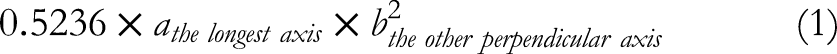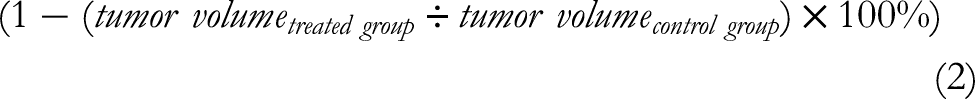Abstract
Introduction
Breast cancer is the most frequently diagnosed cancer and the leading cause of cancer-related deaths in women. 1 Among the various types of invasive breast cancer, triple-negative breast cancer (TNBC) accounts for 10%–20% of cases and exhibits a more aggressive growth and metastasis rate. TNBC cells lack estrogen and progesterone receptors and do not produce sufficient human epidermal growth factor receptor 2 (HER2) protein, rendering hormone therapy or targeted HER2 drugs ineffective. 2 Consequently, TNBC has limited treatment options and a poor prognosis. A study revealed that although patients with TNBC were more likely to receive an early diagnosis, they exhibited the lowest five-year relative survival rate (78%) for female breast cancer across all racial groups. 3 While chemotherapy remains the first-line treatment for TNBC patients, novel therapeutic options and strategies have emerged, such as the antibody-drug conjugate of Sacituzumab govitecan-hziy, immunotherapy of PD-L1 inhibitors, and peptide-decorated solid lipid nanoparticles for paclitaxel.4-8 Ongoing research seeks to identify additional targeted treatments and improve the efficacy of cytotoxic chemotherapy in TNBC.9,10
In recent years, cancer-targeted therapy has gained increasing attention due to its advantages of higher specificity, lower drug dosages, and reduced toxicity. 11 The tumor microenvironment (TME) plays a critical role in tumor initiation, progression, and metastasis by providing a favorable pH and environment for cancer cell growth and development. The extracellular matrix (ECM) is a major non-cellular component of the TME.12,13 Hyaluronic acid (HA), an important ECM component, is a primary ligand of the CD44 receptor. 14 CD44 is a cell surface glycoprotein involved in various biological processes, including proliferation, pathway regulation, and metastasis.15,16 Studies have shown that CD44 is highly expressed in various cancer cells, including breast cancer, and the CD44-HA interaction represents a potential target for therapy, facilitating communication between drugs and CD44-overexpressing tumor cells.17-20
Naturally occurring compounds have emerged as promising complementary medications to enhance cancer treatment with minimal side effects. 21 Geraniol, an acyclic monoterpene alcohol commonly extracted from aromatic plants, has been shown to inhibit cell proliferation and cell cycle progression in the G1 phase in human MCF-7 breast cancer cells. 22 Kazemi et al extracted a geraniol derivative from Artemisia kermanensis. It determined strong cytotoxic effects on the breast cancer cell lines MDA-MB-231 and MCF-7. 23 One study showed that Geraniol demonstrates promising antitumor effects in murine breast cancer models by suppressing tumor growth and enhancing 5-fluorouracil chemosensitivity via miR-21/PTEN signaling, highlighting its potential as a complementary therapeutic agent. 24 Geraniol has also exhibited anti-cancer effects in other types of cancer, including prostate, lung, pancreatic, and skin cancer. 25 In this study, the HA-geraniol conjugate was formulated further to explore its potential anti-cancer effects on TNBC. 26
Proteomic studies involve the comprehensive analysis of all proteins, including their identification, quantification, characterization, and modifications. 27 Protein expression levels can vary over time and in response to different treatments. Proteomics is a crucial tool in targeted drug development. It enables researchers to identify specific proteins for further drug discovery and design, potentially leading to more effective and personalized treatments for patients.28,29 Mass spectrometry (MS) is a commonly used technique for proteomic studies due to its high sensitivity and accuracy. 30 In this study, protein profiling was used to examine the impact of HA-geraniol on breast cancer cells and understand the potential mechanism of action. In addition, the anti-cancer activities of HA-geraniol in human breast cancer were investigated in an in vivo xenograft model.
Materials and Methods
Cell Line and Cell Culture
The murine 4T1 and human MDA-MB-231 breast cancer cell lines were purchased from ProCell Life Science & Technology (CL-007) (Wuhan, Hubei, China) and the Peking Union Medical College Cell Culture Center (1101HUM-PUMC000014) (Beijing, China), respectively. Both cell lines were cultured at 37 °C in a humidified incubator with 5% CO2, using RPMI 1640 medium (Corning; Jiangsu, China) supplemented with 10% fetal bovine serum (FBS; sourced from New Zealand) and 1% penicillin-streptomycin (Gibco, Thermo Fisher Scientific, Shanghai, China). Dr Duan's group prepared HA-geraniol conjugate. 26
Cytotoxicity
Cell Counting Kit-8 (CCK-8) was purchased from Elabscience (Wuhan, China) and used for determining cytotoxicity. 4T1 cells and MDA-MB-231 cells were seeded at a density of 2 × 103 cells/well and 8 × 103 cells/well in 96-well plates, respectively. After adherent growth overnight, 10 μL of different concentrations of geraniol (final concentrations: 57.12, 28.53, 5.71, 2.85, 0.58 to 0.28 mM) and HA-geraniol (final concentrations: 6.48, 3.24, 1.62, 0.32 to 0.16 mM) were added, and the plates were continued for 48 h of incubation. Later, 10 μL of CCK-8 solution was added for 4 h of culture and measured at 450 and 650 nm using a microplate reader (VarioskanFlash-4.00.53, Thermo Scientific Inc., Vantaa, Finland).
Mitochondrial Membrane Potential Loss
Cellstain® Rhodamine 123 (Rh-123) was purchased from Invitrogen (Thermo Fisher Scientific, Shanghai, China) to test the potential loss of the mitochondrial membrane. 4T1 and MDA-MB-231 cells were seeded in 6-well plates with a density of 1 × 106 cells/well overnight. Then, 4T1 cells and MDA-MB-231 cells were treated with a concentration of 1 mM and 0.5 mM for 24 h, respectively. Later, the cells were collected and resuspended in 1 mL Rh-123 working solution (20 μM in RPMI-1640). After 30 min of incubating in a 37 °C environment, the cells were washed and resuspended in 500 μL phosphate buffered saline (PBS). Mitochondrial membrane potential (ΔΨm) depolarization was analyzed using a NovoCyte 3000 flow cytometer (ACEA Biosciences, San Diego, CA) with 488 nm excitation. Data processing utilized FlowJo v10.8.1 (BD Biosciences, Franklin Lakes, NJ) and NovoExpress 1.5.0 (ACEA Biosciences).
Cell Apoptosis Induction
Annexin V-FITC Apoptosis Detection kit was obtained from Dojindo Molecular Technologies (Shanghai, China). The apoptotic induction of geraniol and HA-geraniol was examined after treatment with 1 mM in 4T1 and 0.5 mM in MDA-MB-231 cells. The cells were collected after treatment for 24 h, washed with PBS, and resuspended in 1× Annexin V Binding Solution. Later, 5 μL of Annexin V-FITC conjugate and 5 μL of PI solution were sequentially added to the cell suspension. The suspension was put in the dark for 15 min at room temperature and then tested by flow cytometry.
Cell Cycle Arrest
PI/RNase Staining kit was purchased from Dojindo Molecular Technologies for cell cycle distribution detection. After treatment with geraniol and HA-geraniol in 4T1 cells (1 mM) and MDA-MB-231 cells (0.5 mM) for 24 h, the cells were collected and fixed with 70% ethanol at 4 °C for 2 h. Then, the cells were washed and resuspended in 0.5 mL of working solution. The mixture was then incubated at 37 °C and 4 °C sequentially for 30 min each. Finally, the cells were filtered through a nylon mesh and determined by flow cytometry.
Proteomics Analysis
Sample Preparation for Proteomics
The MDA-MB-231 cells (1 × 107) were seeded in a T150 flask overnight and then treated with geraniol and HA-geraniol at a concentration of 1.30 mM for 24 h. Then, the cells were washed with pre-cooled PBS twice. Later, 0.25% trypsin-EDTA from Gibco was used to detach the adherent cells, and the cells (>5 × 106) were collected. Finally, the cells were frozen in liquid nitrogen for 5 min and kept in a −80 °C refrigerator. Five parallel samples were established for each group.
Protein Extraction and Digestion
Proteins were extracted using the SDT lysis method, which involved a mixture of 4% SDS, 100 mM Tris-HCl (pH 7.6), and 0.1 M DTT. The protein concentration was determined using a BCA protein assay kit from Bio-Rad (USA). Trypsin was used for protein digestion, following the filter-aided sample preparation (FASP) protocol. 31 Briefly, the sample was incubated at 37 °C for 1.5 h after adding 10 mM DTT. Then, an iodoacetamide solution was added, and the sample was further incubated for 30 min in the dark. The sample was washed with 100 μL of 8 M urea solution and 100 μL of 25 mM NH4HCO3 buffer. Next, trypsin was added, and the sample was digested overnight at 37 °C. After digestion, the peptides were desalted using C18 Cartridges (Empore™ SPE Cartridges C18, standard density, bed I.D. 7 mm, volume 3 mL, Sigma). The peptides were then concentrated using vacuum centrifugation and reconstituted in 40 µl of 0.1% (v/v) formic acid.
TMT Labeling and Fractionation
A total of 100 μg of peptides was labeled using the TMT reagent kit (TMT Mass Tagging Kits and Reagents, Thermo Fisher Scientific) and fractionated using the high-pH reversed-phase fractionation kit (Thermo Scientific) following the manufacturer's instructions.
Mass Spectrometry and Data Analysis
The experiments were conducted using a Q Exactive mass spectrometer (Thermo Scientific) coupled with an Easy nLC system (Proxeon Biosystems, now Thermo Fisher Scientific). Chromatographic separations were performed using a reverse phase trap column (Thermo Scientific Acclaim PepMap100, 100 μm × 2 cm, nanoViper C18) connected to a C18-reversed phase analytical column (Thermo Scientific Easy Column, 10 cm long, 75 μm inner diameter, 3 μm resin) at a flow rate of 300 nl/min. The mobile phase for the positive ion mode consisted of buffer A (0.1% formic acid in water) and buffer B (84% acetonitrile and 0.1% formic acid). Mass data was acquired using a data-dependent top 20 method, where the most abundant precursor ions from the survey scan (300-1800 m/z) were dynamically selected for higher-energy collisional dissociation (HCD) fragmentation. The main acquisition parameters were set as follows: automatic gain control target of 1e6, maximum injection time of 50 ms, dynamic exclusion duration of 30 s, survey scans of 60000 at m/z 200, resolution for HCD spectra of 15000 at m/z 200, isolation width of 1.5 m/z, and normalized collision energy of 30 eV. The instrument was operated with peptide recognition mode enabled.
The mass spectra data were searched using the MASCOT engine (Matrix Science, London, UK; version 2.2) integrated into Proteome Discoverer 2.4 software (Thermo Fisher Scientific). The key parameters used were as follows: enzyme: trypsin; maximum missed cleavages: 2; fixed modifications: carbamidomethyl (C), TMT 6/10/16 plex (N-term), TMT 6/10/16 plex (K); peptide mass tolerance: ± 20 ppm; fragment mass tolerance: 0.1 Da. A volcano plot was utilized to identify significant protein changes, considering fold changes and p-values. The Gene Ontology (GO) annotation was performed using Blast2GO software. For KEGG (Kyoto Encyclopedia of Genes and Genomes) annotation, the differentially expressed proteins were matched against the KEGG database online. Enrichment analysis was conducted using Fisher's exact test, with categories and pathways having p-values below the threshold of 0.05 considered statistically significant.
Animal Experiments (in Vivo Study)
The reporting of this study conforms to ARRIVE 2.0 guidelines. 32
Female BALB/c mice aged 6–8 weeks were obtained from the Zhejiang Experimental Animal Center (Hangzhou, China) with a weight range of around 20 g. Mice were divided into 3 groups (control group, geraniol group, and HA-geraniol group), with 6 mice in each group. Animals were maintained in a pathogen-free condition with a regular 12-h light/dark cycle and fed with autoclaved water and sterilizable food. All mouse studies were approved by the Experimental Animal Ethics Committee (approval no. WIUCAS22121501).
After 4 days of acclimatization, the mice were subcutaneously injected near their left mammary gland with 4T1 cells (1 × 106 cells per mouse) suspended in PBS. The cancers were allowed to accumulate for one week, and a two-week subcutaneous administration experiment was carried out. The mice were injected with corn oil in the control group, 100 mg/kg geraniol (diluted in oil) in the geraniol group, and 100 mg/kg geraniol equivalent HA-geraniol in the treatment group every 48 h. The tumor volumes were calculated every 48 h using the formula (1).
33
The formula (2) predicted the tumor growth inhibitory ratio. 34
Hematoxylin and Eosin (HE) Staining
Three lung tissues from each group were randomly chosen and dissected after two weeks of treatment. The tissues were infiltrated with 4% paraformaldehyde (PFA) over 24 h for further use. HE staining was applied to observe the histological changes of lung metastasis. Following standard histological protocols, tissues underwent ethanol gradient dehydration (65%-100%), xylene clearing, and paraffin embedding using a Leica Histocore Arcadia H system. Sections of 4–7 μm thickness were prepared with cryofixation at −20 °C, mounted on slides, and stained with hematoxylin (5 min) and eosin (10 min) using acid-alcohol differentiation. Dehydrated slides were cleared in xylene, mounted with neutral balsam, and imaged under a ZEISS AxioVert.A1 microscope.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 10.0.0 (GraphPad Software, San Diego, USA). Data are presented as mean ± standard deviation (SD) or standard error of the mean (SEM). Comparisons between groups were conducted using the Student's t-test, with significance set at p < 0.05 (*).
Results
Cytotoxicity
The cytotoxicity of murine 4T1 and human MDA-MB-231 breast cancer cells was tested after treatment with geraniol and HA-geraniol. In 4T1 cells, the IC50 values were 1.82 mM under geraniol treatment and 211.35 μM under HA-geraniol treatment. After treatments with geraniol and HA-geraniol in MDA-MB-231 cells, the IC50 values were 1.04 mM and 202.27 μM, respectively (Figure S1). The anti-cancer effects were significantly enhanced after treatment with nearly a 9-fold lower dose and a 5-fold lower dose of HA-geraniol treatment. Moreover, HA did not show cytotoxicity in both cell lines (Figure S2).
Mitochondrial Membrane Potential Loss
Rh-123 is a lipophilic cation dye that can penetrate the cell membrane and accumulate in the mitochondria of living cells with yellow-green fluorescence. The dye was used to assess mitochondria's membrane potential loss rate after geraniol and HA-geraniol treatments in 4T1 cells and MDA-MB-231 cells. The membrane potential loss rate increased from 7.26 ± 0.23% of the control group to 12.53 ± 0.06% of the geraniol group and 39.97 ± 1.11% of the HA-geraniol group in 4T1 cells. In MDA-MB-231 cells, the membrane potential loss rate of the HA-geraniol group was significantly higher, with a value of 30.07 ± 0.76% compared with 1.52 ± 0.12% of the control group and 1.62 ± 0.04% of the geraniol group (Figure 1).

Representative Flow Cytometry Histograms Showing Mitochondrial Membrane Potential Loss in 4T1 (A) and MDA-MB-231 (B) Cells Treated with Geraniol or HA-Geraniol, and Quantification of Membrane Potential Loss in 4T1 (C) and MDA-MB-231 (D) Cells (Mean ± SD, *p < 0.05, n = 3).
Cell Apoptosis Induction
Flow cytometry was used to determine the apoptotic effects of geraniol and HA-geraniol in both 4T1 and MDA-MB-231 breast cancer cells. The cells were treated with an Annexin V-FITC Apoptosis Detection Kit, which contained propidium iodide (PI) dye that could only penetrate the membrane of late apoptotic and dead cells and Annexin V that binds specifically to the membrane of early apoptotic cells. The apoptotic rate was nearly 10-fold higher, with a value of 58.27 ± 0.32% in the HA-geraniol group than the control group of 6.07 ± 1.48% in 4T1 cells. The apoptotic rate in the geraniol treatment was 26.75 ± 1.29% in the 4T1 cells. Under the treatments of geraniol and HA-geraniol in MDA-MB-231 cells, the apoptotic rates were enhanced from 8.71 ± 2.32% of the control group to 15.32 ± 2.74% of the geraniol group and 69.33 ± 0.32% of the HA-geraniol group. The results strongly demonstrated the cell apoptosis-induction effect of HA-geraniol (Figure 2).
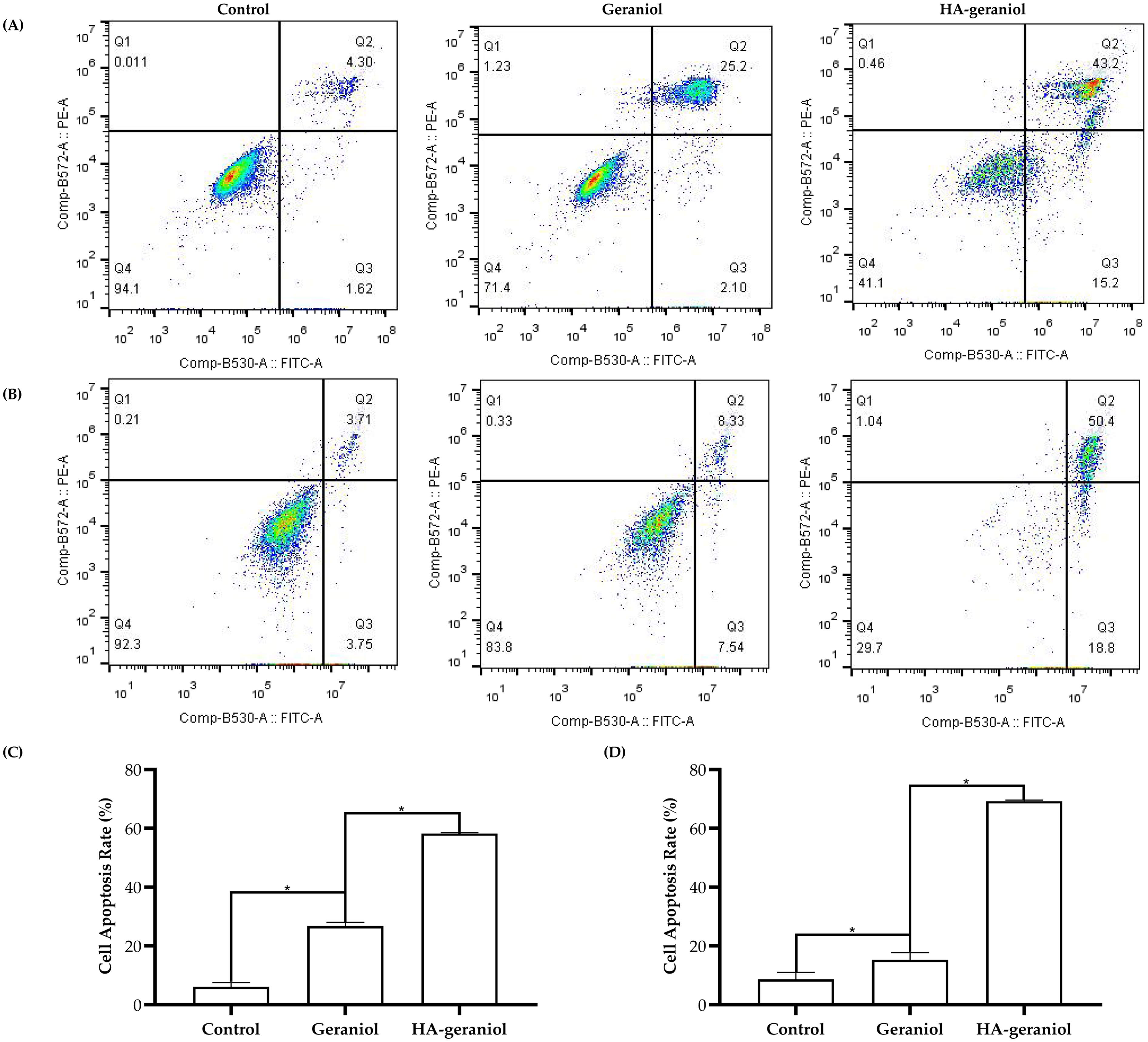
Representative Flow Cytometry Results Demonstrating Apoptosis Induction in 4T1 (A) and MDA-MB-231 (B) Breast Cancer Cells Treated With Geraniol or HA-Geraniol, and Quantification of Apoptotic Rates in 4T1 (C) and MDA-MB-231 (D) Cells (Mean ± SD, *p < 0.05, n = 3).
Cell Cycle Arrest
The cell distributions of 4T1 cells and MDA-MB-231 cells were studied using PI/RNase staining kits (Dojindo, C543) after treatments with geraniol and HA-geraniol. PI was used as a fluorescent DNA dye to obtain the proportion of G0/G1, G2, and S phase cells in the cell cycle by flow cytometry. After geraniol and HA-geraniol treatments, the cells were arrested in the G1 phase at 32.57% in the geraniol group and 38.87% in the HA-geraniol group, compared to 24.83% in the control group in 4T1 cells. Furthermore, geraniol and HA-geraniol treatments arrested the cells in the G2 phase with 31.89% of the geraniol group and 38.97% of the HA-geraniol group, compared with 29.84% of the control group in MDA-MB-231 cells (Figure 3).

Representative Cell Cycle Analysis Results in 4T1 (A) and MDA-MB-231 (B) Breast Cancer Cells Treated with Geraniol or HA-Geraniol.
Differential Expression of the Proteins
To investigate the antitumor mechanism of HA-geraniol on MDA-MB-231 cells, we employed mass spectrometry-based quantitative proteomics in this study. A total of 7227 proteins with unique peptide sequences were identified and quantified. Three hundred eighty-eight differentially expressed proteins were detected in the HA-geraniol group compared to the geraniol group. Among these, 173 proteins were up-regulated and 215 proteins were down-regulated, as illustrated in Figure 4. The top 10 up-regulated and down-regulated proteins are presented in Table 1.

Volcano Plot Analysis of Differentially Expressed Proteins. The Plot Displays 173 Significantly Up-Regulated Proteins (Red Dots, Fold Change >1.2, p < 0.05) and 215 Down-Regulated Proteins (Blue Dots, Fold Change <0.83, p < 0.05). The X-axis Represents log2 Fold Change Values, While the Y-axis Shows -log10 Transformed p-values.
The Top 10 Up- and Down-Regulated Proteins in MDA-MB-231 Cells After HA-Geraniol Treatment.

Gene Ontology Functional Enrichment and Pathway Enrichment Analysis of Differentially Expressed Proteins
Annotation and enrichment analyses of the differentially altered proteins using GO and KEGG were performed to analyze the functional characteristics and signaling pathways further. The GO analysis included three categories: biological process, molecular function, and cellular component. The top 10 significantly enriched items are presented in Figure 5. Regarding biological process annotation (Figure 5A), the proteins were primarily involved in response to stress and tissue development. In the molecular function category (Figure 5B), the three most enriched terms were signaling receptor binding, signaling receptor activity, and molecular transducer activity. The cellular component analysis revealed that the three predominantly enriched terms were plasma membrane part, endoplasmic reticulum lumen, and cell surface (Figure 5C).

GO Enrichment Analysis of Differential Proteins. The Top 10 Enriched Terms are Shown Across Three Categories: Biological Process (BP), Molecular Function (MF), and Cellular Component (CC). Notable Findings Include: “Response to Stress” (BP, 141 Proteins), “Signaling Receptor Binding” (MF, 45 Proteins), and “Plasma Membrane Part” (CC, 63 Proteins).
For KEGG pathway analysis, pathways with a p-value < 0.05 were significantly enriched. Figure 6 displays the top 20 KEGG pathways, with the complement and coagulation cascades identified as the most enriched category. The differentially expressed proteins are prominently featured in the signaling pathway map (Figure 7). It can be observed that most of the altered proteins, including coagulation factor V, plasminogen activator inhibitor 1, clusterin, vitronectin, prothrombin, alpha-2-macroglobulin, C4b-binding protein alpha chain, coagulation factor X, antithrombin-III, and complement C4-A, were up-regulated after exposure to HA-geraniol.

KEGG Pathway Enrichment Analysis of Differential Proteins. The Bubble Plot Highlights Complement and Coagulation Cascades as the Most Significantly Enriched Pathway (p = 2.84 × 10−7, 11 proteins). The X-axis Represents the Enrichment Factor, the Bubble Size Indicates Protein Count, and the Color Gradient from Yellow to Red Reflects Decreasing p-values.

Pathway “the Complement and Coagulation Cascade” from KEGG, with Altered Proteins Mapped. This Includes 10 Up-regulated Proteins (Indicated in red) and 1 Down-Regulated Protein (Highlighted in Green), All Labeled with Pentagrams.
In Vivo Study
HE staining of lung tissues was conducted to investigate the potential metastasis. Diffuse inflammatory cell infiltration was observed in all groups, primarily neutrophils and macrophages. The control group displayed small focal lung metastases, while the geraniol group showed single-cell metastases (Figure 8A and B). However, in the HA-geraniol group, no apparent metastasis was observed (Figure 8C). The tumor volumes were also measured to assess the inhibitory effects of geraniol and HA-geraniol treatments on tumor growth. Following two weeks of treatment, we observed that geraniol led to a 31.68% inhibition of tumor growth compared to the control group. Remarkably, the HA-geraniol group exhibited a tumor inhibitory rate of 75.02% (Figure 8D).

In Vivo Studies of Lung Metastasis and the Anti-Tumor Effect. Representative HE Staining of Lung Tissue from Control (A), Geraniol (B), and HA-geraniol (C) Treated Mice Following Subcutaneous Injection of MDA-MB-231 Cells. Representative Tumor Metastases are Indicated with Circles. Scale Bars = 50 µm for 40X Magnification. (D) A Line Chart Illustrating Tumor Volumes among Control and Drug-Treated Mice (n = 4). Error Bars Represent the mean ± SD (* p < 0.05).
Discussion
TNBC is an aggressive subtype with a poor prognosis, and current research efforts are focused on developing effective treatment strategies. 35 Targeted drug delivery systems have emerged, promising to enhance drug efficacy while minimizing side effects. One such delivery system utilizes HA as a ligand to target CD44, a cell surface receptor overexpressed in many types of cancer, including TNBC.36,37 Previous studies have demonstrated the anti-cancer effects of geraniol, a natural compound, on human MCF-7 breast cancer cells by inhibiting cell growth and inducing cell cycle arrest. 25 In this study, we developed an HA-geraniol conjugate via a carbon-carbon bond and evaluated its efficacy on high CD44-positive expression in human MDA-MB-231 and murine 4T1 TNBC cell lines. Our cytotoxicity studies revealed a dose-dependent toxic effect of geraniol and HA-geraniol on both cell lines, with the HA conjugate demonstrating a more potent anti-cancer effect at lower dosages. Additionally, treatment with HA resulted in minimal cytotoxicity in both cell lines, supporting the high selectivity and biocompatibility of the HA delivery system.
Apoptosis is a highly regulated form of cell death that can be initiated through two main pathways: the extrinsic and intrinsic pathways. The extrinsic pathway is activated when cell-surface death receptors, such as Fas, bind to their ligands. Conversely, the intrinsic pathway is triggered by pro-apoptotic proteins from the BCL-2 family, which cause the permeabilization of the mitochondrial outer membrane. 38 Mitochondrial membrane potential (ΔΨm) can be monitored by suppressing Rh-123 fluorescence, which depends on mitochondrial energization. 39 One of the key characteristics of apoptosis is the externalization of phospholipid phosphatidylserine (PS) through translocation from the inner to the outer layer of the plasma membrane. 40 Previous studies have shown that plant essential oils can induce apoptosis through the mitochondria. 41 This study found that the HA-geraniol conjugate significantly led to a loss of ΔΨm in both cell lines. Annexin V/PI staining also revealed a dominant effect of HA-geraniol in inducing cell death. Our results support the involvement of HA-geraniol in the mitochondria-mediated apoptotic pathway. Furthermore, geraniol induced cell cycle arrest in the G1 phase of 4T1 cells and the G2 phase of MDA-MB-231 cells, leading to cell death.
Proteomics, which involves the large-scale study of proteins within a biological system, provides valuable insights into drug development, efficacy, toxicity, and resistance evaluation. 42 This study identified 388 differentially expressed proteins in the HA-geraniol treatment group compared to the geranial group. The GO analysis revealed a significant enrichment of differentially expressed proteins involved in stress response. This finding suggests that HA-geraniol treatments have a more pronounced impact on cellular stress responses, increasing the sensitivity of cells to stress stimuli. Among the differentially expressed proteins, a stress-induced transcription factor called Cyclic AMP-dependent transcription factor ATF-3 (ATF3) exhibited the most significant fold change, as shown in Table 1. ATF3 belongs to the ATF/cAMP response element-binding family of transcription factors and plays important roles in various cancers, including prostate, breast, colon, lung, and liver. 43 Recent studies have reported that ATF3 can act as a tumor suppressor by promoting apoptosis and cell cycle arrest, inhibiting proliferation, and enhancing immune responses against cancer cells.44-47 This study observed an up-regulation of ATF3 in breast cancer cells treated with HA-geraniol compared to the geranial group. It suggests that HA-geraniol treatment may enhance the expression of ATF3, potentially leading to cell apoptosis. However, further research is needed to elucidate the mechanism by which ATF3 induces cell death in HA-geraniol treatments.
Next, based on the proteomic data, an enrichment analysis of KEGG signaling pathways was performed. The results revealed that the main differential pathway in HA-geraniotreated breast cancer cells was the complement and coagulation cascades. These two interconnected pathways play critical roles in immune responses, inflammation, and hemostasis.48,49 It is important to note that cross-interactions between these cascades can be simultaneously activated.50,51 According to the findings depicted in Figure 7, a significant number of differentially quantified proteins within the complement and coagulation cascades exhibited upregulation. This indicates pathway activation and the facilitation of cell membrane lysis. This observation aligns with the work of Sarma and Ward, who reported that complement activation leads to the opsonization of pathogens, their elimination by phagocytes, and cell lysis.52,53 Furthermore, our GO cellular component (CC) analysis supported these findings, as the most enriched term was associated with the plasma membrane part. This suggests that the differentially expressed proteins within the complement and coagulation cascades are primarily localized to the plasma membrane.
The in vivo study provided strong evidence for the significant tumor-inhibitory effects of HA-geraniol treatment, indicating its potential to suppress tumor growth effectively. Additionally, the HE staining study demonstrated the notable inhibitory ability of HA-geraniol in preventing metastasis, thereby impeding the spread of cancer cells to other areas of the body.
While our study provides strong evidence of the enhanced anti-cancer effects of HA-conjugated geraniol, several limitations should be acknowledged. Firstly, our research primarily focused on in vitro experiments using cell lines, which may not fully replicate the complexities of in vivo tumor environments. Although we conducted an in vivo study, it's essential to note that the murine cell line utilized may not perfectly mimic human physiology. Additionally, further investigation is needed to elucidate the specific mechanisms underlying the observed effects, particularly regarding the involvement of complement and coagulation cascade pathways in apoptosis induction and cell cycle arrest.
Conclusions
Our results provide solid evidence for the therapeutic potential of HA-geraniol, a CD44-targeted hyaluronic acid conjugate of natural geraniol, demonstrating dual-action therapeutic innovation through electively inducing mitochondrial apoptosis in TNBC cells while suppressing metastasis, which isa dual mechanism not previously reported for geraniol formulations. Proteomics revealed novel ATF3-driven complement pathway activation, providing a blueprint for targeting therapy-resistant TNBC. With validated efficacy in a translational 4T1 model and a favorable safety profile, this work establishes HA-geraniol as a candidate, highlighting its clinical promise for reducing systemic toxicity while addressing both primary tumors and disseminated lesions. Future efforts will explore synergistic combinations with immune checkpoint inhibitors to translate this phytochemical-engineered hybrid into precision oncology pipelines.
Supplemental Material
sj-tif-1-npx-10.1177_1934578X251344985 - Supplemental material for Hyaluronic Acid Conjugated Geraniol for Targeted Breast Cancer Therapy
Supplemental material, sj-tif-1-npx-10.1177_1934578X251344985 for Hyaluronic Acid Conjugated Geraniol for Targeted Breast Cancer Therapy by Han Yu, Lingling Jiang, Yindu Bai, Jing Wu, Na Ning, Shaofeng Duan and Yunqi Zhao in Natural Product Communications
Supplemental Material
sj-tif-2-npx-10.1177_1934578X251344985 - Supplemental material for Hyaluronic Acid Conjugated Geraniol for Targeted Breast Cancer Therapy
Supplemental material, sj-tif-2-npx-10.1177_1934578X251344985 for Hyaluronic Acid Conjugated Geraniol for Targeted Breast Cancer Therapy by Han Yu, Lingling Jiang, Yindu Bai, Jing Wu, Na Ning, Shaofeng Duan and Yunqi Zhao in Natural Product Communications
Footnotes
Acknowledgements
This work was financially supported by the General Program of Zhejiang Provincial Natural Science Foundation (ZCLTGY24C1001), Wenzhou-Kean University Internal Faculty Research Support Program, (IRSPG202102), Wenzhou-Kean University International Collaborative Research Program (ICRP202201), Wenzhou-Kean University 2023 & 2022 Student partnering with Faculty Research Program (WKUSPF2023023 & WKUSPF202226).
Ethical Considerations
The animal study protocol was approved by the Institutional Review Board of the University of the Wenzhou Institute, Chinese Academy of Sciences (protocol code WIUCAS221221501, December 15, 2022).
Author Contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the General Program of Zhejiang Provincial Natural Science Foundation (ZCLTGY24C1001), Wenzhou-Kean University Internal Faculty Research Support Program, IRSPG202102; Wenzhou-Kean University International Collaborative Research Program, ICRP202201, Wenzhou-Kean University Student Partnering with Faculty Research Program, WKUSPF2023023 & WKUSPF202226.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are contained within the article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.