Abstract
The composition of root chemicals was studied for 7 samples of Ligularia tongolensis collected in Yunnan and Sichuan Provinces of China. The structures of 2 new 3β-angeloyloxy-6β-acyloxyfuranoeremophilan-15-oic acids were determined. It was found that the plant harbors chemical diversity in the acyloxy groups in 3,6-bis(acyloxy)eremophilan-15-oic acids. The presence of a 3-methylpentanoate moiety at C-3 appears geographically differentiated to a degree. Consistent with this low diversity, results of DNA analysis indicated little genetic differentiation, although introgression was inferred for one of the samples.
The genus Ligularia is highly diversified in the Hengduan Mountains area of China. We have been studying the chemical diversity within each species while referring to DNA data as an index of genetic diversification. To date, we have found various degrees of intraspecific diversity in many species. 1,2 Sesquiterpenoids of the eremophilane type have been found in many species; furanoeremophilanes, in particular, are the major components in major Ligularia species. Ligularia tongolensis (Franch.) Hand.-Mazz. is found in northwestern Yunnan, western Sichuan, and southeastern Tibet. 3 We previously reported that L. tongolensis in northwestern Yunnan Province (Shangrila County) and southwestern Sichuan Province (Daocheng County) produced 3,6-bis(acyloxy)furanoeremophilan-15-oic acids and 3-acyloxyfuranoeremophilan-15,6-olides. 4 Related eremophilane-(12,8),(15,6)-diolides were isolated by Han et al. 5 The chemical composition, judged from TLC patterns, was very similar among our L. tongolensis samples except for the presence of tetradymol in some samples collected in Shangrila County. 4 Hybrids of L. tongolensis and L. cymbulifera were also analyzed and the ability to produce tetradymol in L. tongolensis was inferred to have been introgressed from L. cymbulifera. 6
In the course of our subsequent search in the field, we collected 32 additional samples of L. tongolensis. Seven of the samples (samples 1-7), collected in 2015 to 2017 at locations shown in Table 1 and Figure 1, were analyzed in detail. Samples 1 and 2 (4 km apart from each other) were collected in Wenchuan County, Sichuan, which is about 500 km from the collection area of our previous L. tongolensis samples 4 (Figure 1, red circle). Sample 3 was collected at Pachahai, Shangrila County, Yunnan, where the above hybrid samples were collected. 6 Sample 4 was collected at Tianchi, Shangrila County, where we collected various Ligularia hybrids, for which the chemical outcomes of hybridization were described. 7 -9 Samples 5, 6 (4 km apart from each other), and 7 were collected in Muli County, Sichuan.

Collection locations of the present Ligularia tongolensis samples (squares). Circles indicate major cities. Solid and dotted lines indicate rivers and province boundaries, respectively. The red circle indicates the collection area of our previous 17 samples. 4
Collection Locales and Isolated Compounds of Ligularia tongolensis Samples.

aCounty and province in parentheses.
The EtOH extract of each root sample was subjected to Ehrlich’s test on TLC, a facile method to detect furanoeremophilanes. 10,11 All the samples showed TLC spots similar to those of our previous samples, suggesting that the composition of furanoeremophilanes was similar. Sample 4 showed an additional strong Ehrlich-positive spot of tetradymol at R f = 0.66 (hexane/EtOAc 7:3).
Compounds in each sample were separated with such standard methods as silica-gel column chromatography (CC) and HPLC, while air oxidation and decomposition were carefully avoided. From the Wenchuan samples (samples 1 and 2),

Isolated compounds from Ligularia tongolensis. The asterisks denote new compounds.
Attempts to isolate the new compounds

Key COSY and HMBC correlations for the acid part of 1c and 1d at C-6.
1H and 13C NMR of New Compounds 1c and 1d (C6D6). a
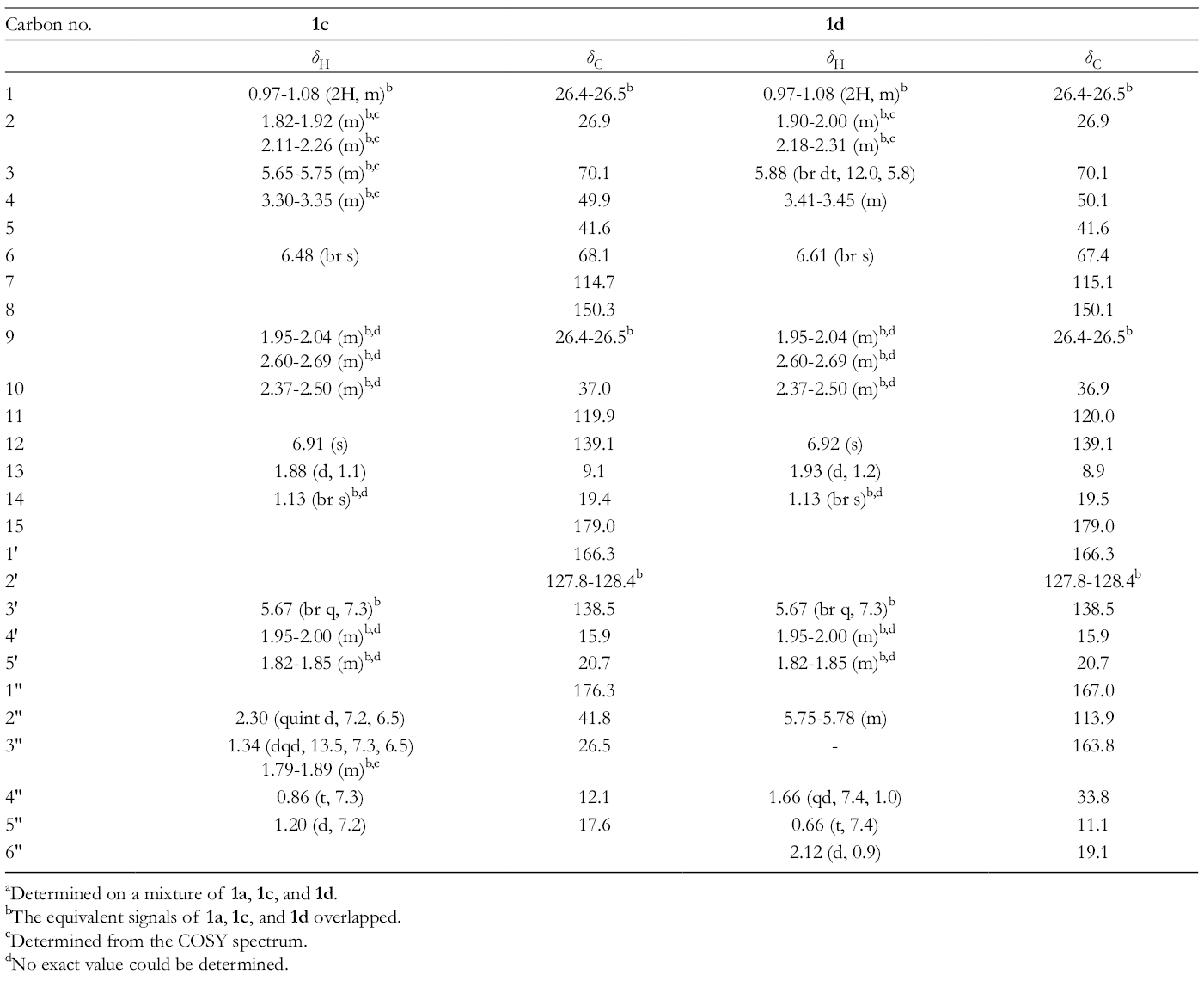
aDetermined on a mixture of
bThe equivalent signals of
cDetermined from the COSY spectrum.
dNo exact value could be determined.
The chemical composition was also analyzed for each sample by LCMS. The total ion chromatograms (TICs) of samples 1 to 7 are shown in Figure 4. In samples 1 and 2,

Total ion chromatograms of samples 1 to 7. The differences in the retention time among the samples are within experimental error.
The LCMS data indicate that the ratio of 3,6-bis(acyloxy)-eremophilan-15-oic acids,

Total ion chromatograms of samples 8 to 11.

Lactonization of 3,6-bis(acyloxy)furanoeremophilan-15-oic acids. See Figure 2 for A, B, and C.
Supposing that the rate of the conversion of
The base sequence of the ITS1-5.8S-ITS2 of the ribosomal RNA gene in the nuclear genome was determined for samples 1 to 11. The results are shown in Table 3. Sample 6 has an exceptionally large number (27) of multiple-base sites. As we discussed previously, it indicates hybridization with some other species. 6 Since the morphology of the sample is of L. tongolensis, it must have undergone backcrossing; namely, it is introgressed. The bases at these sites cannot be accounted for by the superposition of L. tongolensis and L. cymbulifera sequences (Table 3). The hybridization partner is currently unknown because database search using a putative introgressive sequence found no clear candidate with a very good agreement. The sequences of the other samples are essentially the same, indicating that they are not differentiated genetically.
Differences in the ITS1-5.8S-ITS2 Region Among Ligularia tongolensis Samples. a

aThe base numbering is according to the database entry DQ272337 of L. tongolensis. K = G + T; M = A + C; R = A + G; W = A + T; Y = C + T.
bA sequence with C6 in place of C5 at 154 to 159 of ITS1 was also present.
cA sequence with T3 in place of T2 at 156 to 157 of ITS2 was also present.
dA L. tongolensis sequence in the database (DQ272337).
eA L. cymbulifera sequence in the database (DQ272330).
Tetradymol (
In conclusion, L. tongolensis harbors some chemical diversity in the acyloxy groups of 3,6-bis(acyloxy)eremophilan-15-oic acid. This range of variation within the same terpenoid framework suggests that the diversity in L. tongolensis is limited, which appears consistent with DNA sequence data.
Experimental
General
NMR, JEOL ECX-400 spectrometer (400 MHz for 1H; 100 MHz for 13C) or Varian Unity Plus 500 spectrometer (500 MHz for 1H; 125 MHz for 13C); IR, JASCO FT/IR-230 spectrometer; MS, JEOL JMS-700 MStation or CMATE II. CC was performed on silica gel (Wakosil C-200 or C-300). Analytical TLC was carried out on Merck Kieselgel 60 F254, 0.2 mm thickness, with either Ehrlich's reagent (p-dimethylaminobenzaldehyde and HCl) 10,11 or p-anisaldehyde/AcOH/H2SO4 as visualizing agent. HPLC was carried out by use of either a Shimadzu LC-20AT pump with an SPD-20A Prominence UV/VIS detector, a GL Sciences GL-7410 pump with a GL-7450 UV detector, or a JASCO 880-PU pump with an 875-UV detector, and a Hitachi D-2500 Chromato-Integrator, a Shimadzu C-R8A Chromatopac, or a SOMA OPTICS MDL-102 recorder, with either a GL Sciences Inertsil PREP-ODS column (20 × 250 mm), a Kanto Mightysil Si60 (10 × 250 mm) column, or a Nacalai Tesque Cosmosil 5SL-II (10 × 250 mm) column. LCMS was measured on an Agilent 1100 series LC/MSD mass spectrometer. See Reference 21 for details. High-resolution LCMS (ESI) was measured on a Waters SYNAPT G2-Si HDMS with a UPLC-I Class system and an electrospray ionization interface. UPLC was carried out by use of an ACQUITY UPLC BEH C18 column (2.1 × 100 mm, Waters).
Plant Material
Samples were collected in August 2015, 2016, and 2017 at locations shown in Table 1 and Figure 1. Each sample was identified by X. G. (author). Voucher specimen numbers are 2015-04, 2015-05, 2016-220, 2016-221, 2017-02, 2017-05, and 2017-27 for samples 1 to 7, respectively (Kunming Institute of Botany).
Isolation of Compounds
EtOH extraction of dried root of sample 1 (23.7 g) afforded an extract (2.24 g). CC (n-hexane-EtOAc, gradient) of the extract resulted in 9 fractions. CC (n-hexane-EtOAc) of fraction 5 (eluted with n-hexane-EtOAc 90:10) afforded a mixture of
EtOH extraction of dried root of sample 2 (19.6 g) afforded an extract (1.25 g). CC (n-hexane-EtOAc, gradient) of the extract afforded 8 fractions. From fraction 4 (eluted with n-hexane-EtOAc 90:10),
EtOAc extraction of dried root of sample 3 (1.9 g) afforded an extract (116.2 mg). CC (n-hexane-EtOAc, gradient) of the extract afforded 6 fractions. From fraction 3 (eluted with n-hexane-EtOAc 95:5 to 90:10),
EtOH extraction of dried root of sample 4 (6.9 g) afforded an extract (630.5 mg). CC (n-hexane-EtOAc, gradient) of the extract afforded 9 fractions. From fraction 3 (eluted with n-hexane-EtOAc 96:4 to 94:6),
EtOH extraction of dried root of sample 5 (13.0 g) afforded an extract (1021.0 mg). CC (n-hexane-EtOAc, gradient) of the extract afforded 13 fractions. From fraction 2 (eluted with n-hexane-EtOAc 96:4),
EtOH extraction of dried root of sample 6 (1.7 g) afforded an extract (54.1 mg). CC (n-hexane-EtOAc, gradient) of the extract afforded 2 fractions. From the polar fraction (eluted with n-hexane-EtOAc 80:20 to 50:50),
EtOH extraction of dried root of sample 7 (4.2 g) afforded an extract (122.8 mg). CC (n-hexane-EtOAc, gradient) of the extract afforded 4 fractions. From fraction 3 (eluted with n-hexane-EtOAc 91:9 to 86:14), a mixture of
Compounds 1c and 1d
1H and 13C NMR (C6D6): see Table 2.
HR-LCMS (ESI):
DNA Analysis
Purification of DNA from dried leaves, amplification of the ITS1-5.8S-ITS2 region with polymerase chain reaction, purification of the amplification product, and DNA sequencing were carried out as previously described. 22
Supplemental Material
Supplementary material - Supplemental material for Diversity of Furanoeremophilane Composition in Ligularia tongolensis
Supplemental material, Supplementary material, for Diversity of Furanoeremophilane Composition in Ligularia tongolensis by Yasuko Okamoto, Yumi Nakadozono, Kosuzu Shiojiri, Sayaka Suehiro, Yoshinori Saito, Yosuke Matsuo, Takashi Tanaka, Chiaki Kuroda, Motoo Tori, Xun Gong and Ryo Hanai in Natural Product Communications
Footnotes
Acknowledgments
The authors thank Dr Takayuki Kawahara, Forestry and Forest Products Research Institute, Dr Yoshinosuke Usuki, Osaka City University, and Dr Katsuyuki Nakashima, TokushimaBunri University, for their help in sample collection and valuable discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by a Grant-in-Aid for Scientific Research from JSPS (No. 25303010), the Japan-China Scientific Cooperation Program from JSPS and NSFC, and a Strategic Research Foundation Grant-Aided Project for Private Universities from the MEXT, Japan.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
