Abstract
Introduction
Ganoderma lucidum (Reishi) has been the subject of extensive research into its numerous physiological activities, leading to the development of numerous health food products. The active ingredients in Ganoderma lucidum include triterpenes and polysaccharides. Of these, the water-extractable polysaccharides have a variety of pharmacological properties, including immunomodulatory, anti-metabolic syndrome, anti-inflammatory, and anticancer effects.1–5 The active polysaccharide present in Ganoderma lucidum has a unique chemical structure comprising β-glucan with various β-glycosidic bonds and a branching point of β-(1 → 6). Ganoderma lucidum was reported as a water-soluble polysaccharide that has an antitumor effect with a molecular weight of 40 kDa from the fruiting bodies. 6 They proposed that the underlying structure of this polysaccharide's antitumor activity is a branched glucan core, which involves (1 → 3)-β-, (1 → 4)-β- and (1 → 6)-β- linkages. The biochemical and clinical studies that have been conducted have demonstrated that the polysaccharides present in Ganoderma lucidum, with particular emphasis on β-1,3;1,6 glucan, have a significant effect on the modulation of the immune system.7,8
The observed biological activity of this β-D-glucan is dependent on its specific β-structure. The chemical structure of the polysaccharide is dependent upon the methodology employed during the manufacturing process and the specific culture medium used. 3 In controlled environments, Ganoderma lucidum can be cultivated in greenhouses or bioreactors for metabolite production. 9 Submerged cultivation of mushroom mycelium has been demonstrated to be an effective method for producing extracellular bioactive metabolites. 10 Ganoderma lucidum can be cultivated using biotechnology to produce active polysaccharides. Although polysaccharides can be obtained from the fruiting bodies of Ganoderma lucidum, it should be noted that this is not the only source of these compounds. Exopolysaccharides are polysaccharides produced by the culture broth during the submerged fermentation of mushroom mycelium.11,12 Exopolysaccharides are more widely used than polysaccharides obtained from mushroom mycelium and fruiting bodies due to their ability to produce more polysaccharides in a shorter period of producing time and their reduced susceptibility to contamination. 13
Various commercially available products are derived from Ganoderma lucidum, including capsules, powders, dietary supplements, and teas. 14 The physiological activity of pure active ingredients has been evaluated in most studies. In practice, formulation technology is required to produce products that are easy to ingest and are suitable for use in the health food industry. However, activity is usually reduced after the addition of an excipient or formulation. 15 The composition of many commercial products derived from traditional techniques of Ganoderma lucidum cultivation is complicated to standardize effectively, which makes it challenging to assess the results of stabilizing the extracts into products. In this study, the in vitro metabolic syndrome, tumor proliferation inhibition, and anti-inflammatory activities of Ganoderma lucidum polysaccharides produced by an innovative metabolic synthesis technology and the powder produced by the addition of excipients were investigated simultaneously.
Results and Discussion
The active polysaccharides of Ganoderma lucidum (GAP) were processed using metabolic synthesis technology and contained about 95.9% carbohydrates and 51.7 ± 1.0% water-soluble polysaccharides (Table 1). It was also found that no lipids were detected in the GAP and the protein content was 0.4%. As shown in Table 1, Ganoderma polysaccharides contain glucans different from those of grains, mainly β-1,3;1,6-glucan. This study analyzed β-1,3;1,6-glucan content of fermentation broths from a strain-optimized cultivation system and found it to be 73%.
Composition of Ganoderma lucidum Active Polysaccharides.

Bioactive polysaccharides are a new strategy for treating metabolic disorders or syndromes, 3 as most of the polysaccharides that are effective in anti-metabolic syndrome are β-glucan with 1,3, 1,6, or 1,3; 1,4 linkage. 16 Among the two specimens for the study, both FI and SI had inhibitory activity for ACE, α-amylase, and lipase in a dosed manner (Figure 1). The findings indicated that GAP, which is abundant in β-1,3;1,6-glucan, exerts an inhibitory effect on metabolic enzyme activities. Figure 1 also illustrates that the sterilization process did not disrupt the activity of the enzymes involved in metabolic syndrome when comparing the direct freeze-dried FI and sterilized SI. Ganoderma lucidum and its components have been demonstrated to possess therapeutic potential for Alzheimer's disease and may slow the progression of dementia in several ways. 17 The postulated mechanism of action includes the inhibition of acetylcholinesterase (ACE). The results of Figure 1(C) showed that FI and SI could inhibit ACE, suggesting that the GAP produced by the present technique has the potential to prevent dementia in addition to the hypotensive effect. As shown in Figure 2, the inhibitory activities of all three enzymes were lower than those of FI and SI at the same concentration when added fiber as an excipient to produce MSD and MSI powdered products. This suggests that the inhibitory activity of metabolic enzymes is mainly derived from GAPs. In any case, the results in Figures 1 and 2 indirectly indicate that GAP produced by the metabolic synthesis technology is effective in reducing blood pressure, blood glucose, and blood lipids.
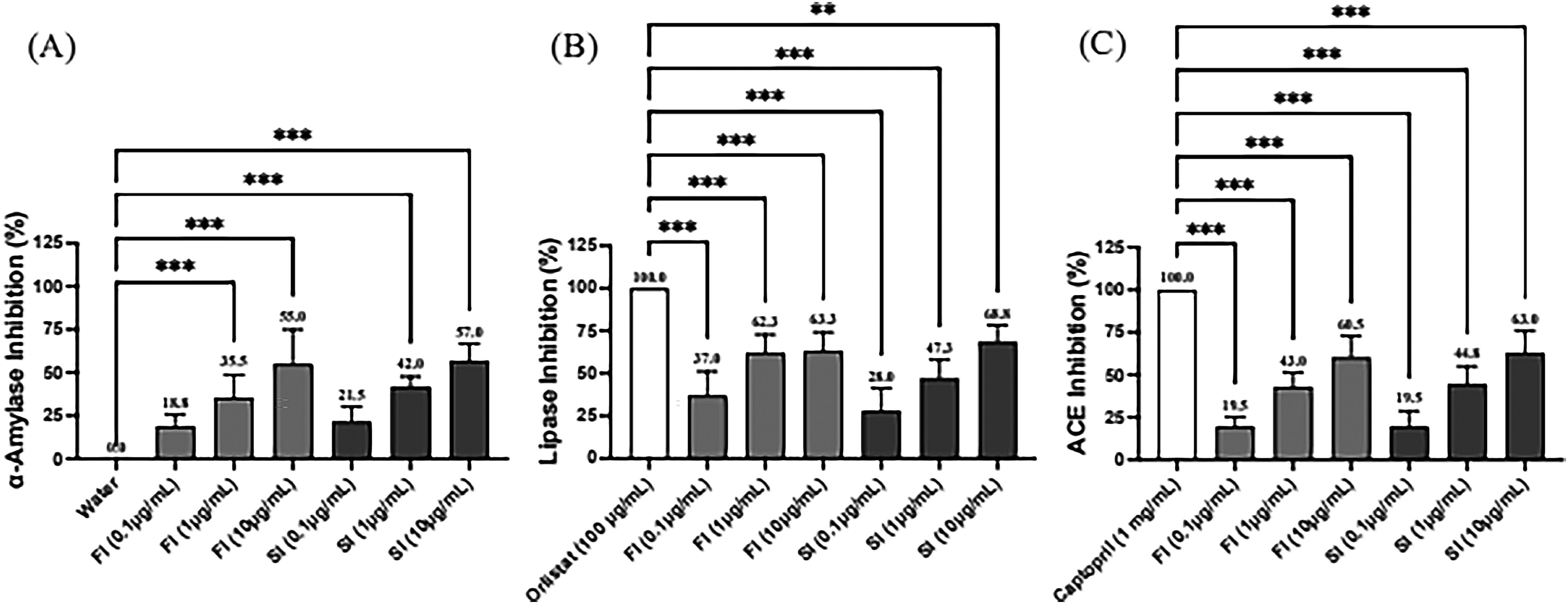
Enzymatic inhibition by Ganoderma lucidum polysaccharides (GAP) across varied concentrations. (A)Data showed the dose-dependent inhibition of α-amylase by freeze-dried (FI) and sterilized (SI) forms of GAP at concentrations of 0.1 μg/mL and 1 μg/mL compared to water control. The results demonstrate significant inhibition, particularly at the higher concentration. (B) The inhibitory effects of FI and SI on lipase are compared against Orlistat, a standard lipase inhibitor. FI exhibits stronger inhibition across both tested concentrations, underscoring its potent lipase inhibitory activity. (C) The inhibitory impact of FI and SI on the angiotensin-converting enzyme (ACE), benchmarked against Captopril. The data were presented as mean ± SD of three experiments. ***P < .001.
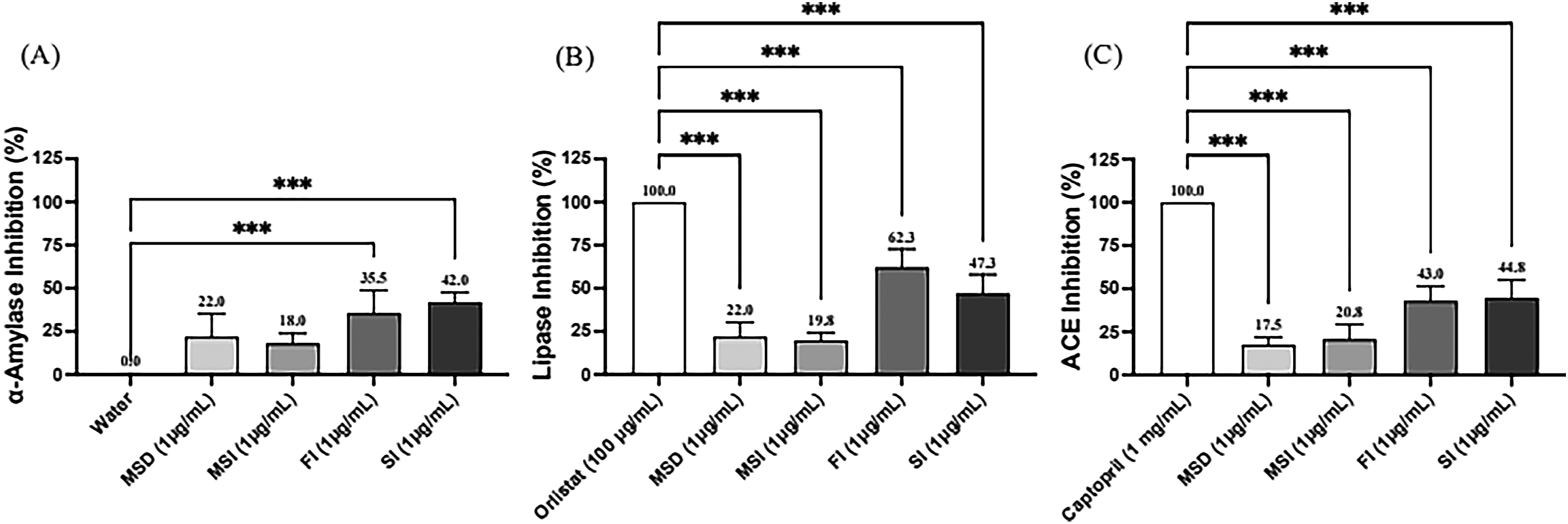
Comparison of the inhibitory activities of the same concentrations of GAP (FI and SI) and powdered products (MSD, MSI) on the three metabolic enzymes. (A) Data showed the dose-dependent inhibition of α-amylase by GAP and powered products at concentrations of 1 μg/mL compared to a water control. (B) The inhibitory effects of GAP and powered products on lipase are compared against Orlistat, a standard lipase inhibitor. (C) The inhibitory impact of GAP and powered products on the angiotensin-converting enzyme (ACE), benchmarked against Captopril. (A) α-amylase, (B) lipase and (C) ACE. The data were presented as mean ± SD of three experiments. ***P < .001.
β-1,3;1,6-glucan has been shown to have tumor suppressor or anticancer activity.18,19 The tested SW620 and HCT116 colorectal cancer cell lines were found to exhibit a significant inhibition of proliferation when subjected to FI, as demonstrated in Figure 3. It is interesting to note that FI at a low concentration of 0.1 μg/mL still had the same antitumor effect. However, the sterilized SI was unable to inhibit the proliferation of SW620 cells and only inhibited the proliferation of HCT116 cells. This may be attributed to the fact that the elevated temperatures utilized in the sterilization process result in the breakdown or deformation of certain glucans, thereby impairing their ability to inhibit the SW620 adenocarcinoma. Nevertheless, it is noteworthy that the sterilized GAP was still capable of inhibiting the proliferation of malignant HCT116 adenocarcinoma. A comparison was made between the SI and the MSI product prepared with the excipient. The MSD product prepared with the three mushrooms was also included for comparison. Figure 4 shows that MSD and MSI with the same concentration of FI and SI can inhibit the proliferation of two types of cancer cells. Although SI has been formulated into a powder with an excipient, which leads to a decrease in its concentration, it retains a significant antitumor effect, as seen in Figure 4. This also provides indirect evidence that the addition of excipients did not diminish the antitumor activity of SI and even enhanced it. The co-culturing of Flammulina velutipes with Ganoderma lucidum resulted in a decrease in cellular biomass production but an increase in exopolysaccharide (EPS) production in comparison to the monoculture of Ganoderma lucidum. 13 MSD is a co-culture of Ganoderma lucidum, Agaricus subrufescens, and Phellinus linteus, 17 which has been observed to exhibit an inhibitory effect. This merits further investigation in the future.

Inhibitory effect of the various concentrations of GAP on colorectal cancer SW620 or HCT116 cell line proliferation. (A) Data showed the inhibition of SW620 cell line proliferation by freeze-dried (FI) and sterilized (SI) forms of GAP at concentrations of 0.1 μg/mL, 1 μg/mL, and 10 μg/mL compared to untreated controls. (B) Data showed the inhibition of HCT116 cell line proliferation by freeze-dried (FI) and sterilized (SI) forms of GAP at concentrations of 0.1 μg/mL, 1 μg/mL, and 10 μg/mL compared to untreated controls. The results are presented as mean ± SD of three experiments. **P < .01, and ***P < .001.

Inhibitory effects of the same concentration of GAP on colorectal cancer SW620 or HCT116 cell line proliferation. (A) Data showed the inhibition of SW620 cell line proliferation by powdered products (MSD and MSI) and freeze-dried (FI) and sterilized (SI) forms of GAP at a concentration of 1 μg/mL compared to untreated controls. (B) Data showed the inhibition of HCT116 cell line proliferation by powdered products (MSD and MSI) and freeze-dried (FI) and sterilized (SI) forms of GAP at a concentration of 1 μg/mL compared to untreated controls. The results are presented as mean ± SD of three experiments. **P < .01, and ***P < .001.
Macrophages respond to inflammatory signals by increasing the secretion of NO and cytokines to kill pathogens. 20 GAP was added to lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages to examine the production of nitric Oxide (NO), interleukin 6 (IL-6), and tumor necrosis factor alpha (TNF-α). A prophylactic model was designed in which GAP was added before LPS induction and an inflammatory model in which GAP was added after LPS induction. The results showed that both FI and SI could effectively inhibit NO production (Figure 5A) and significantly inhibit the production of the cytokines IF-6 (Figure 5B) and TNF-α (Figure 5C) in the prophylactic model. Both FI and SI showed a dose-dependent manner, with higher concentrations being more effective. Similar results were observed in the inflammatory model. In addition, SI at high concentrations (1 μg/mL and 10 μg/mL) was not less effective than FI in inhibiting NO (Figure 5A) and was even better than FI in inhibiting IF-6 (Figure 5E) and TNF-α (Figure 5F). The results in Figure 5 show that GAP has anti-inflammatory activity and that the sterilization process does not reduce the activity of GAP. Figure 6 shows that there is no statistically significant difference in the anti-inflammatory activity of MSI and MSD prepared after sterilization of GAP polysaccharides, probably due to the lower concentration of active polysaccharides. However, regarding the excellent inhibitory effect of SI on IL-6, the same concentration of MSI still has statistically significant inhibitory activity in the prophylactic model (Figure 6B).

Inhibition of various concentrations of GAP on the production of NO and cytokines in LPS-stimulated macrophage cell line RAW264.7. The prophylactic model: (A) NO, (B) IL-6, (C) TNF-α and the inflammatory model: (D) NO, (E) IL-6, (F) TNF-α. LPS (0.5 µg/mL) was added to all groups at time 0 h for 24 h until assay, except the control group. In the LPS-stimulated macrophages, the cells were incubated in the medium without/with various concentrations of GAP samples. The data were presented as mean ± SE of three experiments. *P < .05, **P < .01, and ***P < .001. NO: Nitric oxide; LPS: Lipopolysaccharide.
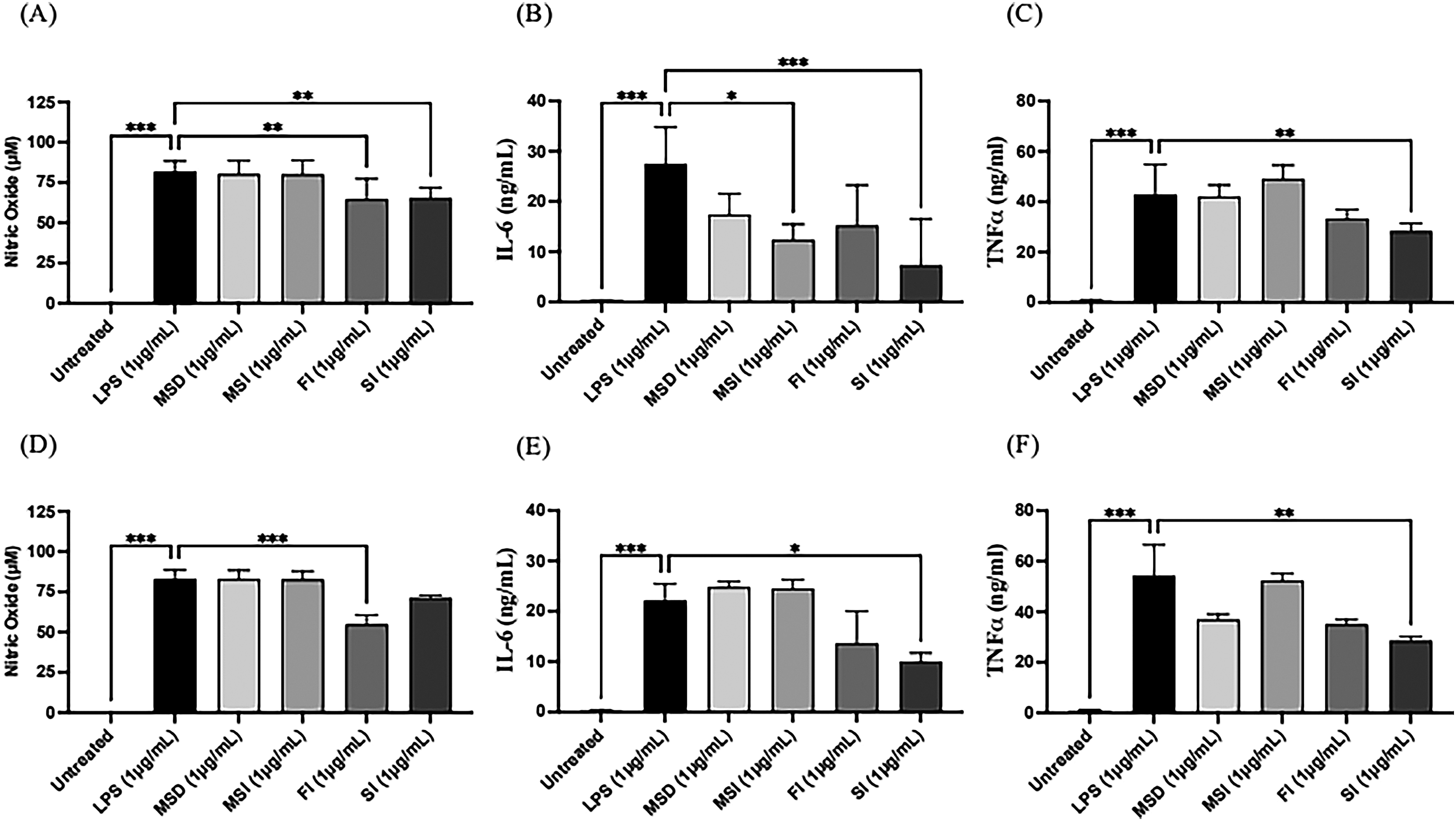
Inhibition of same concentrations of powdered products and GAP on the production of NO and cytokines in LPS-stimulated macrophage cell line RAW264.7. The prophylactic model: (A) NO, (B) IL-6, (C) TNF-α and the inflammatory model: (D) NO, (E) IL-6, (F) TNF-α. LPS (0.5 μg/mL) was added to all groups at time 0 h for 24 h until assay, except the control group. In the LPS-stimulated macrophages, the cells were incubated in the medium without/with various concentrations of GAP samples. The data were presented as mean ± SE of three experiments. *P < .05, **P < .01, and ***P < .001. NO: Nitric oxide; LPS: Lipopolysaccharide.
It has been suggested that high concentrations of nitric oxide (NO) produced by activated macrophages can facilitate apoptosis (programmed cell death) and impede the progression of cancer. 21 However, relatively low concentrations of NO (for example, concentrations measurable in samples from many different types of clinical cancers) can instead stimulate tumor growth and proliferation. IL-6 is a pro-inflammatory cytokine produced by immune cells and has been implicated in several diseases, including chronic inflammation and cancer. 22 The IL-6 signaling pathway is important in the field of cancer biology, particularly in its involvement in cancer metastasis formation. 23 TNF-α is one of the major pro-inflammatory cytokines in the immune system. Furthermore, TNF-α is involved in the development of tumors in several ways, including the transformation and proliferation of cells. However, it can also induce apoptosis and necrosis at specific therapeutic doses. 24 Figure 5 indicates that both FI and SI are more effective in inhibiting the production of IL-6 in both prophylactic and inflammatory modes but less effective in the production of NO and cytokine TNF-α. The NO and TNF-α have antitumor effects in specific environments. Based on the antitumor proliferation results of all GAPs and their products presented in Figures 3 and 4, it can be concluded that GAPs may exhibit an immunomodulatory effect rather than merely inhibiting the production of NO and cytokines. Ganoderma lucidum polysaccharide chains can form a stable triple helix structure through hydrogen bonds. 25 It is hypothesized that Ganoderma polysaccharides do not use a key and lock model but rather an immunomodulation through signal transmission to control tumor progression and progression. Further studies are recommended to confirm this hypothesis. The limitations of this study were that it was conducted using enzyme activity and cellular assays, primarily to determine the effects of the GAP-produced product, which was a preliminary experiment to the ongoing human trial.
Conclusion
In this research work, the active polysaccharides of Ganoderma lucidum from a new metabolic synthesis technology and their powdered products were investigated for the first time for anti-metabolic syndrome, anti-inflammatory, and anticarcinoma activities. Based on current results, it can be concluded that active polysaccharide, which contains abundant β-1,3;1,6-glucan, is a promising source of nutraceuticals with broad-spectrum drug therapeutic potential. The findings of this study suggest that the developed Ganoderma lucidum mycelium extract has the potential for industrial production of health food products, thereby contributing to the field of biotechnology and pharmaceutical technology. Nevertheless, further clinical studies on humans are required for future research.
Materials and Methods
Sample Preparation
Ganoderma lucidum CCRC 36792 was obtained from the Bioresource Collection and Research Center (Hsinchu, Taiwan). The strain was optimized using a strain-optimizing system (TWN Patent M640778) to prepare the inoculum and the mycelium production for producing polysaccharides. The system comprises an activation receptacle, an acclimatization receptacle, a mycelium production receptacle and a fermenting receptacle. This innovative technology is based on the premise that no animal culture medium is used and that the fermentation process is operated using anti-adverse management. The product was cultivated using a mycelium liquid fermentation system under specific conditions, including 80-150 rpm, 24-30 °C, and 0.5-1.0 vvm of aeration. The exopolysaccharide was stably produced using metabolic synthesis technology and then purified defined as Ganoderma lucidum active polysaccharides (GAP). The fermentation broth was then purified and extracted (TWN Patent M M646622) and then either freeze-dried (FI) or sterilized at 121 °C for 15 min (SI). The SI was mixed with Fibersol-2 (Matsutani Chemical Industry Co., Ltd) in a 3:2 proportion and then freeze-dried to produce the active polysaccharide powder, defined as MSI. To compare the effects of different fungi, the polysaccharide powder produced by the combination of Ganoderma lucidum, Agaricus subrufescens and Phellinus linteus was used as a reference group, referred to as MSD. DISAM Biotechnology Co., Ltd (Taipei, Taiwan) supplied the four active polysaccharides used in this study.
Cell Culture
The murine macrophage RAW264.7 cell line was cultured in Dulbecco's Modified Eagle Medium (DMEM), supplemented with 10% fetal bovine serum (FBS). Another two cancer cell lines, HCT116 and SW620, were cultured in Roswell Park Memorial Institute (RPMI) 1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin (100 U/mL penicillin and 100 μg/mL streptomycin). All cells were incubated in a humidified incubator with 5% CO2 at 37 °C. Cells were regularly monitored and maintained at sub-confluent levels to ensure optimal growth conditions. The medium was refreshed every 48 h, and cells were passaged upon reaching 80%-90% confluence. HCT116 and SW620 used 0.25% trypsin-EDTA solution to detach the cells.
Analysis of GAP Composition
The method for detecting protein content was according to the AOAC Official Method AOAC 990.03. The determination of crude lipids, sugars, and ash content is conducted in accordance with the official public methods CNS 5036, CNS 12634, and CNS 5034. The quantity of carbohydrates is calculated by subtracting the amounts of protein, fat, water, and ash from 100. The soluble polysaccharide assay was carried out based on the sulfuric acid-phenol colorimetric method. 26 β-1,3;1,6-Glucan was determined using the K-YBGL β-glucan Assay Kit (Yeast and Mushrooms) (Megazyme, Bray, Ireland) reported by I. Mirończuk-Chodakowska and A. M. Witkowska. 27
In Vitro Anti-Metabolic Syndrome Activity
The spectrophotometric assay (CT2800, Chrom Tech, Singapore) as reported previously determined the inhibitory activities of α-amylase, pancreatic lipase, and angiotensin-converting enzyme (ACE) in the various GAP samples. 28
Carcinoma Proliferation Measurement
For the cancer cell proliferation test, HCT116 or SW620 cancer cells were seeded in a 96-well plate (5 × 104 cells/well). After 24 h, treat with different extracts in the same working concentration (0.1, 1, 10 μg/mL). Incubate for 24 h, Alamar Blue reagent was added directly to the culture medium in each well to a final concentration of 10% of the total volume. Plates were incubated at 37 °C in a 5% CO2 atmosphere for 2 h, allowing viable cells to reduce resazurin to the resorufin product. The products were quantified at wavelengths of 570 and 600 nm using a spectrophotometer. 29
Determination of NO Production and Cytokine Expressions
To determine the effect of GAPs on the production of NO, IL-6, and TNF-α in the prophylactic model, RAW264.7 was seeded in a 24-well plate (5 × 105 cells/well). Different extracts were treated at the same working concentration (0.1, 1, 10 μg/mL) for 1 h and stimulated with LPS (1μg/mL). After 24 h, collect supernatant for IL-6, TNF-α, and Nitric oxide measurement. In the inflammatory model, and vice versa, LPS was stimulated for 24 h before different extract treatments. The concentrations of IL-6 and TNF-α were determined according to the previous report.30,31
Statistical Analysis
Data were presented as triplicate standard deviation (SD). The statistical significance of differences between treated and control groups was determined using one-way ANOVA followed by Dunnett's multiple comparisons test. A p-value of less than 0.05 was considered statistically significant.
Footnotes
Acknowledgments
We thank DISAM Biotechnology Inc. (Taipei, Taiwan) for supplying the study with Ganoderma lucidum active polysaccharides.
Authors’ Contributions
CC and CY conceptualize the study. All authors collected the field data and experimented. CY analyzed the data. CC supervised the work. All authors interpreted the analyzed data, wrote, and reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable to this article. There are no human subjects in this article and informed consent is not applicable. It has not involved any animal experiments.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to DISAM Biotechnology Co., Ltd (Taipei, Taiwan) for providing all samples for the study.
