Abstract
The present review of NMR spectroscopic structural elucidation data of new compounds isolated from Scutellaria species is focused on the title compounds, displaying a peculiar 13-spiro feature. It contains a compilation of 1H and 13C NMR data of these diterpenoids grouped by similar substitution patterns. Comparing shielding effects pointed out not only the identity of some compounds (already reported) but also potential misassignments and convenient revisions to get unambiguous structural proposals.
Interest in clerodane diterpenes has come a long way after the structural elucidation of clerodin, and several reviews already appeared in the 1990s. 1 -3 Structural elucidation of these compounds has improved along with the increasing power of NMR techniques and the interest for them because of their biological activities. Various genera from the plant family Lamiaceae have been identified as rich sources of antifeedant clerodanes, with species of the genus Scutellaria producing some of the most potent known so far. 4,5 Compounds isolated from Scutellaria species possess other interesting pharmacological actions, 6 specifically reviewed for Scutellaria barbata recently. 7,8 The purpose of compiling and revising NMR data of spiro 8β,13-epoxy-neo-clerod-3-en-15,16-olides is 2-fold: serve as background for authors and reviewers in future structure elucidations (with emphasis on precision of reporting data), and to point out (and correct sometimes) apparent inconsistencies through a critical revision.
The Bicyclic 13-Spiro Substructures
The defining structural characteristics of these neo-clerodanes are quoted in a recent review 5 (Section 2.7. Type VII with a 13-Spiro-15,16-γ-lactone Moiety) as follows: a 8,13-ether bridge (creating a tetrahydropyran with C-8, C-9, C-11, and C-13: ring C), and a 15,16-γ-lactone moiety (ring D), linked by C-13 with both (R and S) possible configurations. Parent structures of 13-spiro neo-clerodanes are shown in Figure 1 (R attached position direct insight by the locant as subindex), and structural formulas for the compounds mentioned in this manuscript are shown in Figure 2.
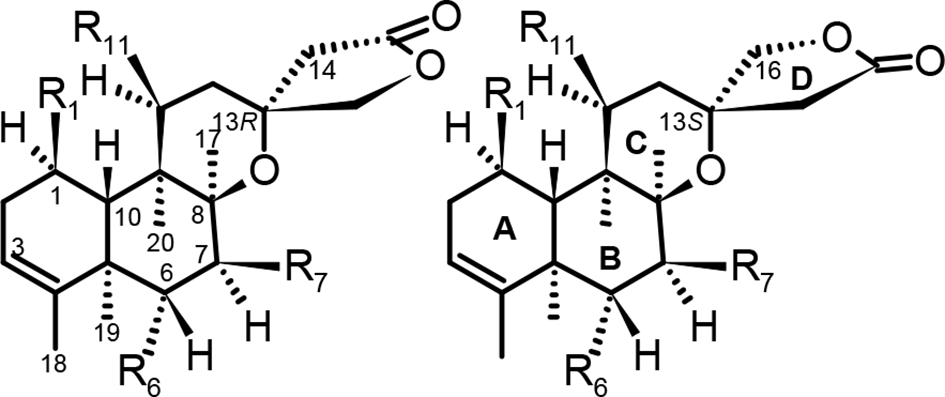
Parent structures, basic locants, and ring systems.

Structures of compounds
Common names and the prefixes to be added to complete their semisystematic neo-clerodane name are shown in Table 1. Four major groups are considered based on 2 different features: C-13 stereochemistry and substitution pattern. The compilation of 1H NMR data is presented in Table 2.
Common and Semisystematic Name for Compounds Mentioned in This Review (prefixes added to)-neo-clerod-3-en-15,16-olide. a

aAdded alphabetically, multiplying affixes do not alter the order
bChange of locant sequence (O, 16, 14 → O, 14, 16 or vice versa) with the same carbon frame (
cRevised structures.
dSpectra provided of known compounds
1H NMR Data of Spiro 8β,13-Epoxy-Neo-Clerod-3-en-15,16-olides. a

Abbreviations: iB, isobutyroyl = 2-methylpropionyl; nr, not reported; N,A, 6-O-nicotinoyl-7-O-acetyl; N,N, 6,7-di-O-nicotinoyl; ov, overlapped.
x = C-2′, y = C-1′ of 2-hydroxy-3-methyl-butyroyl “apparent” couplings (several magnetically nonequivalent nuclei with the same J value): t* ≈ dd; q* ≈ ddd.
aChemical shift values (δ H) in ppm and J values in parentheses (J in Hz).
bReference (compound number in reference)/column: C locant.
cHO coupling (δ H 2.07 d).
dRevised data (printing error?).
eIsochronous methylene?
fIn CD3OD.
Only three 13(S)-clerod-3-en compounds are listed in the comprehensive review mentioned
5
: barbatin A (

Nat Prod Rep 2016 structures. 5
Unfortunately, the 3 drawings of 889 to 891, 1033 to 1035, and 1036 to 1051 (as summarized in Figure 3) represent only 13(S) stereochemistry, whereas scutebarbatine W (
Structure revisions (denoted by →)
5
and identities disclosure were landmarks in the spectroscopic elucidation of these compounds.
10
The originally reported structures of
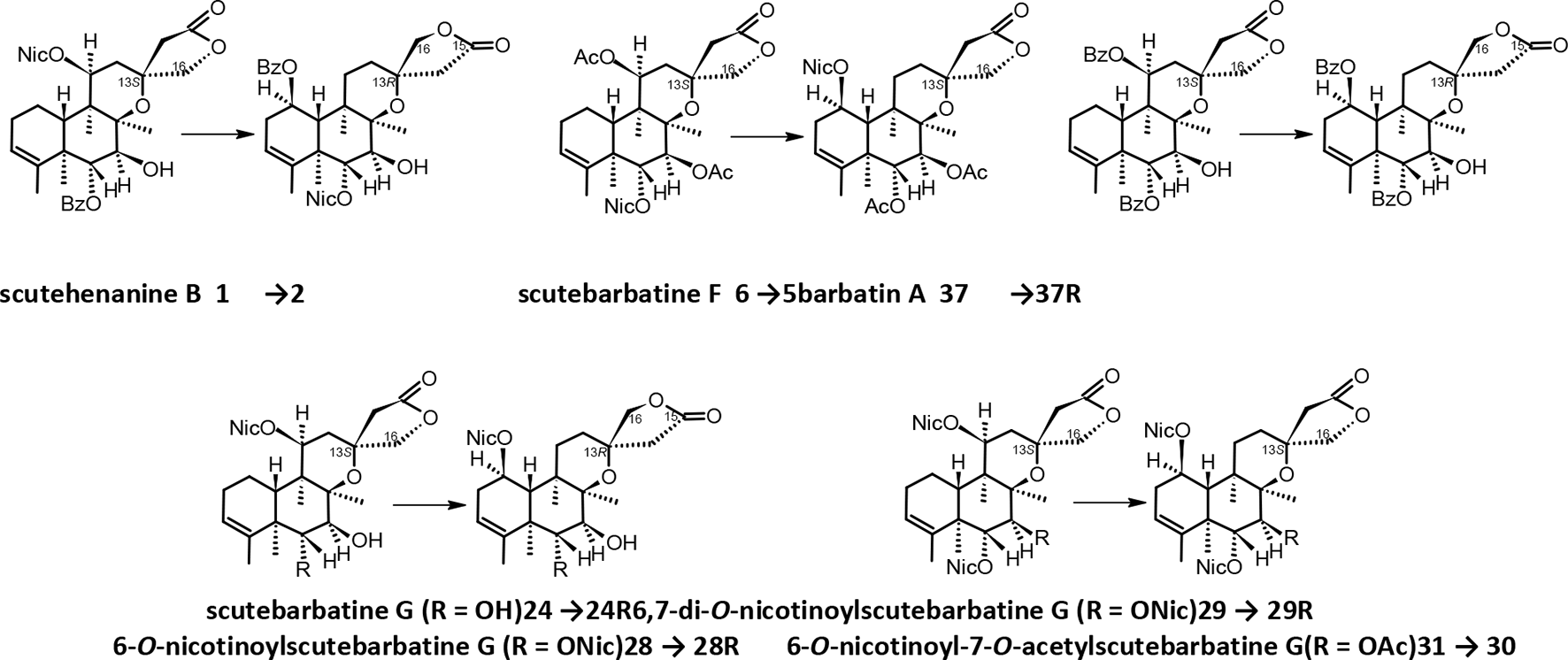
Structure revision of 1, 6, 24, 28, 29, 31, and 37.
Based on the relative configuration established for barbatine B (
It is worth mentioning that the name barbatin (A and B-F) was applied to isolated compounds with no nicotinoyl substituents (featuring or not a 13-spiro carbon, respectively), whereas barbatines (A-D) contained nicotinoyl substituents, but recently a dinicotinoyl compound has been reported as barbatin H (
The Spin Systems of 8β,13-Epoxy-neo-clerod-3-en-15,16-olides
Five spin systems are found in this tetracyclic parent structure of complex signals. Two of them, located in ring D (H2C-14 and H2C-16), appear as geminal simple AB/AX patterns (occasionally collapsing to A2). It was early recognized
10
(“a reliable and more convenient method to determine the configuration at C-13”), the diagnostic character of 1H- and 13C-NMR signals in CDCl3 when no substitution happens in rings C and D. Thus, “doublets around δ
H 2.57, 2.76 (Δδ ≈ 0.20 ppm) and δC 42.2 (t, C-14), 79.6 (t, C-16) are indicative of a 13R* form, while a 13S* analog will display the set of signals at circa δ
H 2.60, 3.15 (Δδ ≈ 0.55 ppm) and δ
C 44.3 (t, C-14), 76.4 (t, C-16).”
10
The chemical shift difference (Δδ) of 2 C-14 protons may be less significantly different and become (almost) isochronous as reported for barbatine B (
Two vicinal isolated methylenes are present in unsubstituted ring B (-H2C-6—H2C-7-) and ring C (-H2C-11—H2C-12-). Thus, complex ABCD multiplets and the need of an unambiguous assignment are expected (to be solved if expanded COSY and HSQC sections are available). However, substitution may result in simplified patterns and the most common is a double O-substitution in ring B. The oxygenated methines [-HβC-6(O)—HαC-7(O)-] appear as another AB system (Δδ < 0.3 if 2 acyloxy groups) and may become isochronous as reported for scutebata O (
The alternative assignment to ring C [-HC-11(O)—HC-12(O)-] would involve a positive correlation of either
Last to be considered here is the more complex ring A system. Signals involve also 2 vicinal methylenes (HaC-1, HbC-1, HaC-2, and HbC-2) with 1 methine at each end (HC-10) and (HC-3). Thus, HC-10 is expected as a dd, while HC-3 is usually reported as a δ ≈ 5.31 ppm broad singlet, pointing out very small couplings with vicinal H2C-2 and allylic H3C-18. Different chemical shifts of HaC-2 and HbC-2 may be observed on examination of the 1H-1H COSY spectrum, throughout the cross signals with HC-3. This spin system is quite common and also found in other neo-clerod-3-en-15,16-olides. It is simplified by C-1 substitution, usually with a β-acyloxy group. Then, HC-10 must become a clear doublet, with 1 large coupling as in scutebata D (
The Splitting Patterns and Accurate Coupling Constants Data
Besides 13C data, the assignment as C-11 vs C-1 substitution (a matter of structural revision as seen) in 1H NMR spectra terms may be envisaged by the expected difference of splitting patterns. While HC-11(O) must appear as a dd as already mentioned, HC-1(O) has 3 surrounding spins yielding a ddd or dt* (1 large and 2 smaller constants: 5 lines with 1:2:2:2:1 ratio if J
large = 2 × J
small). Again, scutebata D (
On the other hand, in the related scutehenanine D (
The peculiar ring A substitution of scutegxbatine A (
Regarding multiplicity, when several magnetically nonequivalent nuclei are coupled with the same J value, the simple (1 J instead of 2) “apparent multiplicity” (t* ≈ dd; q* ≈ ddd; sept* ≈ isobutyroyl qq) is used in this review, whereas overlapped multiplets are simply ov, and complex multiplets (“not easy to describe multiplicity”) are m. Nevertheless, in some instances, obvious multiplet patterns may be sorted out from overlapping data.
The best approach to easy and accurate coupling constants calculation in the first order spectra is to have a display of peaks in Hz. Unfortunately, none is included as part of the available SM. However, if at least 4 decimals are displayed, as occurred in scutebatas D to G
12
(
Also quoted with just 2 decimal figures, the NMR data of scutegxbatine A (
Nor a 3 decimal spectrum is sufficient for accuracy. The Δδ = 0.001 ppm may result from a real range of 0.0009 to 0.0005; in other words, couplings may vary from a high 0.36/0.63 to a low 0.20/0.35 Hz in 400/700 MHz spectra, respectively.
Reporting 1H NMR data usually follows editorial recommendations such as “After each chemical shift, enter in parenthesis number of protons, multiplicity, coupling constants, and assignment in that order. Carbon-13 NMR data should be reported to the nearest 0.1 ppm with the number of attached hydrogens designated as CH3, CH2, CH and C” in Natural Product Communications (NPC). Apparently, there is no recommendation to save table space or improve data accuracy.
Remarks From 13C NMR Data
Table 3 displays a selected set of 13C NMR data to discuss some regular patterns whereas a compilation will be available as Supplemental Material (Supplemental Table SM1). When spectra were available with more than 1 decimal, reported data have been corrected to 2 or more, to point this fact. Significant differences of C-14 and C-16 chemical shifts were pointed out already regarding C-13 stereochemistry: δ C circa 42 (C-14) and 79 (C-16) ppm indicative of a 13(R) form, while δ C circa 44 (C-14) and 76 (C-16) point out a 13(S) analog. C-11/C-12 or C-1/C-2 shifts are good supports for structure elucidation as already commented.
13C NMR Data of Reported 6,7,11-Substituted Compounds.

aReference (compound number in reference)/column: C locant.
A variety of considerations may be formulated with reported 6,7,11-substitution. Whereas data of revised structures to the 1,6,7 pattern, some with additional change of 13C stereochemistry (
Structural Assignment Revisions and Identities Disclosure
Barbatine C, Scutebata F, and Scutebarbatine F Identity
Scutebata F (
Scutebarbatine F was the third compound with the same structure, a conclusion of the revision by Wang et al. 10 The 13C data of all 3 are a remarkable supporting evidence.
Scutebarbatine W and Scutehenanine B Identity
Scutebarbatine W (
The wrong assignment was assumed to be concluded from “not accurately handle the HMBC and ROESY correlations, and (the authors) did not know how to calculate coupling constants of complex signals” (actually, “coupling constants and chemical shifts in second order multiplets”). For instance, the proton signal HC-1 of
However, the apparent integral inconsistency of the 2.63 to 2.83 ppm interval (3 overlapping protons, but 4 reported) was overlooked: a clear doublet, B part of H2C-14 (δ
H 2.75, J = 17.0 Hz), H-10 [δ
H 2.71 (1H, dd, J = 2.3, 12.2 Hz)], and δ
H 2.74 (“2H, m, H-2”). In
Furthermore, the HMBC correlations were the rationale to position the benzoyloxy and nicotinoyloxy groups in
On the other hand, the usual statement “On the basis of the evidence above and comprehensive 2D NMR experiments (1 H-1H COSY, HMQC, HMBC), the structure of 4 was determined to be as shown in Figure 1” was included. Interestingly, correlations supporting C-11 acyloxy position (HC-11 → C-13 and H-11 ↔ H-16 interactions) in “Figure 1. Key HMBC correlations of compounds 1, 4, and 6,” and “Figure 2. Key ROESY,” respectively, should not be observed with the acyloxy in the revised HC-1 position (also applies to all revised structures).
Structural Revisions: Reposition of Substituents and Other Identities Disclosure
In addition, other proposed structures of 13-spiro subtype neo-clerodanes were revised
10
by reanalysis of the published NMR data and “according to the above discussion…the nicotinoyloxy group at C-11 and the spirocarbon configuration of scutebarbatine G (
As a matter of fact, “6,7-di-O-nicotinoyl-scutebarbatine G” and “6-O-nicotinoyl-7-O-acetyl-scutebarbatine G,” with no change of 13(S) stereochemistry, should be renamed accordingly. Either “6,7-di-O-nicotinoyl-13-epi-scutebarbatine G” and “6-O-nicotinoyl-7-O-acetyl-13-epi-scutebarbatine G” or the semisystematic names would avoid confusion between “old” and revised structures. On the other hand, no effort to revise the NMR data was attempted (a simple exchange of C-1 and C-11 data is really enough?).
The Scutelinquanines Structure Revision
Since scutelinquanines A (
The same applies in part to scutelinquanine D (
Then, the multiplicity/coupling constants of HC-10 (
In scutelinquanine E (
NMR Application to Structure Elucidation and Supporting Material
General Comments and Recommendations
A common practice in the abstract of papers is to base structure elucidation “on spectroscopic methods including extensive 1D and 2D NMR analyses,” or closely related expressions. In the full text, “detailed examination of the 1H-1H COSY spectrum indicated…” or “1H-13C long-range correlations observed in the HMBC spectrum…,” “based on the above data and comprehensive 2D NMR experiments (1H-1H COSY, HMQC, HMBC)…,” are used to discuss assignments, and figures may be included “with significant correlations” to support the data compiled in tables. As an example, the relative stereochemistry of chiral centers is usually disclosed by NOE 2D experiments (NOESY, ROESY), fixing the orientation of substituents in the α or β plane of the molecule: H-1, H-7, H-11, H-16, Me-17, Me-19, and Me-20 in α configuration; the cofacial relationship H-6 and H-10 points out β-orientation (Figure 5).
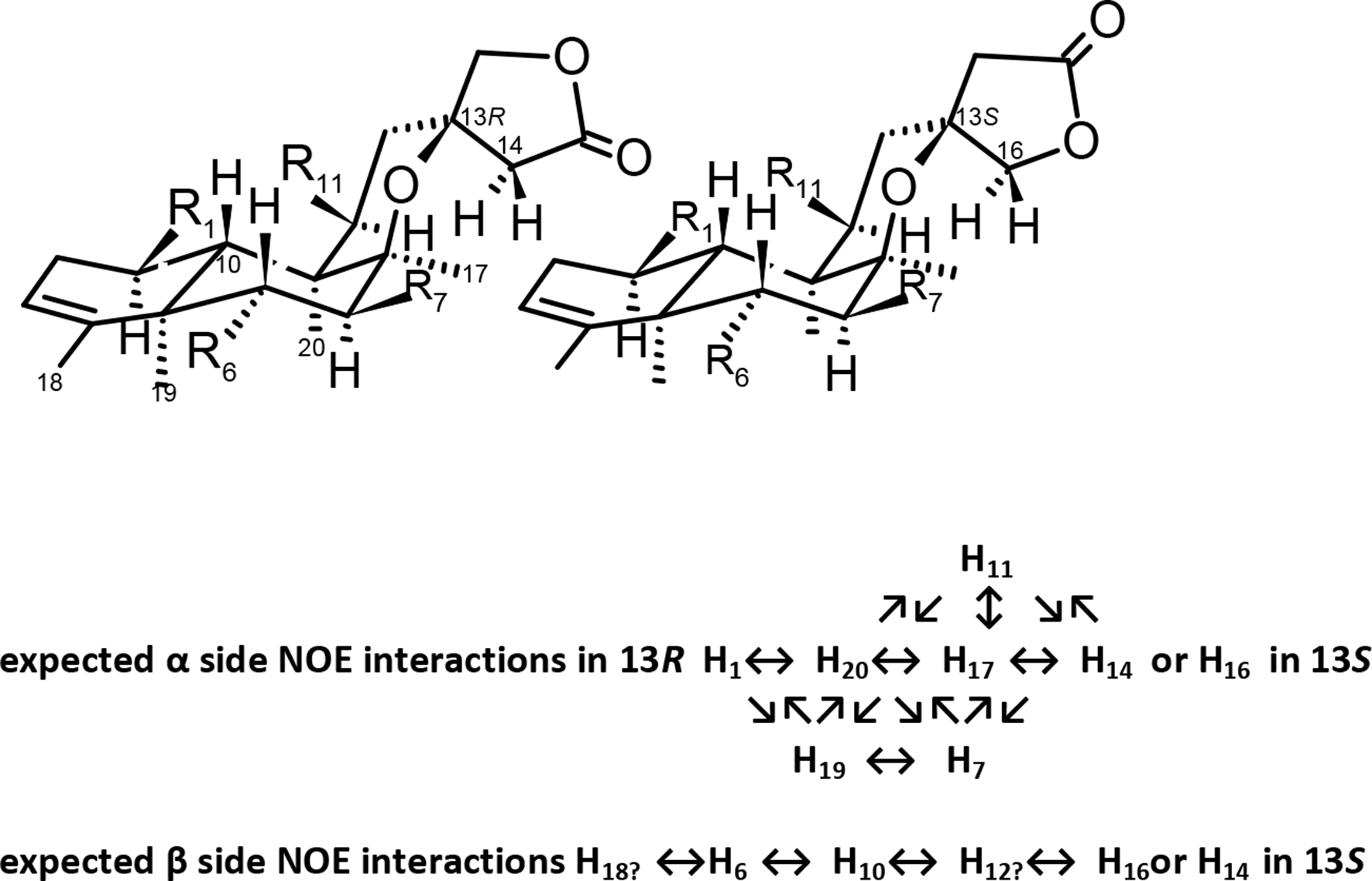
Stereochemistry of α or β substitution.
The trustworthy reported data only may be questioned if “copy/paste or typing errors” are apparent. However, the “real” spectra provided as Supporting Material (SM; only in some instances available online free) may not be consistent with the reported data (usually only minor differences, or apparent typing errors). Unfortunately, not very often expanded sections for improved view of the multiplets or 2D spectra with cross-signals to prove correlations are available. A better report of NMR parameters for spectra simulation (convenient in some overlapping systems) not necessarily may involve changes in the reported structure. The presence of impurities may be pointed out (too frequently!) only if spectra are available to check, leading to misleading integrals or cross-peaks in 2D NMR spectra. Nevertheless, it was recognized that “By chance, scutebarbatine W was found to be contaminated by a trace amount of the C-13 epimer (13S* form) in our current study, in addition, more minor C-13 epimers can be also detectable in the NMR spectra of scutebatas D, E, and F.” 12 A query on (sometimes not so) minor compounds: how their presence is accounted for in bioactivity tests?
Furthermore, a complete list of significant data may not be displayed if a too low peak selection threshold results in an excess of peaks due to insufficient space (in 1D spectra), and in such case, expanded sections may be very convenient. It is worth considering the structural meaning of the aromatic substituents.
The plots may be adjusted to significant windows to improve space use. After optimizing 1D spectra, and securing no information is out of the ranges 8.5 to 0.5 and 180 to 10 ppm, why in HSQC and HMBC there are plots extending beyond 9.0 to 0.5 and 225 to 10 ppm?
There is no reason to omit the splitting pattern and coupling constants of obvious multiplet patterns (for example, the “apparent septet” of isobutyroyl q q) even if partially overlapped. The old-fashioned band width at 50% of height (ω½) may be applied as distinctive information of broad “singlets.”
Scutehenanine B Data Discussion
Chemical shifts are reported with 4 significant decimals and coupling constants were calculated with SF 400.13 MHz (SM 9 “compound 4”), resulting in more precise couplings: H6/H7, J = 9.88 or 9.96 Hz; H2C-14, J = 16.97 or 17.09 Hz; H2C-16, J = 9.123 Hz; and HC-10, J = 9.043. The HC-1 six bands were calculated as 2320.594, 2314.992, 2311.671, 2308.950, 2306.389, and 2300.227 Hz leading to δ H 5.774 ppm; J = 5.602 (1-2), 8.923 (1-3), 8.723 (4-6), or 6.162 (5-6). Whereas H6/H7 doublets are of equal intensity lines (AX system) and the chemical shifts are the center of each doublet, others are AB systems and the true chemical shift has to be calculated [Δδ = √(1 − 4)(2 − 3)].
Scutebata D Data Discussion
In scutebata D (
Scutolide K Data Discussion
Whereas the absolute configuration of scutolide K (
Scutebarbolide M Data Discussion
Some apparent inconsistencies may be considered in the recently reported scutebarbolide M (
Barbatin H Data Discussion
Whereas the drawing of barbatin H (
Scutegxbatine A Data Discussion
The NMR data of scutegxbatine A (
Scutebata O Data Discussion
A very special structural query appears in scutebata O (
Complementary Structure Elucidation Methods
Absolute configurations may be determined by X-ray diffraction (applied to just a couple of compounds reported in this review: scutolide K (
Concluding Remarks
While the structural elucidation of Scutellaria neo-clerodane diterpenoids in NPC has been reported only in a few instances, neither the studied species were barbata nor the compounds displayed any common feature with those commented here. 33 -35 However, in 2 of them, the “Supplementary Data” section contains “Tables of complete spectral data and the 1H NMR, 13C NMR, and 2D NMR spectra (with enlarged detailed sections for multiplets and cross peaks)” as good examples of desirable presentation.
Since the NMR data of revised structures may not involve just a simple exchange of C-1 and C-11 data, it may be taken into account in future isolations of known compounds to report NMR “revised” data if a clear misassignment is detected (or revised already!), instead of the simple (or wrong!) “consistent with those … reported in the literature.”
The C-1 vs C-11 substitution may be best accounted for from the 13C NMR spectra, supported by 1H NMR if clear ddd (sometimes dt*) or dd are observed, respectively. On the other hand, the HMBC HC-1 to C-2 or C-3 vs HC-11 to C-13 or H-20 to C-11 correlations may be required for unambiguous assignment.
Second-order spectra simulators may be found and used to improve the accuracy of spin systems’ data reports, when direct first order is no longer providing correct parameters. 36
Supplemental Material
Table S1 - Supplemental material for The NMR Spin Systems of Scutellaria 8β,13-Epoxy-neo-clerod-3-en-15,16-olides and Revision of 1H and 13C NMR Spectra Reported Data
Supplemental material, Table S1, for The NMR Spin Systems of Scutellaria 8β,13-Epoxy-neo-clerod-3-en-15,16-olides and Revision of 1H and 13C NMR Spectra Reported Data by and Josep Coll Toledano in Natural Product Communications
Supplemental Material
Table S2 - Supplemental material for The NMR Spin Systems of Scutellaria 8β,13-Epoxy-neo-clerod-3-en-15,16-olides and Revision of 1H and 13C NMR Spectra Reported Data
Supplemental material, Table S2, for The NMR Spin Systems of Scutellaria 8β,13-Epoxy-neo-clerod-3-en-15,16-olides and Revision of 1H and 13C NMR Spectra Reported Data by and Josep Coll Toledano in Natural Product Communications
Footnotes
Acknowledgment
The help of Petko Ivanov Bozov (Lecturer in Organic Chemistry, Faculty of Biology, Plovdiv University “Paisii Hilendarski,” Plovdiv, Bulgaria) with some references is gratefully acknowledged.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
