Abstract
Keywords
Introduction
The worldwide application of synthetic pesticides has a remarkable impact on the environment with the emergence of new resistant strains of microorganisms and insects and the ecological conditions associated with them. Given these effects, identifying new plant protection chemicals that are necessary for modern pest management is important.1–3 Bioactive natural products have evolved to inhibit specific cellular targets and have served as lead molecules for health and agricultural applications for the past century.4,5
Artemisia annua L. (Asteraceae) is a fragrant annual herb widely distributed in Asia, Europe, and North America. In 1971, artemisinin, a sesquiterpene with antimalarial properties, has been isolated.6,7 Thus, artemisinin has drawn continuous research attention worldwide, and an increasing number of studies have been reported in recent years.8,9 Since then A. annua has been extensively investigated in recent years.10,11 The search for other active compounds has led to the discovery and separation of many phytochemicals, such as monoterpenoids, sesquiterpenoids, flavonoids, and coumarins, as well as aliphatic and lipid compounds.12,13 Apart from its antimalarial activity, A. annua also shows anti-inflammatory, antipyretic, anticancer, antifungal, antiparasitic, antiulcerogenic, antioxidant, and cytotoxic activities. 14 Previously, studies have focused on the useful constituent qinghaosu (artemisinin) in Artemisia annua L. leaves while ignoring the importance of other potential compounds left in the residual materials of A. annua L. leaves.13,14 For an extended period, residual materials of Artemisia annua after extraction of artemisinin (RMAAEA) has been either used as green manure or discarded in large quantities into the soil and water resources across China.
The carmine spider mite Tetranychus cinnabarinus (Boisduval) (Acarina: Tetranychidae) is an important economic pest mite worldwide, and it has remarkable effects on crop yield and quality.15,16 Given its small size, rapid growth, short life cycle, population density, and high rate of inbreeding, the carmine spider mite can easily resist conventional chemical acaricides. 17 Phytogenic acaricides, also known as botanical acaricides, are derived from plants, and they have shown great application potential in integrated mite management because of their desirable properties. These acaricides are selective, indicating that they target specific mite species while having low toxicity to mammals, making them safer for non-target organisms.18,19 Thus, the identification of reasonable alternatives to chemical pesticides has been the object of considerable global research interest.
During the research on natural product-based pesticides, using a range of chromatographic techniques and other separation procedures concurrently with conducting bioactivity assays is important. This approach ensures that inactive components are discarded, whereas only the active ones are further investigated. 20 Our prior research has validated that the methanol parallel extract from A. annua leaves, which is harvested in July, demonstrated the most potent acaricidal activity against T. cinnabarinus and Panonychus citri. 21 Thus, this study focuses on isolating the acaricidal active compound from the residue of A. annua following artemisinin extraction. A large-scale extraction in a factory setting will also be discussed.
Materials and Methods
Residual Plant Materials
In factory, 900 kg of RMAAEA was collected from Chongqing Heng-star Biotechnologies Co., Ltd., on July 25, 2023. It was identified by Professor X.-Y. Li, College of Horticulture, Southwest University, Chongqing, China. A voucher specimen was deposited in the herbarium of the College of Plant Protection, Southwest University, Chongqing, China.
In the laboratory, the RMAAEA was dried in an oven at 50 °C. Next, the dried materials were crushed into a plant powder using a crushing device and further ground using a grinder with a 5 mm sieve. Subsequently, 500 g of the ground material was soaked in 2500 mL of pure methanol for 72 h. Then, various portions of the extracted solution were dehydrated under vacuum conditions. The resulting RMAAEA extract was transferred to Petri dishes and refrigerated at 4 °C for later use.
Preparation and Bioassay of Mites
The initial culture of the carmine spider mite, T. cinnabarinus (Boisduval), was established from specimens collected from cowpea plants (Vigna unguiculata) in Beibei, Chongqing, China. This culture was preserved by rearing the mites on potted cowpea seedlings (height: 30-40 cm) within an insectary chamber where the temperature was maintained at 26 °C ± 1 °C; the relative humidity was kept between 75% and 80%, and the photoperiod was set at 16 h of light to 8 h of darkness. 21 This colony was preserved for over a dozen years without exposure to any acaricides prior to experimentation. Specimens for reference were contributed to the insect collection at Southwest University in Chongqing, China.
The acaricidal activities of RMAAEA extracts, different groups, scopoletin, pyridaben, and spirodiclofen were tested by using the slide-dip method recommended by the Food and Agriculture Organization of the United Nations.18,21 Female CSM adults were affixed on a double-sided adhesive tape that was preattached to one end of a 10 cm × 2 cm glass slide carefully. The mites were then soaked in different dilutions (concentrations setting based on the screening test protocols of each extracts or components) for 5 s. The slide was removed, and any excess solution was absorbed with filter paper under a biological binocular. All treated mites were cultured in a controlled-climate box under the conditions of 27 °C ± 2 °C, 60% relative humidity, and 16:8 h L/D cycle. Mortality rate was monitored every 24 h. A mite was considered dead if it did not move when lightly touched with a fine brush. All treatments were repeated three times. Data were statistically analyzed using Excel 2003 and SPSS 25.0 for Windows (SPSS Inc., Chicago, Illinois).
Thin-Layer Chromatography (TLC)
Pre-coated TLC plates, particularly Silica gel 60 F254 (type 5554, produced by Merck in Darmstadt, Germany), were utilized using a mixture of ethyl acetate (EtOAc), formic acid (HCOOH), and water (H2O) in a ratio of 90:5:5 (by volume) as the eluent. Once the plates were dried, derivatization was performed using 1% methanolic 2-aminoethyl diphenylborinate (2-APB, CAS No. 524-95-8). For documentation and analysis, a TLC visualization system (Reprostar 3) was used, which is equipped with a digital 12-bit CCD camera and operated with winCATS software, provided by Camag in Berlin.
Silica Gel Column Chromatography
Silica gel chromatography (100-200 mesh) using Qingdao Ocean Chemical silica gel was performed after activation at 110 °C for 30 min. The column was loaded with the dried material. An equal amount of silica gel (10 g) was mixed with 10 g of the RMAAEA extract, which was subsequently layered on top of the silica gel column. During elution, a gradient of petroleum ether:acetone (ranging from 13:1 to 1:3) was used as the eluent at a flow rate of 300 mL/h. Fractions were collected at 20-min intervals, leading to the isolation of a total of 48 sample bottles containing 500 mL effluent components. These fractions were examined by TLC using a petroleum ether:acetone (3:1) mixture as the eluent. Fractions with comparable Rf values were merged on the basis of their volumes and obatined 20 groups (Figure 1). Each groups underwent testing for its acaricidal properties, and those demonstrating remarkable activity were subjected to further purification.
Separation of group 17 is based on the above mentioned protocol. The primary distinction lay in the eluent, specifically a blend of petroleum ether and acetone at a ratio of 1:2
Quantitative Identification of 17-3 by HPLC–MS
To prepare the sample solution, 100 mg 17-3 was weighed out and transferred to a 10 mL beaker. Subsequently, the extract was soaked in 10 mL of 1% hydrochloric acid solution for 12 h. After soaking, the solution was sonicated for 30 min before undergoing centrifugation at 4000 rpm. Subsequently, the supernatant was separated and extracted using 250 mL of chloroform. This process was repeated two times. The organic layer was transferred to a water bath and dried to remove the solvent. The resulting dried residue was then dissolved in an appropriate mobile phase to a final volume of 100 μL, thereby creating the backup solution.
The Waters Alliance 2695 series HPLC system was used as the chromatography system. For detection, a 2487 dual-wavelength UV detector was used to monitor wavelength at 254 and 365 nm. Data acquisition and analysis were handled by the Masslynx V 4.1 workstation. Separation was performed on a Waters Xterra C18 MS column with an inner diameter of 2.1 mm, length of 100 mm, and a particle size of 3.5 µm.
The optimized chromatographic parameters included a column temperature set at 35 °C and a flow rate of 0.3 mL·min−1. The eluent consisted of a mixture of water containing 0.1% formic acid and acetonitrile with 0.1% formic acid. The detection wavelength was set at 254 nm, and the injection volume was set at 10 μL. Gradient elution of the sample was used to achieve separation, which also resulted in a robust response from the mass spectrometry (MS) detection.
The MS conditions were as follows: ionization was achieved using an electrospray ionization source, alternating between positive and negative ion modes; the capillary voltage was set at 3.00 kV; the cone voltage was 40 V; the extraction voltage was 5 V; the ion source temperature was maintained at 100 °C; the desolvation gas temperature was set to 300 °C; the purge gas flow rate was adjusted to 500 L/h, and a full-scan sampling method was used, with a scan range of 50–600 m/z.
The analytical instruments used included a PE-GX infrared spectrometer employing the KBr pellet method, manufactured by PerkinElmer in the United States; a Flash EA-1112 automatic elemental analyzer from Thermo in Italy; an X-6 Precision micro melting point apparatus by Beijing Faucquez Technology Co., Ltd; a BRUKER AVANCE-300 nuclear magnetic resonance spectrometer produced by BRUKER in Germany; and a ZF-1 type three-channel ultraviolet analyzer made by a medical instrument factory in Haimen, Jiangsu, China.
Industrial Process of Scopoletin from RMAAEA
An industrial process of extraction was performed by using instruments and equipment instrument separation for extraction of scopoletin production workshop in Chongqing Heng-star Biotechnologies Co. Ltd.
Following the extraction of artemisinin without high-temperature erosion, 900 kg of RMAAEA was transferred to an extraction tank. Subsequently, 4000 L of methanol was added, and the mixture was heated under reflux conditions at 53 °C and a steam pressure of 0.06 MPa for 3 h. This extraction process was repeated three times. Then, the combined extract was concentrated in a concentration tank at 70 °C, reducing it to a smaller volume of concentrated extract.
The chromatography column was packed with 200 kg of silica gel using a wet method with petroleum ether. After packing the columns, they were allowed to stand for 2 h. The crude extract was mixed with an equal amount of 80 to 120 mesh silica gel, with continuous stirring to ensure uniform contact. If the crude extract was too viscous, then a small amount of petroleum ether or methanol was added to facilitate mixing with the silica gel. The mixed silica gel extract was then dried and ground into a fine, black granular mixture. The separation column was first dried with petroleum ether to coat the silica gel surface, ensuring even distribution of the crude material. This mixture was evenly spread across the packed silica gel column and leveled on the surface. The elution agent, a mixture of petroleum ether and ethyl acetate in a ratio of 6:1, was then added to submerge the surface. After standing for 3 h, the column was washed, with a flow rate controlled at approximately 1000 L per hour.
The eluted liquid was transferred to a concentration tank and vacuum concentrated until no further solution could be extracted. The concentrated material was then removed and placed in a barrel, where ethyl acetate was added to facilitate mixing. After filtration, the mother liquor was drained, leaving the solid material behind. Subsequently, this material was eluted with ethyl acetate to yield a yellow pure form of scopoletin. The mother liquor was pumped back into the concentration tank for further enrichment, and it underwent another leaching and filtration.
Under 365 nm UV light, scopoletin on the silica gel plate exhibited blue fluorescence. During elution, the eluent (petroleum ether:ethyl acetate = 3:1) was used to detect the eluate by TLC.
Results and Analysis
SGCC and Evaluation of the Biological Activity of the Corresponding Components
The methanol extract of RMAAEA was isolated using SGCC, and the bioactivity of all fractions against T. cinnabarinus is presented in detail in Table 1.
Content Percent Rate of Each Group of Methanol Extract of RMAAEA Isolated by SGCC and Their Bioactivity Against T. cinnabarinus.

As shown in Table 1, after detecting them by thin-layer chromatography (TLC) with different Rf values and merging 20 groups, each stage of separation was quantitatively assessed on the basis of the principle “dose intensity ranging.” This method was combined with biometric timing to evaluate the biological activity of various groups against T. cinnabarinus. Considering the relative abundance of each group in the extracts, groups 6, 17, and 14 are the most prevalent, accounting for 18.55%, 13.29%, and 11.45% respectively. From the perspective of biological activity against T. cinnabarinus, groups 17, 8, and 18 exhibit the strongest efficacy, with corrected mortalities of 100.00%, 87.38%, and 84.59% at 48 h post-treatment. Considering the quantitative content and biological efficacy, group 17 shows the most potential for further investigation.
HPLC was used to isolate group 17, which was then analyzed by combining HPLC/MS detection and biological activity assessment, and the results are presented in Table 2.
Content of Group 17 Isolated by HPLC and Their Bioactivity Against T. cinnabarinus.

LC50, the pesticide concentration that causes 50% mortality in a group of test female adults.
Confidence interval, the interval estimation of a population parameter of this sample.
As shown in Table 2, group 17 contains three main components. The high content is found in 17-3 and 17-1, accounting for 77.64% and 16.21%, respectively. The proportion of 17-2 is considerably small (5.31%). TLC analysis of these three components revealed one visible spot for each upon iodine staining, with no signs of tailing. The bioassay results against T. cinnabarinus demonstrated that 17-3 exhibits strong acaricidal activity, with an LC50 value of 101.5 mg/L after 48 h post-treatment. By contrast, the LC50 value of 17-2 against T. cinnabarinus is 701.1 mg/L. Component 17-1 showed an acaricidal LC50 value of 181.3 mg/L, which is similar to the active phase of 17-3.
Purification and Recrystallization of 17-3
The recrystallization experiment for 17-3 was conducted using various solvents, including anhydrous methanol, acetone, chloroform, ethyl acetate, and n-butane, at different temperatures to determine the optimal conditions for crystallization and recrystallization. At 55 °C, ethyl acetate yielded the best crystals with regard to morphology. However, crystals were obtained across all temperatures tested, indicating that 17-3 is not sensitive to temperature when forming crystals. The image below shows the crystals obtained at 55 °C using ethyl acetate for recrystallization.
Crystal Structure Identification
Subsequently, purified 17-3 (Figure 2) was analyzed using HPLC–MS (Figures 3 and 4). Figure 3 shows a prominent main peak detected at 5.47 min at two wavelengths, 254 and 365 nm. Therefore, purified 17-3 corresponds to a compound with a molecular weight of approximately 192 (Figure 4).

Thin-layer chromatography (TLC) detection of 20 groups based on different Rf values.

Acicular structure during crystallization and the purified white powder after purification of 17-3.

HPLC–UV chromatogram of purified 17-3.

ESI–MS chromatogram of purified 17-3.
Following the purification of 17-3, further analytical techniques such as IR and 1H-NMR were used. The crystal form of purified 17-3 was identified as colorless needle crystals with a melting point of 200 °C–201 °C. These crystals exhibited bright blue fluorescence under UV light. The UV absorption maxima (λmax) were determined to be 227, 253, 260 (sh), 297, and 344 nm in ethanol, indicating the presence of a coumarin compound. The 1H NMR spectrum of 17-3 showed a methoxyl group singlet at δ 3.83 ppm and two aromatic proton singlets at δ6.70 and 6.82 ppm which were explained by a 6, 7-disubstitution, indicating scopoletin framework. (Figure 5, Table 3). The IR spectrum showed characteristic bands at 3338 cm−1 (-OH) and 1701 cm−1 (α, β-unsaturated ester), as well as aromatic bands at 1600, 1566, and 1495 cm−1. Additional bands were observed at 1290, 1139, 1015, 920, and 860 cm−1 (Figure 6). ESI–MS analysis revealed a molecular ion peak at m/z 192.04 (100.0%), with minor peaks at m/z 193.05 (11.1%) and 194.05 (1.4%). Literature data 22 for scopoletin indicated that the physical, chemical, and spectral characteristics were consistent, confirming the compound as scopoletin with a molecular weight of 192.17 and a molecular formula of C10H8O4. The chemical structure of scopoletin is presented in Figure 7.
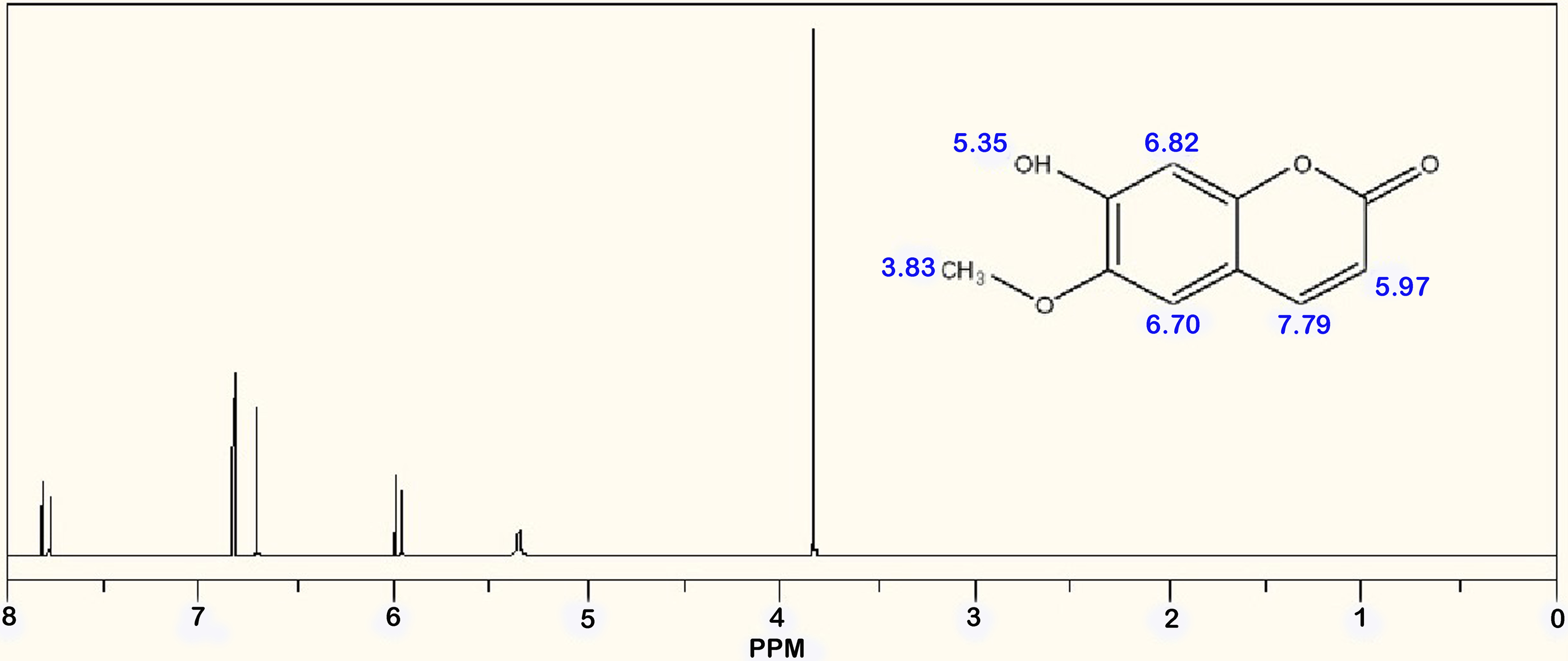
1H-NMR spectrum of purified 17-3.
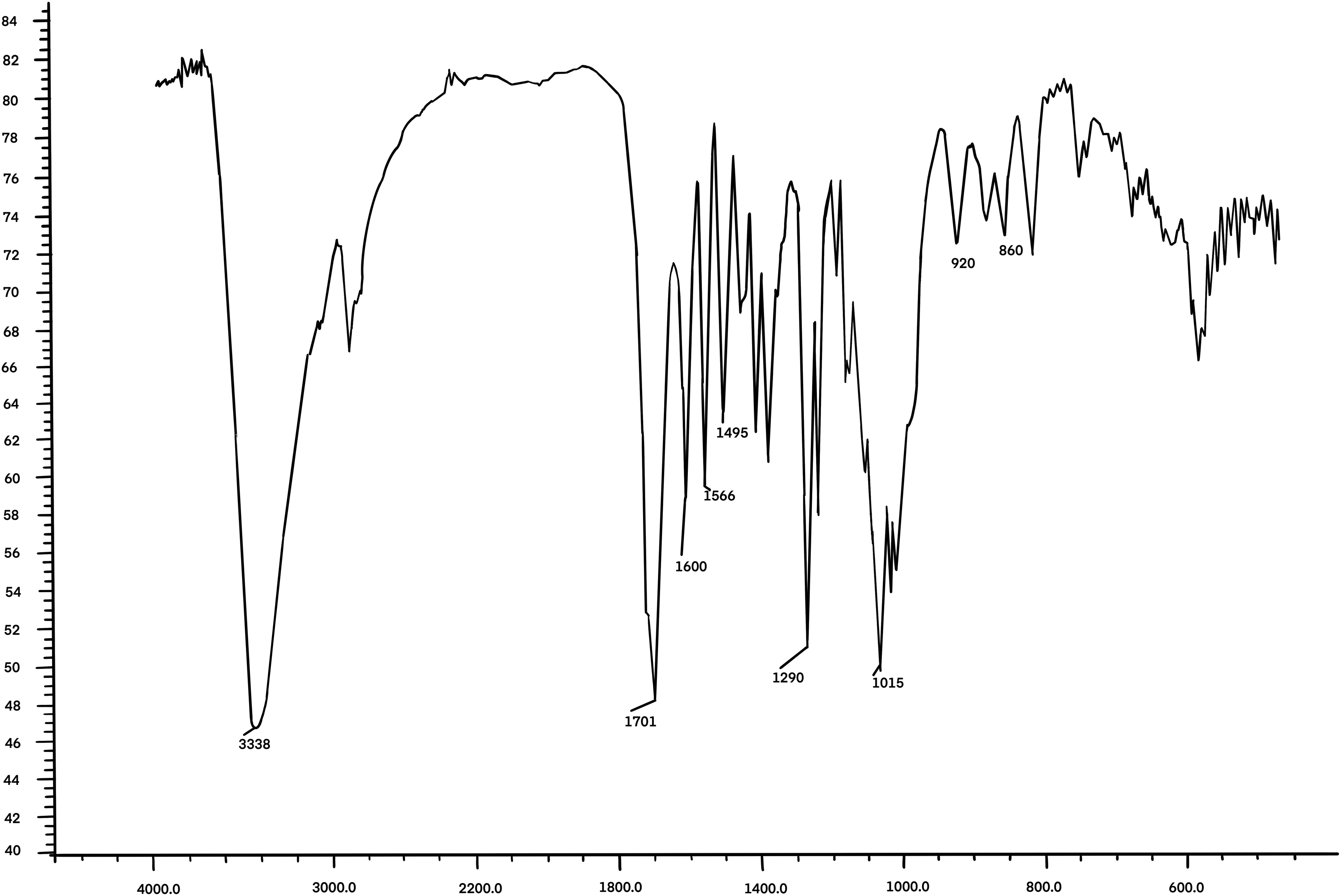
Ir spectrum of purified 17-3. Number of sample scans: 3, Number of background scans:3, Resolution:4,000, Sample gain:1.0, Mirror velocity:0.6329, Detector: DTGS KBr. Beamspliter: KBr, Source:IR.

Chemical structure of scopoletin.
1H-NMR Spectral Data of Purified 17-3.

Bioassay of Scopoletin and Two Commercial Acaricides
A toxicity test was conducted using scopoletin against T. cinnabrinus, and the results were compared with those of pyridaben and spirodiclofen. The findings are presented in Table 4.
Toxicity Analysis of Scopoletin, Pyridaben, and Spirodiclofen Against T. cinnabarinus (48 h).

As shown in Table 4, the biological acaricidal activity of scopoletin, which was further purified from 17-3, was enhanced compared with 17-3 alone. The acaricidal LC50 value of scopoletin was 95.0 mg/L, compared with the LC50 value of 17-3 (101.5 mg/L; Table 2.). However, this difference was not statistically significant (t = 0.370, df = 5, P = .554). By contrast, a significant difference was found when comparing scopoletin to spirodiclofen (t = 3.156, df = 5, P < .01), with the LC50 value of spirodiclofen being 5.96 times higher.
Industrial Process of Scopoletin
The process involved adding 900 kg of RMAAEA to an extraction tank, followed by the addition of methanol four times (4000 L each time, total 16 000 L). After each addition, the mixture was refluxed and extracted at 53 °C and a vapor pressure of 0.06 MPa for 3 h. Then, the liquid extract was filtered and collected, and this extraction step was repeated four times. The combined extracts were transferred to a concentration tank and concentrated under vacuum at 60 °C (with the first and second extracts being concentrated separately, whereas the third and fourth extracts were combined and then concentrated). The methanol solvent was recycled once during this process, thereby obtaining 60 kg of crude extract.
The dried silica gel with a particle size ranging from 80 to 120 mesh was utilized for column chromatography separation, using a mixed solvent of petroleum ether (pure) and ethyl acetate (pure) in a ratio of 7:1 as eluent. During elution, different fractions were collected to ensure the separation efficiency and to identify target compounds using TLC. The elution solution was collected step by step, amounting to a total of 41 000 L. The solvent from the eluate was recovered, and the solvent containing the target compound was concentrated to 300 L before being transferred to a crystallization tank for static crystallization. As the temperature decreased, a significant amount of crystals precipitated. The crystals were then separated via suction filtration and washed with petroleum ether, the crystals information was confirmed by TLC and HPLC-MS with the analysis standard-scopoletin purchased from Macklin Inc. Finally, approximately 1.23 kg of pale-yellow crystalline material was obtained, the purity was 98.3% and the final yield was 1.36‰. The flow-process diagram of scopoletin extraction from RMAAEA has been confirmed (Figure 8).

Flow-process diagram of scopoletin extraction from RMAAEA.
Discussion
Led by bioguided separation, scopoletin extraction from RMAAEA was conducted using the “consistent dose intensity” principle for the quantitative evaluation of each component at various stages. Through comparative analysis and identification of the most potent ingredient, the procedure concurrently aimed for “material separation” and “bioactive separation” to quickly trace the target compounds.23–26 The concurrent separation and evaluation of the physical activity of various components with different potencies were conducted side by side, indicating that an effective separation could be achieved. At present, the discovery of new plant-derived products to be used as food supplements or drugs has been pushed by the exploitation of bioprospection approaches. 27 By regulating the flow rate of the column chromatography and collecting different fractions at specific elution times, a successful separation was achieved. The methanol extract of RMAAEA was isolated using SGCC. After merging and determining various samples by TLC followed by different Rf values, 20 groups were obtained. The corrected mortality of 20 groups against T. cinnabarinus at 48 h ranged from 26.96% to 100.00%. The varying intensity of biological activity among different groups demonstrated that the separation had achieved a satisfactory result. Considering the quantitative content and biological efficacy, group 17 shows the most promise for further investigation.
Several reports have been found on the isolation of phytochemical active substances using column chromatography, for example, the use of high-speed countercurrent chromatography to separate three rotenoids and isoflavones from the seeds of Millettia pachycarpa Benth. 28 The ethanol extracts prepared from the aerial parts and roots of an endemic species, Salvia cerino-pruinosa Rech. f. var. cerino-pruinosa, were fractionated on SGCC and tested to determine their antioxidant activity. 29
Artemisia species have great commercial, medical, and economic value, and they are widely used in the traditional medicine and pharmaceutical industries. 30 Natural artemisinin is unique to the traditional Chinese medicinal plant A. annua L., which remains the major source of this bioactive component. Artemisinin is a compound with relatively low polarity, and it is extracted from A. annua using solvents with low polarity. By contrast, scopoletin has a higher polarity than artemisinin, and it is unlikely to be extracted during artemisinin extraction. 31 Therefore, the content of scopoletin in RMAAEA remains largely unchanged. This study provides an ideal raw material for the extraction of scopoletin from RMAAEA and promotes the reuse of A. annua residue. Furthermore, an industrial process that processed 900 kg of RMAAEA, producing 1.23 kg of pure scopoletin, is explained in detail in this study. However, due to the differences in the nature of artemisinin and scopoletin, and limited by the constraints of the current research conditions, the limitation of this study lies in the failure to achieve the simultaneous extraction of scopoletin during the artemisinin extraction process.
In our future work, the modifying of the process on the same industrial route for artemisinin extraction should be carried out. Simultaneously obtaining scopoletin and artemisinin can remarkably improve the utilization efficiency of A. annua and achieve greater economic benefits.
Conclusion
With the bio-guided isolation, group 17 shows the most promise candidate. Group 17 contains three main components. Component 17-3 exhibits the strongest acaricidal activity, with an LC50 value of 101.5 mg/L after 48 h post-treatment. After further purification and recrystallization from 17-3, a colorless needle crystal should be determined as scopoletin. The biological acaricidal activity of scopoletin was similar to that of pyridaben and stronger than that of spirodiclofen. A total of 900 kg of RMAAEA was industrially process, and 1.23 kg scopoletin was finally obtained, the purity was 98.3% and the final yield was 1.36‰.
Footnotes
Acknowledgements
We extend our gratitude to Zhenguo Yang, Peiyuan Qin, Kai Zhang, and Bingchuan Zhang for their support and aid in the industrial process.
Author Contributions
G.C. contributed to the data collection, methodology, formal analysis, and writing original draft preparation. Y.M.Z. contributed to the data collection, methodology, and formal analysis. X.C. contributed to the data collection, formal analysis, and writing of the original draft. J.X. contributed to the data collection, methodology, and formal analysis. Y.Q.Z. contributed to the conceptualization, methodology, validation, supervision, funding acquisition, and writing review and editing. All authors discussed the results and contributed to the final manuscript. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
The data sources presented in this study are available when requested.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
Ethics approval was not required for this research.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We acknowledge the financial support from Natural Science Foundation of Chongqing Municipality, China (CSTB2023NSCQ-MSX0141) and Mission Statement of Key Special Projects of Science and Technology Help Economy 2020 (SQ2020YFF0405970).
Statement of Human and Animal Rights
This artilce does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent i s not applicable.
