Abstract
A new scordidesin А (
The diterpenoid composition of Teucrium scordium subsp. scordium had been thoroughly studied in the period of 1981‐1985 by different collectives 1 -4 . Ten known clerodane diterpenes in addition to 11 new derivatives were isolated. The decalin moiety in all representatives has 5:10 trans ring fusion. Among them, there is no compound with the 19-nor-clerodane skeleton. Only 2-keto-l9-hydroxyteuscordin 5 contains 18,6-olide.
The diterpenoid fraction of T. scordium L. subsp. scordioides has not been studied. Cytotoxicity and antimicrobial activity of cyclohexane, dichloromethane (CH2Cl2), and methanol (CH3OH) extracts of the plant had been tested. 6 Cyclohexane and CH2Cl2 extracts showed high cytotoxicity against MDA-MB-361 cells (half-maximal inhibitory concentration [IC50] = 130.33 ± 0.1 µg/mL and IC50 = 189.89 ± 3.99 µg/mL). CH2Cl2 extract was more effective against MDA-MB-453 cell line with IC50 = 130.33 ± 0.1 µg/mL. The CH3OH extract possessed no cytotoxicity against breast cancer cell lines, MDA-MB-361 and MDA-MB-453. Herb extracts of the plant showed weak antibacterial activity on Pseudomonas aeruginosa, Klebsiella pneumoniae, Escherichia coli, and Bacillus subtilis and did not show any activity against Staphylococcus aureus, S. epidermidis, Micrococcus luteus, Enterococcus faecalis, and Candida albicans.
As a part of our search of biologically active diterpenoids from Teucrium species,
7
we investigated T. scordium L. subsp. scordioides (Shreb.) Maire et Petitmengin. Herein, we report on the isolation and structural characterization of three furo-clerodane diterpenoids, the new scordidesin A (
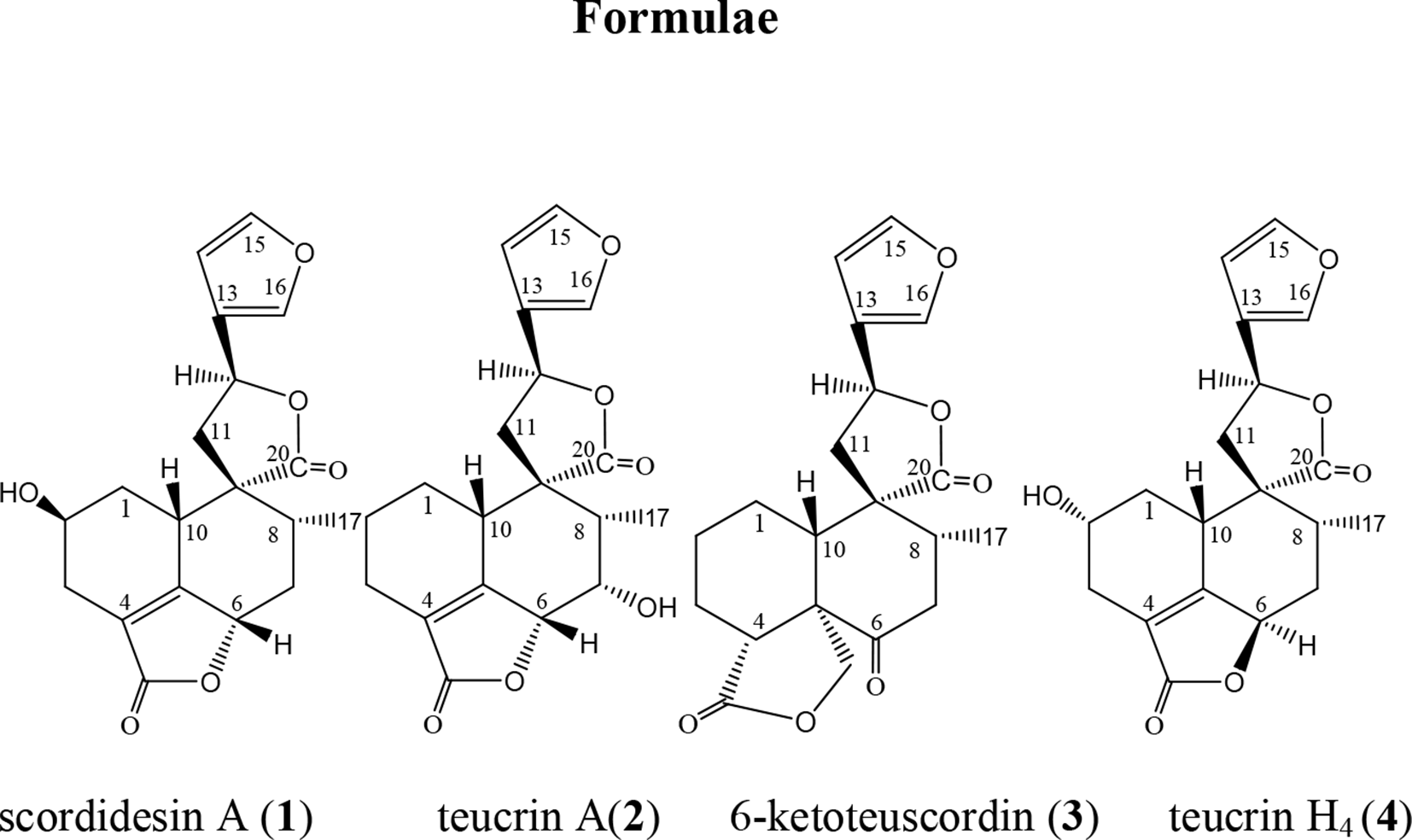
Structures of the 3 compounds (
Results and Discussion
Two homogeneous substances (
The functional groups present in the compound were implied by the observed absorptions in the infrared (IR) spectrum at 1505, 1076, and 874 cm−1 (furan), at 1746 br and 1180 cm−1 (lactones), and at 3435 cm−1 (hydroxyl group).
1H and
11
C nuclear magnetic resonance (NMR) spectra of the substance
The presence of 2 γ-lactone rings was supported by the specific signals for carbonyl groups at δ C 180.6/175.6 (C-20) and 172.5/171.6 (C-18) and 2 oxygenated methine groups at δ C 74.6/71.7//δ H 5.85 m/5.62 t (CH-12) and δ C 80.5/77.7//δ H 4.93 ddd/4.91‐4.84 m (CH-6), forming a C-20, C-12; C-18, C-6 diolide. These assignments were based on the heteronuclear single quantum correlations (HSQCs) and were in agreement with the presence of heteronuclear multiple bond correlation (HMBC) from H-11A to C-12 and C-20, from H-12 to C-11, from the methine proton H-8β to C-6, C-7, C-9, C-10, C-17, and C-20, from methylene protons H2-7 to C-6, C-8 and C-9, and from the methyl protons H3-17 to C-6. Signals of just 1 methyl at δ C 13.2/16.5 and δ H 1.24 d/1.09 d (CH3-17) pointed out the absence of C-19. The 2 unprotonated olefin carbons at δ C 126.7/127.8 (C-4) and 158.3/166.1 (C-5) were part of the С-18 to С-6 α,β-unsaturated γ-lactone ring.
The β-position of the furan ring was established by the nuclear Overhauser effect spectroscopy (NOESY) interactions of the methyl protons H3-17 with H-14 and H-16 and of H-12 with H-11A. The observed NOESY correlation of H-6β with H-8β and H-10β indicated the α bond between C-6 and the oxygen atom of the lactone ring.
The identity of the NMR data for the compound
Abbreviations: Abbreviations: br, broad; ov, overlapped.
aDeuterated acetone, 1H 600.13 MHz, δ ref 2.05 ppm; 13C 150.9 MHz, δ ref 206.26 (CO), 30.60 (CH3) ppm.
bDeuterated chloroform, 1H 600.13 MHz, δref 7.26; 13C 150.9 MHz, δref 77.0 ppm.
cEndo hydrogen with respect to ring B.
dData from heteronuclear single quantum correlation.
The same molecular formula for the other constituent
1
of the mixture
The assignments of the hydrogen atoms to the corresponding carbon atoms were in agreement with the results from the HSQC experiment. The presence of a furan ring in the molecule was confirmed by the correlations between the signals at δ C 108.5/δ H 6.55 dd (CH-14), 144.4/7.62 t (CH-15), and 140.4/7.72 dt (CH-16). The correctness of the assignments was proved by the observed HMBC from H-14 to C-13, C-15 and C-16, from H-15 to C-13, and from H-16 to C-14 and C-15. Other correlations from the signal of proton H-12 (δ H 5.62 t) to carbons C-13, C-14, C-16 and from the signals of the methylene protons H2-11 (δ H 2.78 and 2.65) to C-13 were present. In the correlation spectroscopy (COSY) experiment interactions, H-14/H-15, H-14/H-16, and H-11A/H-12 were observed.
The 1H and 13C NMR spectral data of
The 1H and 13C NMR spectral data of
The recorded 1H and 13C NMR spectra disclosed substance
Absorptions consistent with the presence of a furan ring (1504 and 875 cm−1), lactones (1786, 1760, 1182 cm−1), and hydroxyl group (3436 cm−1) were observed in the IR spectrum.
The 13C NMR spectrum displayed the presence of 20 carbons, and a DEPT experiment identified 1 methyl, 6 methylene (one of them oxygenated, CH2-19), 7 methines (including 3 aromatic and 3 oxidized), and 6 unprotonated (including 2 quaternary, 1 aromatic, and 3 for carbonyl groups—1 fоr a ketone group and 2 fоr γ-lactones).
Furan ring in the structure was confirmed by the signals at δ C 124.5 (C-13), δ C 107.8/δ H 6.40 (br s, CH-14), δ C 144.5/δ H 7.47 (br s, CH-15), and δ C 139.7/δ H 7.48 (br s, CH-16) in the 13C and 1H NMR spectra. Assignments of the aromatic protons to the corresponding carbon atoms were in agreement with the data from the HSQC spectrum and observed HMBCs (see Supplemental Material 1) from H-14 to C-15 and C-16, from H-15 to C-13, and from H-16 to C-14 and C-15. Additional correlations from the signal at δ H 5.44 t (H-12) to carbons C-13, C-14, C-16 and from the methylene proton signals at δ H 2.54 and 2.46 (CH2-11) to C-13 were observed. Correlations between H-14/H-15, H-12/H-11A, H-12/H-11B were represented in the COSY spectra. The presence of 2 γ-lactone rings C-20 to C-12 as well as C-18 to C-19 was supported by the specific signals in the 1H and 13C NMR spectrums: for the carbonyl functions, the signals were at δ C 177.4 (C-20) and 176.8 (C-18), for oxygenated methine group at δ C 72.1/δ H 5.44 t (CH-12), and for oxygenated methylene group at δ C 69.3/δ H 4.80 d and 4.49 d (CH2-19). These assignments were in agreement with the presented HMBCs: for the С-20 to С-12 γ-lactone ring, from H2-11 (δ H 2.46 br t) to C-12 and C-13, from H-12 to C-11 (δ C41.0), C-13, C-14, and C-16, from H-10β (δ H 2.86 dd) to C-20, as well as for the С-18 to С-19 γ-lactone ring, from H-7β (δ H 2.39 dd) to C-5 (δ C 55.4), from H-19A (δ H 4.80, d) to C-18, and from H-19B (δ H 4.49, d) to C-4 (δ C49.2). In the COSY spectrum, cross-peaks between H2-11/H-12, H2-11/H-8, H-2β/H-4, and H-3α/H-4 were observed.
The ketone group, which resonated at δ C 208.1, was located at C-6 on the ground on the downfield shifting of the methylene protons H2-7 signals at δ H 3.41 t and δ H 2.39 dd, which were with the less complicated multiplet structure than the usual dt and ddd in diterpenoids with a hydroxyl group at C-6, due to the lack of adjacent protons at C-6. This conclusion was supported by the observed HMBC corrections to C-6 from the protons H-7α, H-7β, H-19A, and H-19B.
The relative configuration of
Finally, the 1H spectroscopic data of compound
Antimicrobial activity of the isolated diterpenoids was tested against 7 strains, S. aureus ATCC 1805, Streptococcus pyogenes ATCC 12344, Listeria monocytogenes ATCC 8632, E. coli ATCC 3397, Salmonella abony ATCC 6017, C. albicans ATCC 10231, and Candida glabrata ATCC 90030. As seen (Table 2), both tested samples
Antimicrobial Activity of Diterpenoid Constituents of Teucrium scordium L. Subsp. scordioides (Shreb.)
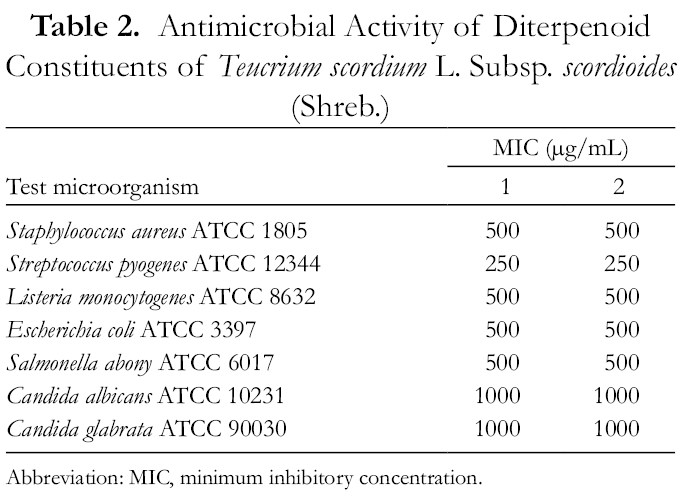
Abbreviation: Abbreviation: MIC, minimum inhibitory concentration.
Conclusion
From the acetone extract of T. scordium L. subsp. scordioides (Shreb.) Maire et Petitmengin, a new neo-clerodane diterpenoid scordidesin A, besides the previously known teucrin A and 6-ketoteuscordin were isolated. Their structures were established by intensive spectral methods. 13C NMR data of 6-ketoteuscordin were published for the first time. Although scordidesin A and teucrin A were isolated as a mixture, their structures were unambiguously assigned, thanks to their varying amounts and by comparing with the published spectral data of the known teucrin A and with the relation teucrin H4.
The substances showed moderate antimicrobial activity against the strains tested.
Experimental
Structural Data
1 H NMR spectra were recorded on Bruker Avance II + spectrometers, operating at 600.130 MHz. 13C NMR spectra were recorded at 150.903 MHz spectrometer. Trimethylsilane was used as an internal standard and deuterated acetone and deuterated chloroform as solvents. Chemical shifts (δ) are expressed in ppm and coupling constants (J) in Hertz. The IR spectra were registered in potassium bromide (KBr) pellet on a Vertex 70 spectrometer from 4000 cm–1 to 400 cm–1 at a resolution of 4 cm–1 with 25 scans. The mass spectra were measured on Hewlett Packard 6890 GC System Plus/5973 MSD.
Plant Material
The aerial parts of T. scordium L. subsp. scordioides (Shreb.) Maire et Petitmengin were collected in June 2019 around the village Staro Orehovo near Varna, Bulgaria, and voucher specimens (n. 7212) were deposited in the Herbarium of the Higher Institute of Agriculture at Plovdiv, Bulgaria.
Extraction and Isolation
Dried and finely powdered material (2.3 kg) were extracted with Me2CO (3 × 8 L) at room temperature for a week. After filtration, the solvent was evaporated to dryness under reduced pressure and low temperature (<40 °C) yielding a gum (28.0 g), which was dissolved in aqueous Me2CO (40% H2O, v/v, 300 mL). This solution was cooled to 4 °C for 24 hours and filtered. The filtrate was extracted with chloroform (3 × 300 mL), and the organic layer was dried (sodium sulfate) and evaporated in a vacuum to afford a residue (7.3 g, bitter fraction). This residue was subjected to column chromatography (100 g silica gel Merck n. 7734, deactivated with 10% H2O, w/w). Pure petroleum ether (15 L), followed by a gradient of CH2Cl2-CH3OH mixtures (10:0-9.7:0.3) were used as eluting solvents. Eluting with CH2Cl2-CH3OH mixtures 9.8:0.2 resulted in the isolation of 15 mg of homogenous substance
Scordidesin A (1 )
Colorless prisms. TLC: Rf 0.40 (ethylacetate [EtOAc]). IR (
Teucrin A (2 )
Colorless powder. TLC: Rf 0.40 (EtOAc). 1H and 13C NMR: see Tables 1 and Table 2s in Supplemental Material 1.
6-Ketoteuscordin (3 )
Colorless powder. TLC: Rf 0.34 (EtOAc). IR νmax (KBr): 3436, 2959, 2928, 1786, 1760, 1504, 1469, 1182, 1165, 1105, 975, 875, 730, 599 cm−1.1H and 13C NMR: see Tables 1 and Table 3s in the Supplemental Material 1. Positive ESI-MS (70 eV, direct inlet) m/z (rel. int. in %): 381 [M + Na]+ (95.2), 301 (15.2), 243 (18.3). HR-ESI-MS m/z 381.1308 [M + Na]+ (calcd. for C20H22O6Na: 381.1314).
Antimicrobial Testing
The antimicrobial effects of the studied compounds were tested against the Gram-positive bacteria S. aureus ATCC 1805, S. pyogenes ATCC 12344, and L. monocytogenes ATCC 8632, as well as the following Gram-negative bacteria E. coli ATCC 3397 and S. abony ATCC 6017. Additionally, antimicrobial testing against 2 species of C. albicans ATCC 10231 and C. glabrata ATCC 90030 was performed. The bacterial strains were stored on nutritional agar (HiMedia Ltd., India) and the yeast strains on Sabouraud dextrose agar with chloramphenicol (HiMedia Ltd.). Stock solutions of the samples for antimicrobial testing were prepared by dissolving the respective compound in dimethylsulfoxide (DMSO; Sigma-Aldrich Co.). Antibacterial activity of the studied compounds was performed according to the Clinical Laboratory Standard Institute (CLSI) M7-A7 reference method for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 15 Anticandidal activity of the studied compounds was performed according to CSLI M27-A3 reference method for broth dilution antifungal susceptibility testing of yeasts. 16 Briefly, stock solutions were added to the Roswell Park Memorial Institute 1640 broth medium buffered to pH 7.0 with 0.165 mol/L 3-N-morpholino-propanesulfonic acid buffer (Sigma-Aldrich, Co) to reach dilutions with final sample concentrations, after inoculation with microbial test suspension, between 2000 and 125 µg/mL. Controls consisting of inoculated medium without tested sample and DMSO, as well as with DMSO were also prepared. Antimicrobial activity determined by broth microdilution tests was expressed as a minimal inhibitory concentration (MIC) in μg/mL. MIC was defined as the lowest concentration of the tested compound at which total inhibition of microbial growth was detected. All tests were performed in triplicate.
Supplemental Material
Supplementary Material 1 - Supplemental material for Diterpenoid Constituents of Teucrium scordium L. Subsp. scordioides (Shreb.) Maire Et Petitmengin
Supplemental material, Supplementary Material 1, for Diterpenoid Constituents of Teucrium scordium L. Subsp. scordioides (Shreb.) Maire Et Petitmengin by Petko I. Bozov, Plamen N. Penchev, Tania D. Girova and Velizar K. Gochev in Natural Product Communications
Footnotes
Acknowledgments
We thank Bulgarian Ministry of Education and Sciences for the funds Grant, National Scientific Program “Innovative low toxic bioactive compounds for precise medicine – Bio Activ Med” D01-217/30.11.2018.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

