Abstract
The volatile oils of fresh and air-dried buds of 3 different varieties of Cannabis, namely, high cannabidiol (CBD) chemotype, intermediate CBD/tetrahydrocannabinol (THC) chemotype, and high THC chemotype were prepared by hydrodistillation. Gas chromatography analysis of the volatile oils resulted in the identification of 71 compounds, of which 33 were monoterpenes and 38 were sesquiterpenes. The volatile oil obtained from the THC chemotype showed an increase in the ratio of the sesquiterpenes to monoterpenes content. The content of terpinolene was dramatically decreased upon drying of THC chemotype. Moderate increase in β-caryophyllene and caryophyllene oxide was observed. However, there was no detectable change in the percentage of monoterpenes and sesquiterpenes content in both the intermediate type and CBD chemotype upon drying. The insecticidal activity of the volatile oils was evaluated. The oil obtained from the fresh and dried high CBD cannabis showed good biting deterrent activity at 10 ug/cm2 compared with N,N-diethyl-meta-toluamide at 4.78 µg/cm2, and good larvicidal activity.
Terpenes are the main fragrant compounds in plant resins and volatile oils responsible for the plants’ distinctive smells. Terpenes in Cannabis plants are responsible for strain-specific variations in the smell of the plants. 1 The distinct scent of a cannabis cultivar is the result of the balance of the different terpenes produced by that particular plant’s breeding. 2
Cannabis sativa plants are particularly rich in terpenes, represented by 120 compounds that are typically monoterpenes and sesquiterpenes. 3 The most common terpenes in cannabis are β-caryophyllene, myrcene, limonene, α-pinene, β-pinene, α-humulene, and linalool. 1 Monoterpenes are predominant in fresh plant material but their yield diminishes upon drying and storage, 4,5 resulting in a higher relative proportion of sesquiterpenes in dried cannabis.
Terpenoids play various ecological roles and their composition is under genetic control. 6 They are considered as a contributing factor that determines plant community properties and ecosystem functions and thought to have evolved primarily as defense compounds. In addition, certain terpenoids in the mixture may be recycled into the plant’s primary metabolism. Other compounds are emitted into the environment, through volatilization, decomposition of the plant, or by exudation, playing a major role in numerous interactions. The remarkable stickiness of cannabis exudates traps insects as it has insecticidal and repellent activities 7 and provides a synergistic mechanochemical defense in combination with the insecticidal phytocannabinoid acids, mainly tetrahydrocannabinolic acid (THCA), cannabidiolic acid (CBDA), and cannabichromenic acid (CBCA). These cannabinoids are biosynthesized from the common substrate, cannabigerolic acid, in the storage cavity of the glandular trichomes. 8
The lipophilic nature of terpenes increases the blood–brain barrier permeability leading to many sites of pharmacological actions. 9 The therapeutic potential of terpenes are numerous, including anti-inflammatory, anti-oxidant, analgesic, anticonvulsant, antidepressant, anticancer, antimutagenic, anesthetic, antinociceptive, antihistaminic, and antispasmodic activities. 1,10
Limonene displayed antinociceptive activity in chemical nociceptive mice models and causes peripheral analgesia through opioid receptors stimulation. 11 Myrcene has potent anti-inflammatory, analgesic, and anxiolytic properties. 12 β-Caryophyllene (BCP) is the most common sesquiterpenoid in cannabis plants and extracts, especially after heat decarboxylation, 13 was shown to be a selective natural CB2 receptor agonist without psychoactivity. 14
Literature reports suggest that besides the individual activities of terpenes, they are responsible for improving the cannabinoids activities, a phenomenon referred to as known “the entourage effect” especially in the treatment of pain. 15
In continuation of our investigation of the cannabis volatile oil, 16 herein, we examined the different chemical composition of the volatile oil of the cannabis chemotypes before and after drying as well as their insecticidal activity.
Results and Discussion
The volatile oils of fresh (F) and air-dried buds (D) of 3 different chemotypes of cannabis plant including CBD chemotype (fiber type), CBD/THC chemotype (intermediate type), and THC chemotype (drug type) were prepared by hydrodistillation. Gas chromatography (GC)–mass spectrometry (MS) and GC/flame-ionization detector (FID) were used to analyze the prepared volatile oils.
The GC–MS analysis resulted in the identification of 71 compounds with 33 monoterpenes and 38 sesquiterpenes. The identification was based on a direct comparison with authentic samples and/or by comparing the retention times and mass spectral fragmentation with the data published by “Başer Library of Essential Oil Constituents” and Wiley GC/MS Library, MassFinder 3 Library. The percentage of each compound was determined depending on its relative response in the FID chromatograms. Table 1 shows a list of all identified compounds, relative retention indices, and their percentage composition.
The Chemical Composition of the Volatile Oils Prepared From Fresh and Dried Cannabis Samples.
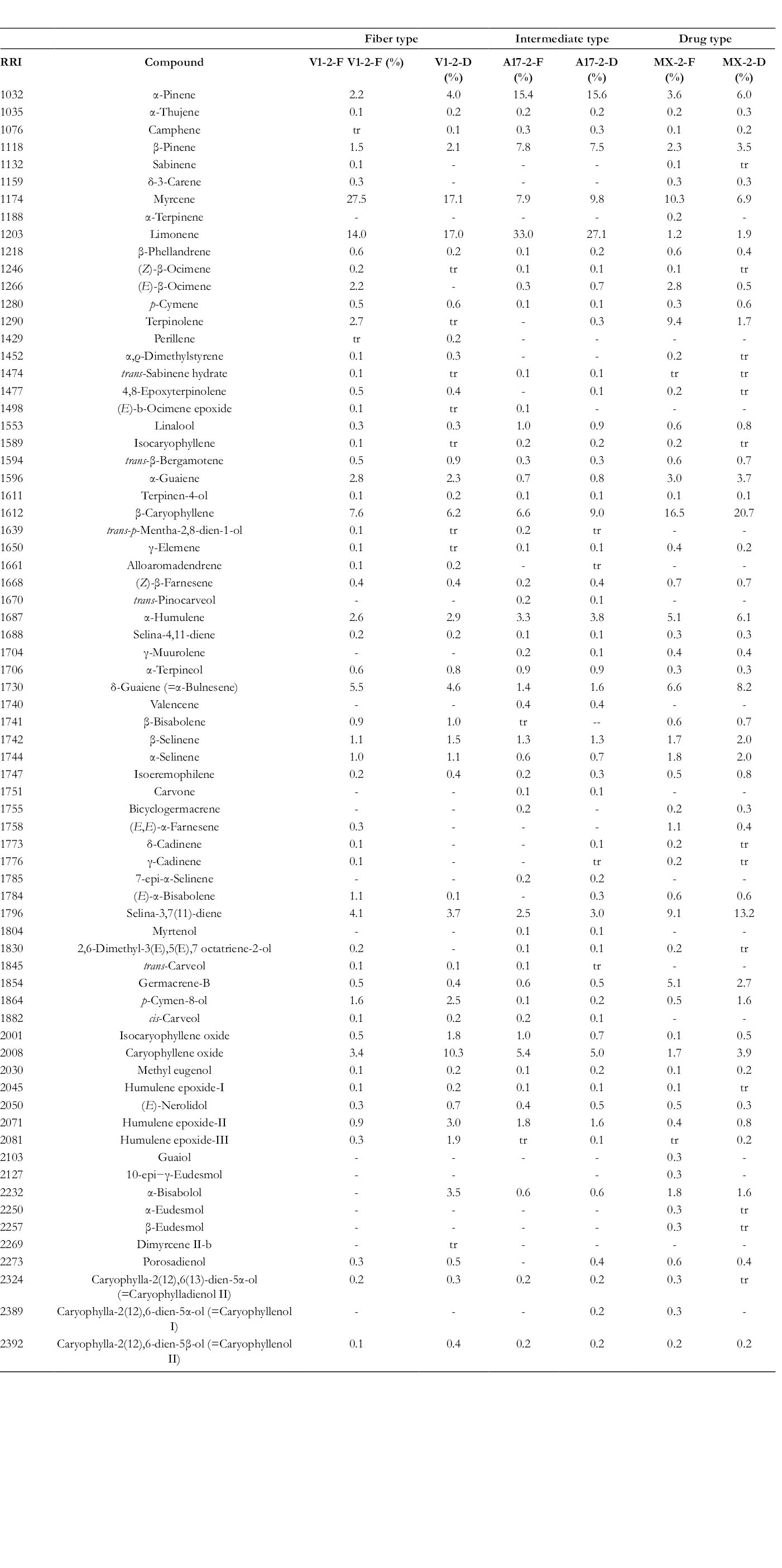
RRI, relative retention indices; tr, traces (<0.1%).
RRI calculated against n-alkanes. Percentage calculated from flame ionization detector data.
Generally, the composition of the different essential oils varied and the percentage of monoterpenes decreased upon drying. The collected data shows the change in the percentage of the monoterpenes and the sesquiterpenes content in each chemotype due to drying.
On drying of CBD chemotype, it was observed that the monoterpenes content of the volatile oil was higher in the oil of the fresh material (58.7%) than in the dried one (46.5%) (Table 2). The oil of the fresh fiber type (CBD chemovar) was rich in myrcene (27.5%) and limonene (14%). An obvious increase in the caryophyllene oxide content from 3.4% to 10.3% along with a 10% reduction in the myrcene content from 27.5% to 17.1% and a slight change of the limonene from 17% to 14% were the most significant changes on drying.
Monoterpenes and Sesquiterpenes Contents of Cannabis Volatile Oils.

An increase in the percentage of sesquiterpenes from 61.0% to 71.6% and a reduction of monoterpene content from 33.7% to 25.5% was observed as a result of drying the THC chemotype plant material (Table 2). The volatile oil of the fresh drug type showed significant content of β-caryophyllene and terpinolene compared with the other 2 types. Terpinolene content was shown to decrease upon drying ranging from 9.4% to 1.7%. Moderate increase in β-caryophyllene and caryophyllene oxide was observed.
However, there was no detectable change in the percentage of the monoterpenes and sesquiterpenes content of the oil of the intermediate chemovar upon drying. Its freshly prepared oil was rich in α-pinene, β-pinene, and limonene. The relative concentration of these compounds only slightly changed on drying. The chemical structures of the major components of the essential oil of cannabis are shown in Figure 1.

Chemical structures of the major components of volatile oils of cannabis.
Biting deterrent activity of these volatile oils was determined by using K&D bioassay. 17 All the essential oils showed higher biting deterrence activity than the solvent control, ethanol. The biting deterrent activity of essential oil of MX-2-F, VI-2-F, and A-17–2-F was similar to N,N-diethyl-meta-toluamide (DEET), whereas the activity of all the other essential oils was significantly lower than DEET (Figure 2). Biting deterrent activity of MX-2-D and VI-2-D was similar to ethanol. The oil obtained from the fresh and dried CBD chemotype cannabis showed good biting deterrent activity at 10 ug/cm2 compared with DEET at 4.78 ug/cm2. Biting deterrent activity of the essential oils is given in Figure 2.
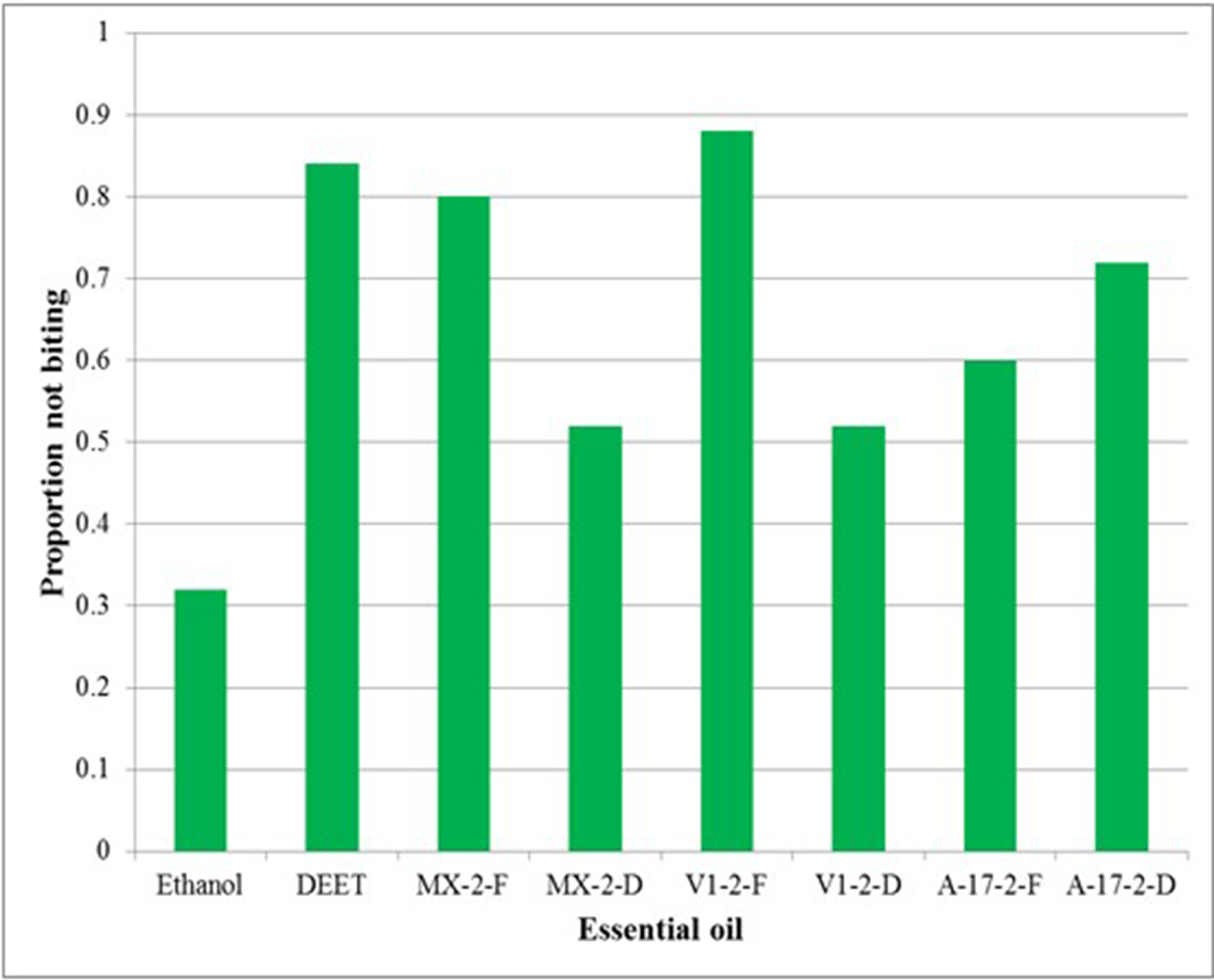
Mean bting deterrence index (BDI) values of essential oils of cannabis chemovars against Aedes aegypti. All the essential oils were tested at 10 ug/cm2. Ethanol was the solvent control and N,N-diethyl-meta-toluamide (DEET) at 25 nmol/cm2 was used as positive control. Means were separated by using Ryan–Einot–Gabriel–Welsch multiple range test (P ≤ 0.05).
Out of 6 essential oils tested in our screening bioassay, only 4 oils showed larvicidal activity against 1-day old Aedes aegypti larvae, whereas the others were inactive at the maximum dose of 125 ppm (Table 3). The 50% lethal concentration (LC50) values for these 4 oils ranged between 21.5 and 27.5 ppm. Based on 95% confidence interval (CI), LC50 values of VI-2-D and A-17-2-F were similar to VI-2-F, whereas these values were significantly lower than A-17-2-F. However, these variations among the essential oils are very small. These data suggest that some compounds or their combinations were responsible for this activity. Further studies should focus on the testing of the pure compounds from these oils to identify the compounds responsible for these insecticidal activities.
Toxicity of Essential Oils of Cannabis Chemovars Against 1-Day-Old Aedes Aegypti Larvae at 24 Hours Post-Treatment.

df, degree of freedom; CI, confidence interval; LC50, 50% lethal concentration; LC90, 90% lethal concentration.
LC50 and LC90 values are in ppm.
aPositive standard, purity 46.1% and 53.2% for cis and trans, respectively.
Experimental
Plant Material
Indoor and outdoor C. sativa plants of 3 chemotypes (high THC, THC/CBD, and high CBD) were grown at the University of Mississippi. Outdoor grown plants were harvested in 2014 and indoor grown plants produced in 2017. Voucher specimens from each variety were kept at the Coy Waller Laboratory, University of Mississippi with codes CFP-MX, CFP-V1, and CFP-A17 for high THC, high CBD, and THC/CBD, respectively. Fresh mature buds of the 3 cannabis chemotypes were harvested and divided into 2 equal parts of each chemotype. One part of each kept fresh, while the other part air-dried at room temperature for 1 week.
Preparation of the Volatile Oil
The volatile oil from each variety of fresh and dried plant material was prepared by steam distillation using the Clevenger apparatus. The terpene content of the prepared volatile oils was determined by GC-FID analysis.
GC-FID Analysis
The GC-FID analysis was carried out using an Agilent 6890N GC system, with the detector temperature set at 300°C. To obtain the same elution order as with the GC–MS method, simultaneous auto-injection was done on a duplicate of the same column applying the same operational conditions. Relative percentage amounts of the separated compounds were calculated from the FID chromatograms. The analysis results are given in Table 1.
Identification of the essential oil components was carried out by comparison of their relative retention times with those of authentic samples or by comparison of their relative retention index (RRI) to a series of n-alkanes. Alternatively, computer matching against commercial 18,19 and in-house “Başer Library of Essential Oil Constituents” built up by genuine compounds and components of known oils, as well as MS literature data 20,21 was also used for the identification.
GC–MS Analysis
The GC–MS analysis was carried out with an Agilent 5975 GC-MSD system. Innowax FSC column (60 m × 0.25 mm internal diameter and 0.25 μm film thickness) was used with helium as carrier gas (0.8 mL/min). GC oven temperature was kept at 60°C for 10 minutes and programmed to 220°C at a rate of 4°C/min, and kept constant at 220°C for 10 minutes and then programmed to 240°C at a rate of 1°C/min. Split ratio was adjusted at 40:1. The injector temperature was set at 250°C. Mass spectra were recorded at 70 eV. Mass range was from m/z 35-450.
Insects
Yellow fever mosquitoes used in these studies were from a laboratory colony maintained at the Mosquito and Fly Research Unit, Center for Medical, Agricultural and Veterinary Entomology, USDA-ARS, Gainesville, Florida, since 1952. Mosquitoes were reared to the adult stage by feeding the larvae on a larval diet of 2% slurry of 3:2 Beef Liver powder (now Foods, Bloomingdale, Illinois) and Brewer’s yeast (Lewis Laboratories Ltd., Westport, CT, USA). The eggs were hatched and the larvae reared to pupal stage in an environment controlled room maintained at 27 ± 2°C and 60 ± 10% relative humidity in a photoperiod regimen of 12:12 (L:D) hour. The adult mosquitoes were maintained in the laboratory using the procedures described by Ali et al. 22
In Vitro K&D Bioassay
Bioassays were conducted using a 6-celled in vitro Klun and Debboun (K&D) bioassay system. 17 Briefly, the bioassay system consists of 6 (3 × 4 cm) wells each of which contains approximately 6 mL of the feeding solution. As described by Ali et al, 22 a feeding solution consisting of citrate phosphate dextrose adenine (CPDA-1) and adenosine triphosphate (ATP) was used instead of blood. All the compounds were tested in this study, and DEET, 97%, N, N-diethyl-meta-toluamide (Cas# 134 – 6 2-3, Sigma-Aldrich, St. Louis, MO, USA) at 25 nmol/cm2 was used as a positive control. All the treatments were freshly prepared in molecular biology grade 100% ethanol. The temperature of the feeding solution in the reservoirs was maintained at 37°C by using a circulatory bath. The reservoirs were covered with a layer of collagen membrane (Devro, Sandy Run, SC, USA). The test samples were randomly applied to 6 (4 × 5 cm) marked areas of organdy (A fine, thin cotton fabric) and positioned over the membrane-covered CPDA-1 + ATP solution. The K&D module containing 5 female mosquitoes per cell was positioned over treated organdy and trap doors were opened to expose the treatments to the females. The number of mosquitoes biting through treated organdy in each cell was recorded after a 3-minute exposure and mosquitoes were prodded back into the cells to check the actual feeding. A replicate consisted of 6 treatments: 4 test samples, DEET, and ethanol-treated organdy as solvent control. Treatments were replicated 5 times.
Larvicidal Bioassays
Bioassays were conducted using the bioassay system described by Pridgeon et al. 23 Further methods and statistical analyses were done as previously described by Ali et al. 22 Dimethylsulfoxide was used as a solvent to prepare the treatments and was also used as a negative control. Permethrin (95.7%) (Chem Service, Inc. West Chester, PA, USA) was used as a positive control.
Statistical Analyses
Proportion not biting (PNB) was calculated using the procedure described by Ali et al. 22

Data on the PNB were analyzed using SAS Proc ANOVA (SAS Institute, Inc., Carry, NC, USA, 2007) and means were separated using Ryan–Einot–Gabriel–Welsch multiple range test. LC50 values for larvicidal data were calculated by using SAS, Proc Probit.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported in part by the National Institute on Drug Abuse, contract # N01DA-15-7793.
