Abstract
A new oxygenated spiroketone, isodonspiroketone (
Isodon ternifolius (D.Don) Kudô is a member of the Isodon genus, which belongs to Lamiaceae family. This genus consists of approximately 150 species, which are primarily distributed in tropical and subtropical Asia. 1 Previous studies examining the chemical constituents in I. ternifolius have identified the presence of diterpenoid, 2 -6 lignan, and phenylethanoid compounds. 7 I. ternifolius has been commonly used in folk medicine for the treatment of enteritis, 8 icterohepatitis, 8 inflammation, 7,8 and cancer. 3,4 Despite the great medicinal value, there has been limited report on chemical constituents and cytotoxic activities against A549, HepG2, and MDA-MB-231 cancer cell lines. Therefore, this work describes the isolation and structural elucidation of isolated compounds as well as the evaluation of their cytotoxic activities against 3 cancer cell lines.
The methanol extract was partitioned with n-hexane, ethyl acetate (EtOAc), and n-butanol (BuOH) to obtain n-hexane, EtOAc, and BuOH-soluble fractions, respectively. Based on bioactivity-guided fractionation (data not shown), the n-hexane and EtOAc fractions were subjected to column chromatography on a silica gel and C18-RP silica gel column to obtain a new compound (
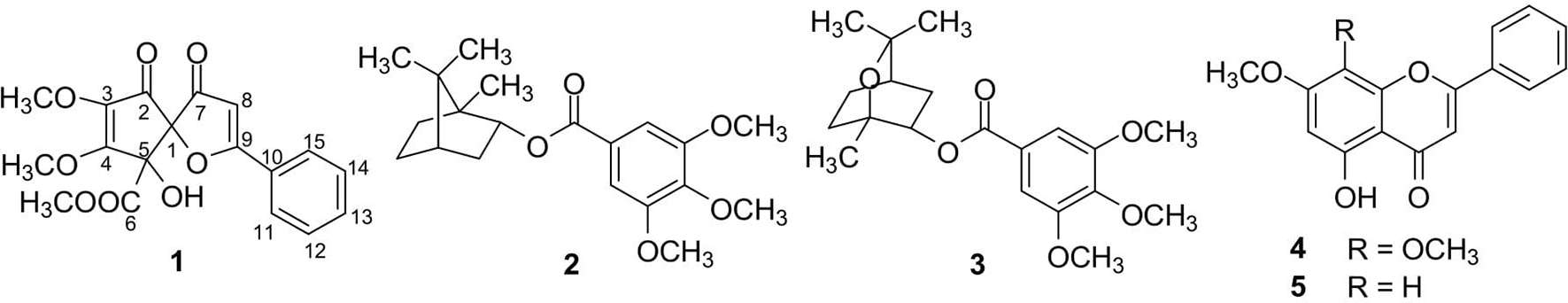
Chemical structure of compounds (
Compound
1H-NMR and 13C-NMR Spectroscopic Data for Compound.
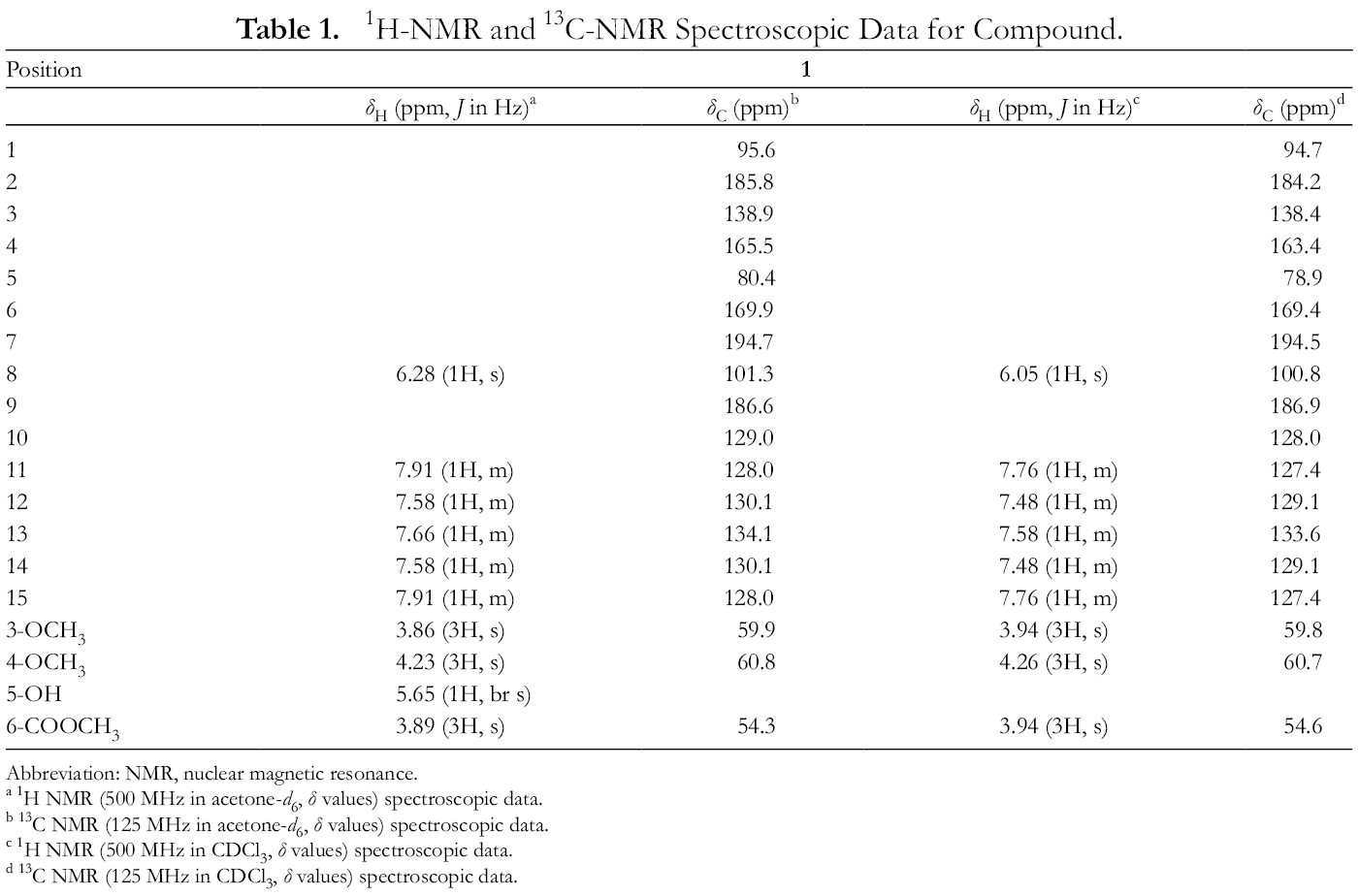
Abbreviation: Abbreviation: NMR, nuclear magnetic resonance.
a 1H NMR (500 MHz in acetone-d 6, δ values) spectroscopic data.
b 13C NMR (125 MHz in acetone-d 6, δ values) spectroscopic data.
c 1H NMR (500 MHz in CDCl3, δ values) spectroscopic data.
d 13C NMR (125 MHz in CDCl3, δ values) spectroscopic data.

Important HMBCs and NOESY spectra of compound
The optical rotation value of compound
The known compounds were identified to be 1,7,7-trimethylbicyclo[2.2.1]heptan-2-yl-3′,4′,5′-trimethoxybenzoate (
Five isolated compounds (
Cytotoxic Activity of Isolated Compounds (

Abbreviation: Abbreviation: IC50, half-maximal inhibitory concentration.
aNot tested.
bUsed as a positive control.
Experimental
General Experimental Procedures
1H-NMR (400 MHz) and 13C-NMR (100 MHz) were measured on a Varian Unity Inova 500 MHz spectrometer. TOF-MS was obtained from a Varian Fourier transform mass spectrometer and MicroQ-TOF III (Bruker Daltonics, Ettlingen Germany). IR spectroscopy was fulfilled on Nicolet Impact 410 spectrometer. UV was performed in spectroscopic V-630 UV-VIS instrument. Column chromatography was carried out on silica gel (Si 60 F254, 40-63 mesh, Merck, St Louis, MO, USA). All solvents were redistilled before use. Compounds were visualized under UV radiation (254, 365 nm) and by spraying plates with 10% H2SO4 followed by heating with a heat gun. Thin-layer chromatography (TLC) was carried out on silica gel F254-precoated glass plates and RP-18 F254S plates (Merck, Germany). Dulbecco’s modified Eagle medium (DMEM), fetal bovine serum (FBS), trypsin-ethylenediamine tetraacetic acid 0.25%, streptomycin, and penicillin were obtained from Hyclone (Logan, UT, USA).
Plant Material
The whole plant of Isodon ternifolius (D.Don) Kudô was collected in Thai Nguyen province, Vietnam, and authenticated by Assoc. Prof. Dr Tran Huy Thai, Institute of Ecology and Biological Resources, VAST. A voucher specimen (IS20171001.HN) has been stored at the Institute of Natural Products Chemistry, VAST.
Extraction and Isolation
Dried, powdered whole I. ternifolius plant (4.0 kg) was extracted using MeOH (3 × 8 L) by refluxing to produce 380 g crude extract. The crude extract was suspended in water (800 mL) and successively partitioned, using n-hexane (4 × 1.5 L), EtOAc (4 × 1.5 L), and BuOH (4 × 1.5 L). An activity-guided fractionation study resulted in the n-hexane and EtOAc extracts being chosen for further study. The n-hexane extract (48.84 g) was chromatographed on an open silica gel column chromatography (CC) and eluted with n-hexane-acetone (20:1 to 0:1), yielding 10 fractions (CMH.1-CMH.10) according to their TLC profiles. Fraction CMH.4 (4.33 g) was further subjected to silica gel CC and eluted with n-hexane-EtOAc (8:1 to 0:1) to afford 11 subfractions (CMH.4-1 to CMH.4-11). Further purification of the subfraction CMH.4-2 (250 mg), using an RP-C18 CC, eluting with acetone:water (3:1), resulted in the isolation of compounds
Isodonspiroketone (1)
White amorphous powder.
UV λ max (MeOH): 238, 276, 336 nm.
IR (KBr) ν max 3384 (OH), 1702, 1632 (C = O), 1568, 1512, 1428, 1012 cm−1.
1H NMR (500 MHz, acetone-d 6) and 13C NMR (125 MHz, acetone-d 6), see Table 1.
HR-TOF-MS m/z 359.0743 [M−H]− (calculated for C18H15O8, 359.0756).
Cytotoxic Activity Assay
The cancer cell lines were maintained in RPMI 1640, which included L-glutamine with 10% FBS and 2% penicillin-streptomycin. Cells were cultured at 37°C in a 5% CO2 incubator. Cytotoxic activity was measured using a modified MTT assay. 18,19 Viable cells were seeded in the growth medium (100 µL) into 96-well microtiter plates (1 × 104 cells per well) and incubated at 37°C in a 5% CO2 incubator. The test sample was dissolved in dimethyl sulfoxide (DMSO) and adjusted to final sample concentrations ranging from 5.0 to 150 µM by diluting with the growth medium. Each sample was prepared in triplicate. The final DMSO concentration was adjusted to <0.1%. After standing for 24 hours, 10 µL of the test sample was added to each well. The same volume of DMSO was added to the control wells. On removing medium after 48 hours of the test sample treatment, MTT (5 mg/mL, 10 µL) was also added to each well. After 4-hour incubation, the plates were removed, and the resulting formazan crystals were dissolved in DMSO (150 µL). The optical density was measured at 570 nm. The IC50 value was defined as the concentration of the sample that reduced absorbance by 50% relative to the vehicle-treated control.
Supplemental Material
Supplementary Material 1 - Supplemental material for Identification of Cytotoxic Constituents from the Whole Plant of Isodon ternifolius
Supplemental material, Supplementary Material 1, for Identification of Cytotoxic Constituents from the Whole Plant of Isodon ternifolius by Minh Quan Pham, Thi-Thuy-Huong Le, Tien-Lam Do, Thi-Hong-Minh Pham, Quoc-Long Pham, Phi-Hung Nguyen and Dao-Cuong To in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Graduate University of Science and Technology under grant number GUST.STS.DDT2017-HH07.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
