Abstract
Black rice bran extract (BRBE), containing various biologically active compounds, such as anthocyanin, has antioxidant activity and numerous pharmacological effects. Here, we aimed to confirm the immunostimulatory effects of BRBE in cyclophosphamide (CP)-induced immunosuppressed cells. Our results confirmed that BRBE exerted an immunostimulatory effect. In vitro, BRBE treatment enhanced cell proliferation, activity of natural killer cells and cytotoxic T lymphocytes, and production of CP-repressed cytokines, such as tumor necrosis factor-α, interferon-γ, interleukin (IL)-2, and IL-12, and immunoglobulins G and A in isolated splenocytes. Additionally, in vivo, BRBE treatment increased the number of immune cells, such as white blood cells, lymphocyte counts, mid-range absolute counts, and neutrophils in CP-induced immunosuppressed rats. Furthermore, BRBE increased the serum levels of abovementioned inflammatory cytokines and immunoglobulins in CP-induced immunosuppressed rats. In addition, BRBE protected against CP-mediated spleen and thymic tissue damage. Our findings suggest that BRBE could be potentially used as a component of functional food for immunity enhancement.
Rice is consumed by more than half of the world’s population as staple food and is majorly produced in Asia. Rice bran contains a variety of bioactive phytochemicals such as phenolic compounds, tocopherol, tocotrienol, vitamins, oryzanol, and dietary fiber. 1 -7 Recently, black rice (Oryza sativa L.) has become increasingly popular and is being widely used in East Asian countries, including Korea. 8,9 Black rice bran (BRB), a by-product of the rice flour milling process, has recently attracted attention for potential applications as a functional food because it contains phenolic compounds such as tocopherol and exhibits antioxidant activity. 6,10 It is also valuable because it contains anthocyanins, such as cyanidin-3-glucoside and peonidin-3-glucoside. 11,12 Black rice bran extract (BRBE) has been reported to show anticarcinogenic, antioxidative, antimutagenic, hypoglycemic, hypolipidemic, and anti-inflammatory effects. 13 -16 However, immunostimulatory effects have not yet been reported.
The immune system is a very complex and sophisticated network of immune molecules, immune cells, tissues, and organs to protect the body from external invasion of bacteria, parasites, viruses, and toxins. 17 The series of steps that act as a body defense against these infections are called immune reactions, and the development of cells involved in immune reactions occur in organs such as the thymus and spleen. 18,19
Immune responses are highly complex biological responses involving macrophages, dendritic cells, and various immune cells, including lymphocytes. 20 Lymphocytes are white blood cells (WBCs) comprising T cells, B cells, and natural killer (NK) cells. Natural killer cells belong to the innate immune system and play a major role in defending the host from cancer cells, bacteria, and virus-infected cells. 17 T cells and secreted cytokines are associated with adaptive or cell-mediated immune responses, while B cells and antibodies are key factors in the humoral immune response. 21 Immune system imbalances are associated with autoimmune diseases and diseases caused by inflammatory responses in various organs. 22
Cyclophosphamide (CP), a commonly used alkylating agent in chemotherapy, has a broad spectrum of activities such as bone marrow suppression, immunosuppression, and cytotoxic effects in a variety of diseases. 23 -25 In the previous studies, high-dose treatment of CP was reported to reduce weight, spleen and thymus weight, total leukocyte count, differential leukocyte, antibodies, bone marrow cells, proliferation of T cells and B cells, and NK cell activity. 26 In particular, administration of CP has been reported to cause immunosuppression by a change in Th1/Th2 ratio. 27 In addition, in multiple studies, the major factor contributing to the immunosuppressive effect of CP was reduced proliferation of T cells and the decreased secretion of Th1 cell cytokines (tumor necrosis factor [TNF]-α, interferon [IFN]-γ, interleukin [IL]-2, and IL-12) and Th2 cell cytokines (IL-4, IL-6, and IL-10). 28,29
Recently, the immunomodulatory properties of natural foods and food products have been studied to develop immune enhancers as a component of functional foods, with a wide range of therapeutic properties and relatively low toxicity. 30 -32 In this study, we investigated the immunostimulatory effect of BRBE in isolated splenocytes and CP-induced immunosuppressed rats.
Experiment 1
In experiment 1, to examine any cytotoxic effects of BRBE on splenocytes, cells were incubated with different concentrations of BRBE (0, 0.01, 0.05, 0.1, 0.5, 1, 3, 5, 10, 30, 50, and 100 µg/mL) for 24 hours. Cells did not show a change of cell viability at concentrations bellow 50 µg/mL of BRBE (Figure 1(A)). To investigate cell proliferation, splenocytes were incubated with different concentrations of BRBE (0, 0.01, 0.05, 0.1, 0.5, 1, 3, 5, 10, 30, and 50 µg/mL) and/or CP (1600 μg/mL) for 24 hours. Black rice bran extract increased the cell proliferation in a dose-dependent manner (Figure 1(B)), suggesting that BRBE could recover from the reduced cell proliferation induced by CP.

Effect of black rice bran extract on the viability and cyclophosphamide-stimulated cell proliferation in splenocytes. (A) Isolated splenocytes were seeded into a 96-well plate with black rice bran extract (0, 0.01, 0.05, 0.1, 0.5, 1, 3, 5, 10, 30, 50, and 100 µg/mL). (B) Splenocytes were treated with black rice bran extract (0, 0.01, 0.05, 0.1, 0.5, 1, 3, 5, 10, 30, and 50 µg/mL) or/and cyclophosphamide (1600 µg/mL) for 24 hours in a 5% CO2 incubator, and then cell viability rates were measured with WST-1 Assay Kit. a- hBars labeled with different superscripts indicate significant differences (P < .05 vs control). Data are presented as means ± standard errors (n = 3).
Experiment 2
In experiment 2, we tested the effect of BRBE on CP-induced reduced expression of cytokine and immunoglobulin (Ig) in splenocytes. Cells were incubated with BRBE (0, 0.5, 1, 3, 5, 10, and 30 µg/mL) and CP (1600 μg/mL) for 24 hours. Black rice bran extract increased cytokine levels in a dose-dependent manner (Figure 2). Our results showed that BRBE treatment suppressed the CP-mediated reduction of levels of TNF-α, IFN-γ, IL-2, and IL-12. Additionally, to confirm the mechanism underlying BRBE immunoregulatory activity, levels of IgA and IgG were determined by enzyme-linked immunosorbent assay (ELISA) in CP-treated splenocytes. As shown in Figure 3, the levels of IgA and IgG were decreased by CP. However, BRBE-treated splenocytes showed increased levels of IgG and IgA, suggesting that BRBE improved the humoral immunity in the CP-treated splenocytes.

Effect of black rice bran extract on cytokine levels in splenocytes. Isolated splenocytes were seeded into 96-well plates, followed by treatment with black rice bran extract (0, 0.5, 1, 3, 5, 10, and 30 µg/mL) and/or cyclophosphamide (1600 µg/mL) and incubated for 24 hours in a 5% CO2 incubator. Levels of cytokines (tumor necrosis factor-α, interferon-γ, interleukin-2, and interleukin-12) of culture medium were analyzed using enzyme-linked immunosorbent assay kits. a-eBars labeled with different superscripts have significantly different values (P < .05 vs control). Data are presented as means ± standard errors (n = 3).

Effect of black rice bran extract on immunoglobulin in splenocytes. Isolated splenocytes were seeded into 96-well plates, followed by treatment with black rice bran extract (0, 0.5, 1, 3, 5, 10, and 30 µg/mL) and/or cyclophosphamide (1600 µg/mL) and incubated for 24 hours in a 5% CO2 incubator. Levels of immunoglobulins A and G of culture medium were analyzed using enzyme-linked immunosorbent assay kits. a-dBars labeled with different superscripts have significantly different values (P < .05 vs control). Data are presented as means ± standard errors (n = 3).
Experiment 3
In experiment 3, we tested the effects of BRBE on NK and cytotoxic T lymphocyte (CTL) cytotoxicity in CP-treated splenocytes. Natural killer cells and CTL play important roles in defense against virus-infected cells and tumor cells. 33 Therefore, we confirmed the effects of BRBE on NK cell and CTL activity. Splenocyte cytotoxicity was tested against NK cell-sensitive tumor cells (AR42J) and CTL-sensitive tumor cells (HL-60). As shown in Figure 4, BRBE treatment resulted in increased NK cell and CTL activity in a dose-dependent manner. The results suggested that BRBE could improve the cell immune response in splenocytes.

Effect of black rice bran extract on splenic natural killer cell and cytotoxic T lymphocyte activity in splenocytes. Natural killer cell and cytotoxic T lymphocyte activity assayed by the WST-1 assay as described in the text. Isolated splenocytes were cocultured with target cells (AR42J) for natural killer cell activity and target cells (HL-60) for cytotoxic T lymphocyte activity in 96-well plates, followed by treatment with black rice bran extract (0, 0.1, 0.5, 1, 3, 5, and 10 µg/mL) and incubated for 24 hours in a 5% CO2 incubator with a ratio of effector to target cells of 25:1. The natural killer cell and cytotoxic T lymphocyte activities were calculated as the survival rate of AR42J or HL-60 compared to that of the control group. a-eBars labeled with different superscripts have significantly different values (P < .05 vs control). Data are presented as means ± standard errors (n = 3).
Experiment 4
In experiment 4, we confirmed the effect of BRBE in rats. First, CP is known to reduce viability of immune cells. Black rice bran extract increased the number of WBCs, lymphocytes, MID (monocyte, eosinophil, basophil), and neutrophils in CP-induced immunosuppressed rats (Figure 5). Next, we confirmed the effects of BRBE on the serum levels of immune-related cytokines such as TNF-α, IFN-γ, IL-2, and IL-12 in BRBE-treated rats and/or CP-induced immunosuppressed rats (Figure 6). The serum levels of cytokines were lower in CP-treated immunosuppressed rats compared with that in normal rats (saline-treated). However, BRBE-treated groups showed a slight increase in plasma levels of TNF-α, IL-2, IFN-γ, and IL-12 compared with the non-BRBE treated group. These results indicate that BRBE improved the reduced cytokine production in CP-treated rats. In addition, we confirmed the effects of BRBE on serum levels of IgA and IgG in CP-induced immunosuppressed rats. In order to confirm the antibody production in CP-induced immunosuppressed rats, the levels of IgA and IgG in serum were determined by ELISA. As shown in Figure 7, the IgA and IgG serum levels were significantly decreased by CP treatment. However, additional treatment of BRBE showed increased serum levels of IgA and IgG, suggesting that BRBE improved the humoral immunity.
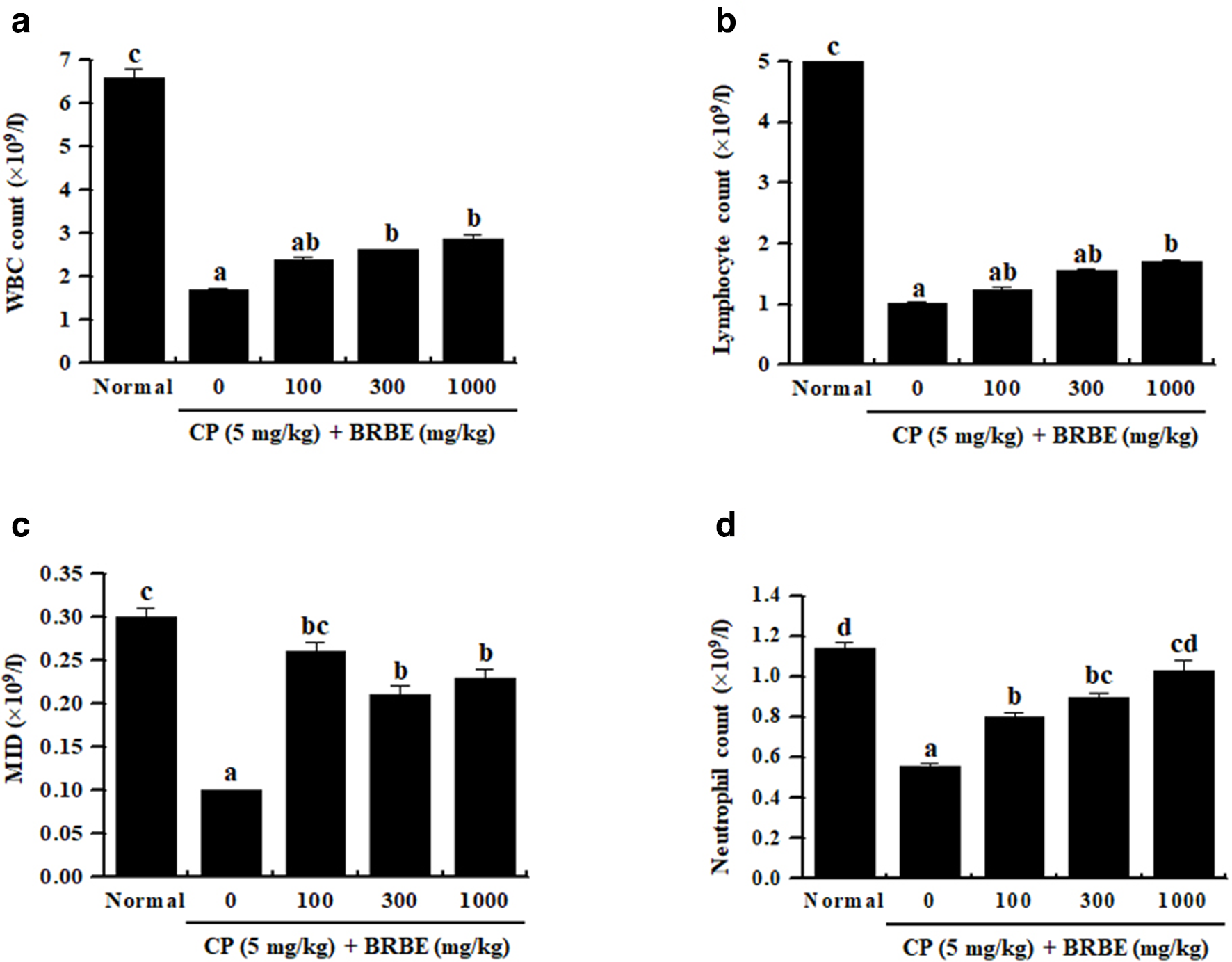
Effects of black rice bran extract on inflammatory cells counts in the whole blood of cyclophosphamide-induced immunosuppression rats. Sprague Dawley rats were administrated with saline, cyclophosphamide (5 mg/kg), and black rice bran extract (0, 100, 300, or 1000 mg/kg) once daily for 28 days, after which whole blood samples were collected. The levels of inflammatory cells (white blood cells, lymphocytes, MID, and neutrophils) in the blood samples were determined using a Hemavet 950 system. a-dBars labeled with different superscripts have significantly different values (P < .05 vs control.). Data are presented as means ± standard errors (n = 7).

Effect of black rice bran extract on the levels of cytokines in the serum of cyclophosphamide-induced immunosuppression rats. Sprague Dawley rats were administrated with saline, cyclophosphamide (5 mg/kg), and black rice bran extract (0, 100, 300, or 1000 mg/kg) once daily for 28 days, after which serum levels of tumor necrosis factor-α, interferon-γ, interleukin-2, and interleukin-12 were quantified using enzyme-linked immunosorbent assay kits. a-cBars labeled with different superscripts have significantly different values (P < .05 vs control). Data are presented as means ± standard errors (n = 7).

Effect of black rice bran extract on the plasma levels of immunoglobulin in the serum of cyclophosphamide-induced immunosuppression rats. Sprague Dawley rats were administrated with saline, cyclophosphamide (5 mg/kg), and black rice bran extract (0, 100, 300, or 1000 mg/kg) once daily for 28 days, after which serum levels of immunoglobulins A and G were quantified using enzyme-linked immunosorbent assay kits. a-dBars labeled with different superscripts have significantly different values (P < .05 vs control). Data are presented as means ± standard errors (n = 7).
Finally, we confirmed the effect of BRBE on spleen and thymus morphology in immunosuppressed rats. The morphological changes in the spleen and thymus were investigated after BRBE treatment using hematoxylin and eosin (H&E) staining. Spleen confirmed that 100, 300, and 1000 mg/kg of BRBE gradually promoted the spleen cell multiplication of white pulp compared with the CP-treated rats (Figure 8(A)). In addition, CP treatment resulted in deformation, nuclear condensation, and fragmentation of the thymus tissue, which could be recovered upon BRBE treatment, which showed reduced separation from the cortex to the surrounding tissue of the thymus (Figure 8(B)). This result suggested that BRBE stimulated innate and adaptive immunity by ameliorating the CP-mediated spleen and thymus tissue damage.

Effect of black rice bran extract on immunity-associated spleen and thymus damage in cyclophosphamide-induced immunosuppression rats. Sprague Dawley rats were administrated with saline, cyclophosphamide (5 mg/kg), and black rice bran extract (0, 100, 300, or 1000 mg/kg) once daily for 28 days, after which damage of spleen (A) and thymus (B) was analyzed histologically. Representative images of the sectioned spleens and thymus of (a) normal rats (saline treatment), (b) control rats (treated with only cyclophosphamide), and (c–e) cyclophosphamide- and black rice bran extract-treated rats [(c) 100, (d) 300, or (e) 1000 mg/kg black rice bran extract]. Scale bar = 100 µm. CV, central vein; LN, lymph nodule; MZ, marginal zone; RP, red pulp; C, cortex; M, medulla.
Recent studies have been actively conducted to confirm the immunostimulatory effects of natural products and food ingredients. 33 -undefined In the current study, we demonstrated that BRBE ameliorated the CP-mediated immunosuppression in isolated splenocytes and immunosuppressed animal models.
Cyclophosphamide is generally used as an important chemotherapeutic drug in tumor therapy; however, it is known to sometimes cause side effects such as bone marrow suppression, immunosuppression, and oxidative stress. 37 Immunosuppression by CP was reported to significantly reduce immune cells, and inhibit spleen NK cell and CTL activity. 38,39 In addition, CP decreases the levels of serum cytokines (IL-2, IFN-γ, and IL-10) and immunoglobulins (IgA, IgM, and IgG). 39,40 Cyclophosphamide damages the spleen and thymus, which are important for the immune response. 41
Splenic cells are composed of various immune cells such as T and B cells, macrophages, and dendritic cells. 42 Splenocyte proliferation ultimately enhances immunity through increased cell-mediated immune response by the expression of cytokines. 43,44 In our results, BRBE increased the immune cell survival rate and suppressed the CP-mediated loss of cell viability (Figures 1(b) and 5). The cytokines expressed by various immune cells play an important role in immune responses such as host defense against bacterial infection, cell survival, apoptotic lymphocyte differentiation, and inflammation regulation. 45 -47 Several types of cytokines secreted by Th1 and Th2 cells are the determinants of cell function. Th1 cells secrete IL-2, TNF-α, and IFN-γ, which are important cytokines in the humoral immune response, 48 -50 while Th2 cells mainly secrete IL-4, IL-6, and IL-10 to induce cell-mediated immune responses. 51 Our study indicated that BRBE plays an important role in inducing Th1 cytokine secretion and subsequent immune response in the immunosuppressed state, confirming the immunostimulatory effect of BRBE (Figures 2 and 6). Also, IgG, IgA, and IgM are major Igs that play a role in the opsonization, complement activation, and neutralization of bacteria or viruses and toxins. 52 Our results confirm that BRBE increases the levels of IgG and IgA in CP-treated splenocytes, suggesting that BRBE improves the humoral immune response.
Natural killer cells and CTLs play a major role in the initial immune response stage to remove foreign and abnormal cells. 53,54 Therefore, measurement of NK cell and CTL activity is a useful method for assessing the cellular immune response of the host. 55 Our results showed that spleen NK cell cytotoxic activity was significantly increased by BRBE treatment (Figure 3).
The immune system is weakened by birth, aging, disease, or a variety of other causes. The number and type of active immune cells is important for the regulation of the immune response. 45 -47 Our results showed that CP-induced immunosuppression reduced WBC, lymphocyte, MID (monocytes, eosinophils, eukaryotes), and neutrophils, consistent with other reports. 56,57 However, BRBE administration could significantly restore the number of immune cells in CP-treated rats. In addition, it was confirmed that the CP-induced damage of spleen and thymus was restored to the normal state by BRBE (Figure 6). These results suggest that BRBE improves immune response by restoring CP-induced impaired immune system function.
In conclusion, the results of this study confirm that BRBE stimulates innate and adaptive immune processes by increasing levels of cytokines, antibodies, NK CTL activity, and immune cell production under immunosuppressive conditions. These results suggest that BRBE could be used as a material for the development of functional food for immunity enhancement.
Experimental
Preparation of Black Rice Bran
Black rice bran extract was manufactured by Jarm Agricultural Co. Ltd (Jindo, Jeollanam-do, Korea). To prepare BRBE, 100 g BRB was extracted in an electric boiling pot for 1 hour with 2000 mL of distilled water at 70 °C temperature, and then filtered through a 55-µm bag filter. The solution was evaporated under natural circulation at 55 °C and sterilized at 90 °C for 10 minutes using a concentrator. After sterilization, the extract was spray dried under the conditions of electric heater at 227 °C, inlet temp 180 °C to 200 °C, outlet temp 80 °C to 100 °C, nozzle press 40 to 70 bar, and then used for each experiment. The physiological activity of BRB is attributed to the active compound, anthocyanins, such as cyanidin-3-glucoside and peonidin-3-glucoside. We found that BRBE contained primarily cyanidin-3-glucoside at a concentration of 4.30 mg/g. Additionally, we confirmed that BBRE contained a concentration of total anthocyanin of 12.6 mg/g.
Animals
Animal experiments were performed as previously described. 58 Briefly, 5-week male Sprague Dawley (SD) (n = 39) rats were purchased from Samtaco Inc. (Osan, Gyeonggi-do, Korea) and adapted to the following conditions for 7 days: 12 hours light/12 hours dark cycle; temperature, 23 °C ± 1 °C; humidity, 50% ± 5%; and illumination, 150 to 300 lux. The animals were allowed ad libitum access to food (Purina diet; Purina Korea, Seongnam, Gyeonggi-do, Korea) and water. Splenocytes were collected from 4 rats for the study. The remaining 35 rats were then randomly assigned to 5 groups (7 rats per group). The protocols used for the animal studies were approved by the Committee on Care and Use of Laboratory Animals of Wonkwang University (Iksan, Jeollabuk-do, Korea; approval no. WKU18-17).
Cell Culture
Cells were cultured as described previously. 58 Briefly, the spleen of an 8-week SD rat was aseptically dissected to obtain splenocytes. The spleen was gently pressed with forceps and then forced through a 70-µm cell strainer (SPL Life Sciences, Pocheon-si, Gyeonggi-do, Korea). The cells were collected and washed 3 times in Roswell Park Memorial Institute (RPMI)-1640 (Invitrogen, Carlsbad, CA, United States) by centrifugation (3000 rpm for 3 minutes at 4 °C). Next, the cells were treated with red blood cell lysis buffer (Sigma-Aldrich, St. Louis, MO, United States). Isolated splenocytes were maintained in RPMI-1640 containing 10% fetal bovine serum (FBS) and 1% antibiotics (penicillin and streptomycin) (Invitrogen) in a 5% CO2 incubator.
Cell Viability
Cell viability was assessed as previously described 58 using a WST-1 Assay Kit (ITSBio, Seoul, Gyeonggi-do, Korea), according to the manufacturer’s instructions. Briefly, splenocytes (2 × 105 cells/well) were seeded into 96-well plates and incubated at 37 °C for 4 hours to allow for cell stabilization. Next, the cells were treated with BRBE (indicated doses in figure) and CP (1600 μg/mL) and incubated for 24 hours in a 5% CO2 incubator. Each experiment was performed in triplicate. Splenocyte viability rate was assessed using the WST-1 Assay Kit and a Sunrise ELISA plate reader (Tecan, Männedorf, Switzerland).
Natural Killer Cell Activity Assay
Natural killer cell activity was examined as previously described. 58 Briefly, AR42J cells obtained from the American Type Culture Collection (ATCC, Manassas, United States) were used as target cells for NK cell activity assay, and splenocytes were isolated from control or BRBE-treated groups for use as effector cells. Splenocytes were cocultured with AR42J cells in 96-well plates at a ratio of effector cells to target cells (25:1) and cultured in a 5% CO2 incubator at 37 °C for 24 hours. AR42J viability rate was assessed using the WST-1 Assay Kit and Sunrise ELISA plate reader (Tecan, Männedorf, Switzerland). The NK cell activity was calculated as the survival rate of AR42J compared with that of the control group.
Assessment of Cytotoxic T Lymphocyte Activity
This assay was performed as previously described 58 using the WST-1 assay kit method as described above. Tumor (HL-60) cells obtained from ATCC (Manassas, United States) were used as target cells to assess CTL activity; the ratio of effector cells to target cells was 25:1. The CTL activity was calculated as the survival rate of HL-60 compared with that of the control group.
Immunoglobulin A, Immunoglobulin G, and Cytokine Levels in Splenocytes
This evaluation was performed as previously described. 58 Briefly, splenocytes (2 × 105 cells/well) were seeded into 96-well plates with RPMI-1640 containing 10% FBS and 1% antibiotics (growth media), after which BRBE (0, 0.5, 1, 3, 5, 10, and 30 µg/mL) and CP (1600 μg/mL) were added to the wells, and then cells were incubated for 24 hours in a 5% CO2 incubator. Each experiment was performed in triplicate. The levels of IgA, IgG, TNF-α, IFN-γ, IL-2, and IL-12 in the culture medium from each well were then measured using Cytokine Activation Analysis Kits (R&D Systems, Minneapolis, MN, United States), according to the manufacturer’s instructions. The results were measured using an ELISA reader.
Complete Blood Count and Immunoglobulin A, Immunoglobulin G, Cytokines Analyses
These analyses were conducted as previously described. 58 Briefly, SD rats were orally administered BRBE (0, 100, 300, or 1000 mg/kg/day) and CP (5 mg/kg, once per day) for 28 days. Rats treated with saline were used as a control group. After the final administration of the various drugs, the rats were weighed and anesthetized via intraperitoneal injection of 2,2,2-tribromoethanol (Sigma-Aldrich). Whole blood was collected through the abdominal vena cava into ethylenediaminetetraacetic acid microcentrifuge tubes. Next, the rats were sacrificed by brief exposure to 100% CO2, followed by cervical dislocation. The numbers of WBCs, lymphocytes, and neutrophils in each whole blood sample were measured using a Hemavet 950 system (Drew Scientific Group, Dallas, TX, United States). In addition, mid-range absolute counts (MID), which generally include monocytes, eosinophils, and basophils, were determined. The plasma levels of IgA, IgG, TNF-α, IFN-γ, IL-2, and IL-12 were quantified using ELISA kits (R&D Systems), according to the manufacturer’s instructions.
Thymus and Spleen Histochemical Analysis
This histochemical analysis was performed as previously described. 58 Briefly, after the animals were sacrificed, their organs (thymus and spleen) were removed, weighed, and fixed in 10% neutral buffered formalin. The organs were then processed for embedding in paraffin, after which they were sectioned into 4- to 7-μm-thick slices using a microtome (Thermo Scientific, Waltham, MA, United States). The sectioned tissues were then stained with H&E. Tissue damage was assessed under an optical microscope (Olympus, Fukuoka, Japan).
Statistical Analysis
Results were analyzed by one-way analysis of variance (ANOVA) and Duncan test multiple comparisons using SAS software (version 9.3; SAS Institute Inc., Cary, NC, United States). P values <.05 were considered statistically significant.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by “Food Functionality Evaluation program” under the Ministry of Agriculture, Food and Rural Affairs and National Research Council of Science & Technology (NST) grant by the Korea Government (MSIP) (No. CAP-16-07-KIOM).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
