Abstract
The methods of extraction, separation, and purification of zizyphusine, spinosin, and 6′′′-feruloylspinosin from Zizyphi Spinosi Semen (ZSS) were investigated. From 31.2 g defatted ZSS, 47.7 mg zizyphusine, 57.8 mg spinosin, and 80.5 mg 6′′′-feruloylspinosin were obtained after ultrasonic extraction, purification with macroporous resin, separation by flash chromatography, and purification by high-pressure preparative chromatography. The purities of zizyphusine, spinosin, and 6′′′-feruloylspinosin were 98.6%, 98.2%, and 99.0%, respectively, and their yields 85.0%, 82.8%, and 83.0%. The methods provide stable samples of these compounds for further study of their physiological function and application.
Zizyphi Spinosi Semen (ZSS), the mature seeds of Ziziphus jujuba Mill. var. spinosa (Bunge) Hu ex H.F. Chow (Rhamnaceae), are used for the treatment of insomnia and anxiety. 1 The seeds are rich in pharmacologically active components, such as saponins, flavones, and alkaloids, which have antioxidant, 2,3 hypnotic, sedation, anti-inflammatory, and antiarrhythmic properties, as well as prevent myocardial ischemia and lower blood pressure. 4
Many studies have proved that aging and some diseases (such as cancer, diabetes, and cardiovascular disease) are closely related to the increase in the number of free radicals and their induced oxidation reactions. 5,6 Many natural products derived from plants have the potential to be used as antioxidants to remove free radicals in the body. 7 Thus, in recent years, research on the antioxidant activity of natural products has developed rapidly. 8,9 Recent studies have found that spinosin could be beneficial to treat learning and memory deficits in patients with Alzheimer’s disease via multitargets. 10 Both spinosin and 6′′′-feruloylspinosin were found to increase the transcription level of GABAAα1, GABAAα5, and GABABR1 mRNA significantly. 11 Most studies were focused only on the identification of different natural antioxidants. However, if there were no specific directions on the separation and purification of these antioxidants, it was difficult to study their biological activities and physiological functions if insufficient antioxidant had been applied. Therefore, it is necessary to study the methods of extraction, separation, and purification of active components according to their properties. 12 -15
Papers have reported the presence of zizyphusine (C20H24NO4), spinosin (C28H32O15), and 6′′′-feruloylspinosin (C38H40O18) in ZSS, 16,17 but these previous studies have only carried out preliminary detection and identification of flavonoids in extracts by LC-mass spectrometry (MS). 7 Therefore, it was difficult to attribute the cumulative observed effects to an individual molecular component, which was detrimental to further research and clinical applications. Our previous study demonstrated that these 3 compounds, especially 6′′′-feruloylspinosin and zizyphusine, have significant antioxidant activity. 18 In this current study, effective and reproducible methods for the simultaneous extraction and purification of zizyphusine, spinosin, and 6′′′-feruloylspinosin were established using ultrasonic extraction and purification through macroporous resin column chromatography, flash chromatography, and high-pressure preparative chromatography. The methods can provide stable samples for further study of the physiological function and application of zizyphusine, spinosin, and 6′′′-feruloylspinosin from ZSS.
Materials and Methods
Materials and Reagents
ZSS (harvested in 2016 in Hebei Province, China) was purchased from Medicinal Materials Market in Anguo (Hebei Province, China).
SEPABEADS SP825 macroporous resin, produced by Mitsubishi Chemical Co. (Japan), had the physical properties as follows: nonpolar, 1000 m2/g of surface area, 57 Å of average pore diameter, 200 to 600 nm particle diameter, 58% moisture.
Standards of zizyphusine, spinosin, and 6′′′-feruloylspinosin (≥98%) were purchased from Shun Bo Bioengineering Co., Ltd (Shanghai, China). HPLC-grade methanol was produced by Fisher Scientific Co. (USA). Pure and ultra-pure water was prepared using a Millipore-Q purification system (USA). Other reagents of analytical grade were purchased from Beijing Beihua Fine Chemicals Co., Ltd (Beijing, China).
Extraction of Zizyphusine, Spinosin, and 6′′′-Feruloylspinosin From ZSS
ZSS (50 g) was oven dried at 80°C and ground into 40 mesh powder (selected by sieve), 41.05 g of which was defatted by refluxing with light petroleum (60°C-90°C) for 8 hours in a Soxhlet extractor and then air-dried. The defatted powder (31.20 g) was extracted with 70% (v/v) ethanol solution (936 mL × 3) using a set of circulation supersonic extraction equipment (CTXNW-2B, Beijing Hongxianglong Biotechnology Developing Co., Ltd, China) for 120 minutes under constant mechanical agitation (1200 rpm) and at a frequency of 59 kHz. At the same time, another supersonic way of multistage shaped charge type ran at 750 W and worked for 3 seconds with ‘‘power on’’ and 3 seconds with ‘‘power off’’ in turn to keep the temperature of the extraction solvent at 40°C. After centrifugation, the supernatant was condensed to quarter volume by rotary evaporation under vacuum at 40°C. The ethanol concentration of the extraction was controlled at 35% (v/v). The concentrations of zizyphusine, spinosin, and 6′′′-feruloylspinosin were about 0.068, 0.16, and 0.096 mg/mL, respectively. The condensed extract solution was stored in a dark place at 4°C. The recoveries of the extraction were all above 95% for zizyphusine, spinosin, and 6′′′-feruloylspinosin.
Purification of Extract With Macroporous Resin
The macroporous resin was pretreated with 1 mol/L HCl and 1 mol/L NaOH successively to remove the monomers and porogenic agents. The resin was dried at 60°C under vacuum. About 500 g macroporous resin was soaked in 95% ethanol for 12 hours and subsequently washed with pure water thoroughly before use. A glass column (300 mm × 35 mm) was wet-packed with 200 g pretreated SEPABEADS SP825 macroporous resin. The bed volume (BV) of the resin was 288.5 mL.
Before sample loading, the resin was conditioned with 5 BV 35% (v/v) ethanol. Then the condensed extract solution (14 500 mL) was applied onto the column at a flow rate of 10 mL/min. After reaching adsorptive saturation, the resin was first washed with 4 BV distilled water and then eluted with ethanol solution. The eluent consisted of water (A) and ethanol (B) in different ratios and the flow rate was 5 mL/min. The gradient elution modes were as follows: 0 to 40 minutes, 35% B; 40 to 80 minutes, 45% B; 80 to 160 minutes, 55% B. The eluent was screened by an online UV detector at 270 nm for reference and all collected in tubes (50 mL ×60). The gradient elution curves were obtained by off-line HPLC-diode array detector (DAD) determination of eluent in each tube. The eluents of 45% and 55% ethanol were evaporated to dryness under vacuum at 40°C, and the obtained dried extracts contained spinosin and 6′′′-feruloylspinosin, respectively. The sample solution through the column from 600 to 3790 mL and the eluent of 35% ethanol were combined together and evaporated to dryness under vacuum at 50°C to obtain the dried extract, which mainly contained zizyphusine. The contents of zizyphusine, spinosin, and 6′′′-feruloylspinosin were measured by HPLC.
Separation by Flash Chromatography
The flash preparative RP-LC apparatus was made up of a flash-150 binary-gradient pump (SSI, Washington, NY), a tee with an injection port, a UV 201 detector (SSI), a BSZ-40 fraction collector (Shanghai Hu Xi Analysis Instrument Factory Co., Ltd, Shanghai, China), and a glass chromatographic column (400 mm × 35 mm, EYELA, Tokyo, Japan) wet packed artificially with particles of RP C18 silica gel (50 mm, 12 nm, Sunchrom ODS-A).
About 2000 mg of dried phytochemical extract was dissolved in 60 mL methanol and applied onto the glass chromatographic column, which was eluted with a solvent system consisting of water (A) and methanol (B). The gradient elution mode was as follows: 0 to 10 minutes, 5% to 25% B; 10 to 25 minutes, 25% to 30% B; 25 to 85 minutes, 30% to 40% B; 85 to 130 minutes, 40% B (v/v, binary gradient). The flow rate was 30 mL/min. The eluent was screened by an online UV detector at 280 nm for reference and all collected in tubes (60 mL ×720). All eluted fractions were determined off-line by HPLC-DAD and pooled according to the results from the chromatograms. As a result, the extract was fractionated into 4 parts, which contained impurity (fraction A), zizyphusine (fraction B), spinosin (fraction C), and 6′′′-feruloylspinosin (fraction D), respectively. The organic solvent in fractions B, C, and D was removed by vacuum drying at 40°C. The remaining liquid was dried by lyophilization. The contents of the 3 compounds in each fraction were detected by HPLC.
Purification of Zizyphusine, Spinosin, and 6′′′-Feruloylspinosin
The high-performance preparative RP-LC apparatus was made up of 2 series Ⅲ high-pressure pumps (SSI), a 7725i 6-way valve, a Model 201 UV detector (SSI), a BSZ-40 fraction collector (Shanghai Hu Xi Analysis Instrument Factory Co., Ltd), a workstation software (ver. 6.14 SP1), and a YMC-Pack ODS-AQ C18 prep-column (250 mm × I.D. 20 mm, S-5 μm, 12 nm) or Grace adsorbsphere XL C18 prep-column (250 mm × I.D. 22 mm, S-5 μm, 12 nm).
Twenty-five milligrams of freeze-dried fraction B or C obtained from flash chromatography, which mainly and separately contained zizyphusine (B) and spinosin (C), was resolubilized in methanol and applied onto a YMC-Pack ODS-AQ C18 prep-column. The mobile phase consisted of water (A) and methanol (B). The gradient elution mode was as follows: 0 to 5 minutes, 5% to 25% B; 5 to 25 minutes, 25% to 30% B; 25 to 45 minutes, 30% to 50% B (v/v, binary gradient). The flow rate was 10 mL/min, and the eluent was screened by an online UV detector at 280 nm for reference. The eluted solutions were collected in tubes (12 mL ×50). Then the eluted fraction in each tube was detected off-line by HPLC-DAD and pooled according to the chromatograms. Fractions containing zizyphusine, spinosin, or 6′′′-feruloylspinosin were combined. Methanol was evaporated under vacuum at 40°C. The remaining liquid was dried by lyophilization. The obtained zizyphusine, spinosin, and 6′′′-feruloylspinosin had purities of >98%, as detected by HPLC-DAD.
Fraction D, mainly containing 6′′′-feruloylspinosin, was separated on a Grace adsorb sphere XL C18 prep-column. The mobile phase consisted of water (A) and methanol (B), and the gradient elution mode was 0 to 15 minutes, 10% to 38% B; 15 to 75 minutes, 38% to 43% B; 75 to 115 minutes, 43% to 50% B (v/v, binary gradient). The flow rate was 12 mL/min. The other processes of purification were the same as for fractions B and C. Spinosin and 6′′′-feruloylspinosin were also obtained with purity of >98% in this separation step.
Chromatographic Analysis of Zizyphusine, Spinosin, and 6′′′-Feruloylspinosin
Off-line analysis of zizyphusine, spinosin, and 6′′′-feruloylspinosin in the prep-LC (middle-pressure or high-pressure) fractions was carried out by HPLC on a Shimadzu 20-AD UFLC system composed of dual LC-20AD UFLC pumps with a degasser, a CTO-20A thermostated column compartment, an SPD-M20A DAD, an SCL-10A controller, and Class-VP workstation software (ver. 6.14 SP1).
Sample analysis was performed with a guard column (10 L × 4.6 mm, Shim-pack GVP-ODS C18) and a YMC C18 column (250 L × 4.6 mm, I.D., S-5 μm, 12 nm, ODS-AQ) at a column temperature of 40°C. The mobile phase consisted of 0.5% (v/v) acetic acid aqueous solution (A) and 0.5% (v/v) acetic acid MeOH solution (B). The gradient elution mode was as follows: 0 to 5 minutes, 10% to 38% B; 5 to 15 minutes, 38% to 42% B; 15 to 18 minutes, 42% to 10% B; 18 to 23 minutes, 10% B. The flow rate was 1.0 mL/min, the oven temperature 40°C, and injection volume 10 µL. All samples were filtered through a 0.45 µm membrane before HPLC analysis.
MS and Nuclear Magnetic Resonance (NMR)
MS and NMR spectra were obtained at the Center of Analysis, Beijing University of Chemical Technology. A Waters (Milford, MA) Quattro Premier Micromass 70-VSE mass spectrometer was used with the selected ion monitoring mode for the pseudomolecular negative ion [M-H]- of zizyphusine, spinosin, and 6′′′-feroylspinosin. The ion source temperature was 100°C, the desolvation temperature 350°C, the desolvation gas flow 602 L/h, and the cone gas flow 45 L/h. NMR spectra were performed in CDCl3 using a Bruker high-resolution AV600 NMR spectrometer at 600 MHz (Billerica, MA).
Statistical Analysis
All statistical analyses were performed using SPSS 12.0 for Windows software.
Results and Discussion
Purification With SP825 Resin
The dynamic breakthrough curves of zizyphusine, spinosin, and 6′′′-feruloylspinosin on an SP825 resin column are shown in Figure 1. The resin had almost no adsorption for the alkaloid zizyphusine. The dynamic adsorption capacities of the 2 flavonoids, spinosin and 6′′′-feruloylspinosin, on the resin were 7.93 and 17.87 mg/g, respectively. Therefore, the characteristic of SP825 resin can be used to purify simultaneously zizyphusine, spinosin, and 6′′′-feruloylspinosin from the extract of ZSS. As shown in the dynamic breakthrough curves of Figure 1, the extract solution was loaded onto an SP825 macroporous resin column for adsorbing spinosin and 6′′′-feruloylspinosin, and the solution through the column from 600 to 3790 mL which mainly contained zizyphusine was collected at the same time. In order to avoid the loss of zizyphusine, the sample solution after loading from 600 to 3790 mL and the 35% ethanol eluent were collected and combined in the practical operation. Therefore, the part mainly containing zizyphusine was obtained after vacuum concentrating and freeze drying. The results for zizyphusine, spinosin, and 6′′′-feruloylspinosin eluted by 35%, 45%, and 55% ethanol solution successively are shown in Table 1, and the elution curves in Figure 1. The recovery of the purification with SP825 resin was above 96%.
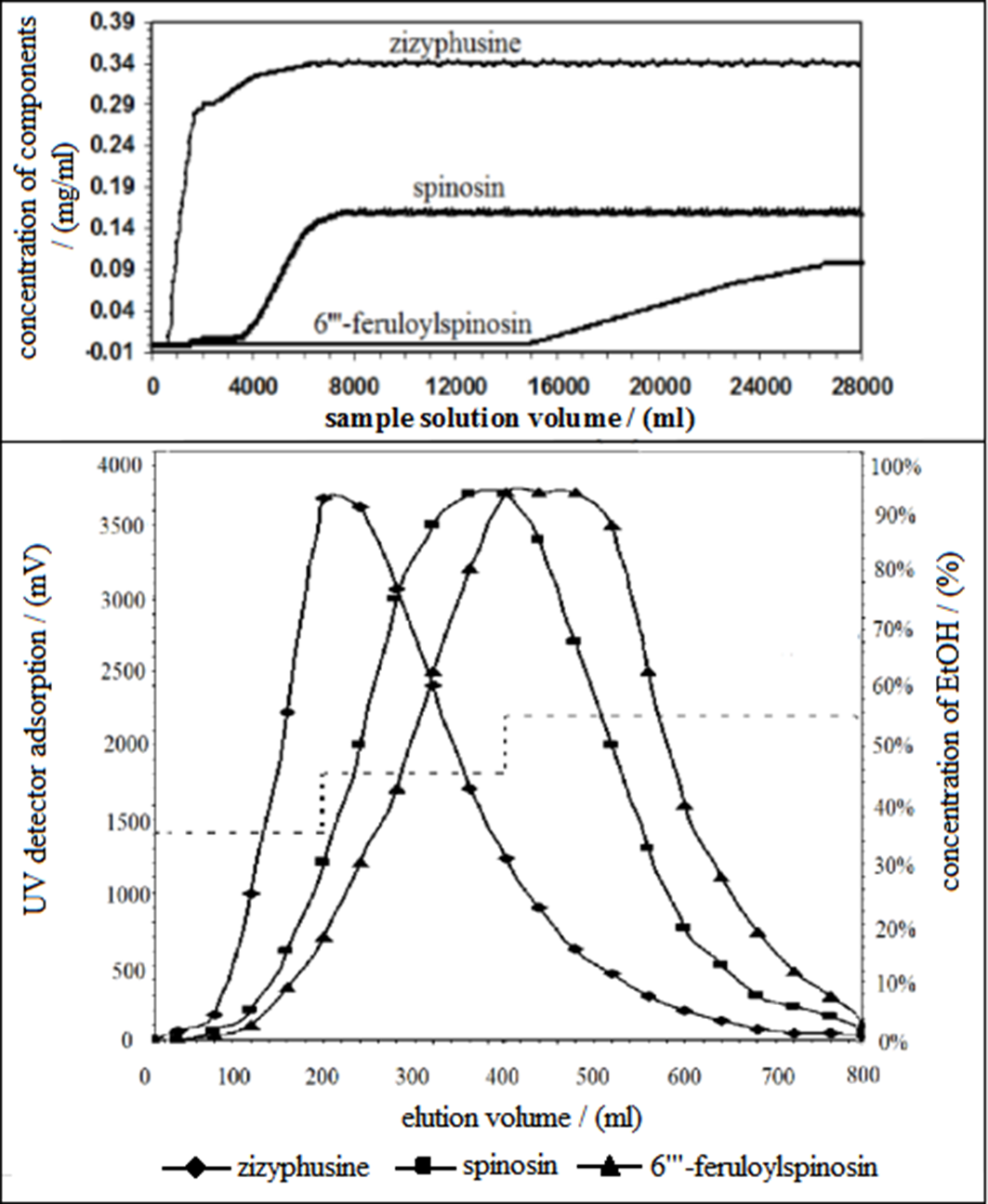
Dynamic breakthrough curves and gradient elution curves of flavones in chromatography column packed with SP825 resin.
Comparison of the Results of Different Elution Modes.
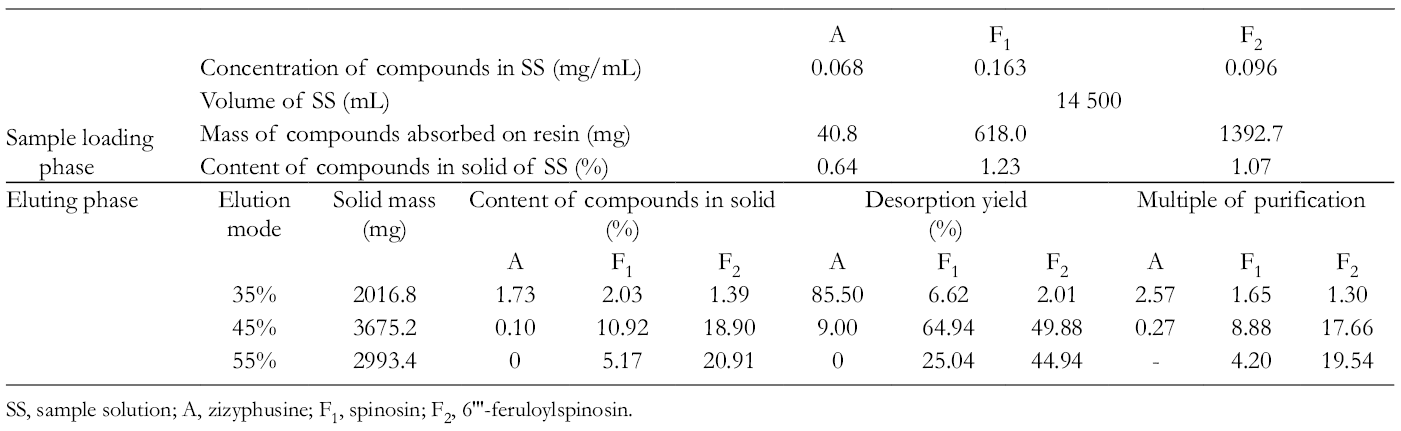
SS, sample solution; A, zizyphusine; F1, spinosin; F2, 6'''-feruloylspinosin.
Preliminary Fractionation by Flash Chromatography
Zizyphusine, spinosin, and 6′′′-feruloylspinosin all had obvious ultraviolet absorption at 270 and 330 nm. In addition, zizyphusine had obvious ultraviolet absorption at 220 nm. These 3 compounds can be detected online using a UV detector during the fractionation. Since the extract of ZSS consisted of complex components, it usually took a few steps to separate the monomers from other compounds. Flash preparative chromatography can be used to fractionate the extract into several fractions, each of which mainly contains one or several components, and then every fraction can be further separated in other ways. The extract of ZSS was fractionated in a prep-column packed with RP C18 medium and the preparative chromatogram is shown in Figure 2(a). As shown, the extract was fractionated into 4 fractions; fractions B, C, and D mainly contained zizyphusine, spinosin, and 6′′′-feruloylspinosin, respectively, and the recoveries for the 3 compounds were all above 95%.

(a) Elution profile of the phytochemical extract on a flash chromatography RPC column obtained with a UV detector at 280 nm (online). Solid line: UV signal at 280 nm; dashed line: gradient curve of MeOH; the fractions that were collected are indicated by the different areas marked with A, B, C, D; chromatography conditions: a glass column (400 mm × 35 mm, 50 mm, 12 nm, Sunchrom ODS-A); flow rate: 30 mL/min; solvent system: ultra-pure water (A) and methanol (B); elution mode: 0 to 10 minutes, 5% to 25% B; 10 to 25 minutes, 25% to 30% B; 25 to 85 minutes, 30% to 40% B; 85 to 130 minutes, 40% B (v/v, binary gradient). (b), (c), and (d) Elution profiles of fractions B, C, and D, respectively, on a prep-HPLC RPC column obtained with a UV detector at 280 nm (online). Solid line: UV signal at 280 nm, dashed line: gradient curve of EtOH, chromatography conditions for fractions B and C: a YMC-Pack ODS-AQ C18 column (250 mm × I.D. 20 mm, S-5 μm, 12 nm), flow rate: 10 mL/min, solvent system: ultra-pure water (A) and methanol (B), elution mode: 0 to 5 minutes, 5% to 25% B; 5 to 25 minutes, 25% to 30% B; 25 to 45 minutes, 30% to 50% B (v/v, binary gradient); chromatography conditions for fraction D: a Grace adsorb sphere XL C18 column (250 mm × I.D. 22 mm, S-5 μm, 12 nm), flow rate: 12 mL/min, solvent system: ultra-pure water (A) and methanol (B), elution mode: 0 to 15 minutes, 10% to 38% B; 15 to 75 minutes, 38% to 43% B; 75 to 115 minutes, 43% to 50% B (v/v, binary gradient).
Purification of Zizyphusine, Spinosin, and 6′′′-Feruloylspinosin
Fractions B, C, and D were separated individually by high-pressure preparative chromatography and detected by an online UV detector. The retention time of zizyphusine, spinosin, and 6′′′-feruloylspinosin can be determined according to the HPLC chromatogram. As shown in Figure 2(b) to (d), zizyphusine, spinosin, and 6′′′-feruloylspinosin were separated from impurities. The 3 compounds were obtained individually after separation, concentration, and drying; all the recoveries of this step were above 98%. The purities of zizyphusine, spinosin, and 6′′′-feruloylspinosin were all higher than 98%, as detected by HPLC-DAD.
Identification of Zizyphusine, Spinosin, and 6′′′-Feruloylspinosin
Under positive ionization mode, there was an intensive molecular ion peak of zizyphusine which had got a H+ at m/z 342.4. Under negative ionization mode, there were intensive molecular ion peaks of spinosin and 6′′′-feruloylspinosin which had lost a H+ at m/z 607.2 and m/z 783.0.
The MS and NMR spectra of the isolated compounds were consistent with those published for zizyphusine, spinosin, and 6′′′-feruloylspinosin. 7,16,17,19 -27
Concluding Remarks
The purities of zizyphusine, spinosin, and 6′′′-feruloylspinosin were 98.6%, 98.2%, and 99.0%, respectively; 47.7 mg zizyphusine, 57.8 mg spinosin, and 80.5 mg 6′′′-feruloylspinosin were obtained from 31.20 g defatted ZSS through 4 steps: ultrasonic extraction, purification with macroporous resin, separation with flash chromatography, and purification with high-pressure preparative chromatography. The yields of the 3 compounds were 85.0%, 82.8%, and 83.0%, respectively. The extraction rate and purity of the 3 compounds were stable after 3 repetitive experiments. The described procedure is an efficient method that can be implemented to obtain gram scale samples of zizyphusine, spinosin, and 6′′′-feruloylspinosin from ZSS in the laboratory and to provide high-quality and stable analytical grade samples for further study on physiological functions and research applications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Beijing Natural Science Fund (KZ201710020014), the National Natural Science Foundation of China (31601658).
