Abstract
The pericarp of
α-Mangostin is a xanthone derivative present in the pericarp of
The use of α-mangostin is limited due to its low water solubility, low bioavailability, and poor blood penetration.
25
As a result, several attempts have been made to improve these limitations, including co-solvation, structure modification, solid dispersion, and nano-sized drug delivery systems.
26
-29
Recently, nanotechnology has allowed effective and sustained delivery of a large number of high-value compounds, either by conjugating or encapsulating them into various carrier systems such as nanopolymerics, nanoliposomes, and metal nanoparticles.
30
-32
Particularly, due to several advantages such as versatility, high stability in various biological environments, high agent-loading efficiency, low toxicity, and long circulation time, liposomes are considered as an excellent delivery agent for several antioxidant and anti-cancer agents of high importance.
33,34
For example, low water-soluble curcumin released from liposome formulations has exhibited significant cytotoxicity against bone cancer cells.
35
Similarly, butyric acid, which is mainly involved in reducing inflammation and cancer in liver, was loaded onto chitosan-coated liposomesand showed improvements in terms of oral bioavailability and cytotoxicity to Hep-G2 hepatoblastoma cells.
36
Previous studies have also shown that liposome formulation has enhanced the solubility and
In the present study, α-mangostin was isolated and its identity was verified by nuclear magnetic resonance (NMR) and Fourier-transform infrared (FTIR) spectroscopy. The compound was then loaded onto newly synthesized liposomes and evaluated for
Materials and Methods
Materials
Healthy mangosteen fruits were collected at Cau Giay District, Hanoi, Vietnam in April 2019. The pericarps were removed from the seeds, dried and milled into fine powder before extraction. Methanol (MeOH), ethanol (EtOH), chloroform, n
Extraction of α-Mangostin
Powdered mangosteen fruit pericarps (1 kg) were macerated with ethanol (2 L × 3 times) at room temperature for 6 days. The combined extracts were filtered and evaporated under reduced pressure to form 144.9 g of dark brown residue. After dispersing in purified water, the mixture was extracted with n
The n

Isolation of α-mangostin from mangosteen pericarps.
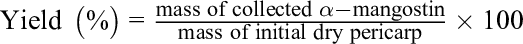
Synthesis of Liposome
Briefly, a mixture was prepared by combining 800 µL stock DOPC (0.025 M; 20 µmol) with 100 µL stock Chol solution (0.01 M; 1 µmol) and 500 µL stock α-mangostin (0.02 M; 10 µmol). The solvent in the mixture was removed slowly by rotary evaporation in a water bath at 30 °C, with a flask pressure of 250 mbar, and rotational speed at level 2, leaving a thin membrane composed of DOPC, Chol, and α-mangostin. Distilled water (4 mL) was added to peel off the membrane, forming α-mangostin-loaded liposomes of varying sizes. In order to reduce polydispersity and assimilate the liposomes into defined size ranges, a mini extruder was used with 100 nm polycarbonate membranes. Each sample was subjected to 50 rounds of extrusion, with each round counted as one passing of the suspension through the polycarbonate membrane. Once extrusion was finished, the samples were transferred to a new Eppendorf flask, which was capped and stored at room temperature (25 °C).
Unencapsulated α-mangostin was removed by dialyzing the liposome suspension obtained from thin film hydration. A square of dialysis tubing (14 000 Da, 10 cm × 10 cm) was hydrated in distilled water and used to cap the dram vial. To prevent leakage of the suspension, circular rubber washers and rubber bands were used to tighten the cap. The suspension was dialyzed for 24 hours against 1 L of distilled water. The distilled water was changed every 2 hours from the start, for 4 times.
Characterization of Liposome
The
To determine the encapsulation efficiency (EE%) and drug loading content (DL%), the samples were transferred to a 100-mL round-bottom flask to perform rotary evaporation (bath temperature of 45 °C, flask pressure of 0 mbar, rotational speed at level 3) until all the water had been removed and a thin film was left in the flask. The lipids/α-mangostin film was dissolved in 3.5 mL of MeOH/CHCl3 (3:1 [v/v]) and sonicated in a bath for 2 minutes to destroy the liposomal suspensions and release the entrapped α-mangostin. Then, 100 µL of this solution was pipetted from the flask, added to 2900 µL of MeOH/CHCl3 (3:1 [v/v]) in an Eppendorf tube, and vortexed for 30 seconds. The mixture was then transferred to a glass cuvette. The absorbance of the sample was determined at 243 nm against a MeOH/CHCl3 (3:1 [v/v]) standard with a Hitachi U-2900 spectrophotometer (Hitachi, Tokyo, Japan). The EE% and DL% could be calculated following the equations.

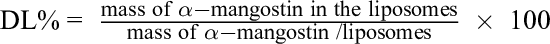
In Vitro Drug Release
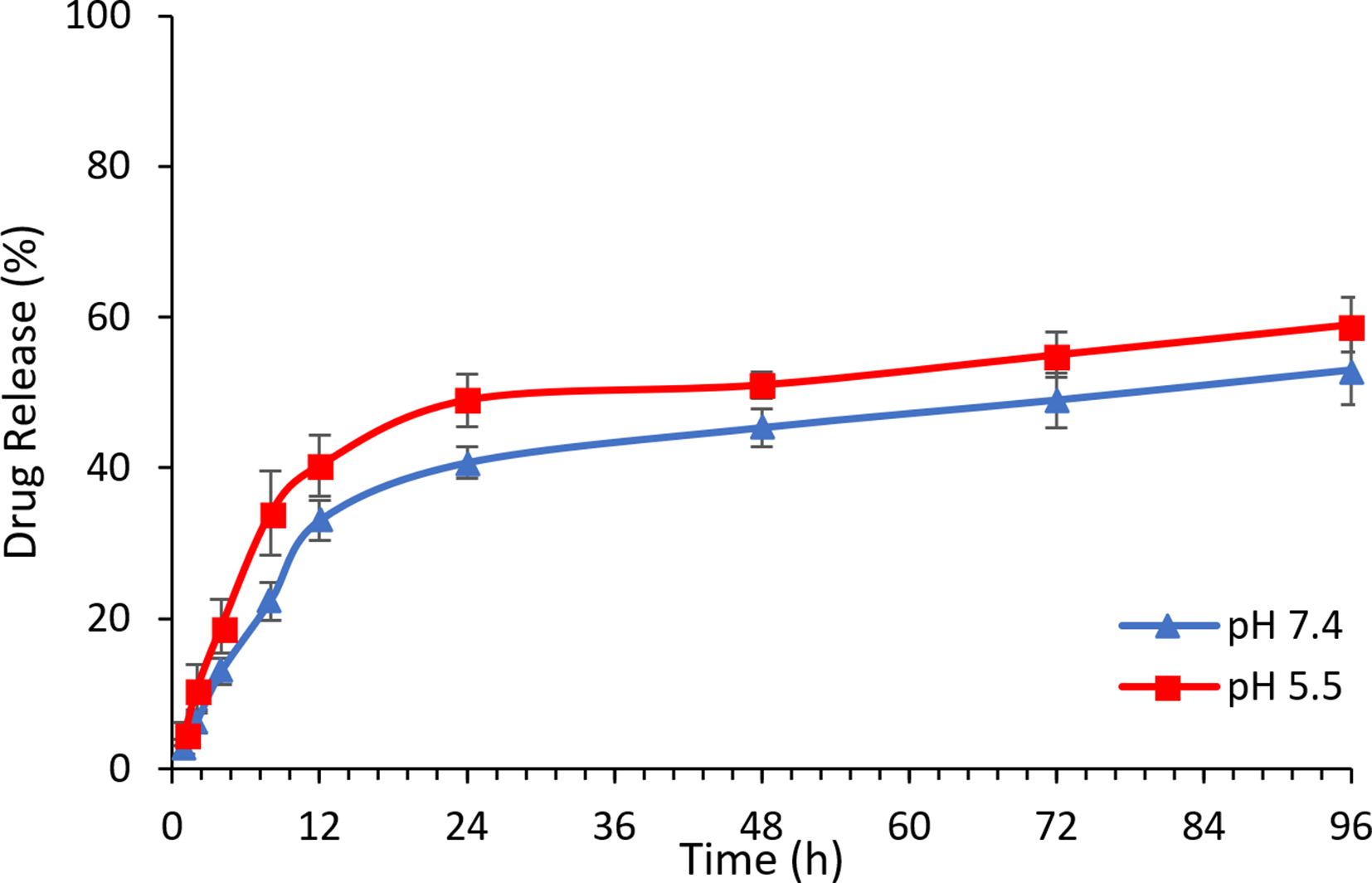
The release profile of α-mangostin from α-mangostin-loaded liposomes was investigated using a dialysis method. Liposomes loaded with α-mangostin were placed into dialysis bags with a molecular weight cutoff of 14 000 Da, then incubated in PBS at pH 5.5 and 7.4% and 0.5% Tween 80 with agitation (100 rotations/min) at 37 °C. At an appropriate time interval, aliquots (1 mL) were withdrawn and replaced with the same volume of fresh buffer. The samples were subjected to analysis using an ultraviolet-visible spectrophotometer to calculate the cumulative amount of released α-mangostin. All data were the mean value of 3 replicates and shown as mean ± SD.
In Vitro Cytotoxicity Assay
A single layer of human hepatocellular carcinoma (Hep-G2) cells (American Type Culture Collection, Manassas, VA, USA) was cultured in DMEM. The conditions to maintain the cells included 37 °C with a 5% CO2 atmosphere. Exponentially growing cells were used for further experiments.
Free α-mangostin, blank liposome, and α-mangostin-loaded liposomes were evaluated for their cytotoxicity by using MTT assay. Briefly, cells were seeded and incubated in DMEM (100 µL) for 24 hours in order to allow cell growth and adherence to the plate surface. The grown cells were treated with various concentrations of free α-mangostin, blank liposome, and α-mangostin-loaded liposomes (0- 20 µM) and incubated for 96 hours. Then, MTT reagent (5 mg mL-1) was added and incubated for another 4 hours at 37 °C. The medium in each well was removed and the formazan crystals were dissolved by dimethyl sulfoxide. Absorbance was measured at 570 nm using a Spectramax M5 Microtiter Plate Luminometer (Molecular Devices, San Jose, CA, USA).
Statistical Analysis
Two-tailed Student’s test was used to compare the statistical significance between pairs of samples.
Results and Discussion
Extraction of α-Mangostin
The yield of α-mangostin isolated from 1 kg of dry pericarp of
Synthesis of Liposome
Liposomes were produced by the thin-film hydration method, combined with extrusion through a polycarbonate membrane (0.1 µm). The physicochemical properties of the optimal blank liposome and α-mangostin-loaded liposome formulation were studied. The mean size of the blank liposome was 106.4 ± 9.1 (nm) and the α-mangostin-loaded liposome 109.3 ± 7.2 (nm). The PDI of the blank liposome was 0.185 ± 0.015 (nm) and the α-mangostin-loaded liposome 0.181 ± 0.017 (nm). The differences between the mean size and PDI of the blank liposomes and α-mangostin-loaded liposomes were insignificantly different.
The DL% and EE% of α-mangostin-loaded liposomes were 2.39% ± 0.23% and 55.3% ± 2.3%, respectively, while the particle size was around 100 nm with a PDI <0.02, indicating high entrapment efficiency and narrow distribution. These findings for the liposome model suggest its potential to improve the therapeutic efficacy of α-mangostin.
As the internal environments of tumors and several cell compartments have been previously shown to have a relatively low pH, this factor was also taken into consideration when assessing the
MTT Viability Assay
The cytotoxicity of α-mangostin-loaded liposomes, blank liposomes, and free α-mangostin was tested on Hep-G2 cells using standard MTT assay during 96 hours of experiment. As can be seen in Figure 3, the blank liposomes showed no apparent cytotoxicity with more than 80% of cells surviving when testing up to 20 µM blank liposome. In contrast, the half-maximal inhibitory concentration value of α-mangostin-loaded liposomes was decreased to 1.9 µM, as compared with 4.6 µM for free α-mangostin. Apparently, there were significant differences in cell viability between α-mangostin-loaded liposomes and blank liposomes, as well as free α-mangostin and blank liposomes at both 10 and 20 µM (

Cytotoxic effects of free α-mangostin, blank liposomes, and α-mangostin-loaded liposomes on HepG2 cells (*
The cytotoxicity investigation of α-mangostin was extended to different time points, including 2 (after incubation), 12, 24, and 96 hours culture (Figure 4). Without treatment, the cell number increased gradually from 2 to 12 hours, then it peaked at 48 hours (Figure 4(A1-C1)). However, cells started to decrease steeply after 96 hours due to confluence and nutritional deficiencies after prolonged culture time.

Effects of different concentrations of blank liposome, free α-mangostin, and α-mangostin-loaded liposome on Hep-G2 cell proliferation during 96 h of incubation. Data presented as mean ± SEM (n = 3).
The blank liposomes exhibited no apparent cytotoxicity to Hep-G2 cells, even at a high concentration (20 µM), and cell viability was not significantly different from the control (Figure 4(A1-6)). In the presence of free α-mangostin (0.1, 20 µM), the cell viability was found to vary with concentrations during the first 24 hours, yet as α-mangostin was absorbed expeditiously, the cells rehabilitated promptly at 96 hours culture (Figure 4(B2-6)). Furthermore, the cells were significantly damaged and unable to recover when treated with 20 µM of free α-mangostin. In the α-mangostin-loaded liposome group, α-mangostin was released slowly and the cells absorbed it gradually (Figure 4(C2-6)). At the same concentration, the α-mangostin-loaded liposomes killed less cells than the free α-mangostin within 24 hours of treatment. Nevertheless, as α-mangostin was continually liberated from liposomes, the cell viability was reduced markedly after 48 and 96 hours, especially at 10 and 20 µM of treatment. Therefore, by loading α-mangostin onto liposomes, α-mangostin exhibited an enhanced cytotoxic effect against Hep-G2 cells.
Conclusions
In the present study, α-mangostin was obtained from mangosteen pericarps with the yield of 1.86%. The obtained α-mangostin was then loaded onto liposomes and showed good results in terms of compound release and EE%. The α-mangostin-loaded liposomes also exhibited a more effective cytotoxic effect against Hep-G2 cells (1.9 mΜ), as compared with free α-mangostin (4.6 mM). Current results suggested that the utilization of liposomes for α-mangostin delivery is a promising method to enable the development of α-mangostin into a more efficient chemotherapeutic agent in the future.
Footnotes
Acknowledgment
The authors gratefully acknowledge the support by project code QTBY01.03/20-21.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
