Abstract
The present study aimed to determine the phenolic compounds of Aristolochia longa root extracts and to evaluate their antibacterial activities on multiresistant strains. Phytochemical analysis revealed the presence of flavonoids, tannins, terpenoids, and alkaloids. The HPLC-DAD analysis of A. longa extracts showed the presence of several major bioactive compounds such as ferulic acid, 4-hydroxycinnamic acid, citric acid, and quinic acid. The agar diffusion method was used for the sensitivity test, while minimal inhibitory concentration (MIC) and minimal bactericidal concentration values were determined by microdilution assay. Different tests were carried out on 3 clinical multiresistant strains and 3 reference strains. The diameter of inhibition of Staphylococcus aureus ATCC 25923 induced by the ethyl acetate fraction at 200 mg/mL was 25 ± 1 mm. Moreover, Escherichia coli ATCC 29522 showed a great sensitivity toward all the concentrations tested. The MICs of the active extracts vary between 12.5 and 100 mg/mL with a bacteriostatic effect on Pseudomonas aeruginosa ATCC 27853, Enterococcus faecalis, and S. aureus ATCC 25923.
The emergence of bacteria resistant to many conventional antibiotics is a major public health problem. 1 The issue of antibiotic resistance challenges the scientific community because it is important to find new natural antimicrobial agents. Medicinal and aromatic plants have been used for centuries to treat human diseases. Extracts of plants were already known and used by different civilizations (Egyptians, Greeks, Chinese, etc.) in traditional medicine. Aristolochiaceae family includes nearly 500 species for most tropical, subtropical, and Mediterranean countries. 2 This family has been reported in the forest of America, Asia, Africa, Europe, and rarely in other continents. Aristolochia longa, Mediterranean specie in North Africa, known as “Barraztam,” was recommended since antiquity against ovarian insufficiency and snake bites. This species was employed to treat different diseases such as cancer, diabetes, asthma and skin, and intestinal affection. Moreover, different parts of this plant are used with several combination with other forms such as honey, milk, and juice. 2 This work aimed to carry out phytochemical investigation of A. longa root extracts growing spontaneously in Morocco, and the investigation of their antibacterial activity.
The yield of the ethyl acetate fraction was 8.4%, the methanol fraction 3.2%, the aqueous fraction 1.2%, and the aqueous extract 4.2%. It has been reported that the polarity of the solvent influences the extraction rate, 3 and the extraction efficiency depend on many parameters, including extraction time and temperature, volume and type of solvents used. 4 The phytochemical analysis revealed that A. longa is rich in phytochemical compounds as summarized in Table 1. These chemical compounds belong to different chemical families such as flavonoids, tannins, terpenoids, and alkaloids.
Phytochemical Screening Results of Root Powder of Aristolochia longa Extracts.

+++, High; ++, average; +, low; −, absence.
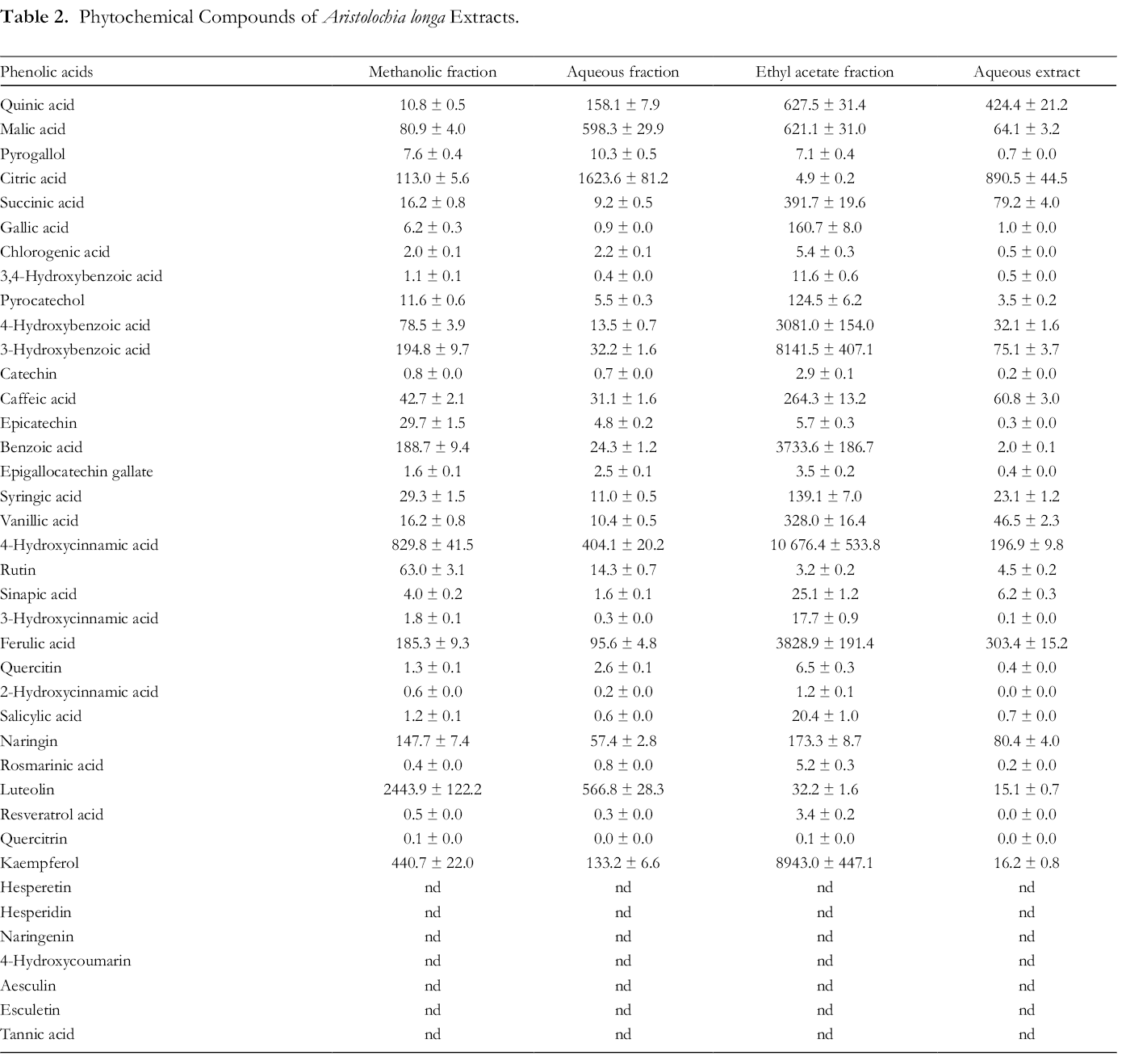
Phytochemical compounds of A. longa extracts were determined using HPLC-DAD/TOF-MS analysis (see Supplemental Data). Phenolic compounds of A. longa extracts are summarized in Table 2. As listed, all extracts of A. longa are rich in phenolic compounds. Citric acid is the main compound of the aqueous fraction (1623.6 ± 81.2 µg/mg extract) and the aqueous extract (890.5 ± 44.5 µg/mg extract). The main compounds of the methanolic fraction were citric acid (113.0 ± 5.6 µg/mg extract) and luteolin (2443.9 ± 122.2 µg/mg extract). Moreover, the major phenolic components of the ethyl acetate fraction are 4-hydroxybenzoic acid (3081.0 ± 154.0 µg/mg extract), 3-hydroxybenzoic acid (8141.5 ± 407.1 µg/mg extract), benzoic acid, 4-hydroxycinnamic acid (3733.6 ± 186.7 µg/mg extract), ferulic acid, and kaempferol (10 676.4 ± 533.8 µg/mg extract). The variability of phenolic compounds in ethyl acetate fraction is certainly related to the solvents of extraction. Other studies have reported that A. longa is rich in bioactive compounds such as limonene, β-carotene, and palmitic acid, which have proven their pharmacological effects. 5,6
Phytochemical Compounds of Aristolochia longa Extracts.
The antibacterial activity of extracts was determined using agar-well diffusion assay and agar-disk diffusion assay (see Supplemental Data). The inhibition diameters of A. longa extracts against tested bacteria determined by disk diffusion assay are shown in Table 3. It was noted that these diameters differ from one bacteria to another and from one extract to another. The variation of the antimicrobial activity of the extracts explains the variations of their chemical compositions. As reported in the literature, we considered that an extract has antibacterial activity if the inhibition zone is greater than 10 mm. 7,8 In well diffusion assay, it is also noted that all bacteria are sensitive to the fraction of ethyl acetate at different concentrations except Proteus vulgaris. The results recorded in Table 3 showed that the fraction of ethyl acetate had a good inhibitory activity at different concentrations tested on bacterial strains with a diameter of inhibition of 25 ± 1.4 and 20 ± 0 mm on Staphylococcus aureus and Escherichia coli, respectively, at the concentration of 200 mg/mL. However, a low sensitivity was observed with the strains of P. vulgaris, Pseudomonas aeruginosa, and Enterococcus faecalis with respective inhibition diameters of 0, 10.5 ± 0.7, and 11.5 ± 0.7 mm at the same concentration (Table 3). These results corroborate with those of Dhouioui et al 9 who indicated that A. longa roots inhibit the growth of various bacterial strains and also the same effect is noted with its essential oil. 10 This is consistent with previous studies indicating that gram-negative bacteria were less sensitive to plant extracts than gram-positive bacteria, and this can be due to the presence of an outer membrane containing very restrictive lipopolysaccharides. 11 According to the results found, E. coli showed high sensitivity to all tested concentrations with diameters ranging from 12.5 ± 0 to 20 ± 0 mm. Moreover, S. aureus was very sensitive with inhibition rings of 23.5 ± 0 and 20.7 ± 1.0 mm around the 100 and 50 mg per disks, respectively. The antibacterial activity of this fraction can be attributed mainly to the major constituents of the plant (flavonoids, tannins, etc.). The absence of antibacterial activity at low concentrations can be attributed to the presence of smaller amounts of antimicrobial compounds. 12 Regarding the antibacterial potency of the methanolic fraction, it was observed only with S. aureus and Klebsiella pneumoniae whose diameters of the inhibition zone are 20.5 ± 0.7 and 13 ± 1.4 mm, respectively. This was similar to the results of Hossen et al 13 who reported that S. aureus and E. coli strains were resistant to Aristolochia indica methanolic extract. Moreover, according to Merouani et al, 5 methanolic extract of A. longa fruit has shown significant inhibitory effects against P. aeruginosa and S. aureus. In contrast, Kumar et al 14 reported that the ethanolic extract of A. indica had low antibacterial activities on the bacteria studied. 14 Our results are still in agreement with those found by Negi et al, 15 who recorded a good antibacterial activity with the ethyl acetate fraction of Aristolochia bracteata roots followed by methanol fraction of an intermediate activity and the aqueous extract was the least effective.
Diameters (mm) of inhibition induced by the various extracts in the disk diffusion method.

Values are expressed as mean ± SD.
*P < .05 compared to control group.
**P < .01 compared to control group.
***P < .001 compared to control group.
****P < .0001 compared to control group.
The method of well diffusion assay allowed us to evaluate the antibacterial activity of the same extracts on the in vitro growth of bacterial strains. The results show that the fraction of ethyl acetate is active only on strains of S. aureus and E. coli whose inhibition surfaces are, respectively, 30 and 18 mm at the maximum concentration. While for the methanolic fraction, S. aureus was the only inhibited bacteria (17.2 and 27 mm). Our study shared the same results with the study of Naik et al 16 who investigated the antibacterial activity of A. indica leaf extract and flowers against gram (+) and gram (−) bacteria also using the diffusion method on wells. This activity could be explained by the disruption of the permeability barrier of the bacterial membrane. 17 Additionally, A. longa contains aristolochic acid, which is known by its mutagenic and carcinogenic effects. 18 This mutagenicity may explain its antimicrobial action. In fact, aristolactam and aristolochic acid, 2 compounds isolated from A. longa, 19 inhibited E. coli, P. aeruginosa, E. faecalis, S. aureus, and Staphylococcus epidermidis which confirm our results. On the other hand, Lee and Han 20 have also shown that these compounds have antibacterial activity against gram-positive bacteria. 20 Therefore, the activity observed with our extracts could be due to the presence of these bioactive compounds. However, the quantitative and qualitative comparison of the results of the extract and the antibiotics is difficult, because the nature of the activity and the molecules composition are not comparable. Therefore, one can still make an overall comparison of antibacterial activity with that of plant extracts. The averages of the antibiotic inhibition diameters used against the targeted strains are presented in Table 3. It is observed that different strains of bacteria studied react differently to the antibiotics tested and the highest activity has been demonstrated against S. aureus. These results are very interesting because this microorganism can be commonly involved in skin infections. 21 In particular, in the high concentrations, the fractions of ethyl acetate and methanol showed an antibacterial activity greater than that of vancomycin with respect to the S. aureus strain, whatever the technique used (disks/wells). As in the disk diffusion assay, the fraction of ethyl acetate had a major activity compared to gentamicin for E. coli. It is obvious that the antimicrobial activity of the extracts depends largely on their concentration, bacterial strains, the solvent used, and the type of plant extract.
The minimal inhibitory concentration (MIC) and the minimal bactericidal concentration (MBC) were determined successively using microdilution method and the culture of bacteria on nonselective medium (see Supplemental Data). The analysis of the experimental data shows that compared to the growth control, there is a decrease in the turbidity that is caused by the growth of seeds in the experimental tubes as the concentration of extract increases. Our results show that the fractions of ethyl acetate and methanol had an antibacterial activity by inhibiting the growth of bacterial germs in a dose-response relationship. This allowed us to determine different antibacterial parameters, namely the MIC and the MBC. The determination of the MIC of A. longa extracts is carried out by the dilutions method in liquid medium. It is a quantitative technique that determines the range of concentrations that effectively inhibit bacterial growth. A very strong antibacterial activity is presented by a very low MIC 22 and the results obtained are shown in Table 4. The analysis of the results showed that both fractions show acceptable antibacterial activity. Indeed, the MIC range of our extracts varies from 12.5 to 100 mg/mL for 3 bacterial strains among the 6 studied (S. aureus, E. faecalis, and P. aeruginosa). The experimental tubes contain both different germs and the extract to be tested. In these tubes, it is noted that increasing concentrations of plant extract cause a gradual and dose-dependent decrease in the turbidity induced by the growth of bacterial strains. According to Table 4, we revealed that the fraction of ethyl acetate is most active, with MIC values that are of the order of 12.5 mg/mL for P. aeruginosa ATCC, 50 mg/mL for S. aureus ATCC, and 100 mg/mL for E. faecalis. Similarly, the methanolic fraction showed a MIC of 50 mg/mL for S. aureus. Our results are in line with those found by Camporese et al 21 who studied the leaves and bark of another species of Aristolochiaceae (Aristolochia trilobata) and found that S. aureus and P. aeruginosa showed the lowest MIC. 21
Minimal Inhibitory Concentration and Minimal Bactericidal Concentration Values of Aristolochia longa Extracts.

MBC, minimal bactericidal concentration; MIC, minimal inhibitory concentration.
The same findings have been observed for the methanolic extract of A. longa fruits. 5 In fact, the active ingredients, alone or in combination, inhibit the vital processes of microbes by binding to their protein molecules, acting as chelating agents, modifying their biochemical systems, and thus preventing the use of nutrients for microorganisms. 23 The activity of a plant substance depends on several factors including the mode of extraction and the concentration of active principles. 24,25 In addition, as noted above, A. longa contains flavonoids, alkaloids, and tannins. Since these compounds have known antibacterial properties, their presence may explain the observed antimicrobial properties. 26 Moreover, the polarity of the organic solvents and the extraction conditions could give high selectivity and antimicrobial components. 27 The bioassays may reveal the presence of new, more potent, compounds in this plant. It follows from our analysis that the fraction of ethyl acetate of A. longa has a bacteriostatic effect on the strains of P. aeruginosa, S. aureus, and E. faecalis, as well as the methanolic fraction has the same power on the strain of S. aureus. However, the aqueous extracts showed no inhibition against bacterial strains at all concentrations tested; this is in agreement with the results of a study reporting that water is less effective than organic solvents in extracting active compounds from plants. 28 The difference between the results of 2 methods (inhibition zones and MIC/MBC) is probably due to the difference in the culture medium. In fact, in the zones of inhibition, we worked with a solid medium, whereas to determine the MICs, the liquid culture medium was used. This also explains why the size of the inhibition zone does not reflect the true antibacterial efficacy of the extract. 29 The sensitivity of different strains to this fraction is of great importance in the treatment of pathologies associated with them because these strains have high resistance to antibiotics used in the current practice.
Supplemental Material
Supplementary Material 1 - Supplemental material for HPLC-DAD/TOF-MS Chemical Compounds Analysis and Evaluation of Antibacterial Activity of Aristolochia longa Root Extracts
Supplemental material, Supplementary Material 1, for HPLC-DAD/TOF-MS Chemical Compounds Analysis and Evaluation of Antibacterial Activity of Aristolochia longa Root Extracts by Nasreddine El Omari, Sanae Akkaoui, Omar El Blidi, Rokia Ghchime, Abdelhakim Bouyahya, Mourad Kharbach, Maâmar Yagoubi, Abdelaali Balahbib, Omar Chokairi and Malika Barkiyou in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
