by cigarette smoke (CS) are not well investigated, and how naringin affects these changes remains unknown. This study aimed to investigate the dynamic changes between the oxidation and antioxidant systems resulting from CS exposure and the effects of naringin on these changes in mice. Mice were chronically exposed to CS for 30 days. The levels of malondialdehyde (MDA), glutathione (GSH), interleukin (IL)-6, and tumor necrosis factor-alpha (TNF-α); the activities of superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px); and the expressions of Nrf2, heme oxygenase-1 (HO-1), and nicotinamide adenine dinucleotide phosphate quinone dehydrogenase 1 (NQO1) in lung tissue were measured on days 2, 7, and 30. The levels of MDA, GSH, IL-6, and TNF-α in the lung were found to increase throughout the exposure. SOD and GSH-Px activities showed an increase on day 2 and a decrease on days 7 and 30. The messenger ribonucleic acid expressions of Nrf2, HO-1, and NQO1 were elevated on day 2 and decreased on day 7; Nrf2 and HO-1 expressions were continually decreased, but NQO1 expression was increased again, on day 30. Naringin restored the levels of these biochemical indices to normal throughout the experiment, suggesting that naringin protected against the CS-induced oxidative damage by suppressing the increase of antioxidants resulting from the early stage of CS exposure, as well as inhibiting the depletion of antioxidants due to long-term oxidative stress. Naringin also suppressed lung inflammation by inhibiting IL-6 and TNF-α. These results indicate that naringin possesses a powerful ability to maintain the balance of the oxidation/antioxidant system in the lung when subjected to CS exposure, probably by regulating the Nrf2 signaling pathway.
Keywords
Cigarette smoke (CS) exposure is a recognized risk factor for acute and chronic respiratory diseases, 1 and cigarette use is the leading cause of preventable death worldwide and remains a serious and growing global health threat. 2 CS contains more than 4000 chemicals, many of which have been associated with deleterious health consequences. 3 Particularly, oxidants, such as nicotine, acrolein, and carbon monoxide, can be detrimental. 4 Oxidative damage resulting from oxidants of CS, such as nitric oxide and carbonyls, plays major roles in the development of many tobacco-caused diseases including respiratory and heart diseases, cancers, and others. 5,6 For example, carbonyls are a major class of powerful oxidants, which can deplete glutathione (GSH), form adducts with deoxyribonucleic acid (DNA) bases, and produce free radicals when metabolized. 7 Kelch-like ECH-associated protein 1 (Keap1)-Factor E2-related factor (Nrf2) pathway plays a key role in regulating oxidative stress. 8,9 As a member of the cap ‘n’ collar protein family, Nrf2 is found in the cytoplasm through its interaction with the E3 ubiquitin ligase Keap1. Under normal conditions, Nrf2 captured by Keap1 is continually ubiquitinated and degraded by proteasomes; when cells are exposed to oxidative stress, thiols of cysteine residues in Keap1 are modified and Nrf2 is stabilized. 10 Nrf2 binds to antioxidant responsive elements to activate downstream target genes, such as nicotinamide adenine dinucleotide phosphate quinone dehydrogenase 1 (NQO1), heme oxygenase-1 (HO-1), and superoxide dismutase (SOD) which together protect against oxidative stress. 8 As a transcription factor expressed predominantly in the alveolar macrophages and epithelium, Nrf2 plays an essential protective role in the lungs by inducing expressions of antioxidant and cytoprotective genes. 11
It has been reported that acute CS exposure led to Nrf2 activation in human macrophages, and Nrf2 expression was decreased in pulmonary macrophages in current smokers and patients with chronic obstructive pulmonary disease (COPD). 12 The early oxidative stress induced by CS exposure has been shown to initiate the activation of Nrf2, and this activation can alleviate respiratory damage. 13 However, long-term CS-induced oxidative stress would lead to an imbalance between the oxidation and antioxidant system in the host, causing a decrease of both Nrf2 level and the expression of its downstream gene. 14 As far as we know, the dynamic changes of the oxidation/antioxidant system, including the changes of antioxidants and expression of the Nrf2 signaling pathway, have not been investigated.
Naringin (Figure 1), a bioactive flavonoid, is widely found in citrus plants of the Rutaceae family. Naringin possesses a variety of biological properties, such as antioxidant, anti-inflammatory, anticancer, hepatoprotective, neuroprotective, and antiatherosclerosis activities. 15,16 In recent years, we have investigated the protective effects of naringin against respiratory diseases; we found that it can attenuate pulmonary inflammation in lipopolysaccharide-induced acute lung injury in mice and beagle dogs 17,18 and showed an anti-inflammatory effect which was responsible for attenuating chronic pulmonary neutrophilic inflammation induced by CS exposure in rats. 19 However, the regulatory effect of naringin on dynamic changes in the oxidation/antioxidant system resulting from CS exposure has not been investigated. In this study, we investigated the dynamic changes in the oxidation/antioxidant system resulting from CS exposure and the regulatory effect of naringin on these changes in a mouse model.

Chemical structure of naringin.
Experimental
Animals and Drugs
All animal experiments were performed with the approval of the Animal Care and Use Committee of the School of Life Sciences, Sun Yat-sen University. Male Balb/c mice with a bodyweight of 19-23 g were purchased from Guangdong Medical Laboratory Animal Center. Animals were maintained on a constant 12 hour light/dark cycle with food and water ad libitum. Naringin (purity, 98.3%) was prepared as described in our previous studies. 20,21 Roflumilast (purity, 99.0%) was purchased from Hubei Yuan Cheng Saichuang Technology Co., Ltd (Wuhan, China). Cigarettes (details obtained from the instructions on the commodity label: 11 mg/cigarette for tar, 1 mg/cigarette for nicotine, and 13 mg/cigarette for CO) were purchased from China Tobacco Guangdong Industrial Co., Ltd (Yeshu, Guangzhou, China) and used for the production of CS.
Treatments
Animals were habituated to maintenance conditions for 1 week and then divided into 6 groups (n = 18): normal group, cigarette smoke exposure (CSE) group, ROF (Roflumilast) group, and the other 3 naringin groups. The normal group was cotreated with air and distilled water; the CSE group was cotreated by CS exposure and distilled water; the naringin groups were cotreated by CS exposure and different doses of naringin (15, 30, and 60 mg/kg/day, respectively); the ROF group was cotreated with CS exposure and roflumilast (5 mg/kg/day, in distilled water). Roflumilast is an oral selective phosphodiesterase-4 inhibitor approved for the treatment of COPD. 22 In this study, roflumilast was used as a positive control. All drugs were administered orally. Six mice in each group were selected randomly and anesthetized on days 2, 7, and 30. Then, the mice were sacrificed by cervical dislocation; whole lung tissues were obtained and frozen immediately at −80 °C.
Exposure of Mice to CS
Mice were subjected to CS exposure utilizing a smoking apparatus (100 cm × 80 cm × 80 cm; Guangzhou Flyde Biotechnology Co., Ltd, Guangzhou, China). Mice underwent CS exposure of 10 cigarettes for 1 hour each time, 2 times per day at 4 hours intervals, 5 days per week for 30 days, including the day before sacrifice.
RNA Extraction From Lung Tissue
Ribonucleic acid (RNA) extraction was performed as previously described. 23 Twenty milligrams of lung tissue was cut into pieces and transferred to a tube. Subsequently, 1 mL of TRIzol reagent was added, mixed, and incubated for 10 minutes. Following the incubation, 0.2 mL of chloroform was added to each tube for mixing and incubated at room temperature for 10 minutes. The mixture was centrifuged at 12 000×g at 4 °C for 15 minutes. The aqueous phase containing the RNA was transferred to a new 1.5 mL Eppendorf tube with added 0.4 mL of isopropanol and incubated at room temperature for 10 minutes. Then, the mixture was centrifuged at 12 000×g at 4 °C for 10 minutes. The supernatant was discarded, and the pellet was resuspended in 1 mL of 75% ethanol for brief mixing and centrifuged at 12 000×g at 4 °C for 10 minutes. The RNA pellet was dried in the air in a fume cupboard and resuspended in 20 µL of RNase-free water for storing at −80 °C.
Reverse Transcription and Reverse Transcriptase Quantitative PCR (RT-qPCR)
Total RNA was quantified using a NanoDrop 2000 spectrophotometer (Thermo, USA). Samples were reverse transcribed using a GoScript Reverse Transcription System (Promega, USA). The polymerase chain reaction (PCR) primer pairs were used to amplify the gene expressions of Nrf2, HO-1, and NQO1. The sequence is shown in Table 1. The reaction was conducted using the Light Cycler 480 SYBR Green I Master kit (Roche, USA), according to the manufacturer’s instructions. The qPCR conditions used were as follows: denatured at 95 °C for 10 seconds; 45 cycles of 95 °C for 10 seconds, 60 °C for 20 seconds and 72 °C for 20 seconds, with melting and cooling. The relative gene expressions were analyzed using the 2−△△Ct method. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as endogenous control.
RT-qPCR Primer Sequence.

GAPDH, glyceraldehyde-3-phosphate dehydrogenase; RT-qPCR, reverse transcription and reverse transcriptase quantitative polymerase chain reaction; NQO1, nicotinamide adenine dinucleotide phosphate quinone dehydrogenase 1; HO-1, heme oxygenase-1.
Measurements of SOD, MDA, GSH-Px, GSH, IL-6, and TNF-α in Lung Tissue
Tissue samples were homogenized in ice-cold normal saline in a ratio of 1/9 and then centrifuged at 4000 rpm for 15 minutes at 4 °C. Total protein concentration was measured with a bicinchoninic acid protein assay kit (Beyotime, Jiangsu, China). The levels of interleukin (IL)-6 and tumor necrosis factor-alpha (TNF-α) in lung tissue were detected using enzyme-linked immunosorbent assay kits (all from USCN KIT INC., Wuhan, China). The levels of SOD, malondialdehyde (MDA), glutathione peroxidase (GSH-Px), and GSH in lung tissue were detected using commercial kits (all from Nanjing Jiancheng Bioengineering Institute, Nanjing, China), according to the manufacturer’s instructions.
Statistical Analysis
Data analyses were performed using GraphPad Prism (GraphPad Prism Software, La Jolla, CA, USA). The results are presented as means ± SD for each group. Significant differences between the groups were assessed by one-way analysis of variance. Results were considered significant at P < 0.05.
Results
Effects of Naringin on CS-Induced Oxidative Stress in Lung Tissue
The effects of naringin on CS-induced oxidative stress are shown in Figure 2. On days 2, 7, and 30, in response to CS exposure, the MDA level in lung tissue in the CSE group was significantly increased compared with that in the normal group. Naringin (60 mg/kg) significantly reduced the MDA level in lung tissue throughout the experiment. In response to CS exposure, the activities of SOD and GSH-Px in lung tissue were significantly increased on day 2 and decreased on days 7 and 30. Naringin (60 mg/kg) restored the activities of SOD and GSH-Px to normal throughout the experiment. However, CS exposure led to increased concentration of GSH throughout the experiment, and naringin (60 mg/kg) restored the concentration of GSH to normal. These results demonstrated that naringin could alleviate CS-induced oxidative stress in the lung in mice.
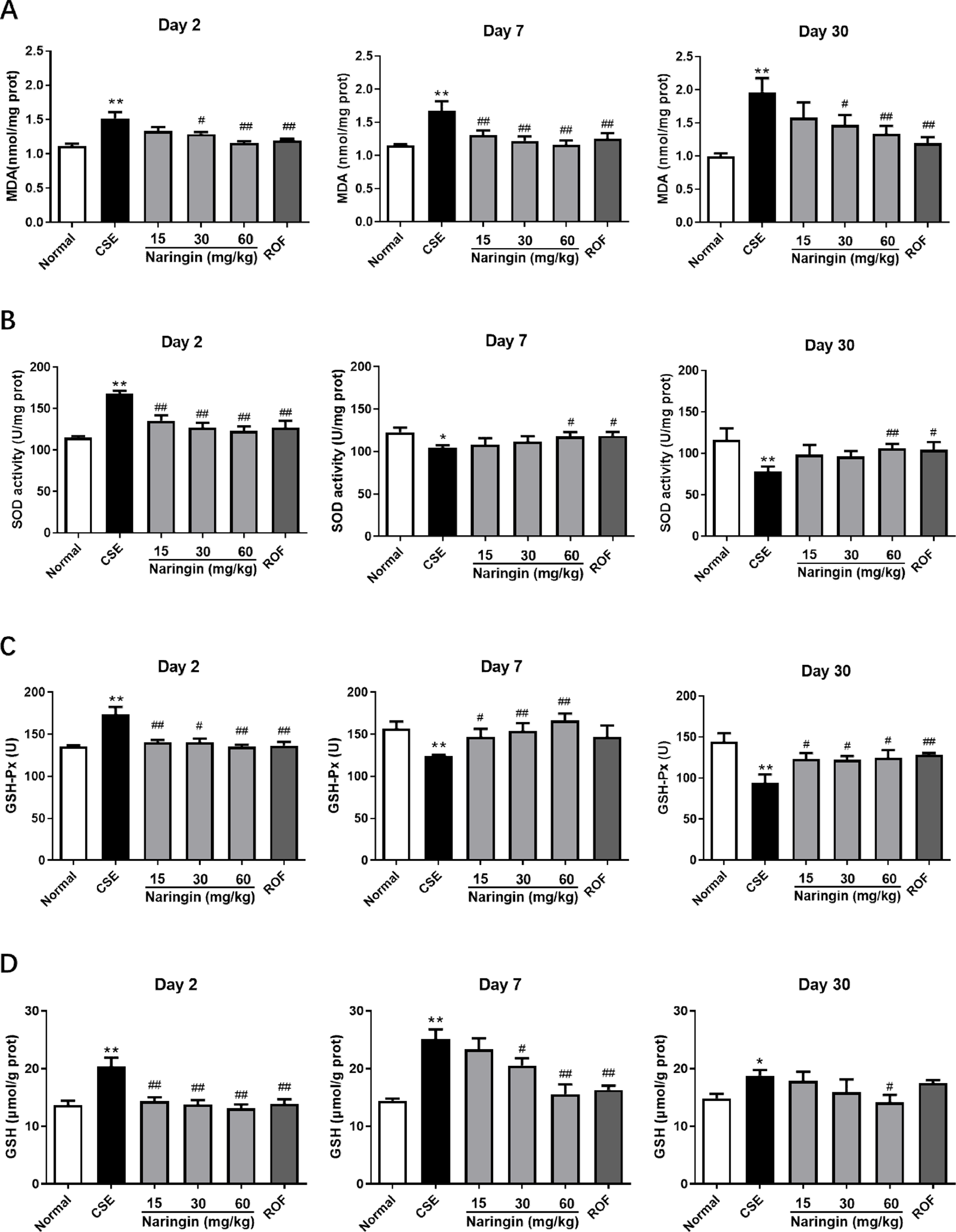
Effect of naringin on the levels of MDA (A), SOD (B), GSH-Px (C), and GSH (D) in the lung of mice exposed to CS on days 2, 7, and 30 (n = 6). Data are expressed as mean ± SD. * P < 0.05, ** P < 0.01 compared with normal group; # P < 0.05, ## P < 0.01 compared with CSE group. Normal, mice cotreated with distilled water and air; CSE, mice cotreated with distilled water and CSE; naringin groups, mice cotreated with different dose of naringin and CSE; ROF, mice cotreated with roflumilast (5 mg/kg) and CSE. CS, cigarette smoke; CSE, cigarette smoke exposure; GSH, glutathione; GSH-Px, glutathioneperoxidase; MDA, malondialdehyde; SOD, superoxidedismutase.
Effects of Naringin on CS-Induced Expressions of Nrf2, HO-1, and NQO1 in Lung Tissue
On day 2, the mRNA expressions of Nrf2, HO-1, and NQO1 were significantly increased in the lung due to CS exposure, and naringin (60 mg/kg) significantly inhibited these increases (Figure 3). However, the mRNA expressions of Nrf2 and HO-1 were significantly decreased on days 7 and 30, and naringin reversed these decreases. Interestingly, the mRNA expression of NQO1 was significantly decreased in the CSE group compared with that of the Normal group on day 7; naringin was capable of inhibiting this decrease. However, the CSE group showed a significant increase in mRNA expression of NQO1 once again on day 30; naringin regulated the mRNA expression of NQO1 to normal.

Effect of naringin on the expressions of Nrf2 (A), HO-1 (B), and NQO1 (C) in the lung of mice exposed to CS on days 2, 7, and 30 (n = 6). Data are expressed as mean ± SD. * P < 0.05, ** P < 0.01 compared with normal group; # P < 0.05, ## P < 0.01 compared with CSE group. Normal, mice cotreated with distilled water and air; CSE, mice cotreated with distilled water and cigarette smoke exposure (CSE); naringin groups, mice cotreated with different dose of naringin and CSE; ROF, mice cotreated with roflumilast (5 mg/kg) and CSE. CSE, cigarette smoke exposure; HO-1; heme oxygenase-1; Nrf2, nuclear factor erythroid 2-related factor 2; NQO1, nicotinamide adenine dinucleotide phosphate quinone dehydrogenase 1.
Effects of Naringin on CS-Induced IL-6 and TNF-α Levels in Lung Tissue
As shown in Figure 4, IL-6 and TNF-α levels in the CSE group were significantly increased compared with the normal group on days 2, 7, and 30. Naringin (60 mg/kg) significantly inhibited the IL-6 and TNF-α levels in lung tissue throughout the experiment. This indicated that naringin was capable of inhibiting the persistent lung inflammation caused by CS exposure.

Effect of naringin on the levels of IL-6 (A) and TNF-α (B) in the lung of mice exposed to CS on days 2, 7, and 30 (n = 6). Data are expressed as mean ± SD. * P < 0.05, ** P < 0.01 compared with normal group; # P < 0.05, ## P < 0.01 compared with CSE group. Normal, mice cotreated with distilled water and air; CSE, mice cotreated with distilled water and cigarette smoke exposure (CSE); Naringin groups, mice cotreated with different dose of naringin and CSE; ROF, mice cotreated with roflumilast (5 mg/kg) and CSE. CS, cigarette smoke; CSE, cigarette smoke exposure; IL-6, interleukin-6; TNF-alpha, tumor necrosis factor α.
Discussion
Most reported studies have focused on the chronic effects of CS exposure, with the rationale that COPD is a tobacco-caused disease that arises from chronic smoking. There have been relatively few studies on the effects of long-term CS exposure on dynamic changes in oxidative stress. The Nrf2 signaling pathway plays a critical role in the antioxidant system. Investigating how this signaling pathway changes in the process of long-term CS exposure is useful to understand the pathogenesis of CS-induced respiratory disease.
In this study, we investigated the dynamic changes of both antioxidants and the expressions of the Nrf2 signaling pathway in mice subjected to CS exposure. The level of MDA in the lung was significantly increased throughout the experiment due to CS exposure, indicating that CS exposure resulted in persistent oxidative damage to the lung. In the early stage of CS exposure (day 2), the antioxidant system in lung tissue had been triggered. These activation events, including the increase of activities of SOD and GSH-Px, an increase in the concentration of GSH, as well as the activation of the Nrf2 signaling pathway, together protected against the oxidative damage. Over time, lung tissue was continuously subjected to free radical attacks generated from CS, which led to an imbalance of the antioxidant system. The activities of SOD and GSH-Px were significantly decreased on days 7 and 30, suggesting that a gradual collapse of the antioxidant system occurred. However, the level of GSH in the lung in the CSE group was higher than that in the Normal group throughout the experiment. A previous study has reported that CS exposure led to the GSH level in the lung epithelial lining fluid of mice peaking at 16 hours, and over repeat exposures were maintained at a higher level for up to 120 days. 24 It has also been reported that the GSH level was increased in rats after being subjected to CS exposure for 4 weeks. 25 Taken together, the upregulation of GSH levels was not accidental since the GSH adaptive response caused by early CS exposure was maintained at a relatively high level throughout the CS exposure. Several studies have suggested that GSH homeostasis may play a key role in the maintenance of the integrity of the lung airspace epithelial barrier, 26 which may help to understand the pathogenesis of COPD. The similarity in changes in expression of the Nrf2 signaling pathway with an increase was observed in the lung in the early CS exposure period. The mRNA expressions of Nrf2, HO-1, and NQO1 showed significant increases in lung tissue on day 2, indicating that the Nrf2 signaling pathway was activated. On days 7 and 30, the mRNA expressions of Nrf2 and HO-1 were decreased due to persistent oxidative damage generated from CS exposure. NQO1 showed a decrease on day 7 and a rebound increase on day 30, indicating that NQO1 cannot simply be depleted but involves a complex fluctuation in response to long-term oxidative stress. NQO1 is a cytosolic flavoenzyme, and its expression is mainly regulated by the antioxidant response element. 27 However, the change of NQO1 expression during long periods of CS exposure has been rarely studied, and how the fluctuation of NQO1 expression occurs is so far unknown, requiring further study to elucidate this process. Taken together, these results indicated that there were dynamic changes in the antioxidant system in the lung, which could be used to investigate the effects of naringin on these changes.
According to our data, naringin could excellently improve the imbalance between the oxidation and antioxidant system caused by acute or chronic CS exposure. Naringin suppressed the increase of antioxidants resulting from early stage CS exposure as well as inhibited the depletion of antioxidants due to persistent oxidative stress. These results demonstrate that naringin could maintain the balance of the oxidation/antioxidant system, which can protect against the oxidative damage induced by CS exposure. HO-1, NQO1, and SOD are the antioxidant genes regulated by Nrf2 binding to antioxidant response elements. 28,29 These genes play important roles in response to oxidative stress and have cytoprotective effects. 30 HO-1 can catalyze the degradation of heme, and the degradation products also have antioxidant functions. 31 NQO1 is an intracellular protective reductase that protects cells from damage by external quinones, such as the conversion of benzoquinones into less toxic hydroquinone and hydroxyl compounds. 32 SOD is a metal enzyme widely existing in the body, which can produce the disproportionation reaction by decomposing superoxide anion radical into hydrogen peroxide and molecular oxygen. 33 MDA is the product of lipid peroxidation and a classic indicator of oxidative stress. Our data indicated that the Nrf2 signaling pathway is involved in the protection of naringin against oxidative stress, while the mechanism of action of naringin in regulating the Nrf2 signaling pathway remains unknown. It has been reported that the expression of Nrf2 could be controlled epigenetically via promoter methylation. 34 In the early stage of CS exposure, the production of Nrf2 was increased in response to oxidative damage; over time CS exposure, a long term of oxidative stress probably modulated the methylation level of the Nrf2 gene promoter region, which would at least partially contribute to the inhibition of Nrf2 expression; naringin probably blocks or reverses these epigenetic alterations, and this hypothesis will be validated in a future study. Previous studies have shown that naringin has good antioxidative activity, 35 but the effects of naringin on dynamic changes of oxidative stress that occur with CS exposure are usually overlooked. To our knowledge, this is the first study reporting the effects of naringin on the dynamic changes of the oxidation/antioxidant system induced by CS exposure.
Additionally, CS exposure led to persistent inflammation in the lung as evidenced by a significant increase of IL-6 and TNF-α levels in lung tissue throughout the experiment. Naringin was capable of inhibiting the inflammation induced by CS exposure in mice. These findings were in line with previous studies that naringin has a distinguished anti-inflammatory activity. 36,37
We noted that the mRNA expressions of Nrf2, HO-1, and NQO1 at 30 mg/kg naringin dose showed the highest expression, indicating that this result is not accidental. Why the protective effect of 60 mg/kg naringin dose was weaker than that in 30 mg/kg naringin dose remains unknown and needs further investigation. As to this result, we think that there are at least 2 possible explanations: (1) as extensively reported in the literature, the reduced forms of flavonoids or other dietary phenolics act as antioxidants; however, the oxidized forms of flavonoids (phenoxyl radicals or quinone/quinone methide intermediates) can have prooxidant activities, and this activity intimately depends on their concentration. 38 -40 The study of Ranawat et al 38 revealed that naringenin, an active metabolite of naringin, led to a dose-dependent reduction in the level of GSH with a concomitant increase in GSSG in mice testis, thus shifting the redox ratio significantly; the activities of CAT, SOD, and GPx were also found to be decreased by naringenin administration in a dose-dependent manner; (2) the mice responded differently to naringin at different stages of CS exposure in this study. Additionally, the protective effect of naringin (30 mg/kg) on day 7 is stronger than that on day 30 for an unknown reason. Over time, mice exposed to CS suffered increasing oxidative damage that was evidenced by the increased MDA level on day 30, while the expression of Nrf2 was further inhibited. Long-term oxidative stress might lead to the hypermethylation of the Nrf2 gene promoter region, which was probably responsible for the inhibition of Nrf2. The inhibition of Nrf2 might limit the antioxidant capacity of naringin, and at least partially contribute to the mice responding differently to naringin in different stages of CS exposure. Taken together, these results show that the dosage level of naringin should be strictly controlled, taking into account the disease progression of the patient.
There are several limitations to the current investigation. In our experiment, the mice were subjected to CS exposure for 30 days, which is not long enough, considering that CS-induced respiratory diseases, such as COPD, take decades to develop. We conducted the detection only 3 times throughout the exposure and the results should therefore be treated as preliminary. A comprehensive and thorough investigation based on a longer period of time with more frequent dynamic monitoring is needed for future study.
Conclusion
The present study has shown that dynamic changes in the antioxidant system occurred in the lung during CS exposure, which triggered a strong antioxidative response in the lung at an early stage. Over time, depletion of the antioxidant enzyme would lead to an imbalance between oxidation and antioxidant systems. However, the relatively high level of GSH was maintained for long periods, even with the oxidant burden of CS exposure; the expression of NQO1 showed a fluctuation in the lung during the period of CS exposure. Additionally, CS led to inflammation in the lung throughout the exposure. Naringin can protect against the CS-induced oxidative damage by suppressing the response increase of antioxidants resulting from early-stage CS exposure, as well as inhibiting the depletion of antioxidants due to long-term oxidative stress. Naringin also showed an anti-inflammatory effect. Taken together, naringin possesses a powerful ability to maintain the balance of the antioxidant system in the lung, probably by regulating the Nrf2 signaling pathway.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Applied Science and Technology R&D Special Fund Project of Guangdong Province (No. 2016B020239003) and the Science and Technology Planning Project of Guangdong Province (No. 2019B090905002).
