Abstract
Malignant melanoma is one of the most aggressive skin cancers, having a very high mortality rate. However, its effective treatment is not clear. Ampelopsin, a plant flavonoid, has been reported to inhibit cell growth and/or induce apoptosis in various types of tumor. In this study, it was shown that ampelopsin significantly inhibits melanoma A375 cell line proliferation in a concentration-dependent/time-dependent manner. The flow cytometric data clearly demonstrated that ampelopsin causes cell cycle arrest in the G2/M phase. Moreover, it also confirmed that growth inhibition mediated by treatment with ampelopsin is related to the decreased expression of Cdc2, Cdc25c, cyclin B1, and activation of caspase-3 and Bax, purportedly by epidermal growth factor receptor (EGFR), extracellular regulated protein kinases, and matrix metalloproteinase-2 (MMP-2) downregulation. As a result of this work, these findings suggest that ampelopsin inhibits human malignant melanoma A375 cell line proliferation by suppressing the EGFR/ERK/MMP-2 pathway.
Skin cancer is the third most common human malignancy and the incidence of malignant skin cancer is rising at an alarming rate 1,2 ; its incidence and mortality rates are increasing in most countries throughout the world, and it accounts for 10% of skin cancers. 3 Besides, melanoma is highly resistant to radiation and conventional chemotherapeutic agents, and this resistance is associated with a poor prognosis for this metastatic disease, which has prompted many investigators to search for effective adjuvant therapy. 2,4,5 Much effort has been focused on the discovery and development of new chemopreventive agents, especially those targeted at mechanisms known to be involved in the process of apoptosis.
Ampelopsin (Figure 1), a flavonoid, is the major bioactive component extracted from the Chinese medicinal herb Ampelopsis grossedentata, which is widely grown in South China, and its tender leaves and stems are used as a healthy tea product. 6,7 More than 27% of the ampelopsin in these plants resides in the tender stem and leaves and >40% is found in the cataphyll. 8 Ampelopsin has very low solubility in water (0.2 mg/mL at 25°C) and very low permeability through the intestinal mucosa. 9 It has been reported that ampelopsin is absorbed rapidly in rats, with a T max at 32.2 minutes after a single oral administration and eliminated with a mean residence time (MRT) lasting 81.4 minutes. Plasma concentrations were below the limit of quantification of 20 ng/mL after 180 minutes. 10 Ampelopsin has been described to possess anti-inflammatory and antimicrobial activity, to relieve coughs, and with antioxidant, antihypertensive, hepatoprotective, and anticarcinogenic effects 11,12 ; the anticancer activity of ampelopsin has been reported in bladder carcinoma and prostate cancer cells. 6,7 However, it is unclear whether ampelopsin possesses effective chemopreventive activities against melanoma. Moreover, no evidence has been reported for the direct effect of ampelopsin on melanoma cell proliferation and the mechanisms of this effect.

Chemical structure of ampelopsin.
In cancer development, the resistance of cells to apoptosis is one of the most crucial steps. 13 Induction of apoptosis of tumor cells has been the main mechanism of action of chemotherapeutic agents, 14,15 and the machinery for the apoptotic action is mediated by many factors, such as Bax and caspases, which are involved in the regulation of apoptotic cascades. 13 -16 Ampelopsin activity is mediated by the induction of apoptosis and cell differentiation, which is regulated by various genes and proteins. 17 In the present study, the effect of ampelopsin upon cell proliferation and apoptosis in the melanoma A375 cell line was assessed. In addition, the regulatory effects and underlying functions of the epidermal growth factor receptor (EGFR)/extracellular regulated protein kinases/matrix metalloproteinase-2 (MMP-2) pathway involved in the apoptotic effect were also investigated.
Results
Ampelopsin Inhibits Cellular Growth and Colony Formation
To investigate the potential cell proliferative inhibition activity of ampelopsin in melanoma cells, we first examined its effect on cell proliferation and clonogenic survival in the A375 cell line. Previous studies have demonstrated that ampelopsin significantly inhibited cell proliferation in a concentration-dependent and time-dependent manner in human breast cancer and ovarian cancer cells. 18,19 In our study, the IC50 value was 0.12 ± 0.02 µM for ampelopsin against the A375 cell line (see supplemental Material). In accordance, we also further observed that ampelopsin significantly inhibited the growth vitality of the A375 cell line, with inhibition peaking at 0.2 µM at 48 hours (Figure 2(a) and (b)). We also showed that exposure of A375 to ampelopsin for 48 hours inhibited the growth of A375 in a dose-dependent manner, resulting in a 23.2% to 84.1% decrease in cell viability (Figure 2(a)). We also examined the effect of ampelopsin on A375 cell line metabolic activity at the concentration of 0.2 µM at different treatment times (12-48 hours) and found that ampelopsin significantly inhibited cell viability in a time-dependent manner from 14.5% to 78.3% (Figure 2(b)).

The effects of ampelopsin on cell proliferative inhibition in melanoma A375. Cell growth inhibition activity of ampelopsin was assessed by 3-(4,5-dimethyl-2thiazolyl)-2, 5-diphenyl-2H-tetrazolium bromide, performed as described in the Results section. Values represent the mean ± standard deviation (n = 3, *P < 0.05). (a) The cells treated with ampelopsin (0, 0.05, 0.1, and 0.2 µM) for 48 hours. (b) The cells treated with 0.2 µM ampelopsin for different times (12 hours, 24 hours, and 48 hours).
We next studied the ability of the A375 cell line to form colonies on 6-well cell culture plates in either the presence or absence of ampelopsin for 2 weeks. The A375 cell line showed the ability to form clones in the untreated control wells. Ampelopsin decreased colony formation at concentrations as low as 0.05 µM (Figure 3(a)). Moreover, with the addition of ampelopsin, a dose-dependent inhibition in clonogenicity was observed, with a >90% inhibition at dosages as high as 0.2 µM ampelopsin in comparison with untreated control cells (Figure 3(b)). These results clearly indicated that ampelopsin greatly decreased cell proliferation.

Ampelopsin inhibits melanoma A375 colony formation in vitro. (a) Colony formation of A375 cells treated with different concentrations of ampelopsin after incubation for 2 weeks. The colony formation assay was performed to determine the proliferation of A375 cells. (b) The data are presented as the percentage of colonies obtained relative to the control (A375 cell without ampelopsin treatment). The data shown are the mean from three independent experiments. Each value is the mean ± standard deviation of 3 determinations.
Ampelopsin-Induced Cell Cycle Arrest at the G2/M Phase and Apoptosis in the A375 Cell Line
Previous studies have reported that ampelopsin causes cell cycle arrest, followed by cell apoptosis in various tumor cells, including human lung adenocarcinoma, human osteosarcoma, and human breast cancer. 20 -22 Accordingly, further biological evaluations focused on this compound to determine whether the antitumor mechanisms in the melanoma A375 cell line were related to cell cycle arrest and apoptosis.
Because the cell cycle is closely related to proliferation, the effect of ampelopsin on cell cycle distribution was examined, and then flow cytometric analysis was performed. After treatment with 0, 0.05, 1, and 0.2 µM ampelopsin for 24 hours, the numbers in the cell cycle were detected by fluorescence flow cytometry. Our results demonstrated that the proportion of cells at G2/M was 0.5% ± 0.1% of the ampelopsin-free culture. However, ampelopsin at 0.05 µM significantly blocked the cell cycle at G2/M. Treating melanoma A375 cells with ampelopsin also increased the percentage of cells in the G2/M phase from 4.5% ± 1.1% to 76.5% ± 6.9% as the concentration increased from 0.05 to 0.2 µM (Figure 4(a)). Also, a concentration-dependent change was observed in the cell cycle pattern, which is shown in Figure 4(b). It was clearly suggested that ampelopsin inhibited the cell growth of the A375 cell line by arresting the cell cycle at the G2/M phase.

Ampelopsin induces cell cycle arrest at G2/M and apoptosis in the A375 cell line. (a) Effects of ampelopsin (0, 0.05, 0.1, and 0.2 µM) on the cell cycle of A375 cells. Cells were incubated for 24 hours and stained with propidium iodide (PI). Their DNA content was analyzed by fluorescence flow cytometry; representative flow cytometric profiles are shown demonstrating the cell cycle distribution in the A375 cell line. (b) The percentage of cells in each phase. Values represent the mean ± standard deviation (SD) (n = 3, *#& P < 0.05 compared with control group). (c) Effects of ampelopsin (0, 0.05, 0.1, and 0.2 µM) on cell apoptosis of the A375 cell line. Cells were incubated for 48 hours and induction of apoptosis was measured by Annexin-V/PI double-staining assay. Representative images of three separate experiments are shown. (d) The rate of apoptosis was calculated and expressed as mean ± SD of 3 separate experiments. *P < 0.05 by Student’s t-test.
In order to explore further whether the growth inhibition was related to the induction of apoptosis, we employed Annexin V/propidium iodide (PI) staining to detect the apoptosis ratio after the A375 cell line had been treated with ampelopsin (0, 0.05, 0.1, and 0.2 µM) for 48 hours. As shown in Figure 4(c), the ratio of apoptotic cells treated with 0.05 µM ampelopsin, including early apoptosis (right lower section of fluorocytogram) and late apoptosis (right upper section of fluorocytogram), was 47.6% ± 4.9%, whereas the control ratio was 37.3% ± 5.5%. Furthermore, treatment of the A375 cell line with ampelopsin (0.05-0.2 μM) resulted in a dose-dependent increase in the number of apoptotic cells, which ranged from 47.6% ± 4.9% to 93.5% ± 8.0%, compared with the non-ampelopsin-treated group (Figure 4(d)). Taken together, these results confirmed that growth inhibition mediated by ampelopsin treatment was related to the induction of apoptosis.
The EGFR/ERK/MMP-2 Pathway is Activated in the A375 Cell Line Treated with Ampelopsin
Apoptosis is usually controlled by two major pathways: the membrane death receptor pathway and the mitochondrial pathway. 23 The majority of antineoplastic drugs appear to activate the mitochondrial pathway. 24 It is well known that caspases and members of the Bcl-2 family, such as Bax, play important roles in mitochondrial pathway induction by various stimulations. 25 -27 Bax is a member of the Bcl-2 protein family that regulates the balance between cell proliferation and apoptosis, which can promote the release of cytochrome c from mitochondria during apoptosis mediated by the mitochondrial pathway. 28 Among the family of caspases, caspase-3 is the main downstream effector caspase that plays an important role in degrading the majority of essential cellular components in apoptotic cells, 29 indicating its crucial role in the cell death process. In consideration of the impressive cytotoxicity of ampelopsin, we next aimed our study at the potential mechanism(s) responsible for its apoptotic effects by examining whether caspase-3 and Bax were activated during apoptosis induced by varying concentrations of ampelopsin. The Western blot analysis data indicated that ampelopsin could increase Bax and caspase-3 expression in a dose-dependent manner (Figure 5(a) and (b)). The results suggested that the apoptotic response induced by ampelopsin may be associated with the mitochondrial pathway and accompanied by increased expression of Bax, and activation of caspase-3. Entry of mammalian cells into mitosis is regulated by cyclin A- and cyclin B1-cdc2 complexes. In these cells, the G2 checkpoint appears to switch off cdc2 activation by upregulating cdc2-inhibitory kinases and/or by downregulating the cdc25C. 30 Indeed, some studies also showed that Cdc2, Cdc25c, and cyclin B1 all can take part in the G2/M arrest in cells and play important roles in cell cycle progression. 31,32 To verify the above finding, we further tested the expression of these marker proteins of G2/M cell cycle arrest using Western blotting (Figure 5(c)). This showed that the expression of Cdc2, Cdc25c, and cyclin B1 was markedly downregulated in cells after treatment with different concentrations of ampelopsin in a dose-dependent manner (Figure 5D). All these results demonstrated that ampelopsin could induce cell cycle G2/M arrest in A375 cells, followed by cell apoptosis.
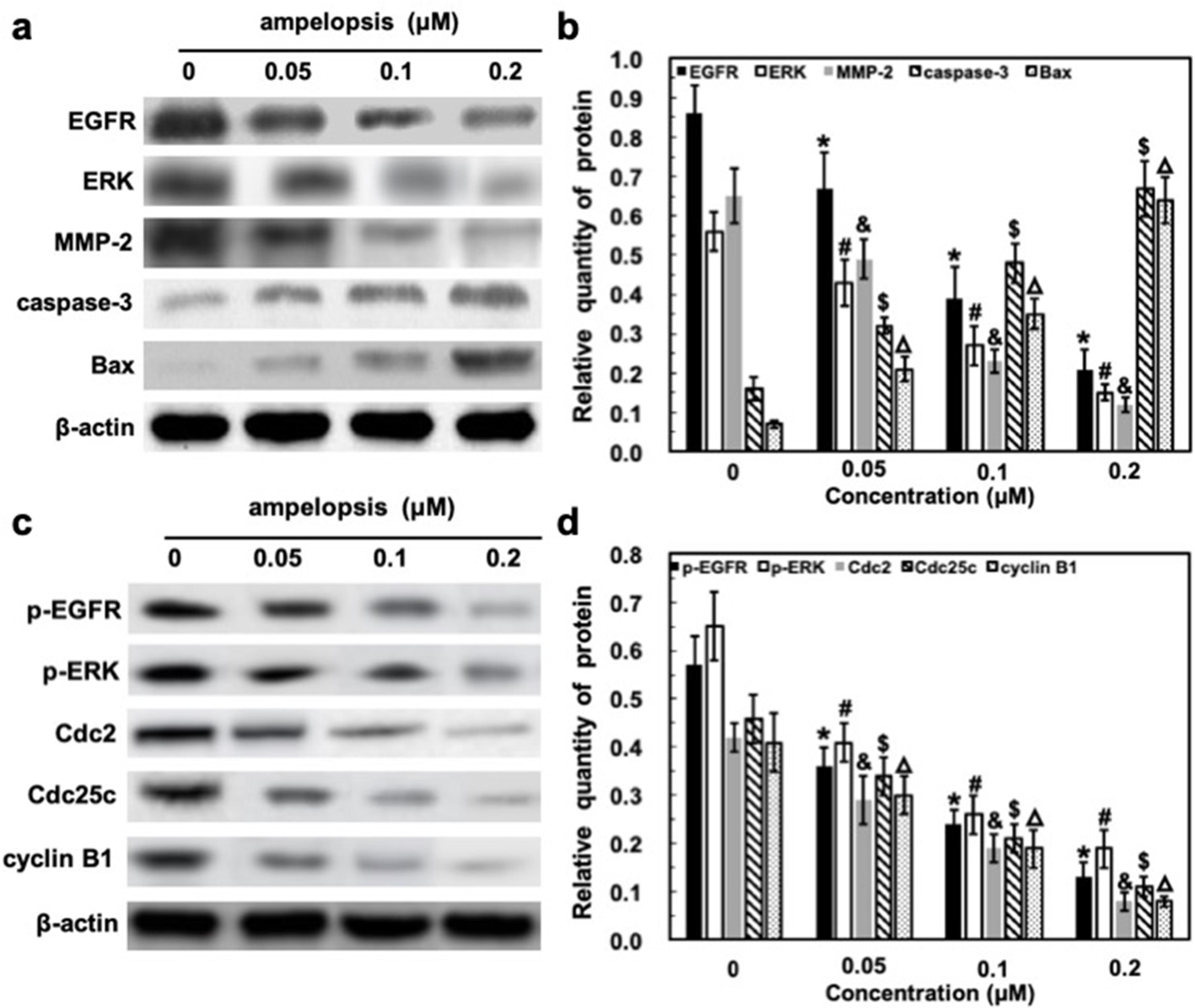
Treatment with ampelopsin suppresses the epidermal growth factor receptor (EGFR)/extracellular regulated protein kinases/matrix metalloproteinase-2 (MMP-2) pathway and increases expression of Bax and caspase-3, with decreasing levels of Cdc2, Cdc25c, and cyclin B1 in the A375 cell. (a and c) Representative images of Western blot analysis. A375 cells were starved overnight in 0.5% fetal bovine serum/Dulbecco’s Modified Eagle Medium and then treated with ampelopsin (0, 0.05, 0.1, and 0.2 µM) in serum-containing medium for another 48 hours. Cell lysates were prepared and protein expression was determined by Western blot analysis. (b and d) Data were analyzed by Modifit software. Data correspond to the means ± standard deviation of at least 3 independent experiments. *, #, &, $, △ P < 0.05, comparison with previous dose according to Student’s t-test.
The receptor tyrosine kinases (RTK), such as EGFR and ERK, play important roles in the proliferation and invasion of cancer cells. 33 -35 Besides, MMP-2 plays an important role in the migration and invasion of cancer cells, which can be regulated by the ERK1/2 pathway. 36 -38 As shown in Figure 5(a) and (c), the expression of these proteins, as well as the level of their phosphorylation were decreased when A375 cells were exposed to ampelopsin (0, 0.05, 0.1, and 0.2 µM) for 48 hours. This response, which was evident even at low doses, was amplified with increasing doses (Figure 5(b) and (d)), implying that the EGFR/ERK/MMP-2 pathway plays an essential role in ampelopsin’s mechanisms of action. In contrast, expression of β-actin, an internal control, was unchanged (Figure 5(a) and (c)). These results demonstrated that after G2/M arrest, the apoptotic response induced by ampelopsin was associated with the EGFR/ERK/MMP-2 pathway, followed by Bax up-regulation and caspase-3 activation.
Discussion
Human malignant melanoma is a cancer that arises from melanocytes. 39 Although melanoma accounts for only 10% of all dermatologic cancers, it is responsible for 80% of deaths from skin cancer. 3 Only 14% of patients with metastatic melanoma survive for 5 years, 2 which has become a major public health problem. Besides, the incidence of melanoma has increased in most countries throughout the world. In particular, the annual incidence rate increased in the order of 3%-7% in fair-skinned populations in recent decades. 40 The intractability of advanced melanoma shows how much we have to learn about the changes that facilitate the vertical growth and deep invasion of melanoma and about the mechanisms that block the effectiveness of chemotherapy.
Ampelopsin (3,5,7,3′4′5′-hexahydroxyl-2,3-dihydrogen flavonol) is a naturally occurring flavonoid isolated from the plant species Ampelopsis grossedentata (Hand-Mazz) W.T. Wang, known as Rattan Tea, a health beverage in China 20. Previous reports have shed light on ampelopsin’s antioxidant, antimicrobial, and anti-inflammatory actions. 11,12 In recent years, ampelopsin has been described to possess activity against various types of cancers. It was found to inhibit the growth of prostate cancer in vivo 7 and to inhibit the growth and invasion of breast cancer cells in vitro. 22 Ampelopsin also showed activity for inhibiting vascular endothelial growth factor and basic fibroblast growth factor, suppressing angiogenesis in hepatocellular carcinoma. 18 Besides, more and more evidence suggests that ampelopsin has the ability to inhibit cell proliferation, migration, and invasion in breast and prostate cancer. 7,22 However, no evidence has been clearly reported for the direct effect of ampelopsin on melanoma cancer cell proliferation, and also, the detailed molecular mechanisms of the action have not been elucidated.
The current study showed that ampelopsin inhibited A375 cell growth in a time-dependent and dose-dependent manner (Figure 2). What is more, the results were in overall agreement with colony formation studies (Figure 3). The compound could also induce cell cycle arrest at the G2/M phase and cell apoptosis of melanoma A375 cells in a concentration-dependent manner (Figure 4). Therefore, the cell cycle arrest and induction of apoptosis in A375 cells may be one of the mechanisms of the antitumor effects of ampelopsin.
To elucidate the molecular mechanisms by which ampelopsin affects the biological behaviors of A375 cells, we measured the expression levels of several growth factors and proteins in these cells. EGFR activation is involved in cancer cell proliferation, migration, and invasion, and it is overexpressed in various cancers, and then can activate several downstream signaling pathways including the PI3K/AKT and ERK pathways. 39 MMP-2 is positively regulated by the ERK pathway and regulates degradation of extracellular matrix components, which play a key role in cancer metastasis. 41 The expression of EGFR, ERK, and MMP-2 in A375 cells exposed to different concentrations of ampelopsin was examined (Figure 5(a) and (b)). The results showed that their expression levels, as well as their phosphorylation, were significantly reduced in the ampelopsin-treated cells in a dose-dependent manner compared with their control groups (Figure 5(c) and (d)). These results suggested that ampelopsin inhibited the growth of A375 cells through EGFR/ERK/MMP-2 signaling pathway suppression.
It has been suggested that apoptosis, an intrinsic cell-suicide program, may represent a protective mechanism against neoplastic development by eliminating genetically damaged cells or excess cells that have improperly been induced to proliferate. 42 The Bcl-2 family consists of both proapoptotic and antiapoptotic members that elicit opposing effects on mitochondria. Bax can promote the release of cytochrome c into the cytosol from mitochondria, which, in turn, activates caspase-3, one of the key executioners of apoptosis. 43 We observed that ampelopsin upregulated the expression of the proapoptotic protein Bax, as well as activation of caspase-3 in the A375 cell line in a dose-dependent manner (Figure 5(a) and (b)). Also, it was found that the expression of cell cycle related proteins, including Cdc2, Cdc25c, and cyclin B1, were downregulated after treatment with ampelopsin in a dose-dependent manner (Figure 5(c) and (d)). These results suggested that ampelopsin caused cell cycle arrest at G2/M and then induced apoptosis, which was related to EGFR/ERK/MMP-2 pathway suppression.
In summary, we report for the first time that ampelopsin can simultaneously inhibit proliferation of melanoma tumor cells. Importantly, we further confirm that the EGFR/ERK/MMP-2 pathway can be involved in the control of ampelopsin-induced proliferation inhibition, followed by cell apoptosis (Figure 6). These inspiring findings have provided us with information that enables a better understanding of the molecular mechanistic basis of the anticancer properties of ampelopsin. Also, it should open up a new exploration for plant lectins as potential antineoplastic agents in the treatment of human malignant melanoma in the future. However, in consideration of the impact of ampelopsin on different kinds of cell proliferation, we think that further research in normal cells is also warranted. It will provide more powerful information indicating differences between melanoma cells and normal cells for ampelopsin, which may be used as strong antitumor agents with high efficiency and low toxicity.

Schematic diagram illustrating the signaling pathways involved in ampelopsin’s inhibition of proliferation in the human malignant melanoma A375 cell line. Ampelopsin acts via direct downregulation of the epidermal growth factor receptor (EGFR)/extracellular regulated protein kinases/matrix metalloproteinase-2 (MMP-2) pathway, which subsequently results in suppression of Cdc2, Cdc25c, and cyclin B1, as well as activation of caspase-3 and Bax.
Materials and Methods
Cell Lines, Reagents, and Antibodies
Melanoma A375 cells highly sensitive for the cell growth inhibitory activity 44 were originally purchased from ATCC (NCC102015) and grown in 25 cm2 culture flasks in Dulbecco’s modified Eagle’s medium (DMEM) with 10% fetal calf serum, 100 U/mL penicillin and 100 g/mL streptomycin) at 37°C in a 95% air and 5% CO2 atmosphere. Ampelopsin (purity ≥98%, Mansite Bio-technology Co, Sichuan, China), from properly sealed glass vials stored in the refrigerator (4°C), was dissolved in dimethyl sulfoxide (DMSO), concentrations of which in the cell culture experiments did not exceed 0.1%. Then the solution could be stored at −20°C. Cells were treated with ampelopsin and the controls with the same amount of DMSO as that used in the corresponding experiments. Anti-EGFR (mouse monoclonal antibody, 1:1000), anti-ERK (mouse monoclonal antibody, 1:1000), and anti-MMP-2 (mouse monoclonal antibody, 1:1000), as well as anti-caspase-3 (mouse monoclonal antibody, 1:1000), anti-Bax (mouse monoclonal antibody, 1:1000), anti-p-EGFR (mouse monoclonal antibody, 1:1000), anti-p-ERK (mouse monoclonal antibody, 1:1000), anti-Cdc2 (mouse monoclonal antibody, 1:1000), anti-Cdc25c (mouse monoclonal antibody, 1:1000), anti-cyclin B1 (mouse monoclonal antibody, 1:1000), anti-β-actin (mouse monoclonal antibody, 1:2000) antibodies, and horseradish peroxidase-labeled goat anti-mouse immunoglobulin G were obtained from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Estimation of Cell Viability
Viability to the in vitro melanoma A375 cells was assessed by the 3-(4,5-dimethyl-2thiazolyl)-2, 5-diphenyl-2H-tetrazolium bromide (MTT) assay. 45 Briefly, the cell line was inoculated onto a series of standard 96-well plates (Wuxi NEST Biotechnology Co., Ltd, Wuxi, Jiangsu, China) at a density of 5 × 103 cells/mL in the absence of the testing agent. After overnight growth, cells were treated with ampelopsin at various concentrations (0, 0.05, 0.1, and 0.2 µM) for 48 hours and also incubated for 0, 12, 24, and 48 hours at 0.2 µM. Following this, the medium was discarded and replaced with 10 µL MTT (0.5 mg/mL, Sigma, St. Louis, Missouri) at 37°C for 4 hours. The culture medium was removed, and the resulting formazan crystals were solubilized in 100 µL DMSO. The absorbance was measured with a multifunctional microplate reader at 490 nm. All assays were performed in triplicate and mean ± standard deviation (SD) values were used to evaluate cell viability.
Cell Colony Formation Assay
Cell colony formation assay was performed as described previously. 46 Melanoma A375 cells were seeded at a density of 1000 cells/well in 6-well culture plates in triplicate containing 2 mL complete medium. After 24 hours, cultures were replaced with fresh medium with 10% fetal bovine serum or the same medium containing ampelopsin (0, 0.05, 0.1, and 0.2 µM) at 37°C in a humidified atmosphere of 5% carbon dioxide (CO2) and 95% air for an additional 14 days to allow colony formation. Then cells were washed twice with cold phosphate-buffered saline (PBS), and the cell clones fixed for an hour with a solution containing 0.5% (w/v) crystal violet (Sigma-Aldrich, St. Louis, MO, USA) in methanol, followed by 3 rinses with tap water to remove excess dye. Colonies were counted only if a single clone contained more than 100 cells by ImageJ software, which is available from the National Institute of Health (http://rsb.info.nih.gov/ij; last accessed 10 October 2018). Each assay was performed in triplicate on 3 independent occasions.
Flow Cytometric Analysis
The effects of ampelopsin on cell cycle were analyzed using flow cytometry, as described by Do et al. 47 Melanoma A375 cells were grown in DMEM at 37°C in a humidified atmosphere of 5% CO2. Cells were treated with ampelopsin (0, 0.05, 0.1, and 0.2 µM). After incubation, 1.5 × 105 cells were harvested from the treated and normal samples. The cells were washed twice with PBS and fixed gently (drop by drop) in 70% ethanol in PBS in ice overnight. The cells were centrifuged at 100× g for 5 minutes, washed by PBS, and incubated in the dark at room temperature for 30 minutes with 100 mg/mL of Rnase A and 40 µg/mL of PI. The cell cycle distribution was analyzed with a flow cytometer (Becton Dickinson, San Jose, California, USA).
Apoptosis analysis was performed according to Ren et al. 48 Melanoma A375 cells were cultured in 25 cm2 flasks and treated with vehicle alone (0.1% DMSO) and various concentrations of ampelopsin for 48 hours. After that, cells were collected by trypsinization and quickly washed with PBS. The cells were resuspended in 200 µL of binding buffer at a concentration of 1.5 × 105 cells/mL. Ten microliters of Annexin V-FITC (1 mg/mL) and 10 µL of PI (2.5 mg/mL) were added to the samples for 15 minutes in the dark. Samples were then analyzed in a flow cytometer (Becton Dickinson, San Jose, California, USA) and evaluated based on the percentage of cells positive for Annexin V. Positive Annexin-V only indicates early-stage apoptosis, while positive Annexin-V and positive PI indicate late-stage apoptosis.
Western Blot Analysis
Western blot analysis was performed as previously described in detail. 49 Melanoma A375 cells were exposed to ampelopsin (0, 0.05, 0.1, and 0.2 µM) for 48 hours. Then, cells were harvested and lysed for 1 hour on ice in lysis buffer (50 mM HEPES [pH 7.4], 1% Triton X-100, 2 mM sodium orthovanadate, 100 mM sodium fluoride, 1 mM ethylenediaminetetraacetic acid, 1 mM egtazic acid, and 1 mM phenylmethanesulfonyl fluoride), supplemented with proteinase inhibitors: 100 µg/mL aprotinin, 10 µg/mL leupeptin, and 100 µg/mL pepstatin. Protein concentration was determined by the bovine serum albumin assay, and then 100 µg protein was fractionated and transferred onto nitrocellulose membranes. After being blocked with blocking buffer (tris-buffered saline containing 0.1% Tween 20% and 5% non-fat milk) for 60 minutes at room temperature, the membranes were incubated overnight at 4°C with appropriate primary antibody at dilutions specified by the manufacturer. After being washed 3 times for 10 minutes each in tris-buffered saline, the membrane was incubated with a diluted horseradish peroxidase-labeled secondary antibody (1:500) in blotting buffer at room temperature for 1 hour. After repeating, the washes in triplicate, the proteins of interest were detected by an enhanced chemiluminescence system (Pierce Biotechnology).
Statistical Analysis
All data represent at least 3 independent experiments and are expressed as the mean ± SD unless otherwise indicated. All the statistical analyses were performed using the SPSS 13.0. Statistical multiple comparisons were made by Student’s t-test on the basis of one-way analysis of variance with post hoc. P < 0.05 considered to represent a statistically significant difference.
Supplemental Material
Supplementary material - Supplemental material for The Inhibition of Cell Growth Through the EGFR/ERK/MMP-2 Pathway Induced by Ampelopsin in the Human Malignant Melanoma A375 Cell Line
Supplemental material, Supplementary material, for The Inhibition of Cell Growth Through the EGFR/ERK/MMP-2 Pathway Induced by Ampelopsin in the Human Malignant Melanoma A375 Cell Line by Fan Yang, Yin-hui Yang and Wei-hui Zeng in Natural Product Communications
Footnotes
Acknowledgments
We wish to thank the editor, the associate editor, and the three anonymous reviewers for their helpful comments and suggestions, which have led to an improvement of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
