Abstract
The use of synthetic chemicals, with harmful effects on the environment and human health, is the principal strategy in the management of stored-product insect pests such as Oryzaephilus surinamensis and Sitophilus oryzae. Various studies in recent years have highlighted the possibility of using plant essential oils as available and low-risk factors in insect pest management. Therefore, in the present study, the possibility of controlling O. surinamensis and S. oryzae was investigated using Eucalyptus camaldulensis and Eucalyptus viminalis leaf essential oils. The essential oils were obtained by hydrodistillation of the leaves of the 2 Eucalyptus species, and the chemical compositions were determined by gas chromatographic-mass spectral analysis. The essential oil of E. camaldulensis was dominated by p-cymene (24.8%), cryptone (18.9%), and spathulenol (12.4%), while the major components in E. viminalis essential oil were 1,8-cineole (51.6%) and α-pinene (15.8%). The essential oils displayed promising fumigant toxicity against insect pests, which was positively dependent on utilized concentrations and exposure times. Oryzaephilus surinamensis, with low median lethal concentrations, was more susceptible than S. oryzae to the essential oils after 24, 48, and 72 hours. Also, E. viminalis essential oil, with a high level of insecticidal monoterpenes such as 1,8-cineole and α-pinene, was more toxic to insect pests than E. camaldulensis oil. According to the results of the current study, E. camaldulensis and E. viminalis essential oils, rich in insecticidal terpenes, can be alternative candidates to synthetic chemicals in the management of O. surinamensis and S. oryzae.
Saw-toothed grain beetle (Oryzaephilus surinamensis L., Coleoptera: Silvanidae) is one of the most destructive insect pests of stored products, including a variety of cereal grains, flour, bran, pasta, nuts, seeds, tobacco, and even historical collections in many countries throughout the world. 1 The small size of the pest allows it to keep hidden in storage conditions, making it difficult to control. 2 The resistance of O. surinamensis to some conventional insecticides has also been reported in recent studies. 3,4
Rice weevil (Sitophilus oryzae L., Coleoptera: Circulionidae) is one of the most destructive coleopteran insect pests of cereal grains, which economically reduces quantity (by direct feeding) and quality (by contaminating and increasing crop moisture) of stored grains. Adults and larvae of S. oryzae feed on the carbohydrate content of endosperm and grain germs. 5,6 The resistance of S. oryzae to some chemical insecticides, particularly to the main fumigant used in storage conditions, phosphine, has been reported recently. 7,8
Due to the availability and high efficiency, the use of chemical pesticides is the main method in pest management strategies. However, their application has caused several side effects, such as destructive effects on the environment, acute and chronic effects on human health and nontarget organisms, including birds, fish, bees, and parasitoid and predator insects, disruption of plant defense mechanisms, and pest resistance. 9 -11 Therefore, the use of alternative, low risk, and, at the same time, effective pest control agents is essential.
The Eucalyptus genus belongs to the Myrtaceae family and has more than 800 different species. Although the origin of these plants is the Australian continent, they have been planted in many tropical and subtropical regions, due to their high adaptability and rapid growth to obtain wood, gum, cellulose, and essential oils. 12 Eucalyptus camaldulensis Dehnh. (river red gum) is one of the evergreen trees that is grown in a purposeful way for use in the wood and paper industries. 13 The essential oils and extracts isolated from the aerial parts of this plant have been used in traditional medicine. 14 The antifungal, antibacterial, antioxidant, and insecticidal properties of E. camaldulensis essential oil, which mainly contain terpenes such as p-cymene, 1,8-cineole, β-phellandrene, and spathulenol, have been recorded. 15 -17 The essential oil extracted from Eucalyptus viminalis Labill. (manna or ribbon gum), which is rich in terpene compounds such as 1,8-cineole, α-pinene, limonene, and aromadendrene, has also shown various biological effects such as antimicrobial, antioxidant, and insecticidal properties in recent years. 18 -20
In order to introduce the biorational and efficient insecticidal agents, the main objective of the present study was to evaluate the fumigant toxicity of essential oils extracted from the leaves of E. camaldulensis and E. viminalis against O. surinamensis and S. oryzae. Because of the importance of clarifying the probable relationship between the toxicity of essential oils with their components, the chemical composition of essential oils was also analyzed using gas chromatography-mass spectrometry (GC-MS).
Result and Discussion
Essential Oil Analysis
The mean yields of E. camaldulensis and E. viminalis essential oils were 2.10 ± 0.16% and 1.03 ± 0.12%, respectively, based on the extraction from 5 separate samples. Forty-four compounds were identified in the essential oil extracted from E. camaldulensis leaves, accounting for 97.4% total oil. Oxygenated monoterpenoids (55.8%) had the highest amount in the essential oil followed by monoterpene hydrocarbons (28.7%), oxygenated sesquiterpenoids (12.6%), and sesquiterpene hydrocarbons (only 0.2%). The most abundant compound in the essential oil was p-cymene (24.8%), followed by cryptone (18.9%), spathulenol (12.4%), terpinen-4-ol (8.5%), 1,8-cineole (6.9%), cuminaldehyde (5.1%), and phellandral (3.8%) (Table 1).
Chemical Compositions of the Leaf Essential Oils of Eucalyptus camaldulensis and Eucalyptus vminalis.

Eucalyptus viminalis essential oil was rich in terpenes, with monoterpene hydrocarbons (18.5%), oxygenated monoterpenoids (64.5%), sesquiterpene hydrocarbons (2.2%), and oxygenated sesquiterpenoids (11.8%) accounting for 97.0% of the total essential oil. Forty-three components were identified, in which 1,8-cineole (51.6%), α-pinene (15.8%), globulol (5.7%), trans-pinocarveol (3.7%), spathulenol (3.1%), and aromadendrene (1.6%) were the main (Table 1).
The chemical composition of E. camaldulensis and E. viminalis essential oils investigated in some previous studies have obvious differences with the present findings. For example, γ-terpinene (42.5%), 1,8-cineole (33.6%), p-cymene (17.5%), and terpinen-4-ol (3.9%) were determined as the main components of E. camaldulensis essential oil in the study of Siramon et al. 13 The quantity of γ-terpinene (0.5%) and 1,8-cineole (6.9%) was very low and, in contrast, p-cymene (24.8%) and terpinen-4-ol (8.5%) have higher percentages in the present work. In another work, 1,8-cineole (4.1%-39.5%), p-cymene (27.8%-42.7%), cryptone (3.2%-10.2%), spathulenol (2.1%-15.5%), and β-phellandrene (3.9%-23.8%) had high percentages in the essential oil of E. camaldulensis from 4 different geographical origins in Italy. 14 1,8-Cineole (6.9%), p-cymene (24.8%), cryptone (2.2%), and spathulenol (12.4%), with approximately equal amounts, were also identified in the present study but β-phellandrene had no trace. Terpenes p-cymene (42.1%), 1,8-cineole (14.1%), α-pinene (12.7%), and α-terpineol (10.7%) had a high amount in the E. camaldulensis essential oil in the study of Dogan et al, 16 while α-pinene was found to be in very low percentage (1.5%) in the present study. In the study Maghsoodlou et al, 19 1,8-cineole (57.8%), globulol (3.1%), limonene (5.4%), and α-pinene (13.4%) were identified as main components of E. viminalis essential oil, while a minimal amount of limonene (0.8%) was identified in our study. In the study of Lucia et al, 23 1,8-cineole (85.0%), globulol (2.5%), aromadendrene (2.0%), p-cymene (1.9%), and α-terpineol (1.7%) were the main components of E. viminalis essential oil. α-Pinene (15.8%) as a main component in the present study had a very low percentage (1.1%) in that research. Furthermore, trans-pinocarveol (3.7%) and spathulenol (3.1%), with high quantities in our study, were not detected in this study.
Fumigant Toxicity
Based on the results of the Kolmogorov-Smirnov test, data on the fumigant toxicity of E. camaldulensis and E. viminalis essential oils against the adults of O. surinamensis and S. oryzae had normal distributions (Table 1). The selected essential oil concentrations and 24-hours, 48-hours, and 72-hours of exposure times had statistically significant effects on the mortality of both insect pests, according to the analysis of variance (ANOVA). However, the interaction between both essential oil concentration and the time on the mortality of S. oryzae was not significant (Table 2).
The Results of the Kolmogorov-Smirnov Test and Analysis of Variance of the Data Obtained From the Fumigant Toxicity of Eucalyptus camaldulensis and Eucalyptus viminalis Essential Oils Against the Adults of Oryzaephilus surinamensis and Sitophilus oryzae.

Abbreviation: Abbreviation: NS, nonsignificant at α = 0.05.
aSignificant at α = 0.05. The number of both tested insects is 480 in each time.
Eucalyptus camaldulensis and E. viminalis essential oils presented notable fumigant toxicity against the adults of O. surinamensis and S. oryzae. A concentration of 14.71 µL/L of either of the essential oils created 100% mortality in O. surinamensis within the 72-hour exposure time. At high tested concentrations of E. camaldulensis (22.06 µL/L) and E. viminalis (26.47 µL/L) essential oils, 100% and 95% mortality, respectively, was also achieved on S. oryzae. According to the comparison of means by the Tukey’s test at α = 5%, the lowest and highest mortalities of both insect pests correlated to the lowest and highest concentrations of essential oils, respectively. In general, increasing the concentration of essential oils and exposure time significantly increased mortality in both pests (Figure 1).

Mortality (%) of Oryzaephilus surinamensis and Sitophilus oryzae adults due to fumigation by Eucalyptus camaldulensis and Eucalyptus viminalis essential oils. Means separated by Tukey’s test at P ≤ 0.05. The difference of means with similar letters is not statistically significant.
The results of the probit analysis of data obtained from the fumigant toxicity of E. camaldulensis and E. viminalis essential oils on O. surinamensis and S. oryzae adults are shown in Tables 3 and 4. The median lethal concentration (LC50) value of E. camaldulensis essential oil was estimated as 7.76 µL/L after 24 hours on the adults of O. surinamensis, which decreased to 3.36 µL/L after progressing the time to 72 hours. These values for S. oryzae were 12.83 µL/L after 24 hours and 6.59 µL/L after 72 hours. On the other hand, the susceptibility of both pests to E. camaldulensis essential oil increased with increasing exposure time. Oryzaephilus surinamensis to the fumigation by E. camaldulensis oil was more sensitive than S. oryzae, although overlapping was found in their 95% fiducial limits at 48 and 72 hours (Table 3).
LC Values and Regression Line Information of the Fumigant Toxicity of Eucalyptus camaldulensis and E. viminalis Essential Oil Against the Adults of Oryzaephilus surinamensis and Sitophilus oryzae.
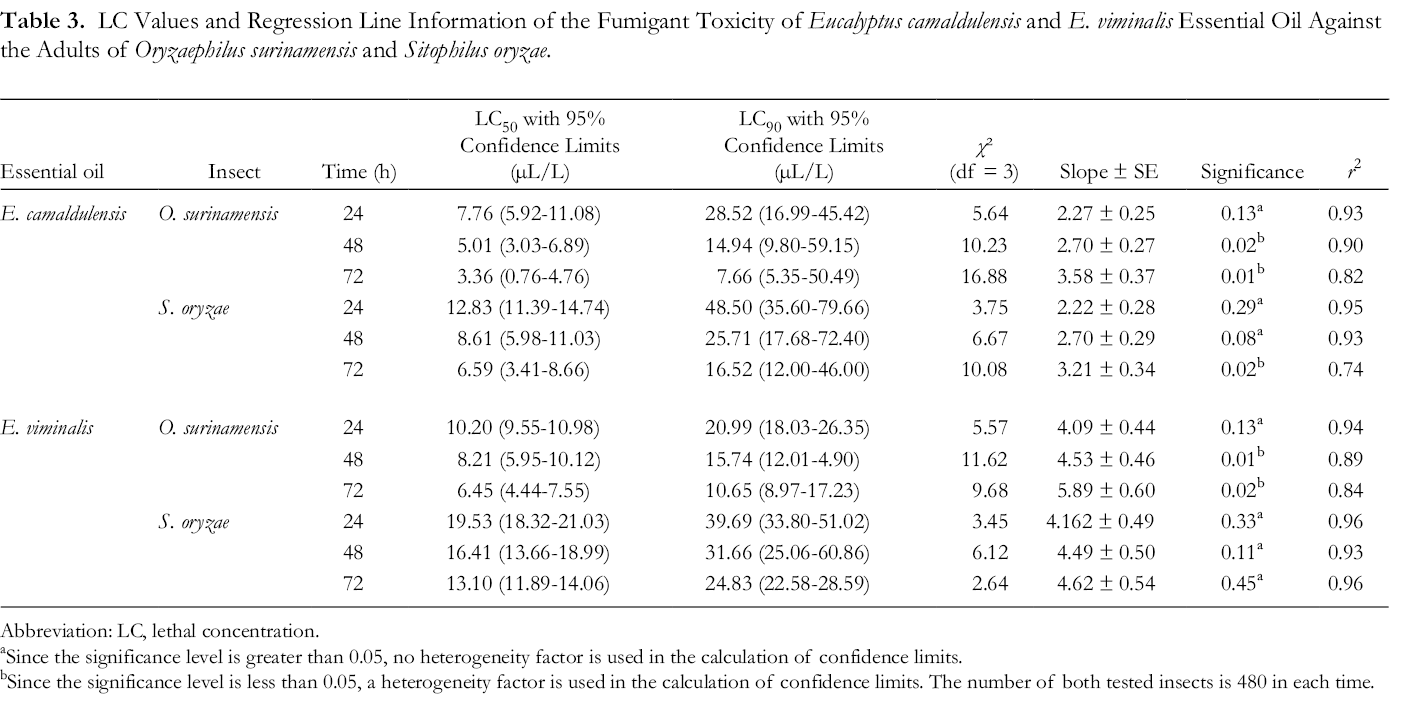
Abbreviation: Abbreviation: LC, lethal concentration.
aSince the significance level is greater than 0.05, no heterogeneity factor is used in the calculation of confidence limits.
bSince the significance level is less than 0.05, a heterogeneity factor is used in the calculation of confidence limits. The number of both tested insects is 480 in each time.
LT Values and Regression Line Information of the Fumigant Toxicity of Eucalyptus camaldulensis and Eucalyptus viminalis Essential Oil Against the Adults of Oryzaephilus surinamensis and Sitophilus oryzae at the Highest Tested Concentration.
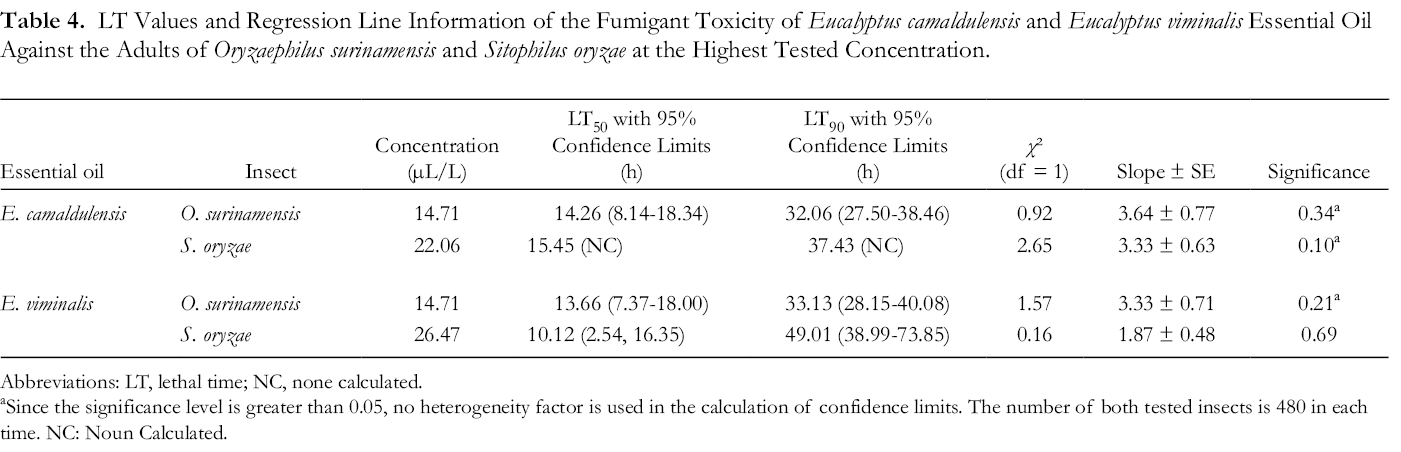
Abbreviations: Abbreviations: LT, lethal time; NC, none calculated.
aSince the significance level is greater than 0.05, no heterogeneity factor is used in the calculation of confidence limits. The number of both tested insects is 480 in each time. NC: Noun Calculated.
The LC50 value of E. viminalis essential oil on O. surinamensis adults was 10.20 µL/L after 24 hours, which was decreased significantly within 72 hours to 6.45 µL/L. These values for S. oryzae were 19.53 µL/L at 24 hours and 13.10 µL/L at 72 hours. In other words, the susceptibility of both pests to the essential oil of E. viminalis was also increased over time. Also, O. surinamensis was more susceptible than S. oryzae to the fumigation by E. viminalis oil (Table 3).
Comparison of LC50 values of E. camaldulensis essential oils (3.36 µL/L) and E. viminalis (6.45 µL/L) at 72 hour-exposure time showed that, despite the partial overlapping in 95% fiducial limits, O. surinamensis was more susceptible to E. camaldulensis than E. viminalis. After 72 hours, the LC50 of E. camaldulensis essential oil for S. oryzae (6.59 µL/L) was statistically lower than the corresponding value in E. viminalis (13.10 µL/L). Consequently, S. oryzae was also more susceptible to E. camaldulensis than E. viminalis (Table 3).
Also, high values of correlation coefficients (r 2) of E. camaldulensis and E. viminalis essential oil concentrations on the mortality of both insects in all exposure times indicate a positive and direct correlation between them (Table 3).
According to median lethal time (LT50) values, 14.26 hours of exposure time will be adequate to 50% mortality in O. surinamensis at a concentration of 14.71 µL/L E. camaldulensis essential oil. This time for S. oryzae was 15.45 hours with 22.06 µL/L of E. camaldulensis essential oil. The LT50 of 13.66 hours by 14.71 µL/L of E. viminalis essential oil recorded for O. surinamensis. The concentration of 26.47 µL/L from this essential oil will kill 50% of the S. oryzae in 10.12 hours (Table 4).
In addition, there have been several investigations into the evaluation of the pesticidal properties of essential oils extracted from many species of the genus Eucalyptus in recent years 24 -26 ; the insecticidal potential of E. camaldulensis and E. viminalis has also been investigated against some stored-product insect pests. For example, the fumigant toxicity of Eucalyptus intertexta R.T. Baker, Eucalyptus sargentii Maiden, and E. camaldulensis essential oils against the adults of Callosobruchus maculatus (Fab.), S. oryzae, and Tribolium castaneum (Herbst) has been reported by Negahban and Moharramipour. 27 Unfortunately, the chemical compositions of the essential oils were not reported. The LC50 values of these essential oils, respectively, were determined to be 2.55, 6.93, and 11.59; 3.87, 12.91, and 18.38; 3.97, 12.62, and 33.50 µL/L after 24 hours. The increases in essential oil concentrations and exposure times had also increased insect mortality as in this present study. The LC50 of E. camaldulensis essential oil on S. oryzae (12.91 µL/L) is approximately close to the estimated value in the present study after 24 hours (12.83 µL/L). Also, the 72 hour LC50 value estimated for E. camaldulensis essential oil on S. oryzae in the present study (6.59 µL/L) was lower than all of the above-mentioned essential oils. In the other research, fumigant toxicity of E. camaldulensis essential oil reported on the adults of C. maculatus with LC50 of 26.10 µL/L, which is more than all LC50 values achieved in the present study for O. surinamensis and S. oryzae. It was also determined that the toxicity of this essential oil increased with increasing concentration and exposure time. 15 Toxicity of essential oils isolated from 5 Eucalyptus species, including E. camaldulensis, E. viminalis, E. microtheca F. Muell., E. grandis W. Mill ex Maiden, and E. sargentii was recorded on the larvae of T. castaneum with 48 hours LC50 values of 103.37, 35.48, 87.01, 63.06, and 122.20 µL/L, respectively. Accordingly, the toxicity of E. viminalis essential oil has been higher than the others. 18 The chemical compositions of the essential oils were not determined, however. Furthermore, the LC50 values of E. viminalis essential oil on both O. surinamensis and S. oryzae adults, obtained in the current study, are lower than the corresponding values in the above-mentioned study. Recently, the susceptibility of Blattella germanica (L.) to the essential oil of E. camaldulensis was evaluated. 17 The LC50 values against the first nymphal stage and adults of B. germanica were 19.360 and 21.817 µL/L, respectively, after 24 hours. The essential oil composition of E. camaldulensis was not determined, however. Along with high toxicity, the fumigant toxicity of E. viminalis essential oil against O. surinamensis and S. oryzae, and E. camaldulensis essential oil against O. surinamensis is reported for the first time in the current study.
The insecticidal activities of essential oils are closely related to their components, especially terpenes. 28 -31 Insecticidal effects of the high percentage components identified in the current study are listed in Table 5; monoterpene hydrocarbons (p-cymene and α-pinene) and oxygenated monoterpenoids (1,8-cineole, cuminaldehyde, linalool, phellandral, terpinen-4-ol, pinocarveol, and α-terpineol), are recognized in the essential oils of E. camaldulensis and E. viminalis, indicated promising insecticidal effects against different groups of pests. Indeed, high vapor pressures of monoterpenes candidate them for application in greenhouse and storage conditions as fumigant pesticides. 32,33 However, their contact toxicity was also documented against some other pests. including cockroaches and mosquitoes. 28,34 Due to lower vapor pressures, high contact toxicity, and moderate fumigant pesticidal activities of sesquiterpenes, such as aromadendrene and spathulenol identified in the present research, were also reported. 31,35 Consequently, the promising fumigant toxicity of E. camaldulensis and E. viminalis essential oils may be associated with high percentages of the above-mentioned monoterpenes. However, synergistic, additive, and antagonistic effects all components should be considered in the essential oil activities. 29,31
Review of the Reported Insecticidal Effects for Main Terpenes Existing in E. camaldulensis and E. viminalis Essential Oils.
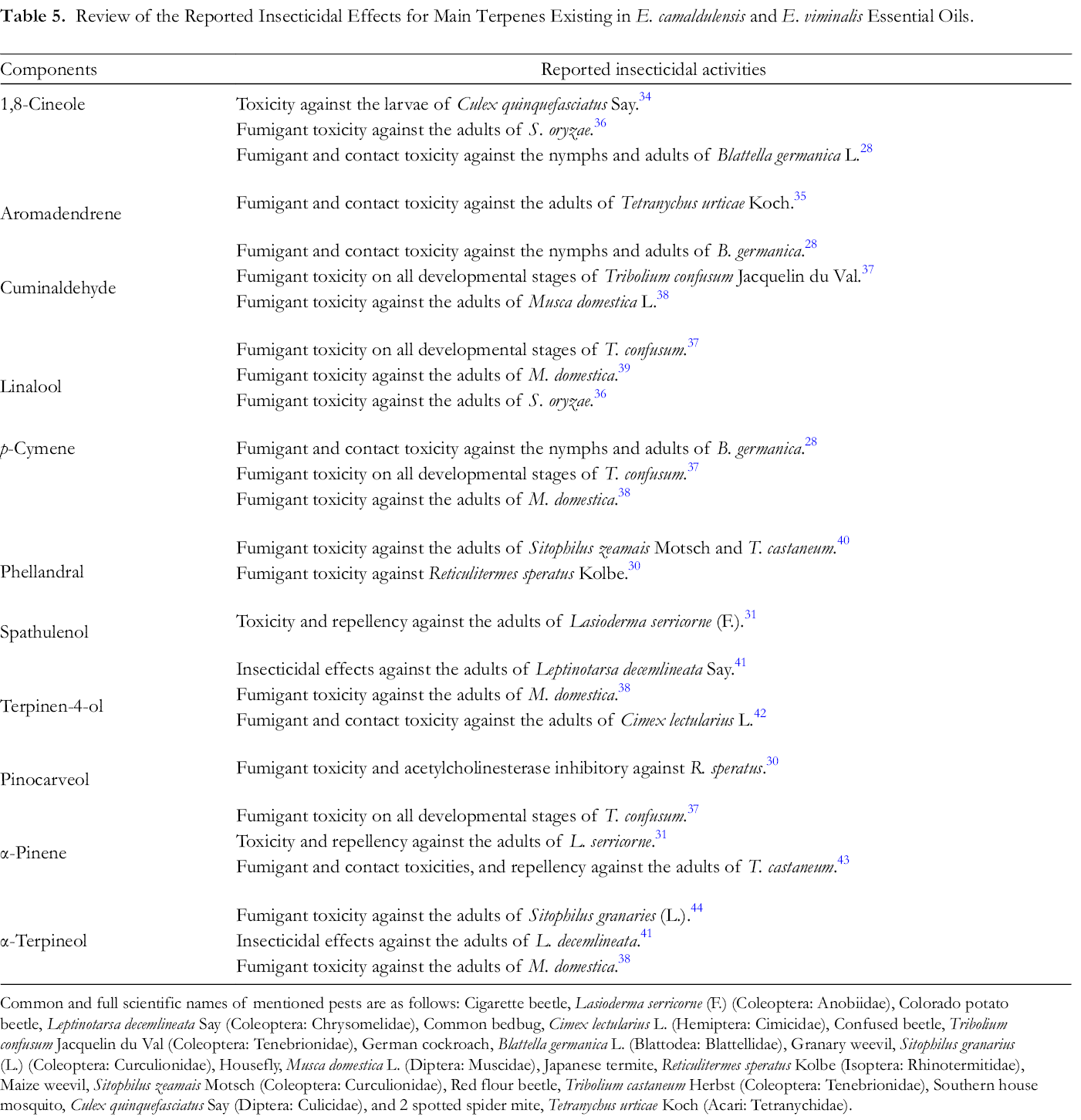
Common and full scientific names of mentioned pests are as follows: Cigarette beetle, Lasioderma serricorne (F.) (Coleoptera: Anobiidae), Colorado potato beetle, Leptinotarsa decemlineata Say (Coleoptera: Chrysomelidae), Common bedbug, Cimex lectularius L. (Hemiptera: Cimicidae), Confused beetle, Tribolium confusum Jacquelin du Val (Coleoptera: Tenebrionidae), German cockroach, Blattella germanica L. (Blattodea: Blattellidae), Granary weevil, Sitophilus granarius (L.) (Coleoptera: Curculionidae), Housefly, Musca domestica L. (Diptera: Muscidae), Japanese termite, Reticulitermes speratus Kolbe (Isoptera: Rhinotermitidae), Maize weevil, Sitophilus zeamais Motsch (Coleoptera: Curculionidae), Red flour beetle, Tribolium castaneum Herbst (Coleoptera: Tenebrionidae), Southern house mosquito, Culex quinquefasciatus Say (Diptera: Culicidae), and 2 spotted spider mite, Tetranychus urticae Koch (Acari: Tetranychidae).
Conclusion
Our findings demonstrate the essential oils isolated from leaves of E. camaldulensis and E. viminalis to be effective biorational insecticides against O. surinamensis and S. oryzae. The essential oils are rich in insecticidal monoterpenes, both monoterpene hydrocarbon and monoterpenoid groups, such as 1,8-cineole, linalool, p-cymene, and α-pinene. The E. viminalis essential oil with a high level of low vapor-pressure monoterpenoids, such as 1,8-cineole, showed relatively more fumigant toxicity against both insect pests than E. camaldulensis oil. Application of such eco-friendly efficient insecticides will support to decrease the side effects of chemical pesticides comprising environmental contamination, human health risk, and development of insect resistance. Further investigations should be focused on the isolation of pure components from such plants and the evaluation of their insecticidal potential and safety.
Experimental
The Plant Materials and Essential Oil Extraction
The leaves of the E. camaldulensis and E. viminalis trees were collected from the Agricultural and Natural Resources Research Center, Moghan Station, Ardabil Province, Iran (47°78ʹN, 39°60ʹE, elevation 69 m) at beginning of blooming in April, which has been cultivated there before the last 10 years. The voucher specimens were also deposited there with their scientific names. After being transferred to the laboratory, the plant specimens were dried on a table in the shade for a week and separately pulverized using an electric grinder. The powder of each plant (100 g) along with 500 mL distilled water was poured into a 1 L round-bottom flask of an all-glass Clevenger apparatus. The extraction of essential oils was completed within 3 hours, and the obtained essential oils were separately poured to glass vials and dried over sodium sulfate. The vials were sealed and stored under refrigeration at 4 °C.
Chemical Characterization of the Essential Oils
The chemical compositions of E. camaldulensis and E. viminalis essential oils were assessed using GC (Agilent 7890B; Santa Clara, CA, USA) coupled with an MS (Agilent 5977A). The analysis was done using an HP-5 MS capillary column (30 m × 0.25 mm × 0.25 µm), according to Ebadollahi and Setzer. 45 The carrier gas was helium (99.999%) with a flow rate of 1 mL/min. Each of the essential oils was diluted in methanol (1:10), and 1 µL of the solution was injected. The temperature of the injector was 280 °C, and the column temperature adjusted from 50 °C to 280 °C. The carrier gas was helium (99.999%), with a flow rate of 1 mL/min. Retention index values computed according to a mixture of homologous n-alkanes (C8-C20), which was analyzed under the same chromatographic conditions. The identification of components was performed by comparing mass spectral fragmentation patterns and retention indices with those reported in the databases. 21,22,46,47
Insect Rearing
The initial colony of O. surinamensis obtained from the Department of Plant Production, Moghan College of Agriculture and Natural Resources, University of Mohaghegh Ardabili, Iran (47°72ʹN, 39°58ʹE, elevation 72 m), where the insect had been reared on wheat grains for several generations. The initial colony of S. oryzae was collected from rice storage at Vizneh village, Hevigh region, Gilan Province, Iran (48°87ʹN, 32°26ʹE, elevation −20 m). Fifty unsexed pairs of insects were separately transferred to uncontaminated wheat and rice grains, respectively, and removed after 48 hours. The grains contaminated with insect eggs were separately kept in an incubator at 26 ± 2 °C, 65 ± 5% relative humidity, and a photoperiod of 14:10 (L:D) hours over a period of at least 3 months. Adult insects (aged 1-10 day) were selected for the bioassays.
Fumigant Toxicity
To determine the appropriate concentrations of essential oils, preliminary range-finding experiments were performed for each essential oil. The final concentration ranges were determined as 5.88-14.71 and 11.76-26.47 µL/L from E. camaldulensis and 2.94-14.71 and 5.29-22.06 µL/L from E. viminalis against O. surinamensis and S. oryzae, respectively. To investigate the fumigant toxicity of essential oils, 20 adults of each insect pest (aged 1-10 days) separately transferred into glass containers (340 mL) as fumigant chambers, and their caps were closed. The calculated concentrations of essential oils poured on filter paper (Whatman No. 1) pieces with dimensions of 3 × 2 cm. The treated filter papers were attached to the inside of the glass container lids, which were then tightly closed. Mortality was documented after 24, 48, and 72 hours of exposure times. The experiments were conducted for control groups without any essential oil concentrations, and each of the treatments was repeated 4 times.
Statistical Analysis
The mortality of both insect pests created by the fumigation of E. camaldulensis and E. viminalis essential oils was checked for normality with the Kolmogorov-Smirnov method. To eliminate the effect of mortality in control groups, the mortality percentage was corrected using Abbott’s formula: Pt = [(Po − Pc)/(100 − Pc)] × 100, in which Pt is the corrected mortality (%), Po is the mortality (%) of insects treated by essential oil concentrations, and Pc is the mortality (%) in the control groups. The data were submitted to ANOVA and the means separated by a Tukey’s test at P ≤ 0.05. The correlation coefficient (r 2), regression lines, LC50 and LC90 values with their 95% confidence limits, and χ² values were determined for each tested essential oil and insect species. Statistical software SPSS version 24.0 (Chicago, IL, USA) was used for all analyses.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study received financial support from the University of Mohaghegh Ardabili, which is 20 greatly appreciated. WNS participated in this work as part of the activities of the Aromatic 21 Plant Research Center (APRC, https://aromaticplant.org/).
