Abstract
One new terphenyl glycoside (
In the current era, marine-derived fungi have attained high focus as a promising source for biologically and pharmaceutically active secondary metabolites and have grown into the third largest source of marine natural products. 1,2 In this perspective, fungi associated with sponges yield numerous molecules with diverse structures and potential biological activities, 3 like glycosides, 4 terpenoids, 5 alkaloids, 6 and polyketides. 7 Terphenyls are a class of aromatic compounds possessing an 18-carbon tricyclic core skeleton, categorized into o-, m-, and p-subclasses. 8 p-Terphenyls are the leading class of terphenyls reported from the natural sources compared with the other terphenyl groups. 9 The majority of terphenyls have been isolated from fungal sources; therefore, they are considered as fungal secondary metabolites, 9,10 although, few of terphenyls have also been reported from moss and actinomyces. 11,12 Moreover, the terphenyl family exhibited extensive activities, including antitumor, antibacterial, antiaging, anticoagulant, neuroprotective, immunosuppressive, and α-glucosidase inhibitory effects. 9 A literature survey revealed that p-terphenyl glycosides exhibited cytotoxic 13,14 and antibacterial activities. 14 For example, gliocladinin C, a p-terphenyl glycoside isolated from a potato-derived fungus Chaetomium subaffine L01 showed strong cytotoxic activity against human tumor cell lines Hep-2 and HepG-2. 13
Large number of terphenyl derivatives have been reported from marine sources. For example, 2 new terphenyl derivatives, terphyl acid, and terphyl diacid, along with 6 known terphenyls, possessing cytotoxic activity were reported from the marine-derived Aspergillus sp. AF119. 15 Ivanets et al reported terphenyl derivative 3″-hydroxyterphenyllin from a fungus Aspergillus sp. collected from an unknown colonial ascidian. 16 Aspergillus candidus IF10 obtained from marine environment produced 3 new terphenyls, 4-deoxy-isoterprenin, 4-de-oxyprenylterphenyllin, and prenyl-terphenyllin, exhibiting strong cytotoxic activities. 17
As a part of our current investigation on exploring for new marine natural bioactive products, 5 p-terphenyl glycosides were isolated from a coral derived-fungus Aspergillus candidus.
18
During our continuous study on new bioactive p-terphenyl glycosides, the chemical profile of the crude extract obtained from Trichoderma reesei also draws our attention due to the presence of p-terphenyl characteristic UV pattern. One new terphenyl glycoside gliocladinin D (
Gliocladinin D (

1H-1H correlated spectroscopy and key heteronuclear multiple bond correlation of compound 1.
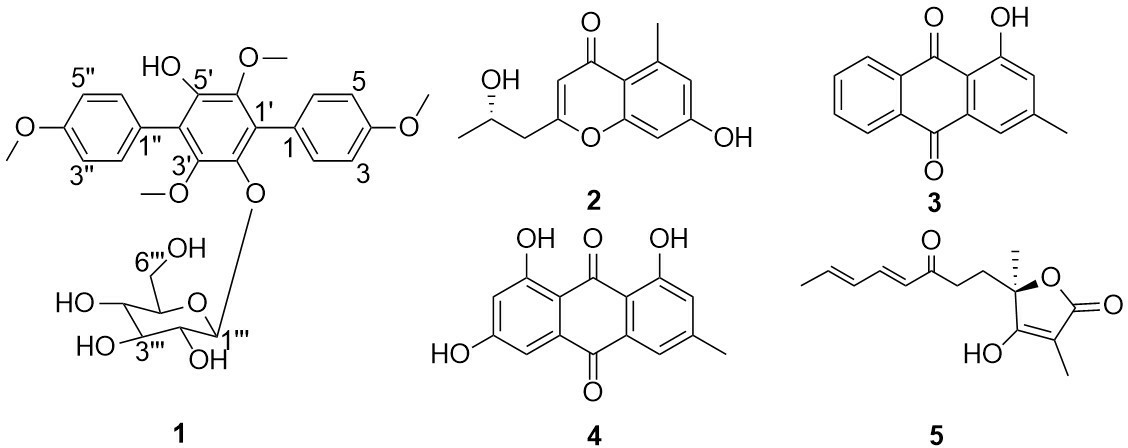
Structures of compounds
The downfield chemical shift of glucose carbon C-1″′ (δ
C 102.3) and the coupling constant of anomeric H-1″′ (J = 7.6 Hz) recommended that the glucose unit was connected with terphenyl ring through β-linkage.
14
To identify the absolute configuration of glucose moiety, acid hydrolysis of
The isolated compounds were evaluated for their cytotoxic, topoisomerase I inhibitory, and antibacterial activities. Unfortunately, all the tested compounds including the terphenyl glycoside gliocladinin D (
General Methods
Optical rotation was measured with a JASCO P-1020 digital polarimeter. Ultraviolet (UV) spectrum was recorded on a UH 5300 UV-Vis spectrophotometer in methanol (MeOH). Infrared (IR) spectrum was recorded on a Nicolet-Nexus-470 FT-IR spectrometer with potassium bromide (KBr) pellets. 1H, 13C NMR spectra were recorded on a JEOLJNM-ECP NMR spectrometer (500/125 MHz for 1H and 13C), using tetramethylsilane as an internal standard. High-performance liquid chromatography (HPLC) separation was attained by utilizing a Hitachi LA-2000 preparative-HPLC system, along with a Hitachi L-2455 photodiode array detector. ESIMS spectra were measured on a Thermo DSQ EImass spectrometer and HR-ESIMS on MAT95XP high-resolution mass spectrometer. Silica gel (200-300 mesh size) (Qing Dao Hai Yang Chemical Group Co.) and Sephadex LH-20 (Amershm Biosciences) were used for column chromatography. Precoated silica gel (GF254) plates (Yantai Zifu Chemical Group Co., Yantai, China) were used for thin-layer chromatography. Sea salt (Qingdao Salt Industry Co., Ltd.) was used in fermentation.
Fungal Materials
The fungus T. reesei (HN-2016-018) was isolated from a piece of inner fresh tissue of an unidentified sponge, which was collected from the South China Sea in October 2016, and recognized on the basis of its morphological characteristics and by amplification and sequencing of the DNA sequences of the ITS region. The fungus was identified as T. reesei whose 500 base pair ITS sequence had 99% sequence identity to that of T. reesei. The strain was deposited in the Key Laboratory of Marine Drugs, the Ministry of Education of China, School of Medicine and Pharmacy, Ocean University of China, Qingdao, PR China, with the GenBank (NCBI) accession number MN209898.
Extraction and Isolation
The fungal strain T. reesei (HN-2016-018) was cultivated on a solid rice media using 100 1 L Erlenmeyer flasks for 28 days (each flask containing 100 g of rice, 110 mL of water, and 3.3 g of sea salt) at 28°C. After 28 days, each flask of the fermented medium was extracted repeatedly (3 times) with 300 mL of EtOAc. The collected organic extract was dried by means of a rotary evaporator to attain a residue (114 g). The residue was fractionized into 5 fractions (Fr. A–Fr. E) by subjecting it into vacuum liquid chromatography eluting with EtOAc–petroleum ether (PE) (0%-100%) and methyl alcohol (MeOH)–dichloromethane (CH2Cl2) (0%-100%). Fr. B was further processed by Sephadex LH-20 column chromatography (CC) eluting with a mixture of CH2Cl2–MeOH (1:1). The ultimate purification was carried out on HPLC eluted with 70% MeOH/H2O to attain compounds
Gliocladinin D (1)
White amorphous powder.
UV (MeOH) λmax (logε): 220 (4.35), 256 (4.43) nm.
IR (KBr) v max: 3344, 2929, 2360, 1612, 1540, 1458, 1246, 1031 cm–1.
1H-NMR and 13C-NMR: see Table 1.
Nuclear Magnetic Resonance Data (500/125Mz) for Compound 1 in Dimethyl Sulfoxide-d 6.

(–)-HR-ESIMS m/z 543.1872 [M – H]‒ (calcd for C28H31O11, 543.1872).
Determination of the Configuration of the Sugar Residue
Compound
DNA Topo I Inhibitory Activity Assay
Topoisomerase I inhibitory activity was judged by evaluating the easing of the super-coiled pBR-322 plasmid of DNA. Camptothecin was used as a positive control. 23 The gel was envisioned under UV radiation and immediately photographed with the support of the gel imaging station system after being stained with Gelred. The MICs were determined at the lowest concentrations of compounds above which no higher stripe was detected.
Antibacterial Activity Assay
Compounds
Cytotoxicity Assay
The cytotoxicity was measured with the sulforhodamine B method, 25 by using liver hepatocellular carcinoma (HepG2), human cancer of the colon (HCT116), human lung carcinoma (A-549), human cervical carcinoma (HeLa), and human pancreatic cancer cells (PANC-1), human colon cancer cell (SW480). The half-maximal inhibitory concentration value was analyzed by software GraphPad Prism 5. The adriamycin was used as a positive control. 26
Supplemental Material
Supplementary material - Supplemental material for One New Terphenyl Glycoside From a Sponge-Derived Fungus Trichoderma reesei (HN-2016-018)
Supplemental material, Supplementary material, for One New Terphenyl Glycoside From a Sponge-Derived Fungus Trichoderma reesei (HN-2016-018) by Saif Ur Rehman, Jing-Shuai Wu, Lu-Jia Yang, Shi Ting, Chang-Lun Shao and Chang-Yun Wang in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. U1606403); the Scientific and Technological Innovation Project Financially Supported by Qingdao National Laboratory for Marine Science and Technology (No. 2016ASKJ08), and the Taishan Scholars Program, China
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
