Abstract
Dermatophytosis is a fungal infection of the skin caused by a group of pathogenic fungi known as dermatophytes. Therefore, we investigated those medicinal plants that were being used by local people and traditional healers to treat skin infections in the Mopani District, Limpopo Province. A survey was conducted through the use of a semistructured program to gather information on the common names, plant parts used, methods of preparation, and administration of the medicine. Findings of the survey revealed that over 30 plant species were used for the treatment of skin infections in the area. The most common local mode of medicinal preparation to treat skin was decoction (37%), followed by paste (21%), infusion (19%), poultice (9%), smoke (7%), maceration (5%), and steam (2%). Of the species, only 12 (Ficus sur L., Peltophorum africanum Sond., Vangueria infausta Burch. subsp. infausta, Diospyros mespilliformis Hochst. ex. A. DC., Ziziphus mucronata Willd. subsp. mucronata, Euclea divinorum Hiern, Ximenia caffra Sond., Dombeya rotundifolia Hochst., Ficus sycomorus L., Sideroxylon inerme L. subsp inerme, Parinari curattellifolia Planch. ex Benth., and Maytenus undata (Thunb.) Blakelock) were selected based on literature and ethnobotanical information. We further investigated the antifungal activity of acetone and aqueous extracts of the above mentioned selected plant species using serial dilution assay against Trichophyton rubrum, Microsporum canis, and Candida albicans. All plant extracts were active against the tested microorganisms with minimum inhibitory concentration (MIC) values ranging between 0.02 mg/mL and 1.25 mg/mL. In the bioautography assay, more active compounds were visible in acetone and water extracts of E. divinorum. No active compounds were observed in some plant extracts with excellent antifungal activity, as shown in the microdilution assay. Findings, in general, suggest that the identified plant species, especially those with extracts showing relatively low MIC values, are playing a big role in treating skin infections in Mopani District.
Keywords
Background
Dermatophytosis is a cluster of diseases caused by certain fungal pathogens known as dermatophytes. Three genera of fungi, Trichophyton, Microsporum, and Epidermophyton, account for most dermatophytic infections. Trichophyton rubrum and Microsporum gypseum are a group of organisms with the ability to degrade keratin in humans and consequently digest tissues. They also cause superficial infections, affecting the outer layers of the skin, hair, and nails. 1 Dermatophytosis may be treated by topical application on the infected areas. Several drugs such as Flucytosine and Echinocandis are used as antifungal agents to combat skin infections in humans. However, fungal infections have been reported to be quite difficult to treat and the fungi develop resistance to the currently available antifungal drugs. 2 Most of the available antifungal drugs used to reduce the spread of skin infections are produced by microorganisms. The mode of action of these drugs is complicated and some may cause damage to the permeability of the cell membranes.
Skin infections are increasing due to the misuse of antibiotics, especially in immunocompromised patients. 3 The increased resistance of the currently available drugs such as amphotericin B, fluconazole, and azoles is a major threat to the public health. Skin fungal infections can become severe, remain a great threat to people, and are more likely to cause death if they are either not treated or poorly controlled. 4 In South Africa, the prevalence of dermatophyte mycosis has also increased due to the increase in immunosuppressed human immunodeficiency virus patients. This has prompted the search for plant sources of antimicrobial agents. Plant extracts of several plant species have antimicrobial properties that are attributed to the presence of secondary metabolites that may serve as part of the plant mechanisms against predators and pathogenic attack. As such, research is increasingly focusing on medicinal plants, which can provide mankind with potential antifungal agents to combat various skin infections and related ailments.
Medicinal plants possess active compounds with the ability to treat fungal infections. Therefore, the screening of plants for fungicidal agents is of great importance due to the increase in resistance of fungal pathogens. 5,6 In South Africa, there are many plants used to treat various skin diseases, including fungal infections. However, information is limited to local communities within a specific area. 7 Such information, which is important in the preservation of indigenous knowledge, is transmitted orally from one generation to another.
An ethnobotanical survey is necessary for the identification and documentation of new plant species, which may lead to the discovery of plants with the potential for use as antifungal agents. This is an important step in the development of drugs since it could lead to the production of useful information of unknown and unidentified biologically active compounds in plants. Ethnobotanical surveys are also important for anthropological reasons, as they may assist in preserving cultural heritage. In this article, we investigate medicinal plants that are used by Xitsonga-speaking people in the treatment of skin infections in Mopani District and determine the antidermatophytic activity of selected plants against pathogenic fungi.
Materials and Methods
Location and Demographics of Study Area
The study was conducted in Mopani District, Limpopo Province, South Africa. Data were collected from 10 villages in the surrounding Tzaneen areas: Mariveni, Zangoma, Bordauex, Burgersdorp, Shiluvani, Khangela/Mafarana, Ntsako, Maake, Tickyline, and Sasekani (Figure 1). There are approximately 3 million Xitsonga-speaking people in Mozambique and South Africa. There is currently limited ethnobotanical information on Xitsonga-speaking people in Mopani District, which falls under the savanna biome. The Tzaneen area is located in Mopani District in the humid Lowveld bioregion and has a mean annual rainfall of 850 mm and midday temperatures ranging from 15°C to 28°C. The dominant vegetation is Tzaneen sour Bushveld and Lowveld.
Map showing Mopani district, Limpopo Province.
Data Collection
Ethnobotanical survey and interviews
The ethnobotanical survey was conducted in Greater Tzaneen Municipality, Limpopo Province (Figure 1). Permission to conduct the survey in the area was granted from the traditional council. Traditional healers to be interviewed were required to sign a consent form before providing information on the medicinal plants. Thirty Xitsonga-speaking traditional healers from different areas were selected using a snowball sampling method. This sampling method involves primary data sources (initial traditional healers) nominating others to be interviewed in the survey. In other words, the snowball method is based on referrals from initial traditional healers to generate a list of additional traditional healers. When applying this non-random sampling method, members of the sample group are recruited via chain referrals. Three traditional healers were interviewed using a semistructured questionnaire and guide fieldwork in each village. The information gathered included local name/s of plants used to treat different ailments, localities of these plants, plant part/s used to treat skin infections, methods of remedy preparation, diagnosis of different ailments, and other relevant information.
Plant Collection and Identification
Voucher specimens were collected from their natural habitat along the Tzaneen dam in 10 local villages during summer with the help of traditional healers (Table 1). Such specimens were identified using the literature and verified at the Larry Leach herbarium of the University of Limpopo. Plants were identified by comparing the vernacular names with records on plants used by Xitsonga-speaking people of South Africa. 8 The herbarium curator, Dr Egan, confirmed the identification of plant species. Voucher specimens were collected and given unique accession codes (Table 1) and then stored in the herbarium. Plant materials were separated according to the different parts used. The plant materials were allowed to dry at room temperature (25°C), ground into a fine powder, and stored in the dark in sealed containers until extraction.
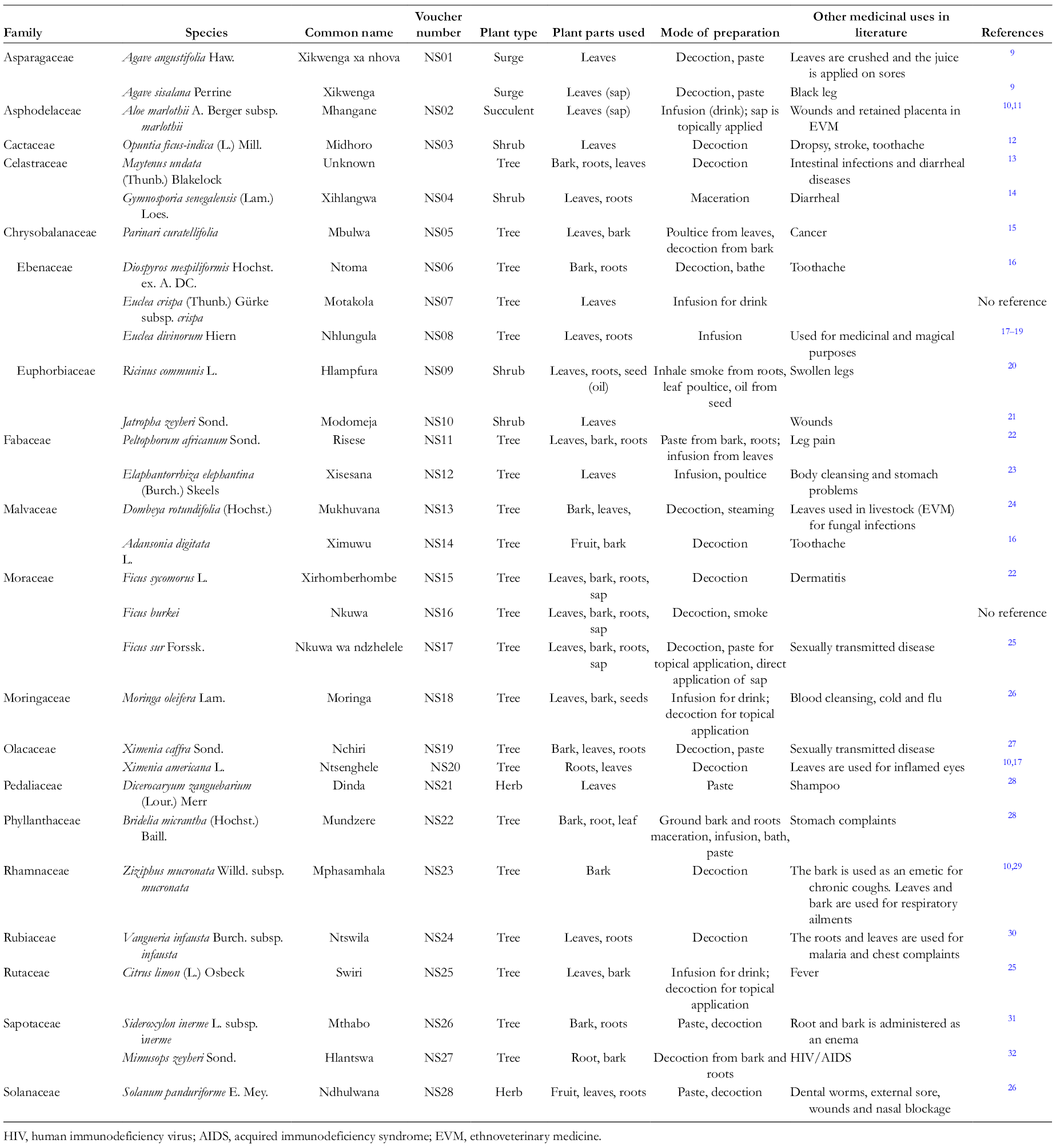
Medicinal Plants Used to Treat Skin Infections in Mopani District, Limpopo Province, South Africa.
HIV, human immunodeficiency virus; AIDS, acquired immunodeficiency syndrome; EVM, ethnoveterinary medicine.
Plant Extraction
Twelve plant species were selected based on information provided by the traditional healers for further phytochemical analysis and microbiological assays. Finely ground roots, leaves, stems, and roots (3 g) were extracted with 30 mL acetone and water in polyester plastic tubes while shaking vigorously for 3-5 minutes on a shaking machine at a high speed of 3500 rpm. Acetone was chosen as a solvent since it is not toxic to microorganisms, 33 while traditional healers use water to prepare their medicine. The plants were filtered using Whatman No.1 filter paper. After centrifuging at 3500 rpm for 5 minutes, the supernatants were decanted into labeled beakers. The process was repeated 3 times and the extracts combined. The solvents were removed under a stream of cold air at room temperature. Aqueous extracts were evaporated using a Specht Scientific freeze dryer. The crude extracts were redissolved in acetone prior to carrying out the biological assay.
Phytochemical Analysis
Thin-layer chromatography (TLC) was used to analyze the chemical components of the plant extracts. Each plant extract (10 µL) was loaded onto TLC plates and developed using 3 different eluent systems. Ethyl acetate:methanol:water (EMW, 40:5:1) polar), chloroform:ethanol:formic acid (CEF, 5:4:1) (intermediate polarity: acidic), benzene:ethanol:ammonium hydroxide (BEA, 90:10:1) (non-polar/ basic). 33 Chemical compounds were visualized under ultraviolet (UV) light (354 and 360 nm) and by spraying with vanillin-sulfuric acid spray reagent to detect compounds not visible under UV light. 34
Fungal Strains and Inoculum Quantification
Candida albicans (ATCC 10231), T. rubrum (MTCC 2961), and M. canis (ATCC 36299) were obtained from the Department of Paraclinical Sciences at the University of Pretoria. These pathogens cause skin infections in humans. For the quantification of fungi, the hemocytometer cell-counting method was used to count the number of cells for each fungal culture. 35
Microdilution Assay
Antifungal activity of plant extracts was determined using serial dilution assay. 36 The plant extracts were tested in triplicate in each assay, and the assays were repeated 4 times to confirm the results. Each plant extract (10 mg/mL) was serially diluted 50% with water in 96-well microtitre plates, 36 and 100 µL of fungal culture was added to the wells. Amphotericin B was used as a reference antibiotic and acetone as a negative control. As an indicator, 40 µL of 0.2 mg/mL p-iodonitrotetrazolium (INT) violet dissolved in water was added to each well. The microplates were covered and incubated for three to 3-5 days at 35°C at 100% relative humidity after sealing in a plastic bag to minimize fungal contamination in the laboratory. The MIC was recorded as the lowest concentration of the extract that inhibited antifungal growth.
Bioautography Assay
TLC plates were loaded with 10 µL of each extract and developed using different solvent systems: CEF, BEA, and EMW. The chromatograms were dried under a stream of cold air overnight to remove the solvent. The plates were sprayed until wet with a fungal culture containing 1.0 × 106 cells/mL and incubated at 37°C in a clean chamber overnight. The TLC plates were sprayed with a solution of INT violet again and incubated for 2-6 hours for fungal growth. White areas indicated where the reduction of INT to the colored formazan did not take place due to the presence of compounds that inhibited the growth of the fungi.
Data Analysis
The documented data were analyzed using descriptive and inferential statistics with Microsoft Excel 2010. Percentages and frequencies were portrayed in tables and figures to interpret the findings of the study. The frequency index of each plant species was calculated using the formula: FI = FC/N × 100, where FC is the number of traditional healers who mentioned the use of the plant and N is the total number of informants (30 traditional healers). 37
Results and Discussion
Plant Parts Used
An ethnobotanical survey was conducted in Greater Tzaneen Municipality to identify plant species used for the treatment of skin infections. In the current study, the most commonly used plant parts were leaves (36%), followed by roots and bark (26%), as well as seeds and sap (12%). Traditional healers reported that leaves were used alone, or in combination with other plant parts such as roots and bark. Some plant species were added to other herbs to increase the efficiency of the traditional remedy. Leaves are commonly used to treat skin infections as they can be applied directly onto the affected area as a paste, powder, or sap.
Roots and bark are commonly utilized in large quantities due to the perceived presence of more healing properties than the entire plant, which was confirmed by the findings of previous researchers. 38 Moreover, it was reported that underground parts are believed to be the most potent. Other plant parts used are tree sap, seeds, seed oils, and fruits. However, these plant parts are utilized in small quantities by traditional healers. This could be due to seasonal variation that occurs in some plants and lack of an efficient storing system for sap. The use of the whole plant is restricted to some herbaceous species.
The choice of plants and plant parts used depends on indigenous knowledge that is passed from generation to generation. Traditional healers make infusions to bathe and decoctions to drink.
Preparation and Treatment
Traditional healers preferred various methods of preparing their medicine. Our findings suggested that decoction is the most popular method of preparation (37%), followed by paste (21%), infusion (19%), poultice (9%), smoke (7%), maceration (5%), and steam (2%). This is because it is believed that some skin ailments are a manifestation of internal illness. Therefore, the decoction is prepared to treat the illness, but topically they apply a poultice, paste, or smoke to treat the symptoms. For this reason, one plant or plant part can be administered in more than one way to treat the same ailment at the same time. Prepared medicine is only suitable for consumption for 7 days. 39 The plant material is ground to prepare some medicines, as grinding facilitates better surface contact with the extraction solvent.
A decoction is prepared with plant parts cut into small pieces. It can be administered orally or applied topically onto the affected area on the skin. Pastes are prepared from the ground plant material and may be burned to ash prior to administration. The paste is applied directly to the affected area. Infusions are prepared using powdered plant materials and are administered orally. A poultice can be prepared by using fresh or dried leaf material.
Dried leaves are soaked in water prior to preparing the poultice. The duration of the poultice depends on the type of plant used and the skin ailment being treated. A fresh poultice can be applied for 2 days daily or 7 days. Smoke is used by burning the plant material and placing it in the affected area directly and is usually applied to treat warts and boils. Maceration is typically used to prepare a mixture to bathe, but it can be administered either topically directly onto the affected area, or orally. Furthermore, it is prepared using plant material that has been cut into small pieces. Steam is administered by placing the affected area directly above the steam produced when boiling the plant material.
Fresh leaves are usually administered by crushing before being placed on the affected area and held in place by a bandage or cloth. Poultice and bandage are changed daily. Smoke is used by burning the plant material and placing the affected area directly above the smoke. Smoke is usually used to treat warts and boils, which was also found to be the case with Tshivenda-speaking people of Limpopo Province, South Africa. 39 Tree sap (latex) from Ficus spp. is reported to be directly applied to ringworms. The sap from the leaf of Aloe spp. and seed oil from Ricinus communis and Moringa olifeira are directly applied to the affected areas. Juices from the fruits of Solanum panduriforme and Adansonia digitata are also applied directly to the wound.
Plant Collection
A survey revealed that 57% of the participants collect their own plants, while 24% would request somebody to collect them. Some of the traditional healers are old and, therefore, find it very difficult to travel long distances for plant collection. Most of the traditional healers indicated that they prefer to cultivate plants that are becoming extinct at home. The remaining 19% either preferred cultivating their own plants or buying them from vendors. Self-collection is mostly preferred over the cultivation and preferring somebody to collect. The traditional healers believed that cultivated plants may not be as effective as wild plants. Furthermore, it is also believed that plants collected from certain localities are more effective than others, and thus the collection is preferred. Cultivation and self-collection are preferred methods as this ensures that there are always sources of plants. These methods also prevent other people from utilizing the plant species.
Identified Medicinal Plants
In the current study, 30 plant species belonging to 20 families were identified as being used for the treatment of skin infections. Table 1 shows the family names of the plants, their vernacular names, the plant parts used, the plant forms, and the mode of administrating the treatment. The most dominating families were Ebenaceae and Moraceae. Among 20 species recorded, 71.4% were trees, followed by shrubs (14.3%), herbs (7.1%), and succulents and surges (3.4%). Citrus limon, M. olifeira, R. communis, Opuntia ficus-indica, Agave angustifolia, and Agave. sisalana are exotic plants.
Microdilution Method
The microdilution method was used to determine the antifungal activity of the selected plant extracts against C. albicans, M. canis, and T. rubrum. The MIC value is the lowest concentration of a plant extract that has the ability to inhibit fungal growth and the value also indicates whether the plant extract could be used to develop a new novel antifungal agent. All plant extracts were active against the tested microorganisms with MIC values ranging between 0.02 mg/mL and 2.5 mg/mL (Table 2). The water extract of Diospyros mespiliformis had the lowest activity with a MIC value of 1.25 mg/mL. The positive control (amphotericin B) exhibited good anti-Candida activity with the lowest MIC value of 0.02 mg/mL.
Minimum Inhibitory Concentration of 12 Plant Species Against Candida albicans, Microsporum canis and Trichophyton rubrum Using Different Extraction Solvents.

S = stem, R = roots, B = bark; L, leaves; A, acetone; H, water.
Amphotericin B was used as a positive control. Minimum inhibitory concentration for C. albicans, M. canis, and T. rubrum were recorded after 24 and 48 hours of incubation time.
The results show the average of 3 replicates with a standard deviation of 0 in all cases.
Acetone bark extracts of D. mespiliformis showed moderate activity against C. albicans with a MIC value of 0.16 mg/mL. It was reported previously that acetone extracts had excellent activity against pathogenic fungi. 40 Diospyros mespiliformis has been found to contain terpenes, triterpenoids, and oligomeric naphthoquinone metabolites. 41 Triterpenes are compounds that are promising for medical applications because of their diverse biological activity and low cytotoxicity. 42 Leaf extracts had an excellent activity with the lowest MIC value of 0.02 mg/mL against M. canis. Plant extracts with low MIC values could be good sources of active compounds with antimicrobial potency. 43
In the current study, acetone extracts of D. mespiliformis were active against M. canis with a MIC value of 0.16 mg/mL. The acetone leaf extract had good antifungal activity against this fungal pathogen, with a MIC value of 0.08 mg/mL. The bark and stem extracts of Ficus sycomorus had better antifungal activity against the tested microorganisms than that of the roots. More interestingly, the aqueous extract was more active against the selected fungal pathogens. However, the acetone extracts had moderate activity against M. canis with a MIC value of 0.63 mg/mL.
Antifungal activity was observed in the acetone bark extracts of Peltophorum africanum, F. sycomorus, and Sideroxylon inerme, with the lowest MIC value of 0.02 mg/mL. Furthermore, acetone and aqueous extracts of F. sycomorus contained compounds exhibiting an antifungal activity, which gives reason for the low MIC value. The bark extracts of F. sycomorus were not active against M. canis. Excellent antifungal activity was observed in the acetone and water stem extracts against the tested microorganisms.
The acetone extracts of Vangueria infausta, D. mespiliformis, and Ziziphus mucronata exhibited better activity against C. albicans. Similar results were observed for the aqueous extract of Dombeya rotundifolia, Ficus sur, and S. inerme. A reduction in the antifungal activity of some plant extracts after 48 hours of incubation could be due to the decomposition of the antimicrobial compounds after a long incubation period. All aqueous extracts of V. infausta exhibited poor antifungal activity against the tested fungal pathogens. However, aqueous extracts were active against M. canis with a MIC value of 0.02 mg/mL. Our findings support the study reported by other researchers that water extracts from other plants had excellent antimicrobial activity against the tested organisms. 37 This indicates the effectiveness of the plant species used by traditional healers using water as an extracting solvent. Previously, it was reported that different plant parts of F. sycomorus contain phenols. 44 The acetone leaf extract of V. infausta had good antifungal activity against C. albicans. Vangueria infausta contains flavonoids and tannins. 45
Bioautograms of Fungal Pathogens
TLC bioautograms of extracts of D. mespiliformis bark, D. rotundifolia bark, Euclea divinorum bark, and F. sur bark and stem were tested against 3 fungal pathogens. Active compounds separated with CEF had Rf values of 0.59, 0.15, and 0.98. Similar compounds were also observed in chromatograms developed in EMW. Antifungal compounds were visible in acetone extracts of D. rotundifolia separated with BEA. No active compounds were observed in acetone extracts of D. rotundifolia developed in BEA against M. canis. Noticeably, the plant extracts showed excellent antifungal activity against the tested microorganisms with serial dilution assay. In chromatograms developed in BEA, 2 compounds were observed in acetone extracts with Rf values of 0.17 and 0.61 against M. canis respectively. The active compounds separated with EMW had Rf values of 0.91, 0.69, and 0.90. The acetone and aqueous extracts of F. sycomorus contained 1 compound with similar Rf values of 0.79.
TLC chromatograms developed in EMW revealed 1 compound visible in the acetone extracts of V. infausta against M. canis with an Rf value of 0.79. The acetone extract of Z. mucronata had active compounds with Rf values of 0.23 and 0.93 against C. albicans and M. canis. No active compounds were observed in some plant extracts of D. mespiliformis and E. divinorum. Antifungal compounds were visible in acetone extracts of D. rotundifolia separated with BEA. The presence of tannins in D. rotundifola has previously been observed and may be responsible for the fungicidal activity 4546. No active compounds were observed in aqueous extracts of D. rotundifolia; the possible reason may be synergy between the compounds present in the plant extract. Another reason may be that some of the active compounds were volatile and evaporated during the drying period of the TLC plates. Among the tested plant species, S. inerme and V. infausta were the most promising.
Conclusion
The current study revealed that 30 plant species from 20 different families are being used by local people and traditional healers to treat skin infections in the Mopani District, Limpopo Province, South Africa. Vangueria infausta is commonly used for the treatment of skin diseases, followed by F. sycomorus. This finding proves that passing on of indigenous knowledge is an important measure in the preservation of useful medicinal plants. All leaf, stem, and root extracts of the selected 12 plant species were active against the tested fungal pathogens with low to moderate MIC values. Aqueous extracts of F. sycomorus showed excellent activity against the tested fungal pathogens, which confirms the efficacy of traditional methods used by local folk in the treatment of skin infections. In TLC chromatograms developed in EMW, more active compounds were observed in extracts of F. sycomorus against M. canis. The results support the traditional use of the selected plants to treat skin infections. Findings suggest that the identified plant species with good antifungal activity, especially those extracts showing relatively low MIC values, are playing a big role in treating skin infections and healthcare in general in the Mopani District. These species may likely act as future sources of advanced remedies for dermatophytosis and dermatophytes. Further investigation in this area is therefore encouraged in order to discover new potent medicinal plant species.
Footnotes
Acknowledgements
We thank the National Research Foundation (NRF) and the University of Limpopo for financial support. We are grateful to the traditional healers and local people who participated in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. The National Research Foundation (NRF) funded the research project.
