Abstract
Studies have shown that lobetyolin (LBT), a component of traditional Chinese herbal medicine, has many very good biological activities and functions. However, its side effects and toxicities have not been evaluated adequately. In this work, we investigated the effects of LBT in transgenic zebrafish. LBT treatments promoted angiogenesis and led to vascular morphological malformation in zebrafish embryos, although they were normal in appearance. Interestingly, our results indicated that LBT has a function of promoting nerve growth in the embryonic stage of zebrafish. We also treated the zebrafish with combretastatin A-4 (which resulted in neuronal apoptosis) and LBT simultaneously and found that LBT promoted nerve growth at higher concentrations. Taken together, our findings clearly display that LBT efficiently promotes angiogenesis, leading to vascular morphological malformation, has low toxicity, and notably promotes neuronal development in zebrafish.
Codonopsis, a traditional Chinese medicinal plant, contains various bioactive components, such as alkaloids, amino acids, phenylpropanoids, saponins, sterols, polyacetylenes, polysaccharides, and triterpenoids. 1 -8 Therefore, it has been used for promoting gastrointestinal function, enhancing immunity, nourishing lungs and spleen, improving microcirculation, helping depressurization, enhancing cognitive functions, and inhibiting tumor growth. 9 -11
Lobetyolin (LBT, Figure 1; IUPAC name: 2-[(4E,12E) −1,7-dihydroxytetradeca-4,12- dien-8,10-diyn-6-yl]oxy-6-(hydroxymethyl)oxane-3,4,5-triol), one of the most important secondary metabolites of Codonopsis pilosula, Codonopsis lanceolata, and Codonopsis radix, has demonstrated a wide range of pharmacological and biological properties, such as estrogenic, antiviral, anti-inflammatory, antitumor, antioxidant, immunoregulation, and improved sexual function activities. 12 -19 However, the side effects and possible toxicities of this compound have not been evaluated adequately.
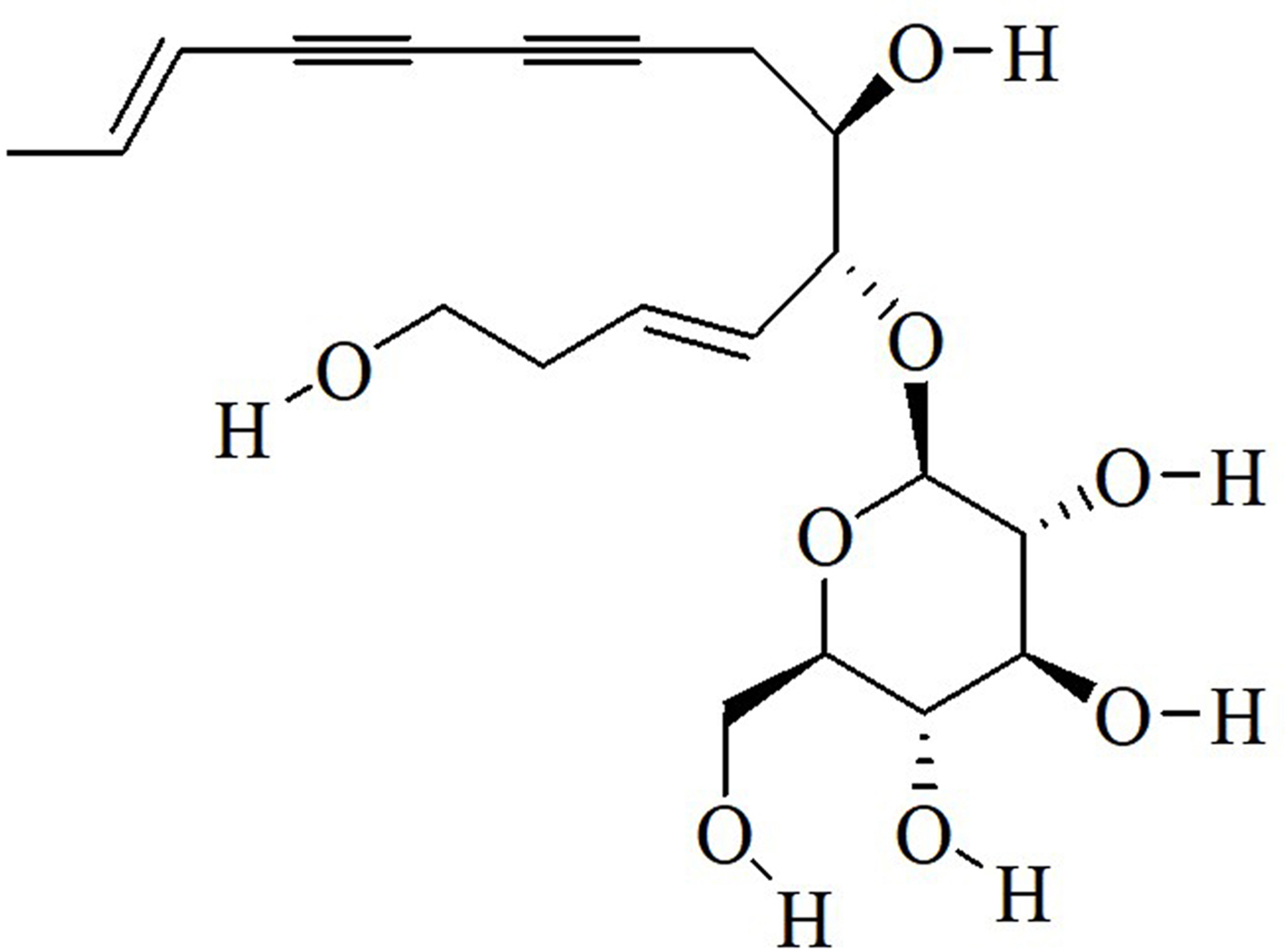
Chemical structure of Lobetyolin.
In recent years, the zebrafish model, which permits whole-animal drug screens with excellent throughput, ease of use, and low cost, has become one of the important vertebrate models for assessing drug effects. 20,21 Early development of the fish occurs rapidly outside the mother and the embryos are transparent, making their observation and manipulation quite simple. Transgenic technology provides proof that a mutant gene has been identified by rescuing the mutant phenotype. 22 The transgenic zebrafish model is suitable for multiple stages of drug discovery pipelines, from lead and target identification to lead optimization and absorption, distribution, metabolism, excretion, and toxicity studies. 23 Until now, no reports about the side effects of LBT have been reported. Therefore, more work might be needed to explore its effects. A study of LBT on the zebrafish model is so far lacking, and assessing the drug’s effect on this model will provide a greater understanding of the function of LBT on angiogenesis and its toxicities and neuroprotective effects. Combretastatin A-4 (CA-4) is a natural product possessing potent antitumor activity. 24 It is known that treatment with this drug impairs zebrafish neural development and induces neuronal apoptosis. 25 Here, CA-4 was used to investigate further the effect of LBT on neural development.
Materials and Methods
Materials
LBT (purity >98%) was purchased from Absin Biological Technology Co. (Shanghai, China), and CA-4 (purity >98%) from Univ Biomedical Technology Co. (Shanghai, China).
Zebrafish and Ethics Statements
The transgenic zebrafish lines of tg (hb9: EGFP) and tg (kdrl: mCherry), which were kindly offered by Dr Jiulin Du (Institute of Neuroscience and State Key Laboratory of Neuroscience) were maintained in the zebrafish center of Jiangsu Key Laboratory of Neuroregeneration. The tg (hb9: EGFP) zebrafish motor neuron gene hb9 is marked with EGFP. The tg (kdrl: mCherry) zebrafish vascular endothelial growth factor receptor gene kdrl is marked with mCherry. All animal experiments were carried out in accordance with the National Institutes of Health Guidelines for the care and use of laboratory animals (http://oacu.od.nih.gov/regs/index.htm) and ethically approved by the Administration Committee of Experimental Animals, Jiangsu Province, China (Approval ID: SYXK (SU) 2007–0021). Care and breeding of zebrafish embryos and adults were carried out under standard conditions in the Zebrafish Center of Nantong University, as previously described. 26 The 2 transgenic zebrafish lines tg (hb9: EGFP) and tg (kdrl: mCherry) were used as described in the previous work. 27
Zebrafish Experiments
Embryos, obtained through natural mating, were maintained at 28.5°C in E3 solution. At 6 hours post-fertilization (hpf), the zebrafish embryos were screened under an anatomical microscope to remove the morphologically abnormal individuals. About 10 healthy embryos were loaded into each well of a 96-well plate, and E3 solutions were replaced with different LBT treatment solutions. The control and treated groups were analyzed at different intervals. The tg (kdrl: mCherry) zebrafish embryos were collected for imaging at 48 hpf and the tg (hb9: EGFP) embryos at 32, 36, and 42 hpf, separately. For confocal imaging, embryos were anesthetized with E3/0.16 mg/mL tricaine/1% 1-phenyl-2-thiourea (Sigma) and embedded in 0.8% low melt agarose. Confocal imaging was performed with a Leica TCS-SP8 LSM. The analysis was performed using Imaris software.
Locomotion Analysis of Zebrafish
Different drug treatments was carried out as previously described to measure the swimming distance of zebrafish. 28 The fish were raised in a 24-well-culture plate with 1 larva in each; each well was filled with 1 mL E3 medium. The 24-well-culture plate was transferred to a Zebralab Video-Track system (Zebrabox, France) equipped with a sealed opaque plastic box that was kept insulated from the laboratory environment, an infrared filter, and a monochrome camera. After adapting for 30 minutes, the traveled distances of the zebrafish were videotaped, with every 5 minutes forming a movement distance and trajectory by the linked software (ESOvision behavioral analysis software for swimming distance analysis).
Statistical Analysis
Intersegmental vessel (ISV) and motor neuron axon length were measured with Imaris (version 7.2.3). These data were statistically analyzed with GraphPad Prism 5. All data were expressed as mean ± SEM. Statistical analysis was performed using a 1-way analysis of variance (P < 0.05).
Results
Effects of LBT Treatment on Embryonic Angiogenesis and ISV Phenotypes
To study the effects of LBT on embryonic angiogenesis in zebrafish, we used transgenic line tg (kdrl: mcherry), in which endothelial cells were labeled in mcherry. The results showed that LBT treatment with 1 µg/mL, 10 µg/mL, 100 µg/mL, 1 mg/mL, and 10 mg/mL at 48 hpf significantly inhibited ISV branching angiogenesis and led to partial vascular malformations to differing extents (Figure 1(A)–(F) and (A')–(F’)). The length of ISV in the LBT-treated group was obviously shorter than that of the control group (Figure 2(G)).
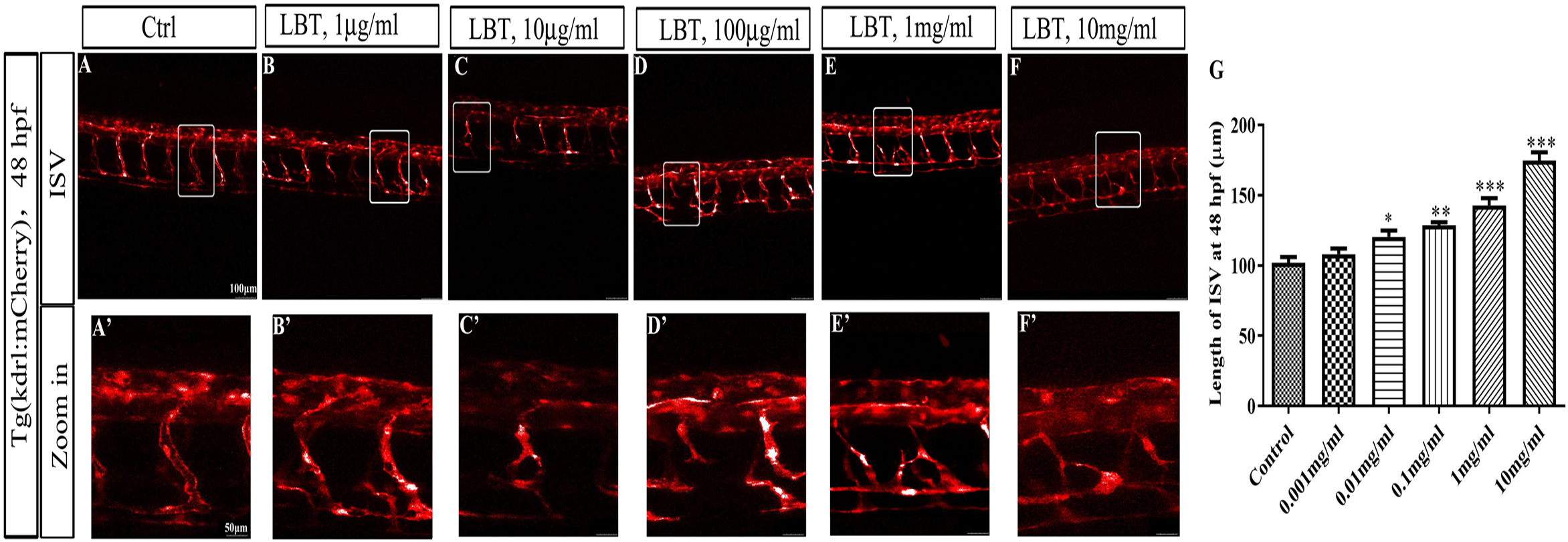
Effects of LBT treatment on vasculature in the trunk of tg (kdrl:mcherry) zebrafish embryos at 48 hpf. (A-F) ISV phenotypes of the control group and 1, 10, 100 mg/mL and 1, 10 mg/mL LBT-treated groups. Scale bar, 100 µm. (A’-F’) Zoomed in images of regions in rectangles of panel (A-F). Arrow shows vascular malformation to different extents. Scale bar, 50 µm. Statistical analyses of ISV length of each group. Data are expressed as the mean ± SEM (n = 8). ***P < 0.001 vs control group. hpf, hours postfertilization; ISV, intersegmental vessel; LBT, lobetyolin.
Effects of LBT Treatment on Zebrafish Embryos
Interestingly, the zebrafish treated at the above concentrations were normal in appearance (data not presented). In order to investigate the toxicity of LBT further, we treated zebrafish embryos with higher concentrations of LBT solution (20 and 50 mg/mL). At 6 hpf, 20 and 50 mg/mL LBT treatment resulted in all embryos appearing normal within 48 hpf (Figure 3). The LBT treatment group had very low mortality and deformity rates.
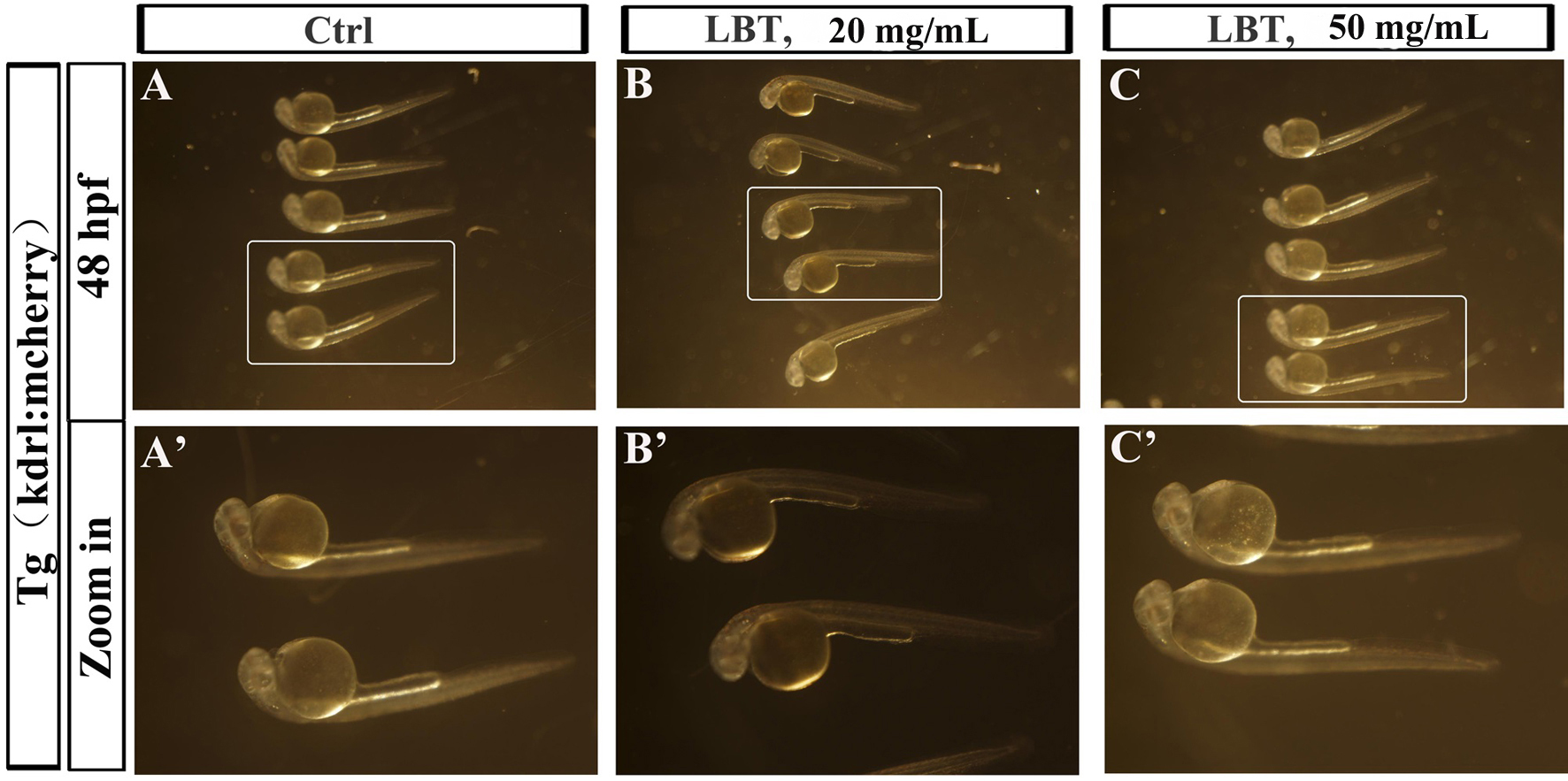
Effects of LBT treatment on appearance of phenotype of zebrafish embryos. (A-C) The bright field images of zebrafish in the control group and 20 and 50 mg/mL of LBT-treated groups at 48 hpf. (A’-C’) Zoomed in images of regions in rectangles of panel (A-C). hpf, hours postfertilization; LBT, lobetyolin.
LBT Treatment Promotes Zebrafish Neural Development
We investigated the effect of LBT treatment on motor neurons using transgenic line tg (hb9: EGFP), in which motor neurons were labeled with green fluorescence. The results showed that the axonal length of the motor neurons was significantly increased by LBT treatment compared with the normal group. LBT treatment at 1 µg/mL, 10 µg/mL, 100 µg/mL, 1 mg/mL, and 10 mg/mL at 32 hpf promoted neuronal development (Figure 4(A)–(F), (A')–(F’) and (S)). At 36 and 42 hpf, LBT treatment also promoted neuronal development at the above concentrations (Figure 4(G)-(R), (G’)-(R’) and (T)-(U)). LBT treatment of motor neurons also showed a concentration-dependent increase in axon length. Combretastatin A-4 (CA-4) was used to investigate further the effect of LBT on neural development. At 8 hpf, we treated the zebrafish embryos with CA-4 (9 ng/mL), CA-4 (9 ng/mL), and LBT 1 µg/mL, 10 µg/mL, 100 µg/mL, 1 mg/mL, and 10 mg/mL, separately. CA-4 treatment impaired zebrafish neural development at 50 hpf. Moreover, LBT at a concentration of 10 mg/mL promoted obvious neuronal development (Figure 5(A)-(D), (A’)-(D’) and (E)). We used ESOvision behavioral analysis software for determining the swimming distance of zebrafish at 72 hpf. The results revealed that LBT can improve swimming distance (Figure 6). These results further confirm that LBT promotes the development of zebrafish motor neurons.

Effects of LBT treatment on motor neurons in the tg (hb9:EGFP) embryonic zebrafish. (A-F, G-L, M-R) Phenotype of motor neuron in control group and 1, 10, 100 mg/mL and 1, 10 mg/mL LBT-treated groups at 32 hpf, 36 hpf, and 42 hpf, respectively. Scale bar, 100 µm (A′-F′, G′-L′, M′-R′). Zoomed in images of regions in dash line rectangles of panels A-F, G-L, M-R. Scale bar 50 µm (S-U). Statistical analyses of the motor neuron lengths in control group and LBT-treated groups at 32, 36, and 42 hpf, respectively. Data are expressed as the mean ± SEM (n = 8). **P < 0.01, ***P < 0.001 vs control group. CA-4, combretastatin A-4 ; hpf, hours postfertilization; LBT, lobetyolin.

Effects of LBT and CA-4 treatment on phenotype of zebrafish embryos. (A-D) Phenotype of motor neuron in control group, CA-4, 9 ng/mL group; CA-4, 9 ng/mL, and LBT 1 mg/mL combined administration group; and CA-4, 9 ng/mL, and LBT 10 mg/mL combined administration group at 50 hpf. Scale bar, 100 mm. (A’-D’) Zoomed in images of regions in dash line rectangles of panel A-D. Scale bar 50 mm. (E) Statistical analyses of the motor neuron lengths in control group, CA-4 group, CA-4, and LBT combined administration group at 50 hpf. Data are expressed as the mean ± SEM (n = 8). ### P < 0.001 vs control group;**P < 0.01 vs CA-4 group. CA-4, combretastatin A-4 ; hpf, hours postfertilization; LBT, lobetyolin.
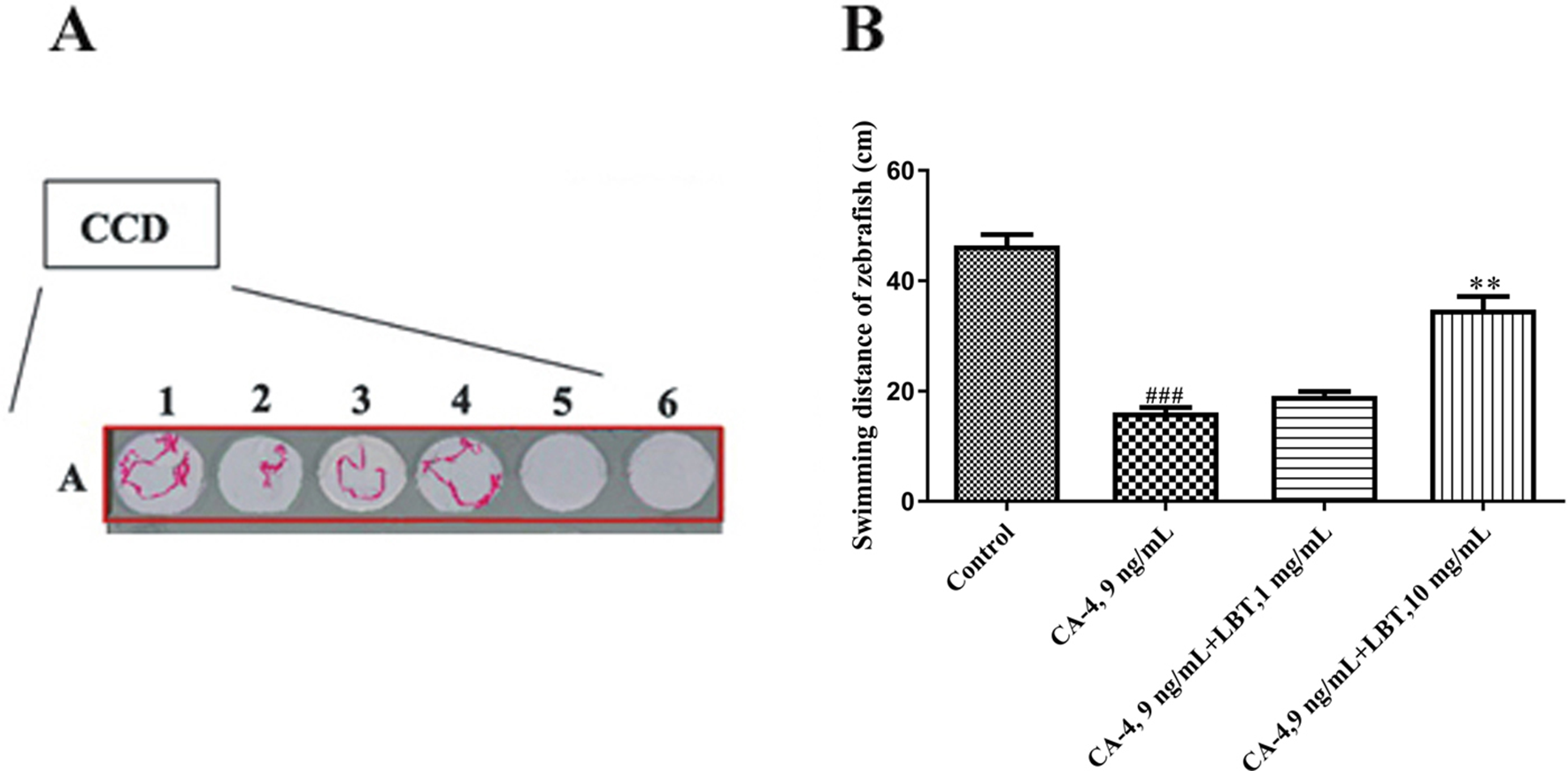
ESOvision behavioral analysis software for swimming distance of zebrafish at 72 hpf (swimming behavior assay [5 minutes]). A1, control group; A2, CA-4, 9 ng/mL group; A3, CA-4, 9 ng/mL, and LBT 1 mg/mL combined administration group; and A4, CA-4, 9 ng/mL, and LBT 10 mg/mL combined administration group (n = 8). CA-4, combretastatin A-4 ; hpf, hours postfertilization; LBT, lobetyolin.
Discussion
Comprehension of the solid tumor microenvironment has made great progress in targeting tumor treatments. 28 The tumor vasculature supplies the requisites for cellular processes and plays an important role in the invasion and eventual metastasis and extravasation of primary tumor cells. The vasculature is commonly thought to be a therapeutic target for anticancer treatment. 29 It has been reported that C. lanceolata, mainly containing LBT, exerted an antitumor effect, and the underlying mechanisms may be involved in promoting angiogenesis, leading to vascular morphological malformation. 10 Our results showed that LBT significantly promoted transgenic zebrafish intersegmental vessel branching angiogenesis, which led to partial vascular malformations to differing extents at 48 hpf (Figure 2). We speculate that the cause of vascular malformation may be due to the rapid development of zebrafish early embryos, and LBT promoting angiogenesis during the early embryonic development of the fish, which in turn leads to vascular growth disorders. More work is needed to confirm this speculation. Interestingly, we treated zebrafish embryos with higher concentrations of LBT solution (20 and 50 mg/mL) at 6 hpf to investigate the toxicity of LBT; all embryos appeared normal within 48 hpf (Figure 3). Our results also demonstrated that the LBT treatment group had very low mortality and deformity rates. These results suggest that LBT may be highly effective, with low toxicity (Figure 3), as a potential therapeutic antitumor and cardiovascular disease drug.
Studies have shown that C. lanceolata can improve cognitive enhancement. 4 This may be due to the neuroprotective effect of LBT. Our results showed that the axonal length of motor neurons was significantly increased by LBT treatment compared with the normal group. LBT treatment of motor neurons showed a concentration-dependent increase in axon length at 32, 36, and 42 hpf (Figure 4). CA-4 was also used to investigate further the effect of LBT on neural development. LBT at a concentration of 10 mg/mL promoted obvious neuronal development (Figure 5) and improved swimming distance of zebrafish (Figure 6). These results further confirm that LBT promotes the development of zebrafish motor neurons and protects them. The effect of LBT on promoting nerve growth may be related to its promotion of angiogenesis as the blood vessels provide nutrients and other substances for nerve growth and development. In addition, CA-4 caused nerve injury by inducing neuronal apoptosis. LBT treatment could reverse this neuronal damage. These results suggest that the underlying mechanism for the neuroprotective effect by LBT may be related to its antineuronal apoptosis. Considering the neuroprotective effect of LBT, it may be considered as a potential and effective therapeutic target for nervous injury-related disease.
Conclusion
In conclusion, our study has shown that LBT can promote angiogenesis and cause vascular malformations during the early embryonic development of transgenic zebrafish. Moreover, LBT showed low toxicity while having strong neuroprotective and nerve growth-promoting effects. Further work will be required to investigate the mechanism of promoting angiogenesis and neuroprotection by LBT.
Footnotes
Acknowledgements
This work was supported by the Grants from the Scientific Research Programs of Jiangsu “Six Talents Peak” High-level Talent Project (WSW-277), Jiangsu Provincial Commission of Health and Family Planning (H2017054), Nantong Science and Technology Project (JC2018020, JCZ18011, MB2019001), and Excellent Talents Financial Assistance of Jiangsu Province (QNRC2016412).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
