Abstract
The endophytic fungus Aspergillus terreus (JAS-2) derived from Achyranthes aspera possesses multiple bioactive metabolites, one of which is terrein, a potent antimicrobial substance. Here we report the improved biosynthesis of terrein in cultures grown in media with a fixed carbon and modified nitrogen source (4% dextrose, 1% casein) for 15 days, after which pure crystals of terrein were obtained in the culture medium. We suggest that our improved method is effective for improving the quantity and quality of terrein produced. Terrein has shown potent antimicrobial activities against various microbes that may lead to its further use to combat antimicrobial resistance which threatens the community worldwide.
Keywords
Endophytes are attractive sources of natural products and have been used to enhance the synthesis of bioactive compounds. 1 These metabolites have been used as antimicrobial, anticancer and antimalarial agents. 2 The endophytic fungus Aspergillus terreus (JAS-2) associated with Achyranthes aspera contains terrein, a secondary metabolite which has the potential to be developed as an anticancer and antimicrobial drug. 3,4 Terrein has been found to target multiple types of cancers. 5,6
Purification of molecules from natural resources is a time-consuming process and requires high capital investment. Despite substantial developments in the extraction and separation techniques for isolation of natural products, it is still a challenging task. 7 An additional problem is that the desired metabolites may only be produced at a specific developmental stage or under specific environmental conditions. 8 Often also, the production of these bioactive compounds is in very low quantities. 9
Biotransformation of metabolites in a fermentation process by fungal species has led to the development of many significant bioactive molecules which show pharmaceutical importance. 10,11 This strategy could fulfill the increasing demand for bioactive compounds to combat infectious diseases and cancers. This strategy can be used to produce cost-effective drugs. There have been many studies which have aimed at scaling up the production of terrein 12 but none has been able to produce the expected quantity. Herein, we report an improved method for the production of pure crystals of terrein using optimal carbon and nitrogen sources from A. terreus (JAS-2) derived from A. aspera. Our improved protocol was able to produce increased levels of terrein. Further, we have identified, quantified, and characterized the terrein by reverse phase-high performance liquid chromatography (RP-HPLC), thin-layer chromatography (TLC), antibacterial assay, and X-ray crystallography.
Methodology
Culture and Improved Production of Metabolite
Sabouraud’s dextrose broth (SDB, 4% dextrose:1% peptone) culture media was used as a basal medium for the growth of the endophytic fungus A. terreus (JAS-2) for the production of metabolites. 13,14 For improved production of terrein, JAS-2 was grown with a fixed carbon source (4% dextrose), but modified nitrogen source (0.2%-1.8% casein) for 15 days. In brief, JAS-2 was grown in 50 mL culture media with a fixed concentration of dextrose (2 g) and varying concentrations of casein (100, 200, 300, 400, 500, 600, 700, 800, and 900 mg) in Erlenmeyer flasks. To solubilize the casein, 100 mg NaCl (0.2% saline) was added in 50 mL media and the culture was kept at 26°C for 15 days. Then, the filtrate was extracted from the grown mycelium using 3 equal volumes of ethyl acetate, and the combined extracts were evaporated under reduced pressure. The slow vaporization of the fermented metabolite (15 days of incubation period) crystallized the compound at 20°C.
Quantitative Estimation of Metabolite Samples by HPLC
The production of terrein was quantitatively estimated using HPLC following reverse phase chromatography/RPLC. 15 The crude metabolites extracted from the cultures with different concentrations of casein (100 mg to 900 mg) were subjected to HPLC analysis. In brief, all crude compounds were collected and dissolved in methanol with a stock of 1 mg/10 µL. Every 10 µL of metabolite was diluted to 1000 µL and subjected to HPLC for 10 minutes. HPLC analysis was carried out using a C-18 (4.6 × 250 mm) column in which acetonitrile:phosphate buffer (35:65) was used as the mobile phase. Twenty microliters of the filtered sample were injected; the flow rate was 1 mL/ following an isocratic gradient, and the relative retention time was recorded. The detection wavelength was 268 nm and the process was carried out at ambient temperature.
TLC of Metabolite
TLC was performed on 60 F254 silica gel precoated on aluminum plates (Merck). The filtered broths of the cultures grown under 2 different conditions, 4% dextrose and 1% peptone, as well as 4% dextrose and 1% casein, were extracted with ethyl acetate and evaporated under reduced pressure using an IKA Rotary Evaporator at a temperature less than 50°C; 10 µg/µL of the compound was spotted using fine capillary tubes. The mobile phase used for separation was 90% chloroform:10% methanol. Separated compounds were detected in a UV chamber. All the solvents were of pure analytical grade.
X-Ray Crystallography
Single-crystal X-ray data were collected using an Xcalibur Eos (Oxford) CCD diffractometer using graphite monochromatic MoKα radiation (λ = 0.71073 Å). Data integration and reduction were processed using Crys Alis Pro software. The structures were determined by the direct method and then refined on F 2 by the full-matrix least-squares technique with the SHELX-97 set of software using the Win GX (version 1.80.05) program package.
Antimicrobial Activity of Metabolite
The antimicrobial activity of the metabolites extracted from both the media (SDB, 4% dextrose:1% peptone and SDB, 4% dextrose:1% casein) were tested for antibacterial activity against Staphylococcus aureus, Aeromonas hydrophila and Enterococcus faecalis by the disc diffusion assay. 16
Results
For the improved production of terrein, we grew the endophytic fungus A. terreus (JAS-2) using a fixed concentration of a carbon source (4% dextrose), but varying the concentrations of the nitrogen source (0.2%-1.8% casein) at 26°C for 15 days (supplemental Figure S1(a)). The filtrate was extracted with ethyl acetate and evaporated under reduced pressure to obtain the metabolites (supplemental Figure S1(b)). With a casein concentration of 1% as the nitrogen source, the metabolite concentration reached its maximum, which was estimated as 1140 mg/L (Figure 1(a)). HPLC examination showed a single peak at 268 nm from the media having 4% dextrose and 1% casein (Figure 2). TLC showed a single band in the crude metabolites extracted from modified SDB (4% dextrose/1% casein) as compared with the existing SDB, 4% dextrose/1% peptone (Figure 3(a)).

(a) Terrein concentration vs increasing concentration of casein (100 mg, 200 mg, 300 mg, 400 mg, 500 mg, 600 mg, 700 mg, 800 mg, and 900 mg). (b) Production of fungal biomass vs increasing concentration of casein (100 mg, 200 mg, 300 mg, 400 mg, 500 mg, 600 mg, 700 mg, 800 mg, and 900 mg).
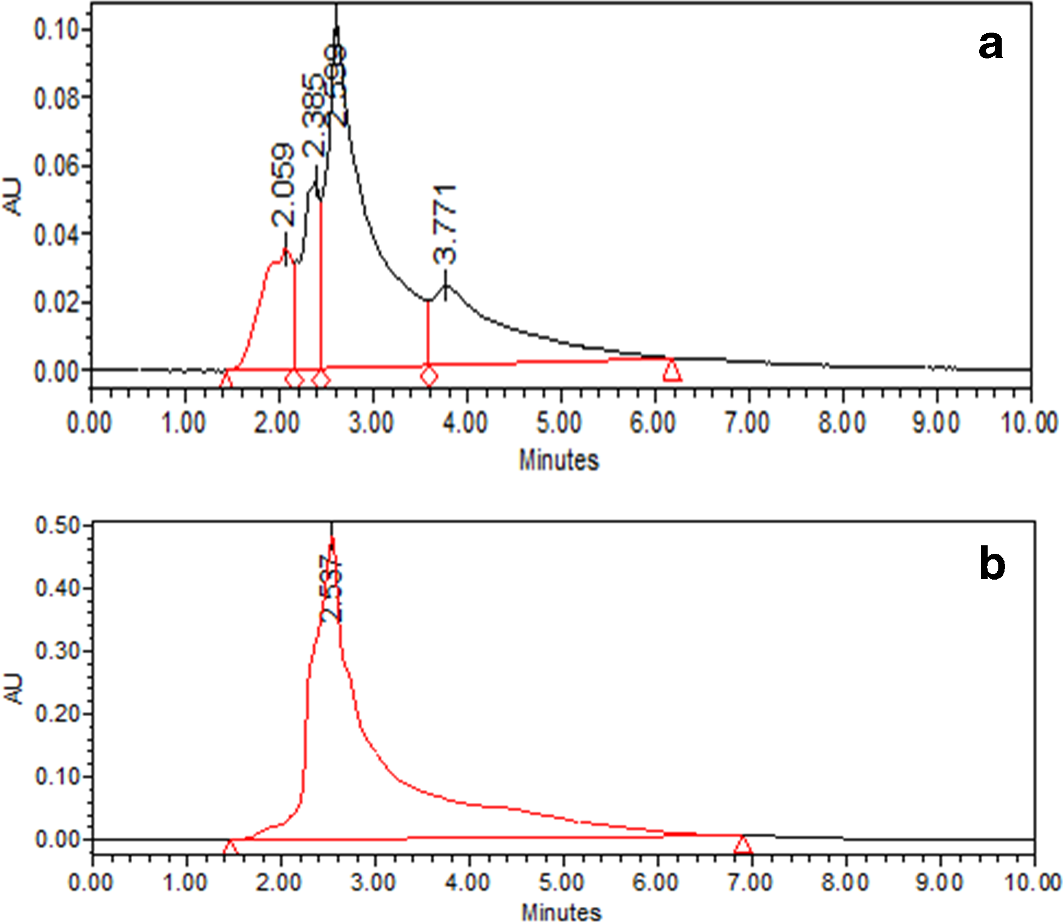
High-performance liquid chromatogram of peak fragmented from crude metabolite (a Metabolites obtained from SDB (4% dextrose and 1% peptone); (b) metabolites obtained from SDB (4% dextrose and 1% casein). SDB, Sabouraud’s dextrose broth.

(a) Comparative chromatograms (thin-layer chromatography bands) of crude metabolites extracted from fermentation broths of fungal endophyte JAS with different nitrogen sources. Lane 1: metabolites obtained from SDB (4% dextrose and 1% peptone). Lane 2: metabolites obtained from SDB (4% dextrose and 1% casein). (b) Crystals formed directly via broth and its structure revealed by X-ray crystallography. (c) Stereostructure of terrein by X-ray crystallographic analysis. SDB, Sabouraud’s dextrose broth.
The slow vaporization of the fermented metabolite (15 days of incubation period at 20°C) in broth crystallized the compound directly. Through X-ray crystallography, we have identified the crystal structure of the metabolite which was crystallized after extraction with ethyl acetate (Figure 3(b)). Molecular structures were determined using ORTEP software (Figure 3(b)) and the stereostructure of terrein is shown in Figure 3(c). All non-hydrogen atoms were refined anisotropically and hydrogen atoms were treated as riding atoms using the SHELX default parameters. Hydrogen bond interactions (C–H…O type) are displayed by Mercury software and ball and stick representation were used for modeling (Table 1). The activities of the metabolite that was isolated from both types of media exhibited antibacterial activity against S. aureus, A. hydrophila, and E. faecalis, as identified in the disc diffusion assay (see in supplemental Figure S2).
X-Ray Crystallographic Refinement Data of the Metabolite Terrein.

Discussion
Previous studies have used optimized fermentation conditions for the fungi secreting various biomolecules, for example, taxol 17 and lovastatin, 18 but all the techniques were complicated. Lee et al 19 invented a 9-step approach to synthesize terrein. However, the chemical synthesis method had drawbacks such as low yield, non-stereoselectivity, extreme reaction conditions, expensive reagents, and most of the products were in a racemic form. On the other hand, biotransformation has its advantages over chemical synthesis as it increases the production of the novel compound, overcoming the above-mentioned problems of chemical synthesis.
A. terreus (JAS-2) associated with A. aspera is the potential source of terrein, a well-known metabolite having multitherapeutic activities. 20 SDB is the commonly used media for the growth of fungi and the production of secondary metabolites. 21 We have employed a modified SDB with fixed carbon and variable nitrogen sources (4% dextrose:0.2%-1.8% casein) for the improved production of terrein. Among the different concentrations of casein (0.2%-1.8%), we found that a 1.0% concentration gave the optimum production of the terrein (1140 mg/L). It was also found that the fungal biomass increased with an increase in the concentration of casein (0.2%-1.8%). During the growth of the fungal culture, the casein was not completely utilized by the fungus, as casein is only partially soluble in water. Hence, the weight of biomass was found to increase as the biomass also contained the remaining amount of unutilized casein. It could also be hypothesized that it is the fungal endophyte (JAS-2) that secretes extracellular enzymes to degrade casein into its constituent components. This information could be supported by the extracellular enzymatic activity of the protease. 22
Comparative HPLC chromatograms indicated that modified SDB (4% dextrose:1% casein) showed the maximum production of terrein with a retention time (RT) of 2.5 minutes as compared with standard SDB (4% dextrose:1% peptone) having multiple RTs, which is reported in the supplemental Figure S1. Each compound should have a characteristic peak under certain chromatographic conditions. 23 We also found the production of purified terrein, which was proven by the presence of a single band in TLC (Figure 2). The crystal structure of the purified metabolite was obtained by X-ray crystallography, which also confirmed its purity. The antimicrobial assay also suggested that the purified metabolite (terrein) from modified SDB (4% dextrose:1% casein) showed better antimicrobial activities in comparison with the standard SDB (4% dextrose:1% peptone) against S. aureus, A. hydrophila, and E. faecalis (data provided in supplemental Figure S2). We assume that the purity of the metabolite (extracted from modified SDB) might be responsible for the better antimicrobial activities.
This study is not concordant with many previous studies but supports the concept of biotransformation as explained in terpene biosynthesis. 24 An earlier study showed the extraction of isopestacin and pestacin (strong antioxidant compounds) that were naturally crystallized from the SDB medium by utilizing an endophytic fungus. 25 Kushari et al 26 reported that the endophytic fungus Fusarium solani obtained from Camptotheca acuminata could produce camptothecin, 9-methoxycamptothecin, and 10-hydroxycamptothecin in SDB. 25 A. terreus strain S020 derived from red sea reports the large-scale production of pure crystalline terrein 537.26 ± 23.42 g/kg extract. 27 Besides these, various derivatives of terrein have been derived from natural resources along with fungi, having an array of impressive activity. Recently, co-culture techniques of endophytic fungi A. terreus EN-539 and Paecilomyces lilacinus EN-531 induced the production of the new derivative asperterrein and a known dihydroterrein having a multibacterial inhibitory effect. 28
In the present study, we describe the improved strategy for the production of pure terrein in a more economical way which leads to higher accessibility to consumers. We discuss the improved production of pure terrein (crystallized form) directly from the modified SDB. In earlier studies, the yield of terrein increased from 0.3 mg to 400 mg/L by microbial fermentation. 29,30 However, in our improved strategy, the average yield of purified terrein was found to be 1.14 g/L, which was higher than the existing reports. Thus, our results have opened a new way to produce and extract terrein in large quantities.
Conclusion
In brief, we have designed an improved strategy for terrein biosynthesis through A. terreus (JAS-2) culture grown in modified SDB culture media (with a fixed carbon source and modified nitrogen source, 4% dextrose, 1% casein). Through our improved strategy, we have obtained a higher yield of terrein (1.14 g/L), which was higher than the existing reports. So we can say that this strategy is better and more economical, leading to higher accessibility for consumers. Terrein has shown potent anticancer and antimicrobial activities which may lead to its further use to combat antimicrobial resistance and cancer that together kill numerous people worldwide.
Supplemental Material
Supplementary material - Supplemental material for Efficient Production of the Potent Antimicrobial Metabolite “Terrein” From the Fungus Aspergillus terreus
Supplemental material, Supplementary material, for Efficient Production of the Potent Antimicrobial Metabolite “Terrein” From the Fungus Aspergillus terreus by Jyoti Goutam, Ravindra Nath Kharwar, Vinod Kumar Tiwari, Ranjana Singh and Divakar Sharma in Natural Product Communications
Footnotes
Acknowledgments
We are thankful to Professor Gopal Nath, Institute of Medical Science (IMS) for providing bacterial pathogens. We also thank S. Bhattacharya for evaluating the crystal data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
