Abstract
Oral squamous cell carcinoma (OSCC) is one of the most common malignant tumors with high incidence, rapid progress, and high mortality. There are still some defects in the treatments of OSCC, which seriously affect the quality of patients’ life. Therefore, it is urgent to find a safer and more effective treatment for OSCC. Low molecular weight fucoidan (LMWF) has various biological activities, such as antitumor, anti-inflammatory, and antithrombotic, and has no obvious side effects, but the effect of LMWF on OSCC has not been reported. In this study, the effects of LMWF on dysplastic oral keratinocyte (DOK) and OSCC cells SCC-9 and SCC-25 were detected by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, flow cytometry, TUNEL, colony formation, and wound healing assay, and the expression levels of Notch-1 and Jagged-1 treated with LMWF were detected by RT-qPCR. The results showed that LMWF could inhibit the proliferation and migration of DOK and SCC-9, and promote apoptosis. Low molecular weight fucoidan upregulated the expression of Notch-1 and Jagged-1 in DOK and SCC-9 cells. It indicated that LMWF might promote the apoptosis of DOK and SCC-9 by upregulating the Notch signal pathway, thereby inhibiting cell proliferation and playing an antitumor role. It provided theoretical basis to develop LMWF as a novel therapeutic drug for oral cancer.
Keywords
Oral cancer is one of the common head and neck malignancies that seriously threaten human health, and its incidence rate ranks sixth in the world. 1 Oral squamous cell carcinoma (OSCC) is the main type of oral cancers, accounting for approximately 90% of oral cancers. 2 It is characterized by high incidence, rapid development, poor prognosis, and early lymph node metastasis. Various exogenous factors (such as mechanical stimuli, chemical factors, biological factors, and nutrition) and endogenous factors (such as psychology, endocrine, immunity, and gene) are the risk factors for its occurrence. 3 Studies have confirmed that smoking and drinking contributed to the development of OSCC. 4 Statistics showed that there were as nearly 150,000 patients died of OSCC per year globally. A total of 60% to 80% of patients were identified in the advanced clinical stage. 5 At present, surgical treatment, radiotherapy, chemotherapy, and comprehensive sequence therapy were the main treatments for OSCC. But the 5-year survival rate of the patients who were treated with those treatment methods mentioned above was not high, 55% to 60% were in the early stage and advanced patients are only 30% to 40%. 2 And the tissue defects, dysfunction, and side effects caused by those treatments seriously affect the physical and mental health of patients. With the development of oncobiology and cancer genetics, it is urgent to find more effective and safer therapeutic drugs to further improve the survival rate and quality of patients’ life, and reduce the recurrence rate. 6
Fucoidan is a kind of fucose-rich sulfated polysaccharide isolated from natural seaweed. It has no toxic and side effects on the human body. Because of its high efficiency and diverse biological activities, such as antitumor, anti-inflammatory, antithrombotic, anti-infection, and immune regulation, it has been widely used in many fields such as medicine, biomaterials, and food processing industry. 7 -9 The activity of fucoidan is related to its molecular weight and the sulfation degree. The lower the molecular weight and the higher the sulfation degree of LMWF are, the higher its activity is, and the content of sulfate is positively related to the ability of antitumor and antithrombus ability. 10,11 The sulfate content of low molecular weight fucoidan (LMWF) (<10 kDa) used in this study is much more than high molecular weight fucoidan, and the biological activity is more efficient. 12 Many studies found that LMWF could promote tumor cell apoptosis by regulating different signaling pathways, thereby further inhibiting the growth and proliferation of cell. 13 It has been confirmed that LMWF inhibited the growth of human cervical cancer cells Hela, 14 human breast cancer cells MCF-7, 15 leukemia cells K562, 16 and lymphoma cells U937, 17 but the role of LMWF in oral cancer has not been studied.
In this study, LMWF was applied to human oral mucosal precancerous cells and OSCC cells to investigate the effects of LMWF on the proliferation, migration, and apoptosis of dysplastic oral keratinocyte (DOK), SCC-9, and SCC-25 cells, and to determine its potential therapeutic effect on oral cancer. It will provide a certain experimental basis to develop new drugs for the prevention and treatment of oral cancer.
Results and Discussion
Fucoidan is the main component of Haikunshenxi Capsule. 18 It has been successfully used in the treatment of clinical nephrotic syndrome and early and middle stage of renal failure. It could effectively reduce blood creatinine and urea nitrogen, improve renal function, and alleviate the progress of disease. 11 The prevention and treatment of fucoidan in cardiovascular and cerebrovascular diseases and tumor got more and more attention. It has been widely used in preclinical trials of liver cancer, breast cancer, colon cancer, and cervical cancer. 19 In oral cancer, Lin et al 20 found that fucoidan decreased the invasiveness of tongue squamous cell carcinoma cell CAL27 by reducing the transcription of MMP-2 and KIF4A. The LMWF used in this study has stronger biological activity due to its high degree of sulfation and low molecular weight, but its effect on oral cancer cells has not been reported. Human oral keratinocyte (HOK) is a primary epithelial cell extracted from the normal oral cavity. Therefore, HOK was used as a control group in this study to investigate the effects of LMWF on oral precancerous cells and OSCC cells. The DOK cells used in this study were cell lines established by Chang et al 21 which are obtained from the epithelia of mild to moderate hyperplasia adjacent to the cancer. Because of its origin and partially transformed phenotype, DOK can be used as a cell model to study the influence of LMWF on oral precancerous lesions. As shown in Figure 1, different concentrations of LMWF had no significant inhibitory effect on the proliferation of HOK (P > .05), which proved that LMWF did not affect the growth of normal cells. For DOK cells, low concentration of LMWF had no obvious inhibitory effect on its proliferation. When the LMWF concentration reached or exceeded 50 µg/mL, it could significantly inhibit the proliferation of DOK (P < .05), indicating that higher concentrations of LMWF had an impact on the proliferation of DOK. As for SCC-9, low concentration of LMWF (5 µg/mL) had a significant inhibitory effect on the proliferation of SCC-9, and as the concentration increases, the inhibitory effect was dose dependent. When the concentration reached 50 µg/mL, there was no significant difference, indicating that LMWF had a significant effect on the proliferation of SCC-9. The proliferation activity of SCC-25 was inhibited only by the high concentration of LMWF (500 µg/mL) (P < .01). These results above proved that LMWF significantly inhibited the proliferation of DOK and SCC-9. In order to study the effect of LMWF on oral cancer further, 50 µg/mL LMWF was selected to intervene HOK, DOK, SCC-9, and SCC-25 cells to investigate the effect of LMWF. Ki-67 is a signature nucleoprotein antigen that exists in all human proliferating cells. It is expressed at all phases of the cell cycle except the G0 phase, and its expression can reflect cell proliferation activity. Immortality and infinite proliferation ability are the main characteristics of cancer cells. Generally, the lower the degree of tumor differentiation is, the higher the degree of malignancy and the expression level of Ki-67 are. Therefore, Ki-67 can be used to evaluate the malignancy and prognosis of tumors. 22,23 In order to further verify the effect of LMWF on cell proliferation, the change of Ki-67 transcription level was detected. It was found that Ki-67 expression levels in DOK and SCC-9 cells were significantly reduced after being treated with LMWF (50 µg/mL) for 24 hours, while Ki-67 transcription levels in HOK and SCC-25 were not significantly changed (Figure 2). The results are basically consistent with the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) experiment, which indicates that LMWF has a significant inhibitory effect on the proliferation of DOK and SCC-9 cells.

Effects of low molecular weight fucoidan on cell proliferation. Cells were treated with low molecular weight fucoidan (0, 5, 10, 20, 50, 100, 200, and 500 µg/mL) for 24 hours. Data are expressed as the mean ± SD. *P < .05, **P < .01, and ***P < .001 vs the control group.

Effects of low molecular weight fucoidan on the expression levels of Ki-67 mRNA in cells. Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours. Data are expressed as the mean ± SD. *P < .05 and **P < .01 vs the control group.
Since adhesion and colony formation are vital steps in tumor distant metastasis, the ability of all 4 cells to form detectable colonies was investigated. Figure 3 indicated that LMWF had no effect on colony formation of HOK. The colony formation assay demonstrated that the numbers of colonies reduced in DOK and SCC-9 cells treated with LMWF (P < .05), indicating that LMWF could suppress cell proliferation of these 2 cells. But there was no significant difference in the colony number of SCC-25 cells compared with the control group. These results are basically consistent with the previous results. Wound healing assay was performed to evaluate the migratory ability of tumor cells. Low molecular weight fucoidan treatment decreased the migration of DOK and SCC-9 cell migration compared with the control group (P < .05). But there was no significant difference in the wound healing rate of HOK and SCC-25 cells (Figure 4). These results indicate that LMWF had no effect on the migration of HOK, but inhibited the migration of DOK and SCC-9 rather than that of SCC-25. In summary, LMWF can inhibit the proliferation and migration of DOK and SCC-9 cells.

Effects of low molecular weight fucoidan on colony formation of dysplastic oral keratinocyte, dysplastic oral keratinocyte, SCC-9, and SCC-25 cells. Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours and the numbers of colonies were counted using Image J software. Data are expressed as the mean ± SD. ***P < .001 vs the control group.

Effects of low molecular weight fucoidan on migration of human oral keratinocyte, dysplastic oral keratinocyte, SCC-9, and SCC-25 cells (100×). Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours and the wound healing rates were calculated using Image J software. Data are expressed as the mean ± SD. *P < .05 and ***P < .001 vs the control group.
In order to investigate the effects of LMWF on cells apoptosis, we performed flow cytometry and TUNEL assay. As shown in Figure 5, there was no significant difference in the apoptosis rate of HOK cells after being treated with LMWF (50 µg/mL) for 24 hours, indicating that LMWF had no significant effect on HOK apoptosis. The apoptosis rate of DOK and SCC-9 cells increased significantly (P < .05), indicating that LMWF promoted apoptosis in DOK and SCC-9 cells. Although the apoptosis rate of SCC-25 cells increased, there was no statistical significance, indicating that the concentration of LMWF did not influence the apoptosis of SCC-25. When apoptosis occurs, DNA endonucleases are activated to cut off genomic DNA between nucleosomes and cause DNA fragmentation. The TUNEL method can mark broken DNA fragments, so that apoptotic cells can be quickly identified at the single-cell level. 24 As shown in Figure 6, no positive cells were detected by TUNEL staining of HOK, indicating that LMWF had no significant effect on HOK apoptosis. The number of TUNEL-positive cells was increased in DOK and SCC-9 cells compared to that of the control group after being treated with LMWF, while no positive SCC-25 cells were detected. It indicated that LMWF could promote the apoptosis of DOK and SCC-9, but had no obvious effect on SCC-25. These are consistent with the results of flow cytometry. These results above showed that LMWF did not affect the proliferation, migration, and apoptosis of normal oral mucosal cells, so was on SCC-25, while it inhibited proliferation and migration, and promoted apoptosis of DOK and SCC-9 cells. SCC-9 and SCC-25 were both tongue-derived OSCC cell lines, but the effects of LMWF on different cell lines were different, suggesting that indications should be considered in the clinical application of LMWF.

Effects of low molecular weight fucoidan on cell apoptosis. Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours. Data are expressed as the mean ± SD. **P < .01 and ***P < .001 vs the control group.
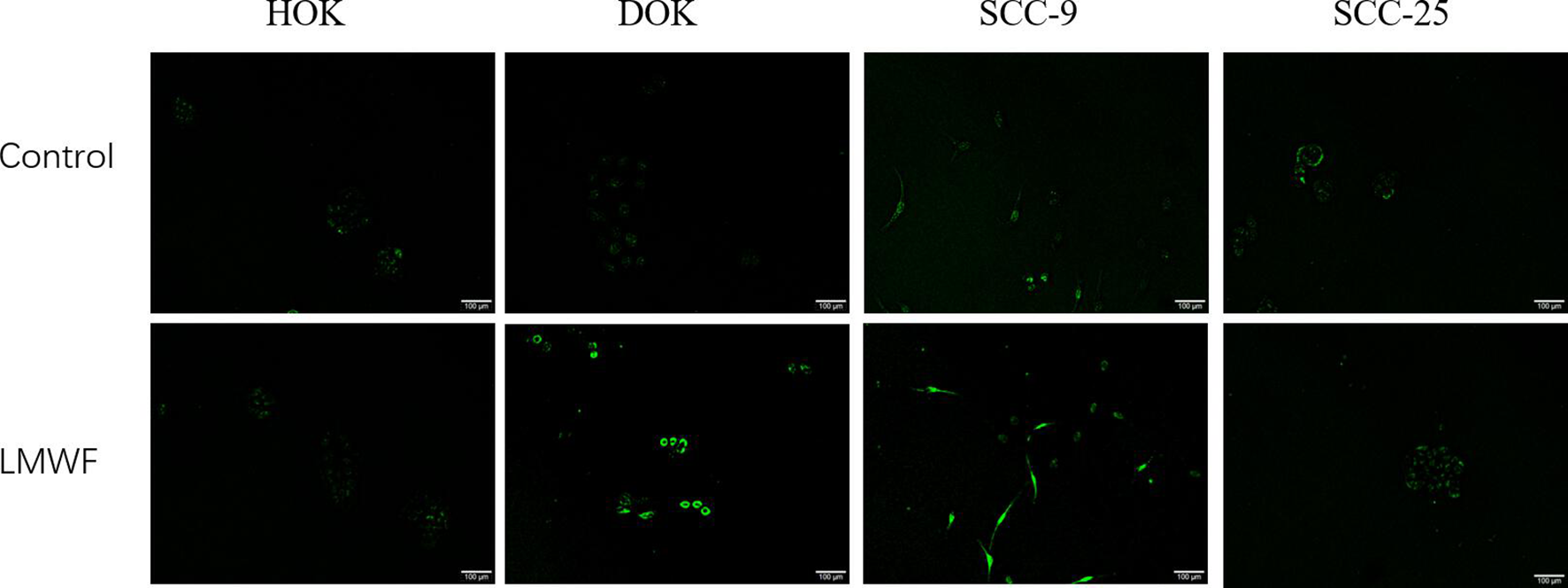
Cell apoptosis was measured by TUNEL staining. Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours. Representative images were photographed and positive cells appeared green (original magnification 100×).
Notch signaling pathway is a highly conserved signal transduction pathway that plays an important role in regulating the growth, proliferation, differentiation, and apoptosis of various tissues and cells. Currently, there are 4 types of Notch signal receptors that have been discovered, namely Notch-1 to -4, and its 5 ligands are Delta-like 1, 3, 4, Jagged-1, and Jagged-2. 25 Notch-1 plays an important role in the occurrence and development of tumors. However, due to different tissue sources and microenvironment of different tumors, Notch-1 exhibited dual effects in promoting and suppressing cancer. In colon cancer and breast cancer, the expression of Notch-1 was increased, which promoted tumor development. 26,27 In esophageal cancer and skin cancer, the decreased expression of Notch-1 inhibited cell differentiation, promoted cell proliferation, and induced tumorigenesis. 28,29 Statistics showed Notch-1 was one of the most frequently mutated genes in the occurrence and development of systemic squamous cell carcinoma. The mutation rates of Notch-1 in head and neck squamous cell carcinoma and OSCC could reach 9% to 17%, and most of them were inactivated mutations. 30 Lacking of the regulatory effect of Notch-1, which mediated the occurrence of tumors, the proliferation and differentiation of keratinocytes in the epithelial basal layer were out of control. 31 At present, the role of Notch-1 in OSCC is still not clear, but most of the studies found that Notch-1 expression was decreased in OSCC and oral epithelial dysplasia. 32,33 Therefore, we picked 2 key factors in this pathway, Notch-1 and Jagged-1, to explore the effect of LMWF on DOK and OSCC cells. Compared with HOK, the mRNA expression levels of Notch-1 in DOK, SCC-9, and SCC-25 were significantly decreased (P < .001), while that of Jagged-1 only decreased in DOK (P < .001). Compared with HOK, the expression levels of Jagged-1 mRNA in SCC-9 and SCC-25 had no significant difference (Figure 7(a)), indicating that the downregulation of Notch-1 may promote the incidence of oral cancer and the malignant transformation of precancerous lesions, and the downregulation of Jagged-1 only effected on precancerous cells. After being treated with LMWF (50 µg/mL) for 24 hours, the expression levels of Notch-1 and Jagged-1 mRNA in DOK and SCC-9 were increased significantly, while Notch-1 and Jagged-1 mRNA were still decreased in SCC-25 (Figure 7(b)). These results showed that LMWF could activate the Notch signaling pathway in DOK and SCC-9, and promote the expression of Notch-1 and Jagged-1 in mRNA levels. It is speculated that LMWF might promote the apoptosis of DOK and SCC-9 and inhibit its proliferation by activating Notch-1 signal pathway. Duan et al 34 also found that overexpression of Notch-1 might inhibit the proliferation and promote apoptosis of tongue squamous carcinoma cell Tca8113 by downregulating the Wnt/β-catenin signaling pathway. Interestingly, the transcription levels of Notch-1 and Jagged-1 in SCC-25 were further reduced after the effect of 50 µg/mL LMWF. As only the high concentration of LMWF (higher than 500 µg/mL) can inhibit the proliferation of SCC-25, we speculated that the effect of LMWF on SCC-25 is related to its concentration.

Expression levels of Notch-1 and Jagged-1 mRNA in cells. (a) Expression levels of Notch-1 and Jagged-1 mRNA in human oral keratinocyte, dysplastic oral keratinocyte, SCC-9, and SCC-25 cells. Data are expressed as the mean ± SD. ***P < .001 vs the HOK group. ### P < .001 vs the dysplastic oral keratinocyte group. (b) Effects of low molecular weight fucoidan on the expression levels of Notch-1 and Jagged-1 mRNA in dysplastic oral keratinocyte, SCC-9, and SCC-25 cells. Cells were treated with low molecular weight fucoidan (50 µg/mL) for 24 hours. Data are expressed as the mean ± SD. *P < .05 and ***P < .001 vs the control group.
Conclusion
Low molecular weight fucoidan could promote the apoptosis of human oral precancerous cells and OSCC cells, inhibit their proliferation and migration activity. It played an antitumor role by activating the Notch signaling pathway and upregulating the expression of Notch-1 and Jagged-1, which proved that LMWF may be a potential new drug for the treatment of oral cancer.
Experimental
Materials
Low molecular weight fucoidan is provided by the Institute of Oceanology, Chinese Academy of Sciences, molecular weight = 8177 Da. The component analysis was authenticated as follows: fucose content 35.07%, sulfate content 36.85%, and uronic acid content 0.039%. Low molecular weight fucoidan was dissolved in distilled water. 35
The following reagents were used: Fetal bovine serum was provided by Biological Industries, Israel. Dulbecco’s modified eagle medium (DMEM, High glucose) and RPMI 1640 medium were provided by Hyclone (UT, United States). Penicillin/streptomycin, trypsin, and MTT Cell Proliferation and Cytotoxicity Assay Kit were supplied by Solarbio Biotechnology Co., Ltd (Beijing, China). Annexin V-FITC Apoptosis Detection Kit and One-step TUNEL Apoptosis Assay Kit were purchased from Beyotime Biotechnology Co., Ltd (Shanghai, China). Trizol Reagent Kit, PrimeScript RT reagent Kit and SYBR Premix Ex Taq II Kit were supplied by TaKaRa Co., Ltd (Dalian, China). All PCR primers were synthesized by Sangon Biotech (Shanghai, China).
Cell Lines
Human oral keratinocytes (HOK, C1366) and dysplastic oral keratinocyte (DOK, C388) cell strains were provided by school of medicine, Wuhan university (Wuhan, China). Human OSCC cell lines SCC-9 (C405) and SCC-25 (C400) were provided by American Type Culture Collection (ATCC, VA, United States). Human oral keratinocyte, SCC-9, and SCC-25 cells were cultured in DMEM supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin. Dysplastic oral keratinocyte was cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin. All cells were cultured in a humidified incubator with 5% CO2 at 37°C.
3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide Cell Proliferation and Cytotoxicity Assay
Human oral keratinocyte, DOK, SCC-9, and SCC-25 cells were seeded in 96‐well plates, respectively, with a density of 5 × 103 cells/well in 100 µL medium. Cells were incubated with LMWF (0, 5, 10, 20, 50, 100, 200, and 500 µg/mL) for 24 hours at 37°C, followed by the addition of 10 µL/well MTT. After incubated for 4 hours at 37°C, the supernatant was discarded and the precipitate was dissolved in 110 µL formazan solution. Optical density was measured using an automatic enzyme mark instrument (Bio-Tek, United States) at 490 nm.
Colony Formation Assay
A total of 50 µg/mL LMWF was selected as the optimal concentration according to the results of half maximal inhibitory concentration (IC50) and MTT assay. To evaluate the ability to form sizable colonies, cells were seeded in 6-well culture plates (1000 cells/well) and left at 37°C for 4 hours to attach. Then, cells were treated with LMWF (50 µg/mL) for 24 hours. The cells were cultured at rest for 10 days and the medium was replaced every 2 to 3 days. After washing with phosphate-buffered saline (PBS) 3 times, the colonies were fixed with methanol for 15 minutes and stained with 0.1% crystal violet at room temperature for 15 minutes. Numbers of colonies were counted using Image J software (National Institutes of Health, MD, United States) and 3 parallel wells were set up.
Wound Healing Assay
Cells were seeded in 6-well culture plates (5 × 104 cells/well) and incubated until reaching the confluence. Two straight vertical wounds were generated using a 200 µL pipette tip. Photographs were taken under an inverted phase contrast microscope at 0 and 24 hours, and the distance between the edges of the wound was measured. The wound healing rate was (width value of initial scratch − width value of current scratch)/width value of initial scratch × 100%. Three parallel wells were set up.
Apoptosis Assay
Cells were seeded in 12-well plates (1 mL/well contained 1 × 105 cells) and treated with LMWF (50 µg/mL) for 24 hours at 37°C. Cultured cells were collected after trypsin digestion and washed in PBS twice. Then, cells were resuspended in 195 μL Annexin-binding buffer and labeled with 5 µL Annexin V-FITC and 10 μL propidine iodide (PI) for 20 minutes in the dark at room temperature according to the manufacturer’s instructions. The apoptotic rate was detected using a DxFLEX flow cytometer (Beckman Coulter, United States) and analyzed using CytExpert for DxFLEX software (Beckman Coulter, United States).
TUNEL Assay
DNA fragmentation in cells treated with LMWF (50 µg/mL) was assessed by One-step TUNEL Apoptosis Assay Kit. After incubated with or without LMWF for 24 hours, the cells were fixed with 4% paraformaldehyde for 30 minutes at room temperature, then permeated with 0.3% Triton-x-100 for 5 minutes. The cells were incubated with 50 µL TUNEL detection liquid which is prepared according to the manufacturer’s protocol for 60 minutes in the dark at 37°C. Fluorescence intensity was detected with a fluorescence microscope and positive cells appeared green.
RT-qPCR Assay
The total RNA was extracted from the untreated cells and cells stimulated for 24 hours with LMWF (50 µg/mL) using a Trizol Kit. The RNA concentration was assessed using a Micro Drop Ultra Micro spectrophotometer (Bio-DL, TX, United States). The RNA was reverse transcribed using a Prime Script RT reagent Kit. RT-qPCR was performed in a CFX96TM Real-Time system (BIO-RAD, CA, United States). The reaction was performed in a 20 µL volume that contained 10 µL of SYBR Premix Ex Taq II, 1 µL of forward primers (final concentration 0.4 µmol/L), 1 µL of reverse primers (final concentration 0.4 µmol/L), and 0.1 µg of cDNA template. Relative expressions were normalized to β-actin for endogenous control. ΔCt was recorded automatically and relative expression levels were calculated by the 2−ΔΔCt method. All the primer sequences used in the study are listed in Table 1.
Primers for RT-qPCR.

Statistical Analysis
The data were presented as mean ± SD. Statistical analysis were performed using Student’s t-test and one-way ANOVA with the statistical software “Graph Prism 8.0” (GraphPad Software, United States). P < .05 was statistically significant.
Footnotes
Acknowledgments
The authors would like to thank Prof. Quanbin Zhang from the Institute of Oceanology, Chinese Academy of Sciences for providing fucoidan samples extracted from Saccharina japonica. They would also like to thank the anonymous reviewers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of China (Grant no. 81902783), the Natural Science Foundation of Shandong Province (Grant no. ZR2017BD035), the Traditional Chinese Medicine Research Project of Qingdao (Grant no. 2019-zyy049), and the Health Science and Technology Project of Qingdao (Grant no. 2018-WJZD102).
