Prostate cancer remains to be one of the big health concerns due to its high prevalence and mortality rate worldwide. Prostate cancer cases (191 930) that account for nearly 21% of all male cancer cases and 33 330 prostate cancer deaths are projected to occur in the United States in 2020.
1
Prostate cancer has been demonstrated to be driven by the androgen receptor (AR)-regulated gene expression that is initiated by the binding of androgens to the AR.
2
Androgen deprivation therapy (ADT) has thus been the therapeutic centerpiece for prostate cancer over 70 years. However, the median duration of ADT response is merely 18-24 months due to inevitably progression to castration-resistant prostate cancer (CRPC).
3
The major driving force for the continued progression of lethal CRPC is the reactivation of AR transcriptional activity.
4,5
The AR therefore continues to serve as the essential therapeutic target for CRPC.
2,3,6
The current Food and Drug Administration (FDA)-approved treatments for CRPC patients that target the AR-signaling axis include abiraterone acetate,
7
enzalutamide,
8
apalutamide,
9
and darolutamide.
10
Each of these treatments can only improve median overall survival by approximately 2-4 months. None is curative and treatment resistance is inevitable.
11,12
Novel drugs with a new structure scaffold targeting the AR-signaling axis are thus needed to treat deadly CRPC.
Our previous studies on the optimization of silibinin as antiprostate cancer agents revealed: (1) that 2,3-dehydrosilybin (2) is more potent in suppressing prostate cancer cell proliferation and (2) that the antiproliferative potency of 2,3-dehydrosilbinin (2) in both AR-positive and AR-negative prostate cancer models could be further improved by introducing an appropriately designed substituent to a phenolic hydroxyl group (Figures 2 and 3).
24
-26,31
The half-maximal inhibitory concentration (IC50) values of the 2 optimal derivatives (3 and 4, Figure 3) fall in the range of 1.40-2.71 µM in prostate cancer cell models, representing a 27-fold to 52-fold improvement in potency compared with silibinin. However, most of the modifications to the phenolic hydroxyl groups did not show the selectivity of suppressing cell proliferation in the AR-positive cell model over AR-negative ones. Very slight selectivity was observed in 20-O-substituted 2,3-dehydrosilbinins.
24
This encouraged us to further investigate the effects of modifications on the alcoholic hydroxyl group of tetramethylsilybin on the potency and selectivity in the AR-positive prostate cancer cell model. This study therefore aims to enhance the selectivity of antiproliferative potency of 2,3-dehydrosilybin (2) towards the AR-positive prostate cancer cells (LNCaP) through chemical modification on its alcoholic hydroxyl group at C-23. The synthesis and antiproliferative activity of these silibinin derivatives are presented in this article.
Experimental
Infrared (IR) spectra were recorded on a Nicolet Nexus 470 FTIR spectrophotometer. High-resolution mass spectra (HRMS)were obtained on a Q-Exactive Orbitrap mass spectrometer with electrospray ionization (ESI). Nuclear magnetic resonance (NMR) spectra were obtained on a Bruker Fourier 300 spectrometer in deuterated chloroform (CDCl3). The chemical shifts are given in ppm referenced to the respective solvent peak, and coupling constants are reported in hertz. Anhydrous tetrahydrofuran (THF) and dichloromethane (DCM) were purified by PureSolv MD7 Solvent Purification System from Innovative Technologies (MB-SPS-800). All other reagents and solvents were purchased from commercial sources and were used as is. Silica gel column chromatography was performed using silica gel (32-63 µM). Preparative thin-layer chromatography (PTLC) separations were carried out on TLC plates loaded with silica gel 60 GF254 (EMD Millipore Corporation). The diastereomeric silibinin (>98%) was purchased from Fisher Scientific (TCI America, CAS: 36804-17-8). Coniferyl alcohol was synthesized using the reported procedures.
35
The purity of compounds 7 and 10-17 was confirmed to be greater than 95% by 1H NMR spectroscopy prior to biological assay.
Synthesis of 2,3-Dehydrosilbinin (2)
Quercetin dihydrate (1.67 g, 4.94 mmol, 1 eq) and coniferyl alcohol (1 g, 5.55 mmol, 1.1 eq) were dissolved in a mixture of toluene and acetone (300 mL in 2:1 ratio). The mixture was stirred for 10 minutes at 80°C prior to the addition of silver carbonate (1.53 g, 5.55 mmol, 1.1 eq). The reaction was allowed to proceed with stirring at 80°C overnight. The cooled reaction mixture was then filtered through a pad of silica gel; the filtrate was concentrated in vacuo. The crude product was sequentially purified through column chromatography (50% ethyl acetate in hexane) followed by PTLC (9% methanol [MeOH] in DCM, developed 3 times). The desired product (354 mg), obtained in 13% yield, still contains minor impurities but is good enough to be used for the next step reaction.
Rf: 0.33 (DCM–MeOH, 91:9, v/v).
1H NMR (300 MHz, CDCl3): 12.13 (s, 1 H, OH), 7.86 (s, 1 H, aromatic H), 7.85 (dd, J = 7.2, 2.1 Hz, 1 H, aromatic H), 7.17 (d, J = 1.8 Hz, 1 H, aromatic H), 7.08 (d, J = 9.3 Hz, 1 H, aromatic H), 7.01 (dd, J = 8.1, 1.8 Hz, 1 H, aromatic H), 6.95 (s, 1 H, aromatic H), 6.90 (d, J = 8.1 Hz, 1 H, OH), 6.58 (d, J = 2.1, 1 H, aromatic H), 6.27 (d, J = 2.1, 1 H, aromatic H), 5.05 (d, J = 8.1, 1 H, H-11), 4.25 (ddd, J = 7.8, 3.9, 2.4, 1 H, H-10), 3.89 (s, 3 H, OCH3), 3.79 (br.d, J = 12.6, 1 H, H-23), 3.55 (br.d, J = 13.5 Hz, 1 H, H-23).
The 1H NMR data of the obtained 2,3-dehydrosiliybinin are consistent with those reported in the literature.
32
Synthesis of 3,5,7,20-O-Tetramethyl-2,3-Dehydrosilybin (7)
Method 1
The suspension of 2,3-dehydrosilybin (2, with minor impurities, 590 mg, 1.23 mmol, 1 eq) in acetone (9 mL) was stirred at 85°C for 5 minutes, to which potassium carbonate (1.17 g, 8.49 mmol, 7 eq) was added. The subsequent mixture was stirred for an additional 10 minutes before dimethyl sulfate (0.82 mL, 8.59 mmol, 7 eq) was added dropwise. The reaction was allowed to proceed with stirring for 30 minutes. Since the reaction was not completed according to the TLC monitor, an additional portion of potassium carbonate (0.17 g, 1.21 mmol, 1 eq) and dimethyl sulfate (0.11 mL, 1.21 mmol, 1 eq) was added and the reaction mixture was stirred for an additional 30 minutes. The reaction was quenched with saturated ammonium chloride solution (10 mL), and the resulting mixture was extracted with 1% MeOH in DCM (30 mL × 3). The combined organic extracts were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was subjected to PTLC purification using 7% MeOH in DCM as eluent to develop the PTLC plate for one time and then using 50% ethyl acetate in hexane as eluent to further develop the PTLC plate for 3 times. The desired product was obtained as a dark brown wax in 61% yield.
Method 2
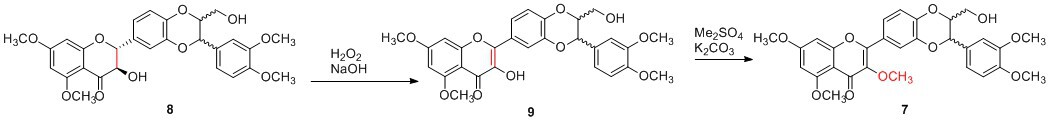
Pure 5,7,20-O-trimethylsilybin (8, 100 mg, 0.19 mmol, 1 eq) was dissolved in MeOH (1.32 mL) and THF (1.32 mL), and the solution was stirred at room temperature for 10 minutes under argon. The solution was then added dropwise into a mixture of 30% hydrogen peroxide (0.57 mL) and 16% sodium hydroxide (0.43 mL) at 0°C under argon. The subsequent reaction mixture was stirred at room temperature overnight. Saturated ammonium chloride (5 mL) solution was added to quench the reaction, and the resulting mixture was extracted with a mixture of DCM and MeOH (15:3, v/v) 3 times. The combined organic layers were dried over anhydrous sodium sulfate and concentrated via reduced pressure to give crude 5,7,20-O-trimethyl-2,3-dehydrosilybin (9) as a yellow solid which was subjected to the next reaction directly. Crude 5,7,20-O-trimethyl-2,3-dehydrosilybin (9, 400 mg, 0.77 mmol, 1eq) was dissolved in dimethylformamide (DMF, 14.5 mL), to which potassium carbonate (1.06 g, 7.69 mmol, 10 eq) was added. The reaction mixture was stirred at 105°C for 15 minutes before the addition of dimethyl sulfate (0.72 mL, 7.69, 10 eq). The reaction was allowed to stir at 105°C overnight prior to being quenched with saturated ammonium chloride (15 mL). The resulting mixture was extracted with a mixture of DCM and MeOH (45 mL [6:1, v/v] × 3). The combined organic layers were dried over anhydrous sodium sulfate and concentrated via reduced pressure. The crude product was subjected to column chromatography purification eluting with 100% ethyl acetate to give 3,5,7,20-O-tetramethyl-2,3-dehydrosilybin (7, 128 mg, 31% yield) as a yellow solid.
MP: 193-195°C.
Rf: 0.62 (DCM–MeOH, 91:9).
IR (potassium bromide [KBr]): 3418, 2932. 2837, 1625, 1607, 1505 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.77-7.73 (m, 2 H, aromatic H), 7.08-7.05 (m, 2 H, aromatic H), 6.98 (d, J = 1.8 Hz, 1 H, aromatic H), 6.93 (d, J = 8.1 Hz, 1 H, aromatic H), 6.48 (d, J = 2.4 Hz, 1 H, aromatic H), 6.33 (d, J = 2.4 Hz, 1 H, aromatic H), 5.03 (d, J = 8.4, 1 H, H-11), 4.14 (dt, J = 8.1, 3.3, 1 H, H-10), 3.95 (s, 3 H, OCH3), 3.93 (s, 3 H, OCH3), 3.92 (s, 3 H, OCH3), 3.90 (s, 3 H, OCH3), 3.87 (s, 3 H, OCH3), 3.90-3.83 (overlapped, 1 H, H-23), 3.58 (d, J = 12.6 Hz, 1 H, H-23).
13C NMR (75 MHz, CDCl3): δ 174.3 (C-4), 164.1 (C-7), 161.1 (C-5), 158.9 (C-8a), 152.1 (C-16a), 150.0 (C-19), 149.6 (C-20), 145.3 (C-2), 143.8 (C-12a), 141.6 (C-3), 128.4 (C-17), 124.5 (C-14), 122.4 (C-15), 120.4 (C-22), 117.3 (C-13), 117.2 (C-16), 111.5 (C-21), 110.2 (C-18), 109.6 (C-4a), 95.9 (C-6), 92.4 (C-8), 78.8 (C-10), 76.5 (C-11), 61.9 (C-23), 60.1 (OCH3), 56.6 (OCH3), 56.2 (OCH3), 56.1 (OCH3), 56.0 (OCH3).
HRMS-ESI: m/z [M + H+] calcd for C29H29O10: 537.1761; found: 537.1761.
Synthesis of 3,5,7,20,23-O-Pentamethyl-2,3-Dehydrosilybin (10)
To a solution of 5,7,20-O-trimethyl-dehydrosilybin (7, with minor impurity) (50 mg, 0.096 mmol, 1 eq) in DMF (0.7 mL) was added sodium hydride (23 mg, 60% dispersion in mineral oil, 0.60 mmol, 6 eq) at room temperature under argon. After stirring the subsequent reaction mixture for 30 minutes, methyl iodide (0.059 ml, 0.95 mmol, 10 eq) was added. The reaction was allowed to proceed with stirring overnight prior to being quenched by adding saturated ammonium chloride (2 mL). The resulting mixture was extracted with 10% MeOH in DCM (11 mL × 3), and the combined organic layers were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was then purified through PTLC eluting with 7% MeOH in DCM to furnish the desired product (45 mg) as a pale yellow solid in 85% yield.
MP: 133-134°C.
Rf: 0.38 (DCM–MeOH, 95:5).
IR (KBr): 3490, 2930, 2838, 1621, 1604, 1579, 1506 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.76 (d, J = 2.1 Hz, 1 H, aromatic H), 7.73 (dd, J = 8.7, 2.1 Hz, 1 H, aromatic H), 7.10 (d, J = 8.4 Hz, 1 H, aromatic H), 7.02 (dd, J = 8.1, 1.8 Hz, 1 H, aromatic H), 6.96 (d, J = 1.8 Hz, 1 H, aromatic H), 6.93 (d, J = 8.1 Hz, 1 H, aromatic H), 6.47 (d, J = 2.4 Hz, 1 H, aromatic H), 6.32 (d, J = 2.1 Hz, 1 H, aromatic H), 5.05 (d, J = 8.1, 1 H, H-11), 4.18 (ddd, J = 8.1, 2.1, 1.8, Hz, 1 H, H-10), 3.94 (s, 3 H, OCH3), 3.92 (s, 6 H, 2 × OCH3), 3.90 (s, 3 H, OCH3), 3.86 (s, 3 H, OCH3), 3.62 (dd, J = 11.1, 2.1 Hz, 1 H, H-23). 3.36 (s, 3 H, OCH3), 2.23 (dd, J = 11.1, 3.9 Hz, 1 H, H-23).
13C NMR (75 MHz, CDCl3): 174.3 (C-4), 164.0 (C-7), 161.1 (C-5), 158.9 (C-8a), 152.2 (C-16a), 149.8 (C-19), 149.5 (C-20), 145.5 (C-2), 143.8 (C-12a), 141.5 (C-3), 128.8 (C-17), 124.3 (C-14), 122.3 (C-15), 120.3 (C-22), 117.5 (C-13), 117.2 (C-16), 111.4 (C-21), 110.3 (C-18), 109.6 (C-4a), 95.9 (C-6), 92.4 (C-8), 78.1 (C-10), 76.4 (C-11), 71.3 (C-23), 60.1 (OCH3), 59.7 (OCH3), 56.6 (OCH3), 56.2 (OCH3), 56.0 (OCH3), 55.9 (OCH3).
HRMS-ESI: m/z [M + H+] calcd for C30H31O10: 551.1917; found: 551.1916.
Syntheses of 3,5,7,20-O-Tetramethyl-23-Dimethylcarbamoyl-2,3-Dehydrosilybin (11), 3,5,7,20-O-Tetramethyl-23-Dimethylythiocarbamoyl-2,3-Dehydrosilybin (12), and 23-Diethylcarbamoyl-3,5,7,20-O-Tetramethyl-2,3-Dehydrosilybin (13)
To a solution of 3,5,7,20-O-tetramethyl-2,3-dehydrosilybin (7) (30 mg, 0.056 mmol, 1 eq) in THF (0.5 mL) at room temperature, sodium hydride (2.9 mg, 60% dispersion in mineral oil, 0.073 mmol, 1.3 eq) was added under argon. The mixture was stirred for 30 minutes before dimethylcarbamoyl chloride (0.007 mL, 0.073 mmol, 1.3 eq) (or thiocarbamoyl chloride [9 mg, 0.073 mmol, 1.3 eq] or diethyl carbamoyl chloride dimethyl [0.0092 mL, 0.073 mmol, 1.3 eq]) was added. The reaction was allowed to proceed with stirring at room temperature overnight prior to being quenched by brine. The subsequent mixture was extracted with 20% MeOH in DCM (12 mL ×5), and the organic extracts were combined, dried over anhydrous sodium sulfate, and concentrated. PTLC purification of the crude product eluting with 5% MeOH in DCM followed by ethyl acetate furnished the respective desired product.
Derivative 11
Yield: 43%.
MP: 103-105°C.
Rf: 0.43 (DCM–MeOH, 95:5).
IR (KBr): 2931, 1702, 1625, 1605, 1505 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.77 (s, 1 H, aromatic H), 7.76 (dd, J = 10.5, 2.1 Hz, 1 H, aromatic H), 7.09 (d, J = 8.4 Hz, 1 H, aromatic H), 6.99 (dd, J = 8.1, 1.8 Hz, 1 H, aromatic H), 6.93 (d, J = 1.5 Hz, 1 H, aromatic H), 6.91 (d, J = 5.1 Hz, 1 H, aromatic H), 6.48 (d, J = 2.4 Hz, 1 H, aromatic H), 6.33 (d, J = 2.1 Hz, 1 H, aromatic H), 4.95 (d, J = 8.1 Hz, 1 H, H-11), 4.41-4.33 (m, 2 H, H-10, H-23), 4.03-4.00 (m, 1 H, H-23), 3.96 (s, 3 H, OCH3), 3.91 (s, 6 H, 2 × OCH3), 3.90 (s, 3 H, OCH3), 3.88 (s, 3 H, OCH3), 2.90 (s, 6 H, 2 × NCH3).
13C NMR (75 MHz, CDCl3): δ 174.0 (C-4), 164.0 (C-7), 161.2 (C-5), 158.9 (C-8a), 155.9 (carbamoyl carbon), 152.1 (C-16a), 150.1 (C-19), 149.7 (C-20), 145.2 (C-2), 143.8 (C-12a), 143.7 (C-3), 128.1 (C-17), 124.4 (C-14), 122.5 (C-15), 120.4 (C-22), 117.5 (C-13), 117.4 (C-16), 111.5 (C-21), 110.2 (C-18), 110.1 (C-4a), 96.0 (C-6), 92.5 (C-8), 77.0 (C-10), 76.5 (C-11), 63.9 (C-23), 60.2 (OCH3), 56.7 (OCH3), 56.6 (OCH3), 56.14 (OCH3), 56.10 (OCH3), 36.7 (NCH3), 36.1 (NCH3).
HRMS-ESI: m/z [M + H+] calcd for C32H34NO11: 608.2132; found: 608.2131.
Derivative 12
Yield: 44%.
MP: 111-113°C.
Rf: 0.50 (DCM–MeOH, 95:5).
IR (KBr): 2935, 2838, 1627, 1606, 1506 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.77 (s, 1 H, aromatic H), 7.76 (dd, J = 10.2, 2.4 Hz, 1 H, aromatic H), 7.08 (d, J = 9.0 Hz, 1 H, aromatic H), 7.00 (dd, J = 8.4, 1.8 Hz, 1 H, aromatic H), 6.93 (s, 1 H, aromatic H), 6.91 (d, J = 6.0 Hz, 1 H, aromatic H), 6.48 (d, J = 2.1 Hz, 1 H, aromatic H), 6.33 (d, J = 2.4 Hz, 1 H, aromatic H), 4.95 (d, J = 8.1 Hz, 1 H, H-11), 4.71 (dd, J = 12.0, 3.0 Hz, 1 H, H-23), 4.48 (ddd, J = 7.8, 4.5, 3.3 Hz, 1 H, H-10), 4.38 (dd, J = 11.7, 4.5 Hz, 1 H, H-23), 3.96 (s, 3 H, OCH3), 3.91 (s, 3 H, OCH3), 3.91 (s, 3 H, OCH3), 3.90 (s, 3 H, OCH3), 3.88 (s, 3 H, OCH3), 3.35 (s, 3 H, NCH3), 3.09 (s, 3 H, NCH3).
13C NMR (75 MHz, CDCl3): δ 187.4 (thiocarbamoyl carbon), 174.1 (C-4), 163.9 (C-7), 161.0 (C-5), 158.8 (C-8a), 151.9 (C-16a), 150.0 (C-19), 149.6 (C-20), 144.9 (C-2), 143.5 (C-12a), 141.4 (C-3), 127.8 (C-17), 124.4 (C-14), 122.4 (C-15), 120.2 (C-22), 120.0 (C-13), 117.2 (C-16), 117.1 (C-21), 111.4 (C-18), 109.9 (C-4a), 95.8 (C-6), 92.3 (C-8), 76.9 (C-10), 76.1 (C-11), 69.4 (C-23), 60.0 (OCH3), 56.4 (OCH3), 56.0 (2 × OCH3), 55.8 (OCH3), 42.9 (NCH3), 37.9 (NCH3).
HRMS-ESI: m/z [M + H+] calcd for C32H34NO10S: 624.1904; found: 624.1900.
Derivative 13
Yield: 40%.
MP: 95-96°C.
Rf: 0.37 (DCM–MeOH, 95:5).
IR (KBr): 2970, 2935, 2838, 2359, 2239, 1698, 1622, 1607, 1506 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.77 (s, 1 H, aromatic H), 7.75 (dd, J = 10.5, 2.1 Hz, 1 H, aromatic H), 7.08 (dd, J = 8.1, 0.9 Hz, 1 H, aromatic H), 6.98 (dd, J = 8.4, 1.8 Hz, 1 H, aromatic H), 6.93 (s, 1 H, aromatic H), 6.91 (d, J = 6.3 Hz, 1 H, aromatic H), 6.47 (d, J = 2.1 Hz, 1 H, aromatic H), 6.33 (d, J = 2.1 Hz, 1 H, aromatic H), 4.94 (d, J = 7.8 Hz, 1 H, H-11), 4.39–4.33 (overlapped, 2 H, H-10, H-23), 4.05-4.00 (m, 1 H, H-23), 3.95 (s, 3 H, OCH3), 3.91 (s, 6 H, 2 × OCH3), 3.90 (s, 3 H, OCH3), 3.87 (s, 3 H, OCH3), 3.35-3.18 (m, 4 H, 2 x NCH2
CH3), 1.14 (t, J = 7.2, 6 H, 2 x NCH2
CH3
).
13C NMR (75 MHz, CDCl3): δ 174.2 (C-4), 164.0 (C-7), 161.2 (C-5), 158.9 (C-8a), 155.4 (carbamoyl carbon), 152.2 (C-16a), 150.1 (C-19), 149.7 (C-20), 145.2 (C-2), 143.7 (C-12a), 141.6 (C-3), 128.2 (C-17), 124.4 (C-14), 122.5 (C-15), 120.3 (C-22), 117.4 (C-13), 117.3 (C-16), 111.5 (C-21), 110.2 (C-18), 109.6 (C-4a), 96.0 (C-6), 92.5 (C-8), 77.0 (C-10), 76.6 (C-11), 63.6 (C-23), 60.1 (OCH3), 56.6 (OCH3), 56.2 (OCH3), 56.1 (OCH3), 55.9 (OCH3), 42.0 (NCH2
CH3
), 41.5 (NCH2
CH3), 14.3 (NCH2
CH3
), 13.6 (NCH2
CH3
).
HRMS-ESI: m/z [M + H+] calcd for C34H38NO11: 636.2445; found: 636.2441.
Syntheses of 3,5,7,20-O-Tetramethyl-23-O-Methanesulfonyl-2,3-Dehydrosilybin (14) and 3,5,7,20-O-Tetramethyl-23-O-Toluenesulfonyl-2,3-Dehydrosilybin (15)
To a solution of 7 (100 mg, 0.19 mmol, 1 eq) in DCM (2 mL) at 0°C, triethylamine (0.037 mL, 0.28 mmol, 1.5 eq) and 4-dimethylaminopyridine (34.2 mg, 0.28 mmol, 1.5 eq) were sequentially added under argon. The solution was stirred for 15 minutes prior to the addition of methane sulfonyl chloride (0.017 mL, 0.224 mmol, 1.2 eq) (or 4-toluenesulfonyl chloride [42.6 mg, 0.224 mmol, 1.2 eq]).The reaction was allowed to proceed with stirring at room temperature overnight before quenching with a brine solution. The subsequent mixture was extracted with DCM, the combined extracts were dried over anhydrous sodium sulfate, and concentrated in vacuo. PTLC (5% MeOH then pure ethyl acetate) purification of the crude product afforded the respective product.
Compound 14
MP: 173-175°C.
Rf: 0.45 (DCM–MeOH, 95:5).
IR (KBr): 2936, 2839, 1625, 1606, 1518, 1506 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.78 (s, 1 H, aromatic H), 7.77 (dd, J = 10.2, 2.1 Hz, 1 H, aromatic H), 7.07 (d, J = 8.7 Hz, 1 H, aromatic H), 7.03 (dd, J = 8.4, 2.1 Hz, 1 H, aromatic H), 6.96 (s, 1 H, aromatic H), 6.95 (d, J = 10.5 Hz, 1 H, aromatic H), 6.48 (d, J = 2.1 Hz, 1 H, aromatic H), 6.34 (d, J = 2.4 Hz, 1 H, aromatic H), 4.99 (d, J = 8.4 Hz, 1 H, H-11), 4.46 (dd, J = 11.7, 2.7 Hz, 1 H, H-23), 4.35 (dt, J = 8.1, 2.7 Hz, 1 H, H-10), 4.14 (dd, J = 11.7, 3.9 Hz, 1 H, H-23), 3.96 (s, 3 H, OCH3), 3.93 (s, 3 H, OCH3), 3.92 (s, 3 H, OCH3), 3.90 (s, 3 H, OCH3), 3.88 (s, 3 H, OCH3), 3.10 (s, 3 H, SO2CH3).
13C NMR (75 MHz dimethylsulfoxide [DMSO]-d6
): 174.2 (C-4), 164.1 (C-7), 161.2 (C-5), 158.9 (C-8a), 151.9 (C-16a), 150.3 (C-19), 149.8 (C-20), 144.6 (C-2), 143.6 (C-12a), 141.7 (C-3), 127.3 (C-17), 125.0 (C-14), 122.7 (C-15), 120.3 (C-22), 117.5 (C-13), 117.2 (C-16), 111.6 (C-21), 110.2 (C-18 & C-4a), 96.0 (C-6), 92.5 (C-8), 76.2 (C-10), 75.9 (C-11), 68.1 (C-23), 60.1 (OCH3), 56.6 (OCH3), 56.24 (OCH3), 56.20 (OCH3), 55.9 (OCH3), 37.9 (SO2CH3).
HRMS-ESI: m/z [M + H+] calcd for C30H31O12S: 615.1537; found: 615.1532.
Compound 15
Yield: 65%.
MP: 165-166 °C.
Rf: 0.57 (DCM–MeOH, 95:5).
IR (KBr): 3004, 2937, 2839, 2239, 1625, 1605, 1505 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.76 (d, J = 8.4 Hz, 2 H, aromatic H), 7.73-7.69 (overlapped, 2 H, aromatic H), 7.34 (d, J = 7.8 Hz, 2 H, aromatic H), 6.96-6.86 (overlapped, 4 H, aromatic H), 6.46 (d, J = 2.4 Hz, 1 H, aromatic H), 6.32 (d, J = 2.1 Hz, 1 H, aromatic H), 4.94 (d, J = 8.1 Hz, 1 H, H-11), 4.26-4.19 (m, 2 H, H-10, H-23), 4.02-3.99 (m, 1 H, H-23), 3.94 (s, 3 H, OCH3), 3.91 (s, 6 H, 2 × OCH3), 3.88 (s, 3 H, OCH3), 3.86 (s, 3 H, OCH3), 2.46 (s, 3 H, Ar-CH3).
13C NMR (75 MHz, CDCl3): δ 174.2 (C-4), 164.1 (C-7), 161.1 (C-5), 158.9 (C-8a), 151.9 (C-16a), 150.1 (C-19), 149.7 (C-20), 145.3 (C-2), 144.7 (toluenesulfonyl), 143.4 (C-12a), 141.6 (C-3), 132.6 (toluenesulfonyl), 130.0 (toluenesulfonyl), 128.2 (C-17), 127.4 (toluenesulfonyl), 124.6 (C-14), 122.5 (C-15), 120.2 (C-22), 117.3 (C-13), 117.2 (C-16), 111.5 (C-21), 110.2 (C-18), 109.6 (C-4a), 95.9 (C-6), 92.4 (C-8), 76.0 (C-10), 75.7 (C-11), 68.2 (C-23), 60.1 (OCH3), 56.5 (OCH3), 56.1 (2 × OCH3), 55.9 (OCH3), 21.9 (Ar-CH3).
HRMS-ESI: m/z [M + H+] calcd for C36H35O12S: 691.1850; found: 691.1846.
Synthesis of 23-Azido-3,5,7,20-O-Tetramethyl-2,3-Dehydrosilybin (16)
To a solution of compound 15 (470 mg, 0.68 mmol, 1 eq) in DMF (4.9 mL), sodium azide (66.1 mg, 1.02 mmol, 1.5 eq) was added. The reaction mixture was stirred at 70°C for 16 hours prior to being diluted with DCM (20 ml). The subsequent suspension was rinsed with brine (2 ml ×5) and dried over anhydrous sodium sulfate. After removal of the organic solvents, the crude product was subjected to PTLC purification eluting with 5% MeOH in DCM first to remove the impurities and then eluting with 100% ethyl acetate to give the desired product (290 mg) in 70% yield.
MP: 92-93°C.
Rf: 0.50 (DCM–MeOH, 95:5).
IR (KBr): 3055, 2936, 2103, 1626, 1607, 1519, 1505 cm−1.
1H NMR (300 MHz, CDCl3): δ 7.78-7.74 (overlapped, 2 H, aromatic H), 7.11 (d, J = 8.7 Hz, 1 H, aromatic H), 7.01 (dd, J = 8.1, 1.8 Hz, 1 H, aromatic H), 6.94 (d, J = 2.1 Hz, 1 H, aromatic H), 6.93 (d, J = 8.4 Hz, 1 H, aromatic H), 6.48 (d, J = 2.1 Hz, 1 H, aromatic H), 6.33 (d, J = 2.1 Hz, 1 H, aromatic H), 4.98 (d, J = 8.1 Hz, 1 H, H-11), 4.26 (ddd, J = 8.1, 4.2, 2.7 Hz, 1 H, H-10), 3.95 (s, 3 H, OCH3), 3.93 (s, 3 H, OCH3), 3.92 (s, 3 H, OCH3), 3.90 (s, 3 H, OCH3), 3.88 (s, 3 H, OCH3), 3.60 (dd, J = 13.5, 2.7, 1 H, H-23), 3.18 (dd, J = 13.5, 4.2, 1 H, H-23).
13C NMR (75 MHz, CDCl3): δ 174.3 (C-4), 164.1 (C-7), 161.2 (C-5), 158.9 (C-8a), 152.0 (C-16a), 150.1 (C-19), 149.7 (C-20), 145.2 (C-2), 144.9 (C-12a), 143.7 (C-3), 127.9 (C-17), 124.7 (C-14), 122.5 (C-15), 120.3 (C-22), 117.4 (C-13), 117.3 (C-16), 111.5 (C-21), 110.1 (C-18), 109.6 (C-4a), 96.0 (C-6), 92.5 (C-8), 68.4 (C-10), 60.5 (C-11), 60.1 (OCH3), 56.6 (OCH3), 56.22 (OCH3), 56.19 (OCH3), 55.9 (OCH3), 51.2 (C-23).
HRMS-ESI: m/z [M + H+] calcd for C29H28N3O9: 562.1826; found: 562.1846.
Synthesis of 23-Amino-3,5,7,20-O-Tetramethyl-2,3-Dehydrosilybin (17)
To a solution of compound 16 (250 mg, 0.45 mmol, 1 eq) in THF (4.4 mL) was added triphenylphosphine (128 mg, 4.89 mmol, 1.1 eq) at room temperature under argon, and the reaction mixture was allowed to stir overnight. Water (1.11 mL) was then added into the reaction mixture, and the reaction was allowed to proceed for an additional 16 hours with stirring. Ethyl acetate (20 mL) and saturated ammonium chloride solution (8 mL) were added to quench the reaction. The resulting mixture was then extracted with ethyl acetate (15 mL ×3). The combined organic layers were dried over anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by PTLC eluting with 9% MeOH in DCM to furnish the amine 17 as a yellow solid in 42% yield.
MP: 112-115°C.
Rf: 0.53 (DCM–MeOH, 91:9).
IR (KBr): 3490, 2937, 1628, 1603, 1518, 1507 cm-=−1.
1H NMR (300 MHz, CD3COCD3): δ 7.72 (dd, J = 8.4, 2.1 Hz, 1 H, aromatic H), 7.69 (d, J = 1.5 Hz, 1 H, aromatic H), 7.15 (d, J = 2.1 Hz, 1 H, aromatic H), 7.08-7.00 (overlapped, 3 H, aromatic H), 6.78 (d, J = 2.4 Hz, 1 H, aromatic H), 6.44 (d, J = 2.1 Hz, 1 H, aromatic H), 5.18 (d, J = 7.8, 1 H, H-11), 4.48 (ddd, J = 7.8, 4.8, 3.3, 1 H, H-10), 3.94 (s, 3 H, OCH3), 3.90 (s, 3 H, OCH3), 3.86 (s, 3 H, OCH3), 3.85 (s, 3 H, OCH3), 3.85 (s, 3 H, OCH3), 3.47 (dd, J = 15.3, 3.0 Hz, 1 H, H-23), 3.27 (dd, J = 15.3, 4.8 Hz, 1 H, H-23), 2.88 (s, 2 H, NH2).
13C NMR (75 MHz, CD3COCD3): δ 173.3 (C-4), 165.1 (C-7), 162.0 (C-5), 159.7 (C-8a), 152.3 (C-16a), 150.9 (C-19), 150.5 (C-20), 146.8 (C-2), 145.0 (C-12a), 142.1 (C-3), 130.4 (C-17), 124.8 (C-14), 122.6 (C-15), 121.4 (C-22), 117.8 (C-13), 117.6 (C-16), 112.6 (C-21), 112.4 (C-18), 110.2 (C-4a), 96.6 (C-6), 93.7 (C-8), 79.3 (C-10), 78.2 (C-11), 59.9 (OCH3), 56.5 (OCH3), 56.4 (OCH3), 56.3 (OCH3), 56.2 (OCH3), 52.5 (C-23).
HRMS-ESI: m/z [M + H+] calcd for C29H30NO9: 536.1921; found: 536.1907.
Cell Culture
All cell lines were initially purchased from the American Type Culture Collection. The PC-3 and LNCaP prostate cancer cell lines were routinely cultured in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin. Cultures were maintained in a high humidity environment supplemented with 5% carbon dioxide at a temperature of 37°C. The DU145 prostate cancer cells were routinely cultured in Eagle’s Minimum Essential Medium supplemented with 10% FBS and 1% penicillin/streptomycin.
WST-1 Cell Proliferation Assay
PC-3, DU145, or LNCaP cells were plated in 96-well plates at a density of 3200 cells each well in 200 µL of culture medium. The cells were then treated with synthesized derivatives or silybin (as a positive reference) separately at different doses (1 µM, 3.125 µM, 6.25 µM, 12.5 µM, and 25 µM for compounds 7, 11-17, and 25 µM, 50 µM, 75 µM, 100 µM, and 125 µM for silybin) for 3 days, while equal treatment volumes of DMSO were used as vehicle control. After the cells were cultured in a carbon dioxide incubator at 37°C for 3 days, the premixed WST-1 cell proliferation reagent (10 µL, Clontech) was added to each well. The cells were incubated for an additional 3 hours at 37°C before mixing gently for 1 minute on an orbital shaker to ensure homogeneous distribution of color. A microplate reader (Synergy HT, BioTek) was used to measure the absorbance of each well at a wavelength of 430 nm. The IC50 value is the concentration of each compound that inhibits cell proliferation by 50% under the experimental conditions. Each IC50 value is represented as the average from triplicate determinations that were reproducible and statistically significant. The IC50 values were calculated based on dose–response curves from at least 5 dosages for each compound.
Statistical Analysis
All data are represented as the mean ± standard deviation for the number of experiments indicated. Other differences between treated and control groups were analyzed using the Student’s t-test. A P value <0.05 was considered statistically significant.