Abstract
Cake tea, a traditional beverage of China, has excellent health benefits. Our study investigated the phytochemical profiles, antioxidant, and antiproliferative activities of cake tea leaves, which were obtained from the same cultivar and processed at different intervals. The effects of bioactive compounds and antioxidant activities of 4 cake tea varieties on a human lung cancer cell line (A549 cells) were systematically examined. The content of total polyphenol, an active ingredient of tea, was significantly higher in green cake teas (14.0% ± 0.4a) than their black (4.8 ± 0.3c), yellow (10.0 ± 0.6b), and white (8.8 ± 0.5b) counterparts. Likewise, the content of total free amino acids in green cake tea (3.6% ± 0.5a) was significantly higher than the other tea varieties. Our results indicated that the extent of fermentation of tea leaves could decrease the antioxidant activities of cake tea leaves. Furthermore, the white tea cake variety demonstrated the maximum antiproliferative activity on A549 cells as opposed to other types of cake tea leaves. Such an observation allows future researchers to narrow down their focus on using specific cake tea types (sourced from the same cultivar) that provide the maximum health benefits.
Tea, a popular beverage made from leaves of the plant Camellia sinensis, has been shown to have numerous health benefits, such as reduction of body weight, alleviation of metabolic syndrome (MetS), cancer prevention, and improved immune system. 1 -3 Besides, the presence of several secondary metabolites, such as catechins, purine alkaloids, and theanine, render excellent taste and aromatic flavor to the tea sourced from C. sinensis, which is a popular nonalcoholic beverage worldwide. 4
In general, tea can be classified into 6 major categories based on its different processing techniques and characteristics special to each individual group, such as unfermented green tea, mildly fermented yellow tea, mildly fermented white tea, semifermented oolong tea, fully fermented black tea, and postfermented dark tea, which determines the final polyphenol content of the product. 5 -7 Fermentation is a multistep process that involves enzymatic oxidation of tea polyphenol by endogenous polyphenol oxidase and peroxidase. 8 Microorganisms are necessary to ferment black and dark tea, which oxidize the polyphenols to theaflavins and rosin, as well as converting catechins into new compounds. As reported, a decrease in the catechin content of green and black tea is accompanied by an increase in their theaflavin and thearubigin contents. Cake tea is produced by steaming, pressing, and drying of loose tea leaves, which result in firm, small-sized, high-density leaves that are convenient for packaging and transportation. 9 Moreover, the prolonged shelf life of cake tea is convenient for long-term storage because of which it has gained immense popularity in China, and, therefore, the demand for different flavors of cake tea such as green, white, yellow, and black cake tea is increasing among consumers. Interestingly, the effect of processing parameters on the antioxidant and antiproliferative activity of the cake tea has not been investigated. Previous studies had thoroughly analyzed the processing technologies of cake tea 9 ; however, the chemical constituents and health benefits of cake tea are largely unknown.
Typically, the quality and health benefits of tea are subject to enormous variations depending on the genetic background and growing environment of tea, appropriate horticultural practices, harvest season, processing, and storage of tea leaves. 8,10,11 In the present study, we implemented traditional manufacturing processes to produce green cake tea (GCT), white cake tea (WCT), yellow cake tea (YCT), and black cake tea (BCT) using fresh tea leaves sourced from the same variety of tea plant, that is, Yinghong No. 9, which is one of the most widely cultivated tea varieties in southern China (Guangdong province). The protocols that are followed for processing the tea leaves are illustrated in Figure 1. The chemical compositions of 4 types of cake tea were determined, and their phenolic compounds were subjected to quantitative and comparative analysis. Additionally, the antioxidant activities of 4 types of cake tea were evaluated using multiple antioxidant assays, including 1,1-diphenyl-2-picrylhydrazyl (DPPH), 2,2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS), and ferric reducing antioxidant power (FRAP). Since tea processing has multiple steps that include withering, rolling, and fermentation, each step plays a vital role in shaping a quality tea blend. Therefore, the objective of our research was to explore the influence of manufacturing processes on the phytochemical profiles, antioxidant, and antiproliferative activities of cake tea sourced from a single cultivar which was subjected to postharvest processing under different conditions. The purpose of this work was to study the oxidation resistance and inhibition of tumor cell growth in vitro of 4 types of tea cakes processed from the same variety of fresh tea leaves. The results of this study can be considered a standard based on which future researchers could choose the most suitable processing technology for cake teas to receive maximal health benefits.

Flow diagram depicting the manufacturing processes used to produce the 4 types of cake tea investigated.
Results and Discussion
Processing methods can change the content of phytochemical components of tea. 6 The contents of the major chemical constituents of 4 types of cake tea samples are shown in Table 1. The content of soluble solid was higher in YCT (35.5% ± 0.9%) than GCT (32.7% ± 2.1%); no statistically significant differences were observed between BCT and WCT. With an increase in the degree of fermentation, the total polyphenol content decreased. 12 However, the total polyphenol content was considerably higher in GCT than those that were not fermented (YCT and WCT). However, the differences between YCT and WCT were not significant. Alternatively, the lower levels of polyphenol in BCT were directly related to the fermentation process. As reported, tea polyphenols can oxidize during the fermentation process, which decreases the content of total polyphenols. 6 It is reported that the fermentation process could result in a decrease of free amino acids in tea. 13 In our study, the content of total free amino acids in GCT was considerably higher than BCT, YCT, and WCT, but they did not show any statistically significant differences. The content of total flavonoids was significantly higher in BCT than the other 3 tea samples, which were not significantly different from each other. Typically, soluble sugar is responsible for the sweet taste of tea soup. 14 However, in our investigation, the total content of soluble sugars in YCT and WCT was significantly higher than that in GCT. Nevertheless, the total content of soluble sugars in BCT was higher than in GCT, but the difference was not statistically significant.
Major Chemical Constituents of GCT, WCT, YCT, and BCT.
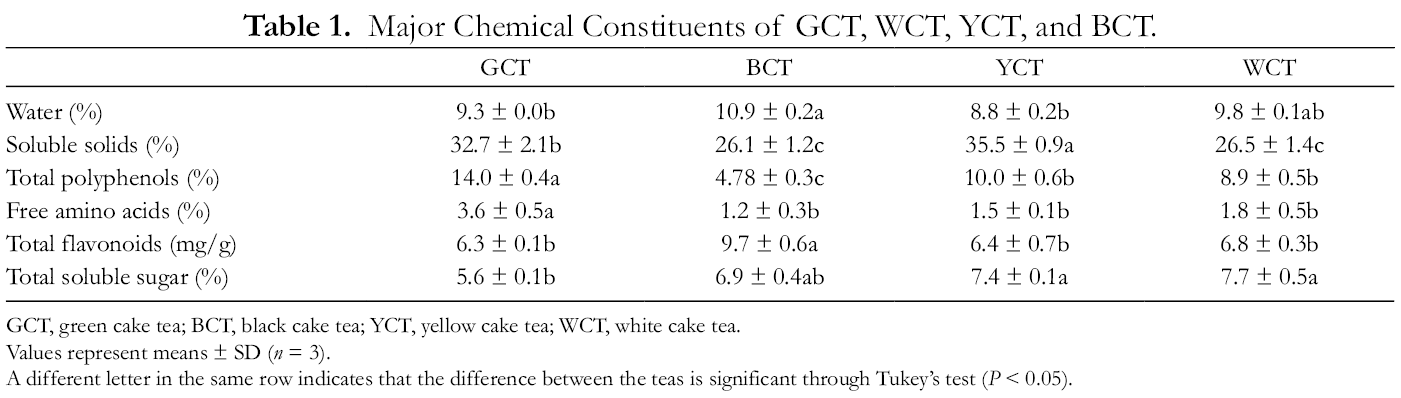
GCT, green cake tea; BCT, black cake tea; YCT, yellow cake tea; WCT, white cake tea.
Values represent means ± SD (n = 3).
A different letter in the same row indicates that the difference between the teas is significant through Tukey’s test (P < 0.05).
Individual phenolic compounds and caffeine content of 4 types of cake tea leaves are shown in Table 2. Caffeine was the primary compound in all the tested samples. Although the caffeine contents were similar in slightly fermented tea leaves (WCT and YCT) and fermented tea (BCT), non-fermented green tea demonstrated the highest caffeine content (GCT). The results were consistent with previous studies, which reported that fermentation could cause a slight loss of extractable caffeine. 15 It was noted that the content of gallic acid in YCT was significantly higher than the other tea samples. The reason for this can be attributed to the type of ester present in catechins, for example, epigallocatechin-3-gallate (EGCG) and epicatechin gallate (ECG), which hydrolyzes into gallic acid and other compounds during the “sealed yellowing” process of making YCT. 5 As shown in Table 2, the content of 8 catechin monomers in BCT was significantly lower than the other cake tea leaves, which could be due to the complete fermentation of tea leaves, which oxidized the catechins into theaflavins, thearubigins, and theabrownin. The contents of gallocatechin (GC), gallocatechin gallate (GCG) in GCT were also considerably higher than those in YCT and WCT; nevertheless, they were not significantly different. With an increase in the degree of fermentation, the content of catechin monomer C and EGCG in cake tea decreased in the order of GCT > YCT > WCT > BCT. Alternatively, epigallocatechin (EGC), EC, and ECG contents were in the order of YCT > GCT > WCT >BCT. The content of bitter catechins consisting of esters and the total catechin levels in GCT were significantly higher than those of the other tea samples.
Content of Phenolic Compounds and Caffeine in 4 Tea Samples (mg/g).
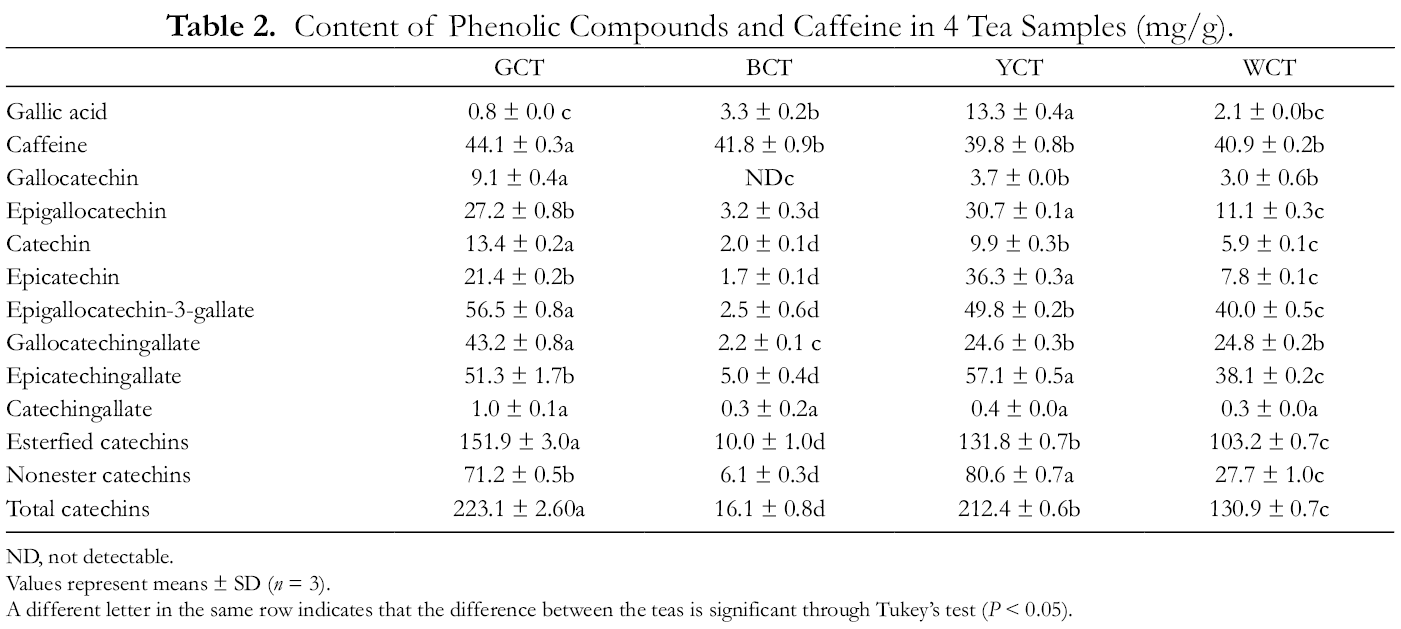
ND, not detectable.
Values represent means ± SD (n = 3).
A different letter in the same row indicates that the difference between the teas is significant through Tukey’s test (P < 0.05).
Typically, the multiplicity method is recommended to evaluate the antioxidant activities of natural antioxidants. 16 Accordingly, the antioxidant activities of 4 types of cake tea leaves were assessed using 3 independent assays (DPPH, ABTS, and FRAP).
The DPPH method is based on the theory that a hydrogen donor could also act as an antioxidant. The antioxidant activities of food and plant extracts are evaluated by the changes in absorbance induced by the reduction of the DPPH radical. 17 The antioxidant activities of GCT, BCT, YCT, and WCT were determined using the DPPH assay, as shown in Figure 2(A); the activities of all samples increased in a dose-dependent manner. At similar concentration levels, BCT showed significantly higher antioxidant activity as compared with all the other tea samples. Similar activities were found in GCT and WCT, whereas the antioxidant activity of YCT was significantly lower than those of the other cake tea leaves.

Antioxidant activities of 4 cake teas. (A) DPPH radical scavenging activity and ABTS radical scavenging capacity of 4 cake teas. (B), Ferric-reducing antioxidant power of 4 cake teas. The different letter in the same row indicate that the difference between the teas is significantly through Tukey’s test (P < 0.05). ABTS, 2,2′-azinobis-3-ethylbenzothiazoline-6-sulfonic acid; BCT, black cake tea; DPPH, 1,1-diphenyl-2-picrylhydrazyl; GCT, green cake tea; Vc, ascorbic acid; WCT, white cake tea; YCT, yellow cake tea.
The ABTS assay is a rapid and reliable method for testing the total antioxidant capacity of tea leaves. This method is useful for both hydrophilic and lipophilic systems. The ABTS radical scavenging activities of the 4 cake tea samples are shown in Figure 2(A), which signifies that antioxidant activity could decrease with an increase in the degree of fermentation. GCT and WCT showed higher antioxidant activities than BCT and YCT at equal concentration.
The FRAP assay is based on the ability of antioxidant compounds to reduce the Fe (III)-2,4,6-tris-(2-pyridyl)-S-triazine (TPTZ) complex to Fe (II)-TPTZ, which gives a blue color. 18 As shown in Figure 2(B), FRAP indicated a similar trend to ABTS but was observed to be inconsistent. Among all the samples, GCT showed the highest FRAP activity, followed by WCT, whereas BCT and YCT showed almost similar activity.
Results from epidemiological studies and laboratory experiments suggested that tea consumption could confer protection against cancer. 19 -22 Most of the studies evaluated the effects of loose tea leaves on lung cancer. Limited research evaluated the efficacy of cake tea. In this study, we evaluated the inhibitory effect of 4 types of cake tea extract on human nonsmall cell lung cancer cell lines (A549 cells). The cells were treated with 200, 400, 800, 1600, and 3200 µg/mL of each cake tea extract for 24 hours and 48 hours, respectively. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was used to depict cell viability, as shown in Figure 3. The 4 types of cake tea extract showed a significant growth inhibition effect on A549 cells in a dose-dependent manner. Moreover, the antiproliferative activity was more robust after 48 hours of treatment following which WCT showed higher anti-proliferative activity than the other cake tea leaves under the same incubation time and concentration (Figure 3B). The correlation analysis (Supporting Information Table S1) was determined between the active ingredient in tea and MTT results in 48 hours. The result with MTT had no significant correlation with a single ingredient of the tea. Therefore, we speculate that the inhibition of growth of A549 cells is not the result of a single component function of tea, but the various components in the mixture.

Antiproliferative activities of 4 cake teas. The A549 cells were exposed tea extract of differentcon centrations for 24 hours (A) and 48 hours (B), respectively, and the cell viability was measured by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay as showed in the Experimental section. The values were represented as the percent viable cells, where untreated cells were regarded as 100% viable. Data were mean ± SD, and different lower case letters (a-d) on the bars within the same concentration denote the significant differences (P < 0.05). BCT, black cake tea; GCT, green cake tea; WCT, white cake tea; YCT, yellow cake tea.
Experimental
Chemicals and Materials
Catechin, catechin gallate (CG), epicatechin (EC), ECG, EGC, EGCG, GC, GCG, and caffeine were purchased from Shanghai yuanye Biotechnology Co. Ltd. (Shanghai, China). All solvents used for high-performance liquid chromatography (HPLC) analysis were of HPLC grade and purchased from Thermo Scientific (Waltham, MA, USA). DPPH in free radical form and ABTS were purchased from Shanghai yuanye Biotechnology Co. Ltd. (Shanghai, China), ascorbic acid from MYM Biotechnology Co. Ltd. (Beijing, China), TPTZ from Mecklin Reagent Co. Ltd. (Shanghai, China), MTT from Sigma-Aldrich (St.Louis, MO, USA), and Dulbecco’s Modified Eagle’s Medium (DMEM) and fetal bovine serum (FBS) from Invitrogen-Gibco (Grand Island, NY, USA). Ultrapure water was obtained from a Milli-Q purification system (Millipore, Bedford, USA). All other chemicals and solvents used were of analytical grade.
Tea Samples and Preparation
GCT, WCT, YCT, and BCT samples were respectively processed at the tea factory of Guangdong Hongyan tea industry Co., Ltd, in Yingde, Guangdong, China. Briefly, the fresh leaves (1 bud and 2 leaves) of C. sinensis L., Yinghong No.9, were plucked from the tea garden of Guangdong Hongyan tea industry Co., Ltd in June 2016. The fresh tea leaves were then processed by using typical manufacturing approaches (Figure 1). Briefly, the fresh tea leaves were first heated at 230 °C to promote inactivation of the endogenous enzymes and rolled for 30 minutes; next, they were dried and crushed to green tea. Steam was used to moisten and soften the green tea leaves following which they were pressed into molded cake tea (GCT). The fresh tea leaves were withered at room temperature for 48 hours and then dried into white tea. Steam was used to moisten white tea leaves, which were then pressed into molded cake tea (WCT). For the manufacturing of YCT, the fresh tea leaves were first heated at 180 °C to inactivate the enzymes, followed by rolling for 30 minutes. Next, they were subjected to 70% relative humidity for 8 H that rendered a yellow color to the leaves. Then, steam was used to moisten the leaves, which were pressed into molded cake tea. For the preparation of black tea, the tea leaves were withered for 6-8 hours, rolled for 30 minutes, and allowed to undergo fermentation with endogenous enzymes at room temperature with 90% relative humidity (8 hours). Steam was used to dry the moist black tea leaves, which were pressed into molded cake tea (BCT).
All the tea samples were ground in an FW100 disintegrator (Taisite Co., Ltd, Tianjin, China) and then passed through a 40-mesh sieve. The tea samples were kept in airtight bags at −20 °C until use. For antioxidant and antiproliferative activity assays, each of the tea powders (5.0 g) was extracted with 100 mL boiling water for 30 minutes. Next, the mixture was filtered, and the residue re-extracted twice. The extracts were combined and reduced to 50 mL under vacuum at 60 °C. Finally, the condensed tea infusion was freeze-dried and stored at −20 °C for assay.
Analysis of Chemical Compositions
The water content of the 4 tea samples was measured by comparing the weight difference before and after oven heating for 3 hours at 130 °C. Soluble solid contents were determined according to the GB/T of Chinese standard (GB/T 8305–2013). According to previous reports, the content of total polyphenols in the 4 tea samples was determined using Folin-Ciocalteu reagent.
23
Briefly, 0.07 g of the tea extract was extracted twice with 5 mL of 70% (v/v) methanol, mixed with 5 mL 10% (v/v) Folin-Ciocalteu reagent, and incubated for 5 minutes followed by the addition of 7.5% (w/v) sodium carbonate (4 mL). The mixture was incubated for 1 hour in the dark. Absorbance was measured at 765 nm, and the content of polyphenols was calculated in terms of gallic acid (GA) equivalents. The content of free amino acids was determined according to the method of GB/T of Chinese standard (GB/T 8314–2013). Briefly, 0.3 g of tea extract was dissolved in 90 mL boiling distilled water. The mixture (1 mL) was mixed with 0.5 mL phosphate-buffered saline (PBS) (pH = 8.0) and 0.5 mL ninhydrin solution, which was incubated in a boiling water bath for 15 minutes, followed by absorbance measurement at 570 nm (
Analysis of Phenolic Compounds and Caffeine
The contents of various catechin monomers (GC, EGC, C, EC, EGCG, GCG, ECG, and CG), caffeine, and gallic acid were determined using HPLC (Agilent 1200, USA) based on the method described in our previous report. 26 The tea supernatant was filtered through a 0.45 µm nylon microporous membrane (Millipore), and 10 µL of each sample was injected into the HPLC, while the detection temperature was set at 28 °C. Gradient elution was performed at a flow rate of 1 mL/minute, during which 100% of solvent A (double distilled water) was utilized for 10 minutes, followed by solvent B (methanol), solvent C (0.05% phosphate acid v/v), and solvent D (acetonitrile). The wavelength was set in the range of 200-400 nm, and an external standard method was used to quantify the peak areas.
DPPH Scavenging Activity
The determination of DPPH scavenging activity was performed as previously reported, with some modification. 27 Briefly, 0.1 mM DPPH solution was prepared freshly before use. Next, 2 mL of DPPH solution was mixed with 1 mL of the sample. After 30 minutes of incubation at room temperature in the dark, the absorbance was measured at 517 nm using an ultraviolet-visible spectroscopy-752N spectrophotometer (Jinghua, China). Ascorbic acid was used as a reference compound. The half-maximal inhibitory concentration (IC50) of the DPPH methodology was calculated using the origin8.0 curve regression fitting.
ABTS•+ Scavenging Activity
ABTS was dissolved in deionized water (7 mM concentration). 28 ABTS radical cation (ABTS·+) was generated by reacting ABTS solution and potassium persulfate, followed by mixing 7 mM ABTS and 2.4 mM potassium persulfate after 16 hours of incubation at room temperature in the dark. ABTS·+ solution was diluted with deionized water, and its absorbance (0.7 ± 0.02) was measured at 734 nm. Next, 3 mL of diluted ABTS·+ solution was mixed with 100 µL of the sample at different concentrations. The reactive mixture was placed at room temperature for 6 minutes, and the absorbance was immediately recorded at 734 nm. Ascorbic acid was used as a positive control, and the IC50 of the ABTS•+ scavenging methodology was determined using the origin8.0 curve regression fitting.
Ferric Reducing Antioxidant Assay
FRAP assay was performed as previously reported, with some modification. 29 The FRAP reagent was freshly prepared before use by mixing 100 mL of sodium acetic buffer (0.3 M, pH 3.6), 10 mL of TPTZ solution (10 mM TPTZ in 40 mM hydrochloric acid) and 10 mL of ferric chloride (20 mM). Tea sample (1 μL) solution was added to 3 mL of FRAP reagent. Next, 300 µL of water was added to the mixture and incubated for 10 minutes at 37 °C. The absorbance of the mixture was recorded at 593 nm. Ferrous sulfate (FeSO4) solutions ranging from 100 μM to 1000 μM were used to perform the calibration curve. The antioxidant efficiency of the sample was expressed as mM FeSO4/g of powder, based on the standard curve.
Cell Proliferation Assay
A549 human lung cancer cells were obtained from the Shanghai Institute of Cell Biology (Shanghai, China). The cell line was cultured in DMEM medium supplemented with 10% FBS and 1% penicillin-streptomycin (Gibco, USA), which was maintained in a humidified atmosphere of 5% carbon dioxide and 95% air at 37 °C. Cell viability was determined by MTT assay, with some minor modifications. Briefly, A549 cells were seeded at a density of 5 × 103 cells per well in clear 96-well plates and incubated for 24 hours under standard culturing conditions. Next, the medium was changed, and the cells were subsequently incubated for either 24 or 48 hours in quadruplicate wells consisting of different concentrations of tea extracts from the 4 cake tea types. Control wells were prepared by the addition of culture medium. Wells containing culture medium without cells were used as blanks. After incubation, 20 µL of MTT (5 mg/mL in PBS) was added to each well and incubated for another 4 hours. The supernatants were discarded, and 150 µL dimethyl sulfoxide was added to each well to dissolve the precipitate, and the absorbance of each well was measured at 570 nm with a microplate reader (Thermo Scientific Multiskan GO, Vantaa, Finland). The results are reported as percentage proliferation (%) relative to the control.
Statistical Analysis
Each experiment was performed in triplicate, and mean values were calculated. The data were expressed as mean ± SD. The statistical differences between the groups were assessed by one-way analysis of variance performed using GraphPad Prism 6 (Graphpad Software, San Diego, CA, USA).
Conclusion
In this study, we used tea leaves from the same cultivar to develop 4 types of tea cakes using different manufacturing techniques. The main composition and the antioxidant and antiproliferative activities of the cake tea leaves were evaluated. The extent of fermentation was observed to influence the quality of the cake tea. The differences in the degree of fermentation of tea leaves determined the ultimate chemical composition of the cake teas. Using similar concentrations, GCT and WCT showed higher antioxidant activities than BCT and YCT. The antioxidant activities of cake teas decreased with an increase in their degree of fermentation (Supporting Information Table S1). The 4 types of cake tea extract showed significant growth inhibition effect on A549 cells in a dose-dependent manner. WCT showed higher antiproliferative activity than other cake tea leaves under the same incubation time and concentration. The MTT result showed that there was no significant correlation with any single ingredient of the tea (Supporting Information Table S1). Therefore, we speculate that the beneficial effects of cake tea in oxidation resistance and inhibition of tumor cell growth in vitro were the result of a combination of ingredients rather than 1 individual compound.
Supplemental Material
Table S1 - Supplemental material for Phytochemical Profiles and Bioactivities of Cake Tea Leaves Obtained From the Same Cultivar: A Comparative Analysis
Supplemental material, Table S1, for Phytochemical Profiles and Bioactivities of Cake Tea Leaves Obtained From the Same Cultivar: A Comparative Analysis by Lingli Sun, Qiuhua Li, Limin Xiang, Xingfei Lai, Wenji Zhang, Ruohong Chen, Junxi Cao and Shili Sun in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was funded by National Natural Science Foundation of China (81803236, 31800295, 81903319). Guangdong Science and Technology program (2017A070702004, 2016B090918118, 2018KJYZ002), Guangdong Basic and Applied Basic Research Foundation (2020A1515011266), Qingyuan Science and Technology Program (DZXQY021, DZXQY016), Shaoguan Science and Technology Program (2018CS11902), Ying De Science and Technology Board (JHXM2018029), and special fund for scientific innovation strategy-construction of high level Academy of Agriculture Science (R2019PY-JX004, R2018YJ-YB3002, R2016YJ-YB3003, R2018PY-QF005, R2018QD-101).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
