Abstract
Sea buckthorn (Hippophae rhamnoides L.) is a medicinal plant widely distributed in Asia and Europe, containing plentiful bioactive substances. Our research aimed to promote the comprehensive utilization of the branches, leaves, and berries of sea buckthorn. Qualitative analysis of chemical constituents in branches, leaves, and berries of sea buckthorn was conducted by ultra-high performance liquid chromatography accurate mass quadrupole time-of-flight mass spectrometry with electrospray ionization (UHPLC-ESI-QTOF-MS). As a result, the branch, leaf, and berry samples could be clearly separated in principal component analysis scores plot, and 24 significant markers were found to distinguish these parts by partial least squares regression discrimination analysis in Mass Profiler Professional software. Meanwhile, the compositional similarity of sea buckthorn leaves and branches was higher than that of leaves and berries. In addition, the inhibition of nitric oxide (NO) production of different parts in macrophage RAW 264.7 cells was carried out. At the concentration of 10 μg/mL, sea buckthorn extracts showed good anti-inflammatory activities with NO inhibition values from 73% to 98%.
Sea buckthorn (Hippophae rhamnoides L.) is a kind of hardy deciduous plant belonging to family Elaeagnaceae, mainly distributed in countries along the Silk Road. As an important economic plant with potential health benefits and high commercial interests, sea buckthorn has drawn worldwide attention. Sea buckthorn has significant pharmacological effects, such as antioxidant, 1,2 antimicrobial, 3 antiviral, 4 anti-inflammatory, 5 -7 and anticancer 4 activities. Sea buckthorn contains plentiful bioactive substances, such as ellagitannins, 8 flavonol glycosides, 8,9 proanthocyanidins, 10,11 carotenoids, 12 sterols, 13 and tocopherols. 13
The UHPLC-ESI-QTOF-MS method is widely used to analyze and identify various chemical components with excellent sensitivity and resolution. Phenolic compounds in sea buckthorn have been analyzed by liquid chromatography - mass spectrometry (LC-MS). 8,9,11,14-16 It has been demonstrated that ellagitannins are the predominant phenolic compounds in leaves, 15 and flavonol glycosides are the main phenolic compounds in berries. 15 Moreover, the content of flavonol glycosides in leaves is higher than that of berries. 9,14 Isorhamnetin and quercetin are the major aglycones in berries 14,17 ; casuarinin and stachyurin are the most abundant ellagitannins in leaves. 8 Phenolic compounds possess a broad range of health-related properties, such as anti-inflammatory, 18,19 antioxidant, 3 and antimicrobial 20 effects. To our knowledge, the researches on the branch of sea buckthorn are still very rare. In order to promote the comprehensive utilization of sea buckthorn, we carried out the analysis on the components and markers of different parts (berry, leaf, and branch) of sea buckthorn by UHPLC-ESI-QTOF-MS. The anti-inflammatory activities of the branches, leaves, and berries of sea buckthorn were determined by measuring the nitric oxide (NO) production in RAW 264.7 cells.
Identification of Compounds in Sea Buckthorn Branch, Leaf, and Berry
Information of all the sea buckthorn samples was listed in Table 1. As shown in Figure 1 and Table 2, a total of 44 peaks were detected in the branch, leaf, and berry of 3 representative samples S7, S8, and S9 by UHPLC-ESI-QTOF-MS. These peaks were identified or tentatively identified by comparing the fragmentation patterns in mass spectra to those of standard compounds or references. The secondary mass spectrometry (MS/MS) spectra of 44 peaks were presented in Supplemental Figure S1.
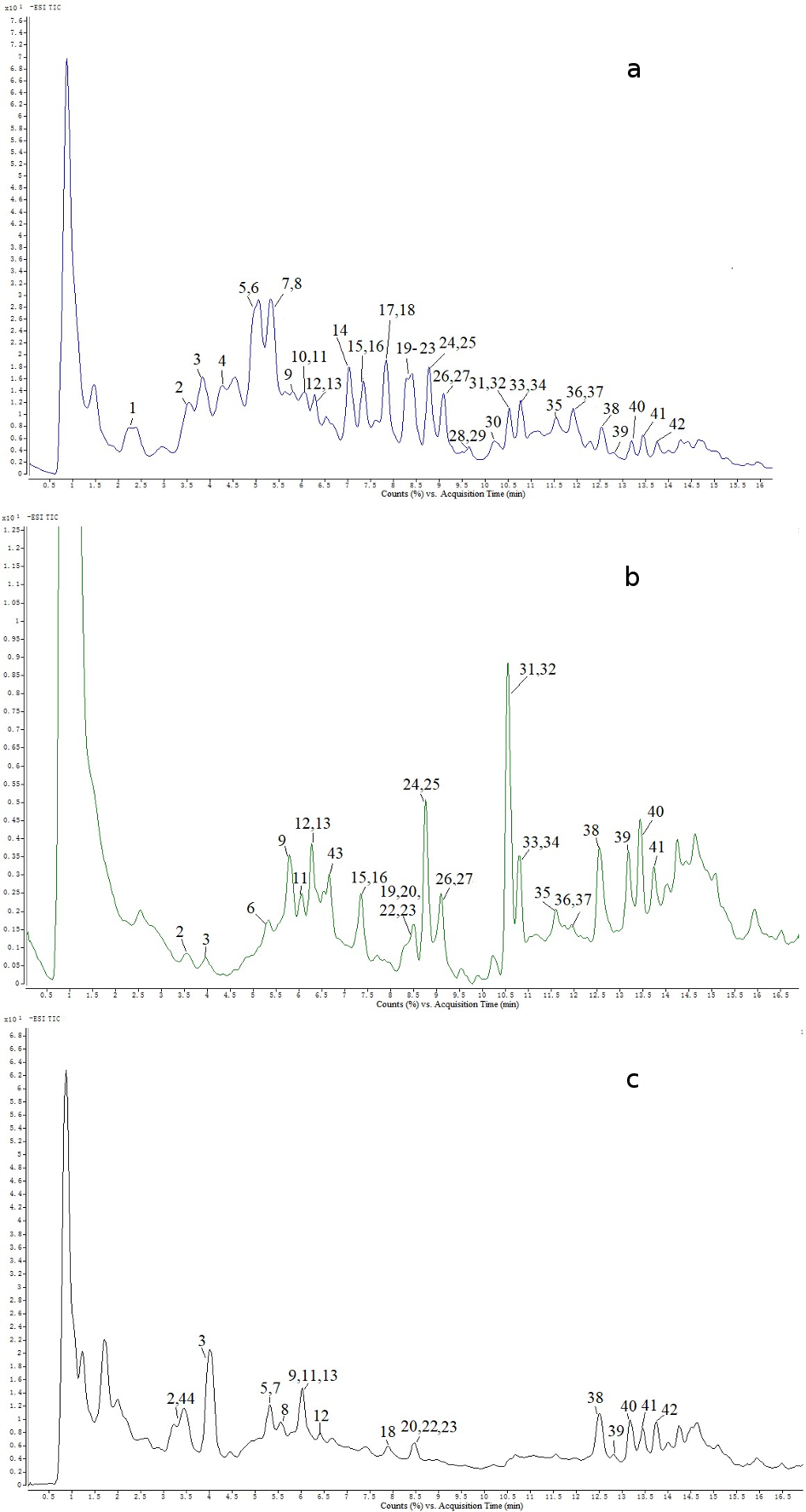
Representative UHPLC-ESI-QTOF-MS total ion chromatograms in negative ion mode of leaf, sample S8 (a); berry, sample S9 (b); and branch, sample S7 (c).
Information of Sea Buckthorn Samples.
Mass Data of Compounds Detected by UHPLC-ESI-QTOF-MS of Sea Buckthorn in Negative Ion Mode.

HHDP, hexahydroxydiphenoyl; Rt, retention time.
Thirteen peaks, pedunculagin (
In the negative ion mode, mass fragment at m/z 301 was an indicator of the presence of quercetin aglycone.
8,21,22
Compound
The fragment ion at m/z 285 was an indicator of the presence of kaempferol aglycone.
8,21
Compound
The fragment ion at m/z 315 was an indicator of the presence of isorhamnetin aglycone.
8
Compound
The mass fragment at m/z 301 (300.999) in the negative ion mode was an indicator of the presence of ellagic acid derivatives, and the hexahydroxydiphenoyl (HHDP) group was lactonized spontaneously to ellagic acid.
8,21,22
Compound
Two compounds (
Comparison of Different Parts of Sea Buckthorn
Unsupervised principal component analysis (PCA) was applied to observe the differences and similarities in chemical constituents between different parts of sea buckthorn (Figure 2). The PCA score plot showed that the branch, leaf, and berry samples were well separated. The first 2 principal components of the PCA model explained 56.10% of the total variance.

The score plot of principal component analysis for different parts of sea buckthorn in negative ion mode (leaf, berry, and branch).
The hierarchical cluster analysis (HCA) of sea buckthorn samples was shown in Figure 3. We can clearly see that all samples of sea buckthorn were classified into 3 significant clusters of berry, leaf, and branch. Cluster I (S3, S6, S9, S12, S15, and S16) consisting of all berry samples showed higher compositional similarity of sea buckthorn berries in Kashi, Aletai, and Gilgit-Baltistan. Cluster I also indicated that the composition of sea buckthorn berries in Youyv, Lvliang, and Dingxi had a certain degree of similarity. Cluster II (S2, S5, S8, S11, S14, and S18) forming from all leaf samples showed higher compositional similarity of sea buckthorn leaves in Aletai and Dingxi. Cluster III (S1, S4, S7, S10, and S13) consisting of all branch samples showed higher compositional similarity of sea buckthorn branches in Youyv and Dingxi. Based on 3 clusters shown in HCA, we can speculate that the compositional similarity of sea buckthorn leaves and branches was higher than that of leaves and berries.

Hierarchical clustering analysis of different parts of sea buckthorn tested in negative ion mode.
A better separation between 3 groups was observed in the supervised partial least squares regression discrimination analysis (PLS-DA) score plot (Figure 4). Cross-validation was used to estimate the predictive abilities of the constructed model. The number of components, folds, and repeats of the model was set to 3, 7, and 200, respectively. Validation matrix accuracy was 100%. The parameters for the model were 0.904 (R 2 Y) and 0.709 (Q 2 Y). Hence, the established model had good fitness and prediction. We work on finding the chemical markers in different parts of sea buckthorn with variable importance in projection (VIP) >1.5 and P < 0.05.
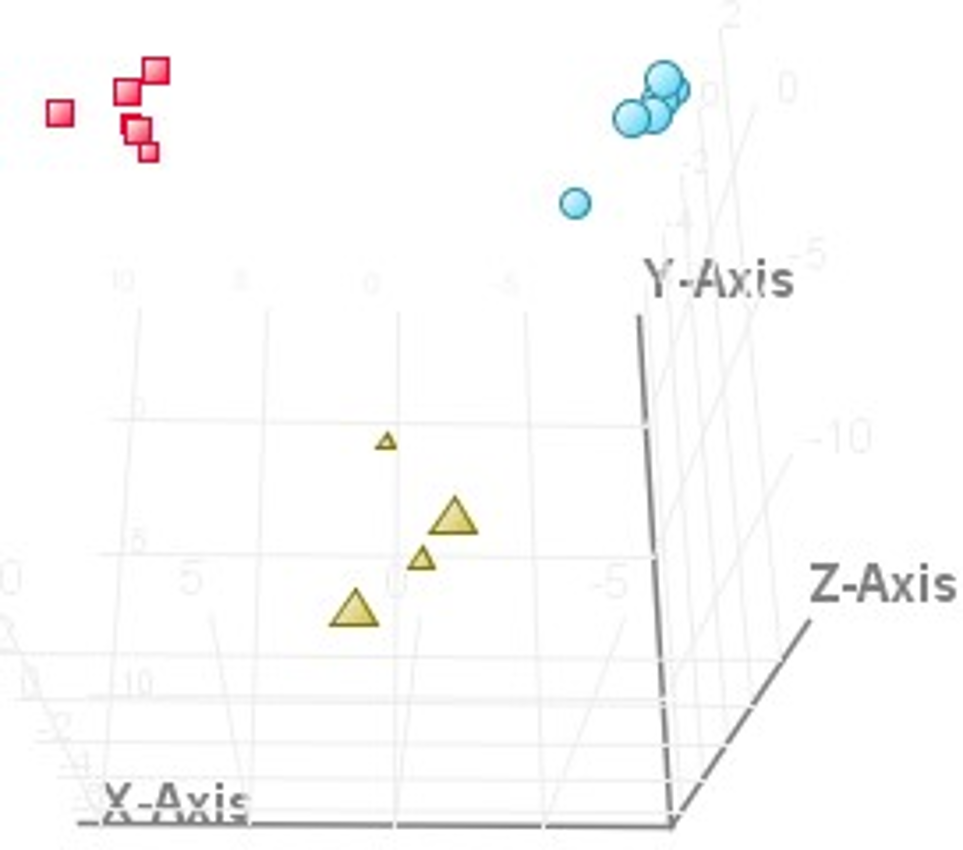
The score plot of 3D partial least squares regression discrimination analysis model in negative ion mode (leaf, berry, and branch).
As a result, a total of 24 significant ions were selected as the chemical markers (
Mass Data of Chemical Markers in Different Parts of Sea Buckthorn in Negative Ion Mode.

Rt, retention time.
Detailed information of 24 marker compounds was clearly shown in the heatmap (Figure 5). The heatmap indicated that the content of

Heatmap of marker compounds in different parts of sea buckthorn in negative ion mode.
Anti-Inflammatory Activities of Sea Buckthorn
As shown in Table 4, the anti-inflammatory activities of sea buckthorn branches, leaves, and berries were measured by the NO production. In addition, at the concentration of 10 µg/mL, sea buckthorn extracts showed NO inhibition rates from 73% to 98%, and cytotoxic effects have not been observed in 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assays (Supplementary Table S1), indicating that sea buckthorn extracts displayed good anti-inflammatory activities on RAW 264.7 macrophages.
The Nitric Oxide Inhibition Rate of Sea Buckthorn (mean ± SD, N = 3, 10 µg/mL).

The NO inhibition rates of S13, S14, S15, S16, and S17 were significantly higher than those of other samples, which showed that the branches, leaves, and berries in Dingxi and leaves and berries in Gilgit-Baltistan have better anti-inflammatory activities. As the samples of Gilgit-Baltistan were taken from the town of Sost (75°25′E, 36°50′N), with an average altitude of 4700 m. The higher elevation may enhance the anti-inflammatory activity of sea buckthorn. In future, we need to collect more samples for the further research.
Experimental
Chemicals and Reagents
The berries, leaves, and branches of sea buckthorn were collected from China, Pakistan, and Tajikistan. All samples were identified by the author of Dr Zhigang Yang and dried under shade until the study began.
Reference compounds of isorhamnetin-3-O-glucoside-7-O-rhamnoside, isorhamnetin-3-O-sophoroside-7-O-rhamnoside, isorhamnetin-3-O-glucoside, and quercetin-3-O-rutinoside were isolated from sea buckthorn berries in our lab. Isorhamnetin, quercetin, ellagic acid, stachyurin, pedunculagin, hippophaenin B, and 1,2,6-tri-O-galloyl-β-d-glucopyranose were isolated from sea buckthorn leaves by the author. Isorhamnetin-3-O-rutinoside and quercetin-3-O-glucoside were purchased from Chengdu Pufeide Biotech Co., Ltd. (China). LC-MS grade methanol and acetonitrile were purchased from Mreda Technology Inc. (United States). LC-MS grade water and formic acid were purchased from Merck Company (Germany).
Dulbecco’s Modified Eagle’s Medium (DMEM), dimethyl sulfoxide, and MTT were purchased from Sigma-Aldrich (United States). Fetal bovine serum (FBS) and antibiotics (penicillin and streptomycin) were purchased from Gibco BRL (Grand Island, NY, United States). Lipopolysaccharide (LPS) was purchased from Beijing LabLead Biotech Co., Ltd. (China). Nitric Oxide Colorimetric Assay Kit (NO kit) was purchased from Beijing BioDee Biotech Co., Ltd. (China).
Sample Preparation
The sea buckthorn samples were milled into powder. Each powdered sample (0.2 g) was precisely weighed and extracted by ultrasonic extraction with 20 mL 70% LC-MS grade methanol, followed by centrifugation with centrifugal filter units. A mixture of stock standard solution containing 13 reference compounds was prepared. All collected sample solutions and standard solutions were stored at 4°C prior to injection.
UHPLC-ESI-QTOF-MS Analysis
Chromatographic analysis was performed on an Agilent 1290 series UHPLC system (Agilent Technologies, United States). The sea buckthorn samples were loaded onto a Waters CORTECS UHPLC C18 analytical column (2.1 mm × 100 mm, 1.6 µm) at temperature of 36°C. The injection volume was 2 µL and the flow rate was 0.3 mL/min. Mobile phase consisted of water (A) and acetonitrile (B) (both containing 0.1% formic acid), and the following gradient program was used: 0 to 2 minutes, 7% B; 2 to 3 minutes, 7% to 12% B; 3 to 8 minutes, 12% to 18% B; 8 to 12 minutes, 18% to 60% B; 12 to 13 minutes, 60% to 100% B; and 13 to 14 minutes, 100% B.
Mass spectrometry analysis was performed on an Agilent 6560 Ion Mobility QTOF MS system (Agilent Technologies, United States) equipped with an electrospray ionization (ESI) source. Mass spectrometry parameters in negative ion mode were as follows: drying gas (N2) flow rate 5 mL/min; drying gas temperature 300°C; nebulizer pressure 35 psig; capillary voltage 3500 V; nozzle voltage 1000 V; fragmentor 400 V; and fixed collision energies 20 eV. The mass range was obtained by scanning ions from m/z 100 to 1700.
Statistical Analysis
All the raw data in negative ion mode collected from the UHPLC-ESI-QTOF-MS were imported into Profinder software (version B.08.00), grouping samples by branches, leaves, and berries. The parameters of Profinder were set as follows: retention time tolerance 0.25 minutes, mass tolerance 10.00 ppm ±2.00 mDa, score filter 90, height filter 5000 counts, compounds minimum filter 100% of file in at least 1 sample group.
Then, the data files in CEF format generated in Profinder were then imported into Agilent Mass Profiler Professional software (MPP, version 14.9) for analysis. All files were grouped into branches, leaves, and berries, and the filter parameter was set to retain the entities that appear in at least 80.0% of samples in at least 1 condition. Principal component analysis, HCA, and PLS-DA were performed in MPP software. The chemical markers in different parts of sea buckthorn with VIP > 1.5 and P < 0.05 were filtered out. The analysis of all compounds was performed by Agilent MassHunter Qualitative Analysis (B.08.00) software.
Anti-Inflammatory Assays
RAW 264.7 macrophages were cultured in DMEM supplemented with 10% FBS and with 1% antibiotics (penicillin and streptomycin). The cells (1 × 105) were added and inoculated in 96-well plates in an incubator with a 5% CO2 atmosphere at 37°C for 24 hours. Then, the culture solution was discarded, and 100 µL of DMEM containing the mixture of sea buckthorn extracts and LPS (1 µg/mL) was added into the plate. A positive control of quercetin (10 µg/mL) was set up. After 24 hours incubation, the cell suspension was taken to determine NO inhibitory according to the instructions of the NO kit. In addition, MTT assays were used to evaluate cytotoxic activity.
Supplemental Material
Supplementary material - Supplemental material for Analysis on the Constituents of Branches, Berries, and Leaves of Hippophae rhamnoides L. by UHPLC-ESI-QTOF-MS and Their Anti-Inflammatory Activities
Supplemental material, Supplementary material, for Analysis on the Constituents of Branches, Berries, and Leaves of Hippophae rhamnoides L. by UHPLC-ESI-QTOF-MS and Their Anti-Inflammatory Activities by Wen-Hui Zheng, Hai-Ying Bai, Shu Han, Fang Bao, Kai-Xue Zhang, Li-Li Sun, Hong Du, and Zhi-Gang Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from the Major International S&T Cooperation Project, Ministry of Science and Technology of the People’s Republic of China (2016YFE0129000); the Key Program for International S&T Cooperation Projects of Gansu Province (18YF1WA115); Research project of Gansu Provincial Administration of Traditional Chinese Medicine (GZK-2015-21); and the Fundamental Research Funds for the Central Universities (lzujbky-2017-k26 and lzujbky-2017-sp40).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
