Abstract
Thiolated hyaluronic acid was synthesized, and a quantification method was established to determine the content of the thiol group. Three thiol compounds, 2-nitro-5-mercapto-benzoic acid (TNB), 2- mercaptonicotinic acid (2-MNA), and mercaptopyridine (MPD), were tested for their efficiency in forming adducts with hyaluronic acid (HA) through disulfide bonds. Our results showed that MPD exhibited the highest disulfide exchange efficiency when the reaction was conducted at the optimal pH value. The synthesized hyaluronic acid-SS-mercaptopurine (HA-SS-MP) was characterized by 1H-nuclear magnetic resonance spectroscopy. The HA-SS-MP micelle showed a particle size of 264.4 nm in aqueous solution. An in vitro release experiment demonstrated that the releasing rate of 6-MP from HA-SS-MP in the release medium containing glutathione (GSH) was much higher than that in the release medium without GSH. Cell uptake experiments demonstrated the CD44 targeting of hyaluronic acid. Cytotoxicity tests in mouse melanoma cells (B16F10) showed that HA-SS-MP can kill melanoma cells effectively and with lower cytotoxicity, compared with the active ingredient, 6-MP.
Nowadays, cancer is one of the leading causes of death. Although various chemotherapeutic agents have been developed to kill cancer cells effectively, they are usually toxic to normal cells, thereby causing side effects such as damage to the liver, kidney, and other organs. Stimuli-responsive drug delivery is a promising approach to solve this problem. The principle of stimuli-responsive drug delivery is to target the release of the drug when the drug carrier arrives at the tumor. Variations in the physiochemical conditions, such as pH, temperature, magnetic field, and oxidation–reduction reactions, can stimulate the release of drug. 1
Stimuli-responsive drug delivery based upon oxidation–reduction reactions has gained a lot of attention in recent years. Glutathione (GSH) is an important antioxidant in human cells. In healthy cells, the intracellular GSH concentration (2-10 mM) is 100-1000 times higher than the extracellular concentration (2-20 µM). 2 GSH plays a significant role in regulating the proliferation and metastasis of cancer cells. 3 The GSH concentration in tumor cells is several times high than that in healthy cells. For example, the GSH concentration in human lung adenocarcinoma cells (A549) is 7-fold higher than that in normal human lung fibroblasts. 4 Notably, GSH is capable of reducing compounds with disulfide bonds into sulfhydryl compounds. This property can be utilized to control the release of drugs from drug carriers in tumor cells. In this study, disulfide-linked polymeric prodrug delivery systems were designed and synthesized based on this guiding principle.
Hyaluronic acid (HA) is a polymer of disaccharides. It is composed of
6-Mercaptopurine (6-MP) is an anticancer drug. It is structurally similar to hypoxanthine and can competitively inhibit the chemical transformation of hypoxanthine to inosinic acid (IMP). It prevents the transformation of guanine into guanylic acid, thereby inhibiting the synthesis of deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). 6-MP is mainly used as an agent in the treatment of acute lymphoblastic leukemia (ALL), choriocarcinoma, acute non-lymphocytic leukemia, and enteritis. However, 6-MP has low water solubility, and its thiol group is prone to interact with thiol groups of plasma proteins to form disulfide bonds. Moreover, the half-life of 6-MP is short (0.5-1.5 hours), and its bioavailability is low (about 16%) in systemic circulation. 11 To solve those problems, researchers have linked 6-MP to polymers via disulfide bonds. Such treatments can enhance the solubility, increase the half-life, and improve the bioavailability of this drug. Several articles have reported 6-MP-based polymeric drug delivery systems. For example, Zheng et al prepared a series of 6-MP-carboxymethyl chitosan (6-MP-CMC) drug delivery systems 12 ; Yu et al synthesized the mPEG-SS-NH-g-PAsp-MP micelle drug delivery system, 13 and Zhuang et al prepared the poly (VBPT-co-PEGMA)-S-S-MP drug delivery system. 14 In this work, we linked 6-MP to HA in a thiol-disulfide exchange reaction and demonstrated that the resulting complex could rapidly release 6-MP to cancer cells with high specificity, owing to the oxidation–reduction reactions mediated by GSH.
Synthesis of HA-SH and Determination of the Thiol Content
Thiolated hyaluronic acid (HA-SH) was synthesized according to Scheme 1. Thiol groups are susceptible to oxidation under alkaline conditions. Thus, the dialysis solution was adjusted to be acidic to prevent the oxidation of thiol groups. HA can be degraded if the pH is too low, so the pH of the dialysis solution was adjusted to 4.5. The thiol content of HA-SH was determined to be 330 μmol/g, and the total thiol content of HA-SH was determined as 438 μmol/g.
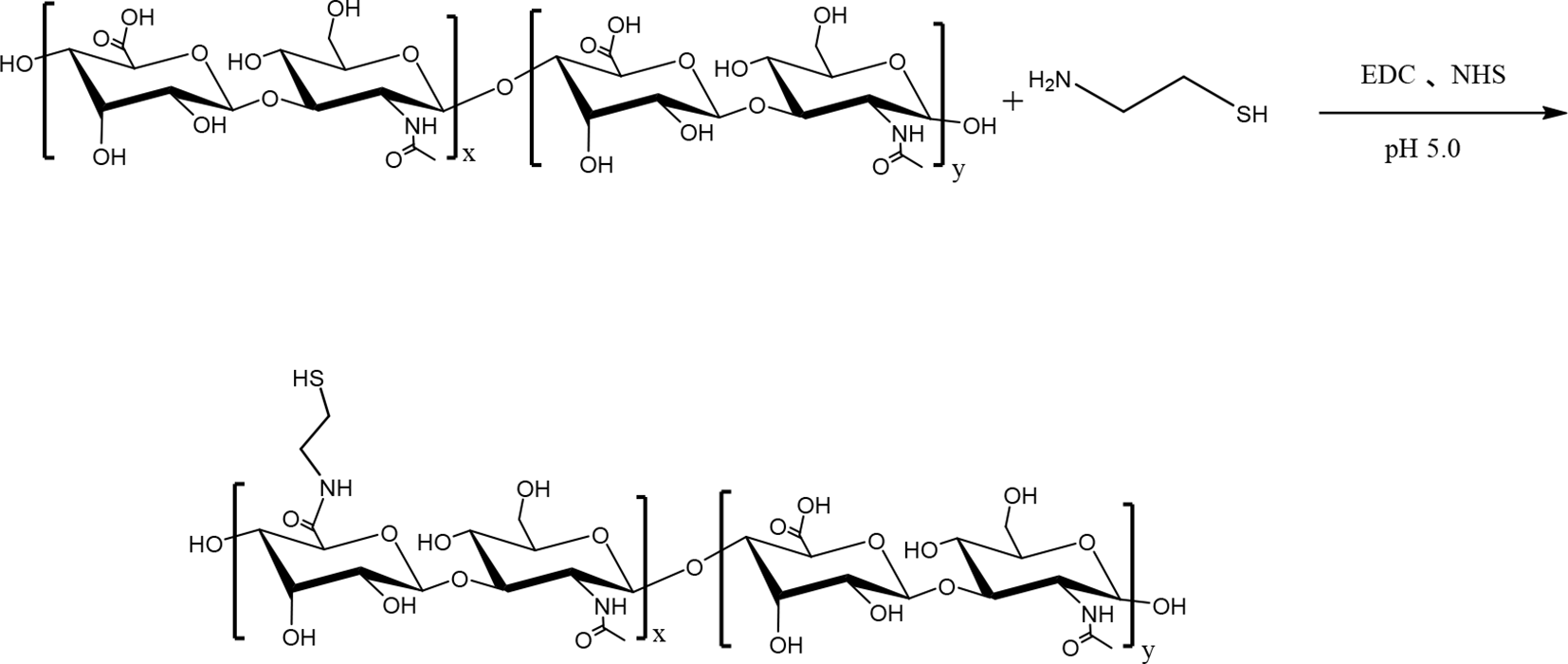
Synthesis of thiolated hyaluronic acid (HA-SH) by carboxyl-to-amine crosslinking using 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC) and N-hydroxysuccinimide (NHS).
Selection of Disulfide Exchange Group
Three different disulfide exchange groups, 2-nitro-5-mercapto-benzoic acid (TNB), 2- mercaptonicotinic acid (2-MNA), and mercaptopyridine (MPD), were selected to synthesize HA-SS-MP. HA-SS-MP was synthesized according to Scheme 2. The acid dissociation constant (pKa) values of TNB, 2MNA, and MPD are 4.4, 15 3.9, 16 and 1.1, 17 respectively. The grafting rate of 6-MP increased as the pKa value of the disulfide exchange group decreased. This observation was caused by 2 major reasons. First, the reaction of thiol-disulfide exchange is affected by the pKa value of the thiol group as the exchange reaction between thiol and disulfide requires the deprotonated thiolate form. Second, the pKa value of the leaving group also has a great influence. The disulfide exchange group with a lower pKa value has higher electrophilicity; thus, the rate constant for the reaction increases as the pKa value of the leaving group decreases. When thiolate anions attack an asymmetric disulfide (ie, R1-SS-R2), the thiol group with the lower pKa value is easier to leave. 18 The content of 6-MP is shown in Table 1.

Synthesis of HA-SS-MP by disulfide exchange reaction; the disulfide exchange group R = 2-nitro-5-thiobenzoic acid (TNB), 2-mercaptonicotinic acid (2MNA), 2-mercaptopyridine (MPD).
The Content of 6-MP in HA-SS-MP From Different Synthesis Routes.
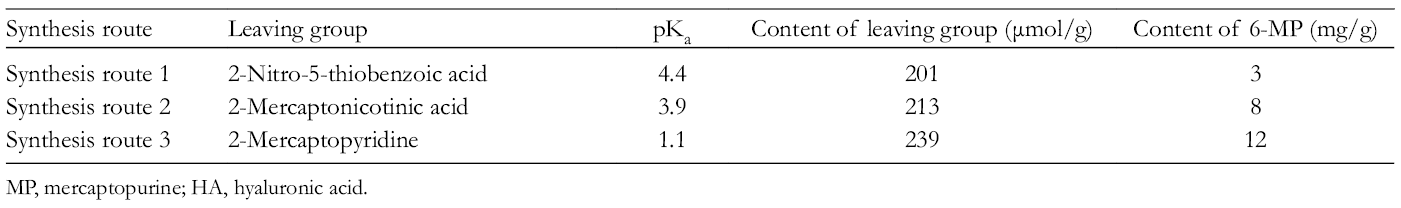
MP, mercaptopurine; HA, hyaluronic acid.
Screening for Optimal pH Value for HA-SS-MP Synthesis
The pH value of the reaction system has a great impact on the disulfide interchange reaction. The exchange reaction between a thiol group and disulfide requires thiolate anions, and the concentration of thiolate anions depends on the pH value of the reaction solution. The pKa value of 6-MP is 7.8. 19 More thiolate anions are participating in the reaction when the pH of the solution is higher. However, MPD is easier to leave under acidic conditions than basic conditions. Our result showed that the highest efficiency of the disulfide exchange reaction was achieved at pH 6.5 (Table 2).
Content of 6-MP in HA-SS-MP Under Different Reaction Conditions of pH.

MP, mercaptopurine; HA, hyaluronic acid.
1H-Nuclear Magnetic Resonance Characterization
HA-SS-MPD: the signals at δ 7.33, 7.88, and 8.43 were assigned to hydrogen atoms of MPD, indicating a successful connection of MPD with HA (Figure 1).
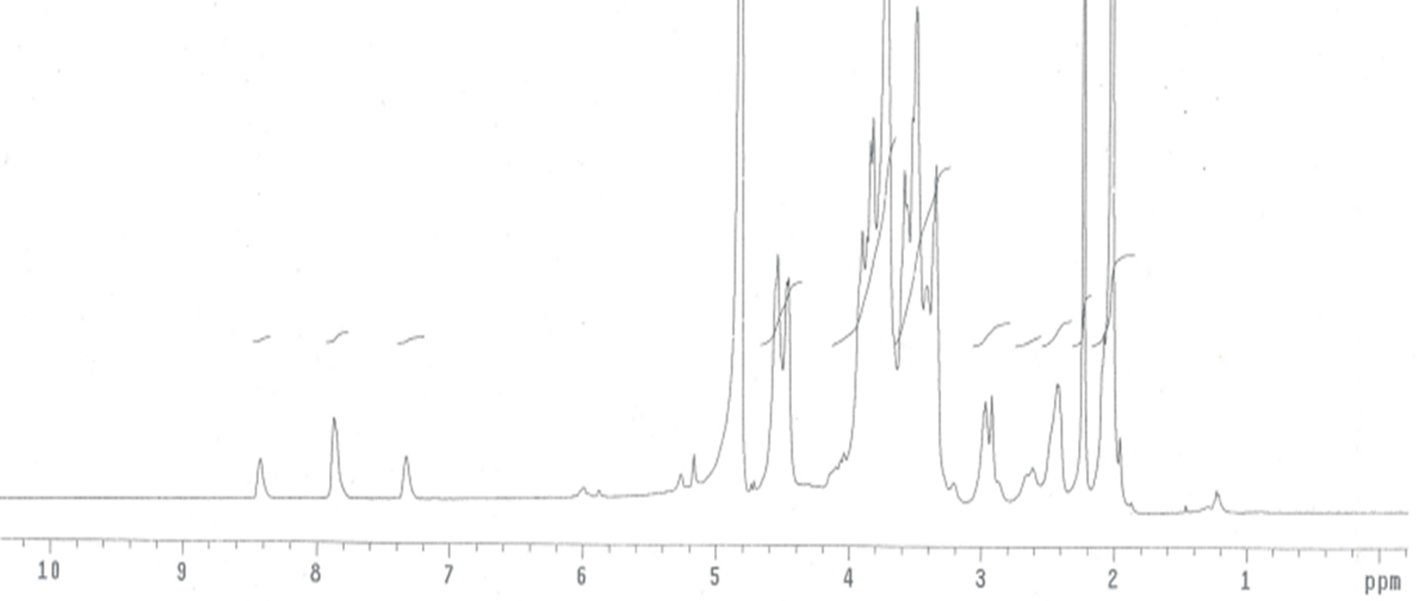
1H-nuclear magnetic resonance spectrum of hyaluronic acid-SS-2-mercaptopyridine in deuterium oxide.
HA-SS-MP: the signals at δ 8.54 and 8.81 were assigned to hydrogen atoms of 6-MP (Figure 2). The signals at δ 7.33, 7.88, and 8.43 were significantly weakened, indicating that the majority of MPD was substituted by 6-MP.

1H-nuclear magnetic resonance spectrum of hyaluronic acid-SS-mercaptopurine in deuterium oxide.
Size of HA-SS-MP
The size of unmodified HA micelles, tested by Malvern Zetasizer, is 177.9 nm, and the size of HA-SS-MP in aqueous solution is 264.4 nm. In the thiolation process, some HA molecules may be crosslinked together, causing an increase in the particle size. HA-SS-MP is composed of hydrophilic HA and hydrophobic 6-MP. HA forms the hydrophilic shell and 6-MP forms the hydrophobic core. HA-SS-MP was able to self-assemble into core-shell micelles in aqueous solution. The distribution of particle size is narrow and in a monodisperse state. The endothelial cells of tumor vessels are looser than those of normal blood vessels, and there are 200 nm to 1.2 µm pores in tumor vasculature 20 ; this indicated that HA-SS-MP could pass through the tumor blood vessels and accumulate in the tumors. The results are shown in Table 3.
The Size of HA-SS-MP (n = 3).

HA, hyaluronic acid; MP, mercaptopurine.
In Vitro Drug Release of HA-SS-MP
When 10 mM reduced glutathione was added into the release medium, more than 90% of 6-MP was released from HA-SS-MP in 1 hour. The intracellular concentration of glutathione was more than 10 mM in cancer cells, suggesting that HA-SS-MP can quickly release 6-MP to kill the cancer cells. When there was no reduced glutathione in the release medium, the release of 6-MP from HA-SS-MP reached about 78% in 48 hours. As we all know, blood circulation is very fast in the human body. The blood volume is about 5 L in an adult body with a weight of 60 kg, the normal heart rate is 75 beats per minute, the blood stroke volume is 70 mL, and the cardiac output is about 5 L/minute. That means one blood circulation can be accomplished generally in 1 minute, so HA-SS-MP can be delivered to tumor tissue within minutes by intravenous injection and accumulated in tumor tissue by passive and active targeting. Therefore, HA-SS-MP can be targeted to tumor tissue quickly. The release profile is shown in Figure 3.

In vitro release profile of hylauronic acid-SS-mercaptopurine in pH 7.4 phosphate-buffered saline medium at 37°C (n=3,  contain 10 mM glutathione (GSH),
contain 10 mM glutathione (GSH),  no GSH).
no GSH).
Cell Uptake Assays
Murine melanoma B16F10 cells highly express the CD44 receptor. 21 HA was labeled with fluorescein isothiocyanate (HA-SS-FITC) to research the cell uptake of HA by flow cytometry (Figure 4). After the B16F10 cells had been pretreated with free HA for 2 hours (B16F10 + HA), the fluorescence intensity of the cells was obviously decreased compared with unpretreated B16F10 cells (*P < 0.01) (Figure 5). These results suggested that the uptake of HA-SS-FITC by B16F10 cells could be competitively inhibited by HA, indicating that HA could bind with the CD44 receptor and show CD44 targeting.
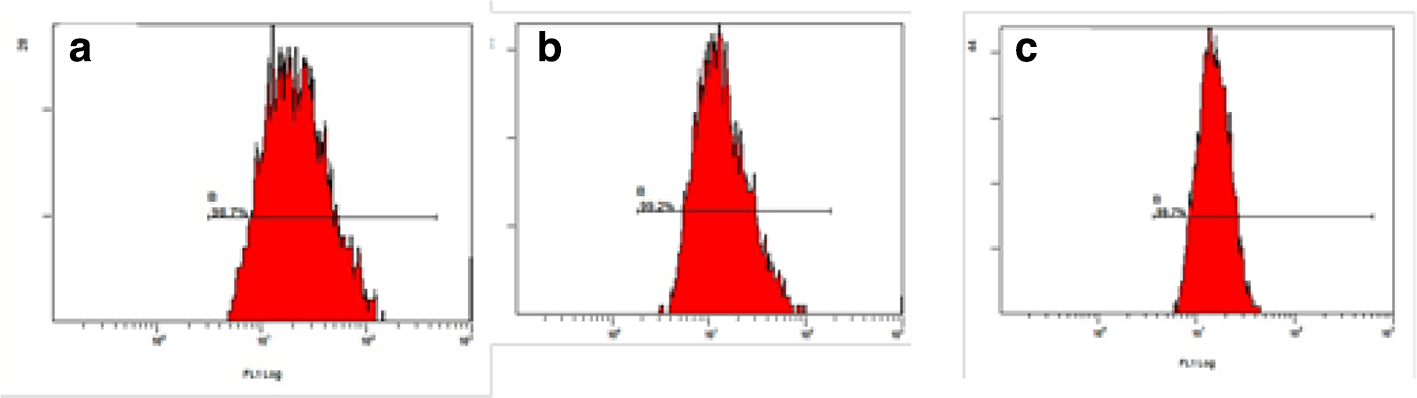
Flow cytometric analysis results of cell uptake. (a) B16F16 cells, (b) B16F10 cells pretreated with free HA, and (c) NIH/3T3 cells.

The fluorescence intensity of hylauronic acid -SS-fluorescein isothiocyanate measured by flow cytometry (n = 3, *P < 0.01, **P < 0.01).
The fluorescence intensity of HA-SS-FITC in B16F10 cells was higher than that in NIH/3T3 cells (**P < 0.01) (Figure 5), because the latter do not express CD44 receptors, and only a small amount of HA-SS-FITC entered the NIH/3T3 cells through nonspecific endocytosis. B16F10 cells express the CD44 receptor; HA-SS-FITC could be uptaken by B16F10 cells rapidly via receptor-mediated endocytosis. These results indicated that the prodrug of HA was more easily uptaken by cancer cells compared with normal cells. Therefore, HA prodrug can reduce the side effects of drugs on normal cells.
In Vitro Cytotoxicity of HA-SS-MP
6-MP can stop the propagation of cancer cells mainly by interfering with the metabolism of purine and impeding the synthesis of nucleic acid. 22,23 HA-SS-MP showed excellent anticancer activity (Figure 6). The cytotoxicity of HA-SS-MP was lower than that of the free 6-MP. This observation is due to the fact that 6-MP is easier to enter cells because of its smaller size. Compared with normal cells, 24 the surface of cancer cells has a higher expression level of the CD44 receptor, 25 -29 thus the targeting ability of HA-SS-MP can be enhanced by HA. 30 -38 Thus, HA-SS-MP is highly specific to cancer cells and can kill them more efficiently, thereby reducing the side effects of the chemotherapy.

Cell viability of mouse melanoma cells in the presence of different concentrations of hylauronic acid-SS-mercaptopurine and 6-mercaptopurine (n = 3).
In this work, HA-SS-MP was designed and synthesized. Its 1H-nuclear magnetic resonance (NMR) spectrum indicates that 6-MP was linked to HA successfully. The cumulative release of 6-MP from HA-SS-MP reached more than 90% in 1 hour when the release medium contained 10 mM glutathione. The release of 6-MP became slower when the release medium contained no glutathione. Thus, it can be predicted that the release of 6-MP from HA-SS-MP is slow in the blood, whereas the release process will be significantly accelerated when HA-SS-MP enters cancer cells. Moreover, the targeting ability of 6-MP to cancer cells is enhanced by HA; this drug delivery carrier may be able to improve specificity for cancer cells and reduce the side effects on normal cells.
Experimental
Materials
Sodium hyaluronate (molecular weight: 5 kDa) was purchased from Bloomage Freda Biopharm Co. Ltd (Shangdong, China), fluorescein isothiocyanate isomer I (FITC), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDC), N-hydroxysuccinimide (NHS), cysteamine hydrochloride, cystamine dihydrochloride, 2MNA, 2,2-dithiodipyridine (DPD), and 6-MP from Aladdin Chemical Reagent Co., Ltd, and 5,5′-dithiobis-(2-nitro benzoic acid) (DTNB) from Sigma-Aldrich. The B16F10 cell line was a generous gift from the Key Laboratory of Drug Targeting and Drug Delivery Systems, West China School of Pharmacy, Sichuan University. The NIH/3T3 cell line was kindly provided by Stem Cell Bank, Chinese Academy of Sciences. The Roswell Park Memorial Institute (RPMI) 1640 medium was purchased from Hyclone Co. Ltd, the Dulbecco’s Modified Eagle’s medium from GIBCO, fetal bovine serum (FBS) from FMG Biotech Co. Ltd (ShangHai, China), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) from Amresco.
Synthesis of Thiolated HA (HA-SH)
HA (0.5 g) was added to 37.5 mL of deionized water. After overnight incubation under constant stirring, the pH of the solution was adjusted to 4.7. EDC (1197.5 mg) and NHS (575.0 mg) were added to the solution, the pH of which was adjusted to 5.5. After 60 minutes of incubation, cysteamine hydrochloride (710 mg) was added to the reaction system under a constant flow of nitrogen. After 6 hours, this reaction mixture was dialyzed against deionized water for 12 hours at pH 4.5 with the temperature maintained at 0-10°C. The dialysis treatment was repeated another 2 times. At last, HA-SH was isolated by freeze-drying.
Determination of Free Thiol Content
HA-SH (10 mg) was added to 2 mL of hydrochloric acid (HCl) solution (1 mM), and the mixture was incubated for 3 hours. Dibasic sodium phosphate (DSP) buffer solution (pH 8.0, 0.1 M) was then added to the mixture, making the final volume to 10 mL. DTNB (200 µL, 10 mM) was added to this newly prepared HA-SH solution (1.5 mL). DSP buffer solution (pH 8.0, 0.1 M) was added to this mixture to make the final volume to 10 mL. This was placed in a water bath at 37°C for 5 hours. The optical absorbance was recorded at 412 nm with an ultraviolet–visible (UV–vis) spectrophotometer. 39
Determination of Total Thiol Group Content
HA-SH (10 mg) was added to the DSP buffer solution (pH 8.0, 0.1 M), and the solution was incubated for 3 hours. Sodium borohydride solution (4%, 2 mL) was added, and the mixture was placed in a water bath at 37°C for 10 minutes. 40 DTNB (200 µL, 10 mM) was then added to this mixture, followed by adjusting the final volume to 10 mL by adding a DSP buffer solution (pH 8.0, 0.1 M). This final solution was placed in a water bath at 37°C for 5 hours. The absorbance at 412 nm was recorded.
Synthesis Route 1
Synthesis of HA-SS-TNB
HA-SH (100 mg) and DTNB (24 mg) were dissolved in 5 mL DSP buffer solution (pH 8.0, 0.1 M). The solution was then purged with nitrogen gas (N2). Afterward, the pH of the reaction solution was adjusted to 8.0 by 0.5 M sodium hydroxide (NaOH). After 12 hours, the reaction mixture was dialyzed against water, 3 times (each time for 12 hours). Finally, the reaction solution was frozen and lyophilized to obtain HA-SS-TNB.
Determination of the grafting rate of HA-SS-TNB
HA-SS-TNB (5 mg) was added to 5 mL of deionized water and incubated under stirring for 1 hour. GSH (3 mg) was added to this solution and incubated for 30 minutes. The optical absorbance of this reaction mixture was recorded at 412 nm on a UV spectrophotometer.
Synthesis of HA-SS-MP
HA-SS-TNB (30 mg) was dissolved in 3 mL of degassed water under an N2 atmosphere and incubated for 4 hours at 37°C. Dimethylsulfoxide (DMSO, 5 mL) was added to the solution, followed by a solution of 6-MP (60 mg of 6-MP, dissolved in 1 mL of DMSO solution). Afterward, acetic acid was added to the reaction mixture to adjust the solution pH to 6.5, followed by 24 hours of incubation at 37°C. The resulting conjugate was dialyzed against deionized water. After the first dialysis, the mixture was centrifuged for 3 minutes at 5000 rpm. The supernatant was dialyzed a further six times, and HA-SS-MP was obtained by freeze-drying.
Determination of HA-SS-MP content
HA-SS-MP (3 mg) was added to 1 mL of water and incubated for 3 hours. Afterward, 4% sodium borohydride solution (0.6 mL) was added to the HA-SS-MP solution, and the reaction mixture was placed in a water bath for 1 hour. HCl (2 M, 0.3 mL) was added to the reaction mixture, and then the solution was placed in a water bath for 10 minutes. At the end, 1.1 mL of DSP solution (pH 8.0, 0.1 M) was added to the reaction system, followed by a 10-minute incubation at 37°C. Potassium permanganate solution (0.6 mL, 1 mM), NaOH solution (0.8 mL 1M), and deionized water (2.2 mL) were added sequentially to the previous reaction solution. After 48 hours of incubation, the fluorescence signal of the final reaction mixture was monitored; the pH of the solution was about 13. The excitation wavelength was 286 nm, and the emission wavelength 397 nm. 41
Synthesis Route 2
Synthesis of 6,6′-dithiodinicotinic acid
2MNA (0.5 g) was dissolved in 25 mL of deionized water, and the pH of the solution was adjusted to 8.7. Hydrogen peroxide (30%, 0.65 mL) was added dropwise to the solution under constant stirring. The solution gradually became colorless and the pH of the solution dropped to 6.0. NaOH solution (1 M) was added to adjust the pH of the solution to about 8-9. NaOH (1 M) solution was added under continuous stirring for about 1 hour to preserve a pH of 8-9. 42 At last, 6,6′-dithiodinicotinic acid was isolated by freeze-drying.
Synthesis of HA-SS-2MNA
HA-SH (200 mg) was dissolved to 10 mL degassed DSP buffer solution (pH 8.0, 0.1 M) under an N2 atmosphere and incubated at room temperature for 6 hours. 6,6′-Dithiodinicotinic acid (120 mg) was dissolved in 10 mL DSP buffer solution (pH 8.0, 0.1 M), and 1 M NaOH solution was added to adjust the pH of the solution to 8.5. 43 The 2 solutions were mixed together under an N2 atmosphere and placed in a water bath at 37°C for 48 hours. After dialysis against deionized water for 7 times, HA-SS-2MNA was isolated by freeze-drying.
Determination of the grafting rate of HA-SS-2MNA
HA-SS-2MNA (5 mg) was dissolved in 1 mL of deionized water. GSH (3 mg) was added to the HA-SS-2MNA solution and incubated for 30 minutes. The optical absorbance of the reaction solution at 354 nm was measured on a UV–vis spectrophotometer. 44
Synthesis and characterization of HA-SS-MP
HA-SS-2MNA (30 mg) was dissolved in 3 mL of deionized water and incubated at room temperature for 4 hours. DMSO (5 mL) was added into the solution, then 1 mL of DMSO solution containing 60 mg of 6-MP was added, the pH was adjusted to 6.5 by acetic acid, and incubated for 24 hours at 37°C. The resulting conjugate was dialyzed against deionized water. After dialysis, the mixture was centrifuged for 3 minutes at 5000 rpm. The supernatant was dialyzed a further 6 times. HA-SS-MP was obtained by freeze-drying.
The procedure for characterizing the 6-MP grafting ratio was the same as described in the Synthesis Route 1 section.
Synthesis Route 3
Synthesis and characterization of HA-SS-MPD
HA-SH (450 mg) was dissolved in degassed PBS buffer (pH 8.0, 0.1 M) and the pH of the solution was adjusted to 6.0. The solution was incubated under an N2 atmosphere and incubated for 4 hours at room temperature. 2,2′-Dipyridyldisulfide (DPD) (220 mg) was dissolved in 12.5 mL of methanol. The 2 solutions were mixed together under an N2 atmosphere, and the mixture placed in a water bath and incubated at 37°C for 72 hours under constant shaking. The resulting conjugate was dialyzed 3 times against deionized water, and HA-SS-MPD was obtained by freeze-drying.
Determination of MPD grafting ratio
HA-SS-MPD (3 mg) was dissolved in 8 mL of deionized water. GSH (3 mg) was added to the solution and the mixture incubated for 30 minutes. The content of MPD was calculated by the absorbance of the reaction mixture at 343 nm. 45
Synthesis and characterization of HA-SS-MP
HA-SS-MPD (30 mg) was added to 3 mL degassed water under an N2 atmosphere and incubated for 4 hours at 37°C. DMSO (5 mL) was added in the solution, followed by 1 mL DMSO solution containing 60 mg of 6-MP; the pH was adjusted to 6.5 by acetic acid, and the mixture was incubated for 24 hours at 37°C. The resulting mixture was dialyzed against deionized water. After dialysis, the mixture was centrifuged for 3 minutes at 5000 rpm. The supernatant was dialyzed a further 6 times. HA-SS-MP was obtained by freeze-drying.
The procedure for determining the 6-mercaptopurine grafting ratio was the same as described in the Synthesis Route 1 section.
Selection of the Optimal pH for HA-SS-MP Synthesis
The synthesis method was the same as described in the Synthesis Route 3 section. 6-MP was added to the solution, and the pH was adjusted to 5.5, 6.5, and 7.5 by the addition of either acetic acid or 1 M NaOH solution, followed by incubation at 37°C for 24 hours.
Characterization of the 1H-NMR Spectrum
HA-SS-MP (5 mg) was dissolved in 5 mL of deuterium oxide. 1H-NMR spectra were recorded on a 400 MHz Varian NMR spectrometer (Bruker AVANCE III 400 NMR spectrometer).
Particle Size Measurement of HA-SS-MP
HA-SS-MP (4 mg) was dissolved in 4 mL of PBS buffer, pH 7.4, and incubated for 30 minutes at 37°C. The particle size of HA-SS-MP was measured using dynamic light scattering on a Malvern Zetasizer Nano-ZS90 (Malvern Instruments, Worcestershire, UK).
Drug Release Experiment
The pH value of blood in the human body is about 7.4 46 ; therefore, a release medium of pH 7.4 was selected. HA-SS-MP (7 mg) was added to 5 mL of potassium phosphate buffer (pH 7.4), and the solution was placed in a dialysis bag. This was put into 2 different release media (20 mL): (1) potassium phosphate buffer, pH 7.4 and (2) potassium phosphate buffer with 10 mM GSH, pH 7.4. The release experiment was performed at 37°C in a water bath at a rotating speed of 50 r/min. A small amount (0.5 mL) of the release medium was withdrawn from time to time with a constant interval. The same amount of fresh release medium was added back to the system to maintain the total volume.
Cell Uptake Assays
Synthesis of HA-cystamine
HA (0.1 g) was added to 8 mL 2-Morpholinoethanesulfonic acid (MES) solution (0.1 M), and the pH of the solution was adjusted to 4.7. EDC (239.6 mg) and NHS (115.1 mg) were added to the solution. After 15 minutes of incubation, cystamine dihydrochloride (562.4 mg) was added, and the pH of the solution was adjusted to 5.5. After 3 hours, this reaction mixture was dialyzed against deionized water for 12 hours. The dialysis treatment was repeated another 4 times. At last, HA-cystamine was isolated by freeze-drying.
Synthesis of fluorescent-labeled HA (HA-SS-FITC)
HA-cystamine (30 mg) was dissolved in 3 mL of sodium bicarbonate solution (pH 9.3) and incubated for 1 hour. DMSO (2 mL) solution containing 13 mg FITC was added and the mixture was incubated for 2 hours. The reaction mixture was dialyzed against water 5 times (each time for 12 hours) and then precipitated with ethanol. The precipitate was dried at 40°C to obtain HA-SS-FITC. The content of FITC in HA-SS-FITC was 39 mg/g.
Cell uptake assays
B16F10 (cultured in RPMI1640, supplemented with 10% FBS, 1% penicillin, and 1% streptomycin) and NIT/3T3 (cultured in DMEM supplemented with 10% FBS, 1% penicillin, and 1% streptomycin) cells were seeded in 12-well plates (2 × 106/well) and cultured for 24 hours. After incubation with 2.5 µg/mL of FITC labeled HA for 4 hours, the cells were washed 3 times with PBS (pH 7.4), then treated with 0.25% trypsin. The complete growth medium (0.5 mL) was added and blown by pipet to form a cell suspension, centrifuged, and resuspended in 0.35 mL of PBS. The fluorescence intensity of the cells was measured by flow cytometry (BECKMAN, FC500). To test whether the cell uptake of HA-SS-FITC was mediated by the CD44 receptor, 2.5 mg/mL of HA was added to B16F10 cells and incubated for 2 hours before the addition of HA-SS-FITC.
Cell Viability Assays
B16F10 cells were selected to test the pharmacological activity of HA-SS-MP. Cytotoxicity of HA-SS-MP and 6-MP against mouse melanoma cells B16F10 was assessed by MTT assays. Different concentrations of HA-SS-MP and 6-MP solutions were prepared. The cells were seeded in 96-well plates to a density of 3000 cells per well. After 24 hours of incubation, the serum-free RPMI1640 culture medium containing different concentrations of either HA-SS-MP or 6-MP (0.1, 1, 2.5, 10, and 25 µg/mL) was added to displace the old cell culture medium. After 24 hours of incubation, 20 µL of MTT PBS (5 mg/mL) was added to the cell culture medium and incubated for another 4 hours. The RPMI1640 culture medium was removed and 150 µL of DMSO was added. Finally, the optical absorbance of the solution in each well was measured at 490 nm, 47 and the cell viability was calculated based on the absorbance.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was financially supported by the National Natural Science Foundation of China (No. 81373338).
