Abstract
Two intergeneric hybrids between Ligularia nelumbifolia and Cremanthodium stenoglossum were examined with respect to the chemical composition of root extracts and the sequences of neutral DNA regions. The DNA data showed that the direction of hybridization was different between the individuals. Eremophilane sesquiterpenes were found in both hybrids and deduced to have come from their Cremanthodium parents, because sesquiterpenes were detected in C. stenoglossum but not in L. nelumbifolia.
More than 100 species of Ligularia (Asteraceae) exist in the Hengduan Mountains area of China, providing us with interesting materials for studies of chemical evolution of plants. We have been examining chemical and genetic diversity in Ligularia by analyzing root chemicals and the DNA sequence of evolutionarily neutral regions. 1,2 Eremophilane-type sesquiterpenes have been isolated from most of the major species of Ligularia. Many species have been found to harbor intraspecific diversity.
Hybridization is an important pathway in plant evolution. 3 Over the course of our Ligularia research, spanning nearly 20 years, we have had opportunities to collect hybrids involving Ligularia. Detailed morphological and genetic characterization has been performed for some hybrids. 4 -8 Chemical characterization has revealed a variety of outcomes of hybridization. 9 -15 In some cases, both parents contributed compounds to their offspring; in some, the chemical contribution was only from one parent; in others, hybridization appeared to have engendered compounds that were absent in their parents. 9,12 -14 In addition, introgression has been revealed by DNA analysis 10,12,15,16 and inferred to be behind the presence of the compounds that are detected only in some populations within a species. 12,17 These results show that hybridization is important in the chemical evolution of Ligularia in the Hengduan Mountains area.
We have found hybrids of Ligularia nelumbifolia (Bureau & Franchett.) Handel-Mazzetti and Cremanthodium stenoglossum Y. Ling & S.W. Liu in Sichuan Province, China. 8 The genus Cremanthodium is taxonomically close to Ligularia and found at altitudes of circa 4000 m in the Hengduan Mountains area. 18,19 Sesquiterpenes such as eremophilanes, 20 -22 bisabolanes, 23 -26 and other compounds 27,28 have been isolated from Cremanthodium. These compounds are found in many Ligularia species as well. In this report, we describe the chemical composition of the root extracts of 2 hybrid individuals (samples 1 and 2) as well as their parents, L. nelumbifolia (sample 3) and C. stenoglossum (sample 4), collected at the same location (Table 1). To the best of our knowledge, this is the first chemical study on an intergeneric hybrid between Ligularia and Cremanthodium and is also the first on C. stenoglossum.
Compounds in Ligularia nelumbifolia, Cremanthodium stenoglossum, and Their Hybrids.

aThose in parentheses were only detected by LC-MS; the others were isolated.
DNA sequencing was carried out for the internal transcribed spacer (ITS)1-5.8S-ITS2 region of the nuclear rRNA gene and the plastid rpl32-trnL intergenic region. The results are summarized in Tables 2,3. Most of the multiple-base sites in ITS1-5.8S-ITS2 in samples 1 and 2 are the superposition of the bases in samples 3 and 4 (Table 2). This result confirms that samples 1 and 2 are hybrids of L. nelumbifolia and C. stenoglossum. The sequence of the rpl32-trnL region was quite different between sample 3 (L. nelumbifolia) and sample 4 (C. stenoglossum), especially by 2 indels (Table 3). The sequence of sample 1 was almost the same as that of sample 3; the sequence of sample 2 was identical to that of sample 4. Because plastids are maternally inherited, the rpl32-trnL sequences show that the maternal parent of sample 1 was L. nelumbifolia and that of sample 2, C. stenoglossum.
Differences in ITS1-5.8S-ITS2 Among the Samples and Reference Sequences. a

aThe base numbering is based on the sequences of L. nelumbifolia (sample 3 and a reference sequence; see note b). R = A + G; S = C + G; W = A + C; Y = C + T; - = deletion.
bTwo sequences with and without CG were present.
cThe sequence of L. nelumbifolia in the database (ID = JF976821).
dThe sequence of C. stenoglossum in the database (ID = AY176136).
Differences in rpl32-trnL Among the Samples. a

aThe base numbering is based on the database entry of sample 4 (see note d), which includes a partial sequence of rpl32. - = deletion.
bThe same as in the database entry of sample 4 (see note d).
cDeposited in the database with ID = LC457553.
dDeposited in the database with ID = LC457552.
Root samples were extracted with EtOH and the extracts were subjected to LC-MS (MeOH/H2O). Their total ion chromatograms are shown in Figure 1. Four peaks were detected for sample 1 in the region of typical terpenoids (t
R = 10-20 minutes). The peaks at t
R = 16.1 (m/z 317), 15.5 (m/z 317), 14.5 (m/z 219), and 14.1 (m/z 219) minutes were identified as

Total ion chromatograms of samples 1 to 4.
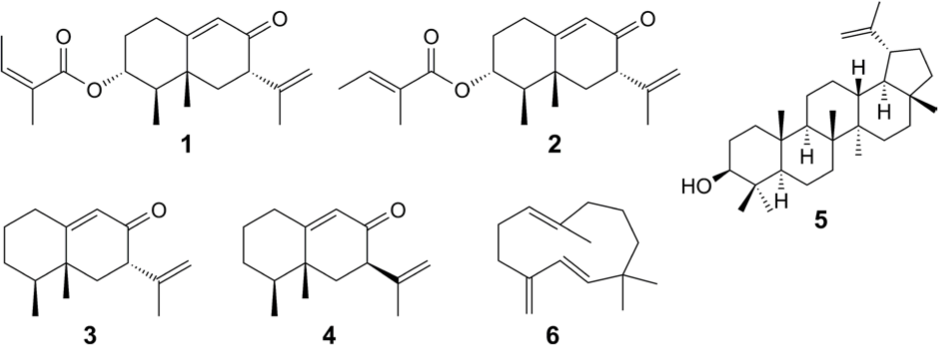
Compounds in the extracts were isolated by use of silica-gel column chromatography and HPLC. From sample 1, eremophilane compounds
The results above indicate that the eremophilanes in the hybrid samples must have originated from C. stenoglossum, since no eremophilane was detected in the parental L. nelumbifolia (sample 3). The plants belonging to what we call the dkn-complex, comprising L. duciformis, L. kongkalingensis, and L. nelumbifolia, can be grouped into 4 chemotypes. The typical compounds in each chemotype are eremophilane sesquiterpenes (type 1), oplopane sesquiterpenes (type 2), phenylpropanoids (type 3), and no sesquiterpenes or phenylpropanoids (type 4), although the entire chemical spectrum is somewhat continuous.
29,35,36
Since lupeol (
Eremophilanes have been isolated from other Cremanthodium species:
In conclusion, the present results indicate ongoing chemical exchange between Ligularia and Cremanthodium, which are undergoing reticulate evolution in the Ligularia-Cremanthodium-Parasenecio complex. 19
Experimental
General
NMR, JEOL ECX-400 (400 MHz for 1H; 100 MHz for 13C) spectrometer; MS, JEOL JMS-700 MStation or CMATE II. Column chromatography (CC), silica gel (Wakosil C-200 or C-300). Analytical TLC, Merck Kieselgel 60 F254, 0.2 mm thickness, using p-anisaldehyde/AcOH/H2SO4 as visualizing agents. HPLC, GL Sciences GL-7410 pump with a GL-7450 UV detector, and a Hitachi D-2500 Chromato-Integrator, with a Kanto Mightysil Si60 (10 × 250 mm) column. LC-MS, Agilent 1100 series LC/MSD mass spectrometer with 5C18-MS-II using a gradient system (MeOH/H2O) as eluent. See Ref. 17 for details of LC-MS.
Plant Materials
The samples were collected in August 2016 on a border between Ganze and Xinlong Counties (elevation: 4700 m), Sichuan Province, China, and were identified by X. G. (author). Voucher specimen numbers are 2016-38 (hybrid), 2016-39 (hybrid), 2016-40 (L. nelumbifolia), and 2016-41 (C. stenoglossum) for samples 1 to 4, respectively (Kunming Institute of Botany).
DNA Analyses
DNA was purified from dried leaves with a commercial kit (DNeasy Plant Minikit, QIAGEN). The amplification of the ITS1-5.8S-ITS2 region by polymerase chain reaction and the sequencing reactions of the region were carried out with the primers described previously. 39 The rpl32-trnL region was amplified and sequenced with a primer rpl32 (5′-CAGTTCCAAAAAAACGTACTTC-3′) and a primer trnL (5′-CTGCTTCCTAAGAGCAGCGT-3′). 40 The sequences were determined by a Model 3500 Genetic Analyzer (Applied Biosystems).
Isolation of Compounds
Dried roots of sample 1 (hybrid) (10.2 g) were extracted with EtOH. CC (n-hexane-EtOAc, gradient) of the resulting extract (556.1 mg) afforded 6 fractions. Fraction 3 (eluted with hexane/EtOAc at 8:2) was subjected to CC (n-hexane-EtOAc) and HPLC (hexane/EtOAc 9:1) and yielded
Dried roots of sample 2 (hybrid) (20.4 g) were extracted with EtOH. CC (n-hexane-EtOAc, gradient) of the extract (797.7 mg) afforded 6 fractions. Fraction 4 (eluted with hexane/EtOAc 7:3) was subjected to CC (n-hexane-EtOAc) and HPLC (hexane/EtOAc 7:3) and
Dried roots of sample 3 (L. nelumbifolia) (9.9 g) were extracted with EtOH. CC (n-hexane-EtOAc, gradient) of the extract (797.7 mg) afforded 6 fractions. Fraction 3 (eluted with hexane/EtOAc 8:2) was further subjected to CC and
Dried roots of sample 4 (C. stenoglossum) (18.6 g) were extracted with EtOH. The extract (1093.9 mg) was subjected to CC (n-hexane-EtOAc, gradient). From the least polar fraction (eluted with n-hexane),
Footnotes
Acknowledgments
The authors thank Dr Takayuki Kawahara, Forestry and Forest Products Research Institute; Dr Yoshinori Saito, Nagasaki University; Dr Yoshinosuke Usuki, Osaka City University; and Dr Katsuyuki Nakashima, Tokushima-Bunri University, for their help in sample collection and valuable discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant-in-Aid for Scientific Research from JSPS (No. 25303010), Japan-China Scientific Cooperation Program from JSPS and NSFC, and Strategic Research Foundation Grant-aided Project for Private Universities from the MEXT, Japan.
