Abstract
This research was conducted to identify the anti-aging effects of gyrophoric acid on the skin, using normal human dermal fibroblasts. The anti-aging effects of gyrophoric acid on dermal fibroblasts were demonstrated through cell viability, verification of collagen, type I, alpha 1 (COL1A1)/COL3A1/matrix metalloproteinases 1 (MMP1) messenger ribonucleic acid (mRNA) expression levels with quantitative real-time reverse-transcription polymerase chain reaction, and protein estimation using type I collagen/MMP1-enzyme-linked immunosorbent assay. Further, the effects of gyrophoric acid on superoxide dismutases (SODs)/catalase were investigated by assessing their mRNA expression. In ultraviolet A (UVA)-treated dermal fibroblasts, gyrophoric acid was observed to increase mRNA levels of COL1A1/COL3A1/SOD2 genes and type I collagen protein levels, consistent with its anti-aging role. Furthermore, gyrophoric acid treatment decreased both MMP1 mRNA and protein expression levels. Therefore, the results of this study demonstrate that gyrophoric acid can be considered as an important natural compound with potent anti-aging effects on the skin. Based on the findings of this study, further research about the mechanism of action of gyrophoric acid should be pursued so as to develop novel anti-aging strategies not only in the field of cosmetics but also for healthcare.
Aged skin is generally thin and fragile, with increased susceptibility to bruising and impaired wound healing. 1 This phenomenon, commonly observed in the elderly, is caused by the degradation and reduction of extracellular matrix (ECM) that is composed of a large amount of type I collagen. Dermal fibroblasts secrete type I procollagen, which is converted into mature collagen. 2 Mature collagen has a half-life of about 15 years and constitutes a stable dermis. However, as aging progresses, mature collagen is gradually degraded by matrix metalloproteinases (MMPs). 3,4 Thus, as the functionality of fibroblasts of the dermis declines, ECM of the dermis degrades gradually and dermal degeneration occurs. 1
Fibroblasts attach to the collagen fibers of the dermis that exert contractile forces on their cytoskeleton. These contractile forces generate mechanical forces within fibroblasts, which largely determine the morphology, signaling pathways, and gene expressions. 5 -7 Collagen denaturation and degradation causes a loss in the attachment site of fibroblasts, which in turn reduces fibroblast proliferation and accelerates skin aging as well. 1,2,8
Skin, being the outermost layer of the human body, is not only exposed to ultraviolet (UV) rays but is also directly exposed to oxygen in the atmosphere that generates excessive reactive oxygen species (ROS). Exposure of the skin to UV can also cause abnormalities in antioxidant enzymes such as superoxide dismutases (SODs), glutathione peroxidases, and catalase (CAT) leading to an imbalance in the antioxidant system. 9,10
To restrict aging caused by UV, several studies have been conducted to discover natural compounds derived from certain organisms, which could act as sunscreens. Particularly, the compounds vulpinic acid and gyrophoric acid, isolated from lichen, have the effect of blocking UV radiation. 11 -13 Gyrophoric acid is a type of polyphenol, composed of 2 or more monocyclic aromatic units connected by ester bonds (Figure 1a). 12,14 Gyrophoric acid is reported to have the ability to block UV rays including UVB. 15 However, currently, there are no studies that have determined if gyrophoric acid can reduce the effects of aging caused due to UV radiation in human dermal fibroblasts and if it can improve the anti-aging effects by regulating antioxidant enzymes.
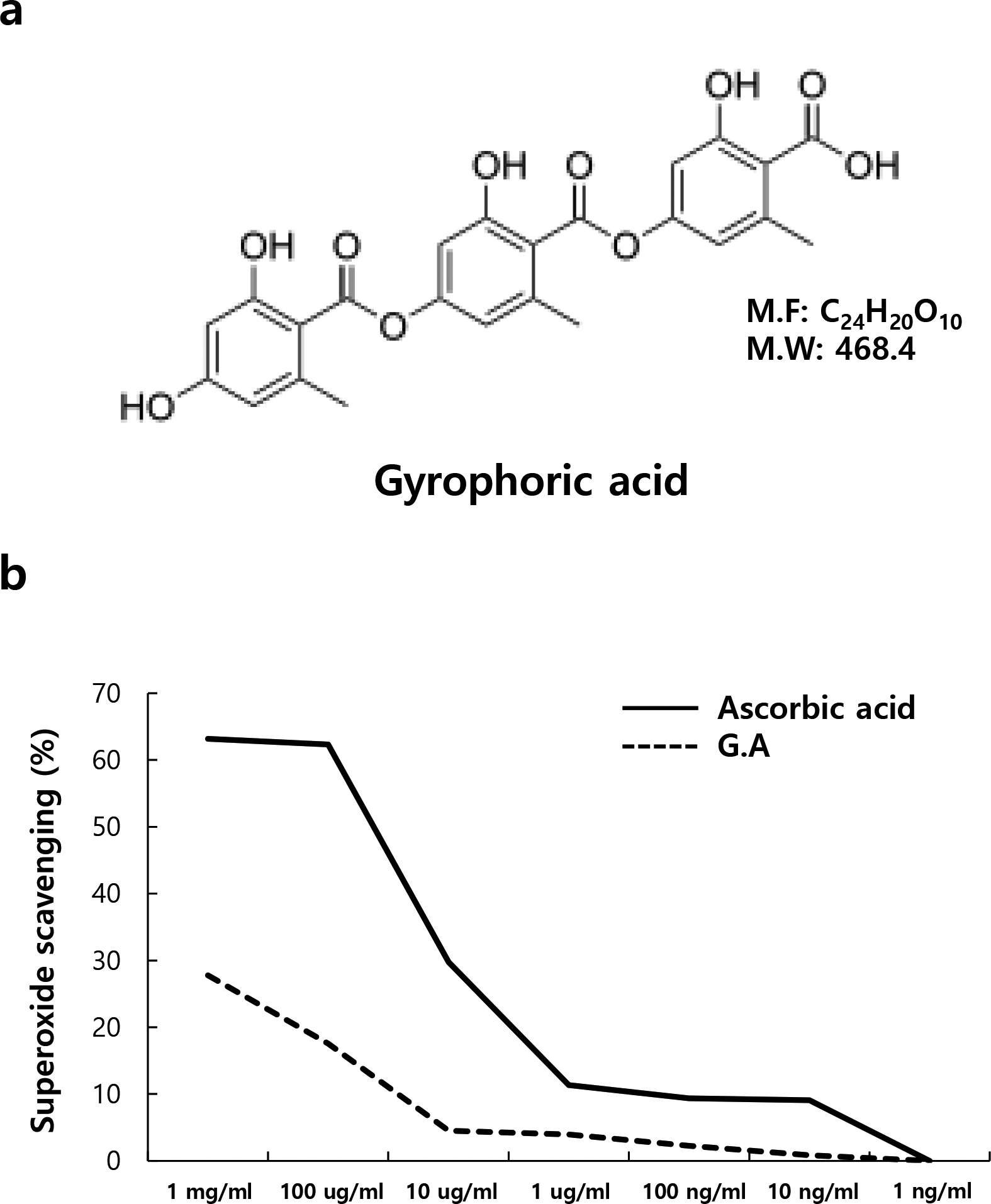
(a) The chemical structure of gyrophoric acid. (b) Free radical scavenging effects of gyrophoric acid. Antioxidant ability of gyrophoric acid was measured by the 2,2-diphenyl-1-picrylhydrazyl assay at the indicated concentrations. Ascorbic acid was used as a positive control. Gyrophoric acid showed free radical scavenging effects at higher concentrations.
Excessive generation of ROS could lead to diseases such as aging and cancer, and ROS has been reported to play an important role in skin photoaging. 16 Gyrophoric acid is a type of depside and has been reported to have antioxidant effects among several other lichen-derived compounds (Figure 1a). 2,2-Diphenyl-1-picrylhydrazyl (DPPH) radical assay is commonly used to evaluate the radical scavenging ability of antioxidants. DPPH is used to measure the electron-donating ability of relatively stable free radicals present in natural antioxidants. Electron donation by the antioxidant quenches the DPPH radical, causing its purple color to turn a yellow non-radical compound. 17 Gyrophoric acid was serially diluted with concentrations ranging from 1 mg/mL to 1 ng/mL and the radical scavenging ability was measured. The results (Figure 1b) show that the electron-donating ability of gyrophoric acid increased the DPPH radical scavenging ability in a concentration-dependent manner. Treatment with 1 mg/mL and 100 µg/mL of gyrophoric acid showed antioxidative effects and is consistent with a previous report. 18
Cell counting kit (CCK-8) was performed to determine the effect of gyrophoric acid on cytotoxicity of normal human dermal fibroblasts. To measure cell viability, the control group was untreated, and the test groups were treated with gyrophoric acid in the following concentrations—100, 10, 1 µg/mL and 100, 10, 1, 0.1 ng/mL (Figure 2a). As cell viability decreased significantly when the concentration was greater than 100 µg/mL, CCK-8 assay was performed with concentrations ranging between 100 µg/mL and 10 µg/mL. The survival rate of the fibroblasts significantly decreased when treated with more than 20 µg/mL of gyrophoric acid (Figure 2b). Therefore, 10 µg/mL of gyrophoric acid treatment was used for further experiments.

Cytotoxicity of gyrophoric acid in normal human dermal fibroblasts (NHDFs). NHDFs (2 × 104 cells) were seeded in 96-well plate and treated at indicated concentrations of gyrophoric acid for 24 hours. Cell viability was measured using cell counting kit assay. The results are represented as mean ± standard deviation of the percentage of control optical density in triplicate (a, b). Asterisk indicates means compared with control *P < 0.05
UV rays are classified depending on their wavelength into UVA (long wavelength, 320-400 nm), UVB (medium wavelength, 290-320 nm), and UVC (short wavelength, 200-290 nm). 19,20 In this study, dermal fibroblasts were treated with UVA to induce extrinsic aging, with gyrophoric acid to check its anti-aging effect and both UVA and gyrophoric acid to understand if the effect of UVA on aging was rescued by gyrophoric acid. Therefore, fibroblasts were irradiated with 5 J/cm2 of UVA and treated with 10 µg/mL of gyrophoric acid in different groups and their effect was measured by analyzing the expression levels of COL1A1, MMP1, and COL3A1 using quantitative real-time polymerase chain reaction (Q-RT-PCR). UVA irradiation has been reported to decrease the expression of fibroblast markers such as COL1A1 and COL3A1 and increase the expression of MMP1. 21 As predicted, the fibroblasts treated with gyrophoric acid showed an increase in the expressions of COL1A1 and COL3A1 by 2.2-fold and 2.56-fold, respectively, compared with the UVA-irradiated group (Figure 3a,c). On the other hand, MMP1 expression showed a 77% reduction compared with the UVA-treated group (Figure 3b). These results suggest that gyrophoric acid induces the expression of type I and III collagen, which occupy most of the ECM of the dermis, and reduces the expression of the MMP1 messenger ribonucleic acid (mRNA) that causes the degradation and degeneration of the ECM.

Characterization of gyrophoric acid (10 μg/mL) treatment on ultraviolet A (UVA)-irradiated normal human dermal fibroblasts. Expression of the dermal fibroblast markers. After UVA irradiation, gyrophoric acid was treated for 24 hours. COL1A1 (a), MMP1 (b), and COL3A1 (c) obtained using real-time reverse transcription-polymerase chain reaction. Values represented as mean ± standard deviation of 3 independent experiments. *Means compared with control, **means compared with UVA-irradiated condition *, **P < 0.05.
To determine whether the production of type I collagen protein also increased in fibroblasts treated with gyrophoric acid, the expression of proteins was measured in the cell culture supernatant using enzyme-linked immunosorbent assay (ELISA) for type I collagen (Figure 4a). The results confirm that type I collagen, which reduced by 39% in UVA-irradiated fibroblasts, in contrast increased by 61% in fibroblasts treated with both UVA and gyrophoric acid. In addition, the production of MMP1 protein in fibroblasts after gyrophoric acid treatment was determined using MMP1-ELISA. This assay confirms that MMP1, increased by 57% in UVA-irradiated fibroblasts as opposed to a 33% reduction in UVA-gyrophoric acid-treated fibroblasts (Figure 4b). Western blot results showed a similar trend as ELISA results (Figure 4c). These results display the same trend as the experimental results obtained using Q-RT-PCR for the expression of COL1A1 and MMP1 mRNAs. Gyrophoric acid not only affects the expression of genes but also exhibits consistent anti-aging effects at the protein levels.

Effects of gyrophoric acid on type I collagen and matrix metalloproteinase 1 (MMP1) protein synthesis in normal human dermal fibroblasts (NHDFs). NHDFs (10 × 104 cells) were seeded in 35 mm culture dish. One day later, ultraviolet A (UVA, 5 J/cm2) was irradiated and treated with gyrophoric acid (10 ug/mL) for 48 hours. Cell culture supernatant was collected and analyzed for type I collagen (a) or MMP1 (b) using enzyme-linked immunosorbent assay. Immunoblotting analysis for COL1 and MMP1 (c). Values are represented as mean ± standard deviation of 3 independent experiments. *Means compared with control, **means compared with UVA-irradiated condition *, **P < 0.05.
The generation of ROS, induced by UV irradiation, is known to be an early signal that promotes the production of MMPs such as MMP1 22 -25 and regulated by antioxidative enzymes such as SODs and CAT. In mammals, there are 3 isoforms of SODs: (SOD1[CuZnSOD]; SOD2[MnSOD]; SOD3[EC-SOD]). SOD1 is present in the cytoplasm and induces antioxidative effects by using copper and zinc as co-factors; SOD2 is mainly present in the mitochondria with manganese as a co-factor; and SOD3 is secreted in the extracellular region and is known as an antioxidant enzyme that also uses copper and zinc as co-factors. SODs catalyze the superoxide (O2 •−) into oxygen and hydrogen peroxide (H2O2). The enzyme CAT then reduces the H2O2 produced by SODs to water and oxygen. 26 Since gyrophoric acid is known to have an antioxidative effect, the following experiment was carried out to determine if gyrophoric acid could regulate the expression of antioxidative enzymes changed by UV irradiation. 18,27 The fibroblasts were treated with UVA/gyrophoric acid and the expression of SOD1, SOD2, SOD3, and CAT genes was examined by Q-RT-PCR. The expression of SOD2, SOD3, and CAT genes significantly decreased after UVA treatment, and upon gyrophoric acid treatment, only the expression of SOD2 significantly increased 2.2 times with respect to the UVA-treated group (Figure 5b). In an animal model, oxidative stress caused by SOD2 deficiency promotes aging in the skin. 28 Recovery of SOD2 expression by gyrophoric acid in human dermal fibroblasts was firstly demonstrated in this study. Further research is needed to determine why gyrophoric acid specifically increases the expression of SOD2 in dermal fibroblasts.

Expression of antioxidant enzymes after gyrophoric acid treatment on ultraviolet A (UVA)-irradiated normal human dermal fibroblasts (NHDFs). After UVA irradiation, gyrophoric acid (10 ug/mL) was treated for 24 hours. Real-time reverse transcription-polymerase chain reaction analysis of the representative antioxidative enzyme markers, (a) superoxide dismutase (SOD)1, (b) SOD2, (c) SOD3, and (d) catalase (CAT). The values on the graphs are shown as mean ± standard deviation of 3 independent experiments. *Means compared with control, **means compared with UVA-irradiated condition *, **P < 0.05.
Aging caused by extrinsic factors, such as exposure to UV rays, blocks the regeneration and recovery of normal skin tissue by decreasing their cell functionality. 29,30 First, this study has determined the optimal concentration of gyrophoric acid on dermal fibroblasts and has demonstrated that 10 µg/mL of gyrophoric acid does not affect the viability of these cells (Figure 2). More importantly, analysis of COL1A1/COL3A1 gene expression and type I collagen protein expression has shown that gyrophoric acid treatment can significantly induce their expression in fibroblast even after UVA irradiation. On the other hand, the expression of the MMP1 gene and protein revealed a decrease after treatment with gyrophoric acid and a corresponding increase upon UVA irradiation (Figures 3 and 4). In addition, gyrophoric acid treatment revealed an induction in the gene expression of SOD2, an antioxidant enzyme that regulates the availability of free radicals which in turn determines the activity of ECM peptidases such as MMP1 (Figure 5). Based on these results, we can conclude that gyrophoric acid could be considered to act as a potential and a novel skin anti-aging natural material.
Experimental
Cell Culture
Normal human dermal fibroblasts (NHDFs; Lonza, adult abdominal skin) were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Welgene) containing 10% fetal bovine serum (FBS; Welgene), 100 U/mL of penicillin, and 100 µg/mL of streptomycin (Invitrogen, USA) in a 5% carbon dioxide and 37°C incubator. NHDFs were used for experiments at passages 3 through 8. The gyrophoric acid used in this experiment was purchased from ChemFaces Co.(CFN98919, purity: ≥98%). According to the manufacturer, gyrophoric acid was extracted from herbs of Parmelia tinctorum Despr.
UVA Irradiation and Sample Treatments
Before UVA irradiation, NHDFs were washed 2 times with phosphate-buffered saline (Welgene) and protected from drying by adding phenol red-free DMEM (Welgene). After inoculating 1 mL of phenol red-free DMEM into 35 mm tissue culture dish, UVA of 5 J/cm2 was irradiated with a UVA irradiation device (BioLink). To assess the effects of UVA/gyrophoric acid on COL1A1, MMP1, and SOD2 expression of NHDFs, NHDFs were cultured in serum-free DMEM in the presence of gyrophoric acid after UVA irradiation. Cells were treated with 10 ug/mL of gyrophoric acid for 24 hours (for Q-RT-PCR) or 48 hours (for ELISA and western blot).
DPPH Assay
For the DPPH radical (Sigma-Aldrich) scavenging assay, DPPH was dissolved in methanol:water (3:2) at 0.1 mM concentration. Diluted gyrophoric acid (50 µL) was added to the DPPH solution (500 µL) and allowed to react at room temperature (RT) for 20 minutes, followed by measurement of absorbance using a microplate spectrophotometer (Epoch; BioTek Instruments) at 517 nm wavelength. Ascorbic acid (Sigma-Aldrich) was used as a positive control.
Cell Viability Assay
Cell viability was evaluated by CCK-8 solution (EZ-Cytox, DoGen). NHDFs were seeded with the appropriate culture medium at a density of 2 × 104 cells/well of a 96-well plate and treated with gyrophoric acid. NHDFs treated with gyrophoric acid were incubated for 24 hours and then incubated with CCK-8 solution for 30 minutes at 37°C. Absorbance was measured at 450 nm using a microplate reader (BioTek) and cell viability was calculated using the absorbance of the control group without cells.
Reverse Transcription and Q-RT-PCR
Total RNA was isolated with Trizol (Thermo Fisher Scientific) using manufacturer’s protocol. The concentration of RNA was determined spectrophotometrically (BioTek). Four micrograms of RNA were reverse-transcribed into complementary deoxyribonucleic acid (cDNA) using ReverTra Ace reverse transcription kit (Toyobo). The reverse transcription was stopped by adding Tris–ethylenediaminetetraacetic acid buffer (pH 8.0) to a total of 200 µL of cDNA solution. The TaqMan Gene Expression Assay sets were purchased from Applied Biosystems. Q-RT-PCRs were done according to the manufacturer’s instructions. Briefly, 20 µL of Q-PCR mixture contained 10 µL 2× TaqMan Universal Master Mix, 1 µL 20× Taqman expression assay, and 50 ng cDNA. Q-RT-PCR was performed in StepOnePlus System (Applied Biosystems). The gene identification numbers for the TaqMan expression assay used in the Q-RT-PCR analyses are presented in Table 1. Human glyceraldehyde-3-phosphate dehydrogenase (GAPDH, Applied Biosystems) was used for normalizing the variation in cDNA quantities from different samples.
Gene Name and Assay ID Number in Real-Time Reverse-Transcription Polymerase Chain Reaction Analysis.

Enzyme-Linked Immunosorbent Assay
Procollagen type I C-peptide (PICP) was measured to estimate type I collagen synthesis in NHDFs. PICP levels in cell culture supernatant were determined using a PICP ELISA kit (Takara Bio) and MMP1 was determined using an MMP1-ELISA kit (R&D Systems) according to the manufacturer’s protocol.
Western Blot
NHDFs were lysed on ice for 1 hour with RIPA lysis buffer (Merck Millipore) with a protease inhibitor (Merck Millipore). After incubation, NHDFs lysates were centrifuged at 12 000 rpm at 4°C for 15 minutes, and the supernatants were collected into new E-tubes. For western blot analysis, 30 µg of protein was separated on 4%-12% gradient Bis-Tris gels and transferred to polyvinylidene fluoride (PVDF) membranes (Thermo Fisher Scientific). The PVDF membranes were blocked by incubation in tris-buffered saline with Tween (TTBST) buffer containing 5% skim milk (Becton Dickinson) for 1 hour at RT and incubated for 2 hours at RT with the GAPDH primary antibody (1:2000; Thermo Fisher Scientific), anti-COL1 primary antibody (1:1000; Abcam), or the MMP1 primary antibody (2 μg/mL; R&D Systems) in TBST containing 5% skim milk. Blots were washed thrice with TBST and incubated with horseradish peroxidase-conjugated secondary antibodies (1:5,000; Bio-Rad Laboratories, Inc.) for 1.5 hours at RT. Finally, the blots were developed using Clarity Western ECL blotting substrate (Bio-Rad Laboratories).
Statistical Analysis
Statistical analyses were used by the one-way analysis of variance. Results are expressed as the means ± standard deviation of at least 3 independent experiments.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 201700280004).
