Abstract
Highlights
Active compounds, biological activities (antimicrobial, antioxidant, anti-leishmanial) and cytotoxicity from aerial parts aqueous extract and essential oil of Artemesia herba alba (Asso) were resolute and supported by careful comparison with results reported in the literature; Antimicrobial efficacy of the above extracts especially against Enterococcus feacalis ATCC 29; Anti-leishmanial efficacy of the above extracts against L. infantum and L. major promastigotes; Antibacterial compounds of the aqueous extract were isolated by combining TCL/ Autobioautography techniques and identified by CG-MS analysis.
Introduction
In cultures around the world, aromatic plants have historically served as remedies, nutritional sources, and cosmetics. Nowadays, scientific research increasingly validates the medicinal use of these plants. Numerous studies aim to isolate and purify the bioactive compounds responsible for these effects and elucidate their modes of action. The genus Artemesia (Asteraeae) contains around 500 species, most of which are herbaceous perennials, biennials or annuals and are silvery-green, dark-green or blue-green in color. Four subspecies (chitachensis, kabilica, valentina, and herba-alba) and three varieties (densiflora Boiss., laxiflora Boiss., and tenuiflora Boiss.) had been distinguished within the species. 1 The species Artemisia herba alba (Aha), known as “Chih” in Arabic and “Armoise blanche” in French, is a dwarf silver-green shrub between 20 and 40 cm tall. Aha grows wild in arid areas of southern and Western Europe, the Mediterranean basin and the steppes of North Africa, the Middle East, and Western Asia. 2 They are characterized by their excessively bitter taste and pungent aroma attributed to the presence of sesquiterpene terpenoids and lactones. 3
This plant is known as “absinthe” in commemoration of its traditional use as a remedy for intestinal worms and, more broadly, it exhibits notable economic (ornamental use, beverages, and spices) and biological properties. This plant species, Aha, contains numerous biologically active compounds that offer various health benefits. Additionally, it holds significant concentrations of essential macro- and micro-elements, with values close to those recommended by the FAO. 4 It has been reported that non-pharmaceutical products, such as herbal teas prepared from this plant, could be effective in treating or preventing malaria and COVID-19. This species has been described in the literature as having excellent therapeutic properties: antimicrobial, antitumor, antihelminthic, insecticidal, antioxidant, antimalarial, anti-leishmania, anxiolytic, hepatoprotective, repellent, antimutagenic, antitubercular, anti-ulcerogenic, vasorelaxant, anti-hyperlipidemic, antidiabetic, analgesic, and antidepressant.3,5 These medicinal properties stem from its abundant variety of polar and non-polar phytochemical metabolites such as terpenoids, flavonoids, coumarins, lignans, miscellaneous compounds, phenolic acids, polyacetylenes, lactones, fatty acids, and fatty esters hydrocarbons. 5
Our investigation of Aha leaves aligns with several Sustainable Development Goals (SDGs) established by the United Nations. Indeed, this research supports SDG 3 (Good Health and Well-being) by exploring new potential treatments for infectious diseases, SDG 12 (Responsible Consumption and Production) through the sustainable use of local plant resources, and SDG 15 (Life on Land) by emphasizing the importance of preserving biodiversity and using natural resources sustainably. Several studies have demonstrated the promising therapeutic properties of Aha extracts, thus contributing to SDG 3.6,7 The sustainable production and use of this plant, as explored by Mohammad et al, 8 are essential for achieving SDG 12. Finally, Aha plays a crucial role in biodiversity conservation for the maintenance of ecosystems, thus contributing to SDG 15.6,8
Aha EOs predominant compounds are roughly typical of one or a group of countries. For example: Moroccan Aha are characterized by a predominance of α-Thujone, Sabinyl acetate, and β-Thujone; 9 Iran Aha by are predominance of β-Thujone, Camphor, 1,8-Cineole, and α-Thujone; 10 Tunisian Aha with a preponderance of β-Thujone, α-Thujone, Camphor, and Chrysanthenone;1,11 Jordan Aha by dominance in α-pinene, β-Thujone, 1,8-Cineole, Artemesia alcohol and Caryophyllene acetate 12 ; the one in Canada with trans-Sabinyl acetate, Myrcene, and trans-Thujone. 13 Algerian Artemesia herba-alba EOs corresponds to the camphor/ thujones/ chrysanthenone chemotype. They are characterized by a high percentage of Camphor (19% to 48%), 1,8-Cineole (5% to 20%), Chrysanthenone (5% to 22.5%), α-Thujone (1.0% to 26.7%), β-Thujone (1.65% to 9.3%) and Camphene (1.7% to 7.9%). 14
Phytochemical characterization of the genus Artemisia growing wild in different Algerian regions such as Boussaâda, Djelfa, Batna, Oum-Lebwaki, and Tiaret has been undertaken in previous studies.15–18 Until now, no study has identified any active substances from Algerian Aha demonstrating confirmed biological activities, nor has any study described the phytochemical characterization of Aha grown in the ‘Tebessa’ region of Algeria. This study aims to achieve the following objectives: (1) characterize the phytochemical compounds, evaluate toxicity, and assess specific biological activities of aerial part extracts (AE; EO) from wild Aha growing in the ‘Tebessa’ region, and (2) identify antibacterial molecules in the AE.
Experimental Part
Plant Material and Crude Extract Preparation
The wild-grown plant (Aha) was sourced from the ‘Tebessa’ region in northeastern Algeria (latitude 35.4; longitude 8.116) and was collected at the late flowering stage in November 2022. The species was identified by Professor Abderazzak Smaoui from the Extremophile Plants Laboratory, Biotechnology Center of Borj-Cedria (CBBC) in Tunisia. A sample model (AHA-CBBC-LSBA19022) was kept at the Bioactive Substances Laboratory of the same Centre.
The washed aerial parts of the plant were stored in a well-ventilated area away from direct sunlight for one week. Afterward, the dried plant material was cut into small pieces and ground in a blender. The obtained powder (20 g) was mixed with preheated distilled water (250 mL) and shaken at 150 rpm for 24 h at 25 °C (Edmund Buhler-KS-15). The solution was subsequently filtered using Whatman paper (No.4), and the resulting filtrate was centrifuged and then freeze-dried using an Alpha 1-2 LD Freeze Dryer apparatus. The extraction yield was then calculated.
Essential Oil Extraction and GC–MS Analysis
The dried herbal material (150 g) were hydro distilled in distilled water for 150 min (optimal time retained after a kinetic study (60, 90, 120, 150, and 180 min) using a Clevenger-type apparatus. The recovered EO was kept at - 20 °C in opaque glass vials until further analysis. The EO yield was calculated. GC–MS analysis was conducted using an HP 7890 (II) gas chromatograph and an HP 5975 mass spectrometer, both from (Agilent Technologies in Palo Alto, CA, USA). A capillary column, HP-5MS (30 m × 0.25 mm, 0.25 μm) film thickness was utilized. The split ratio was set at 30:1 and Helium carrier gas was used at a flow rate of 0.9 mL/ min at average velocity of 34.574 cm/sec. The oven temperature was programmed to increase from 40 to 280 °C at 5 °C/ min. Temperatures of each the injector, the transfer line and the quatrupole were 250 °C, 230 °C, and 150 °C respectively. Mass scan range from 50 to 500 m/z at an electron impact ionization of 70 eV, and run time was 37.333 min. EO identification was accomplished by comparing the recorded mass spectral with those archived in the Wiley 09 NIST (National Institute of Standards and Technology) 2011 mass spectral library within the GC–MS system data.
Phytochemical Compounds Analysis
Total phenolic content (TPC), flavonoids content (FC) and condensed tannins content (CTC) were determined using the reagents of Folin-Ciocalteu, aluminium chloride and vanillin, respectively. The protocols applied were based on our previous work. 19 The TPC was expressed as mg Gallic Acid Equivalents per gram dry weight (mg GAE/g DW) using the equation from Gallic acid calibration curve. FC and CTC were expressed as mg Catechin Equivalents per gram of dry weight (mg CE/g DW) using the equation from Catechin calibration curve. Analysis was performed in triplicate and the results are expressed as mean ± standard deviation.
Antioxidant Activity
DPPH Radical-Scavenging Assay
The procedure was performed according to that adopted by us. 19 Briefly, the concentration of the DPPH solution (0.04 mM in methanol) was adjusted so that its absorbance was between 0.7 and 0.8 at 517 nm. A sample (25 μL) at various concentrations in methanol was mixed with DPPH solution (975 μL) and left for 30 min at room temperature, protected from light. In the control tube, the plant sample was substituted with methanol solvent. After incubation, absorbance measurements were performed at 517 nm using a Ref spectrophotometer. The ability to scavenge the DPPH• radical was calculated using the formula: % Inhibition = [(A0 – A) /A0] × 100
Where A0: absorbance of the control solution and A: absorbance of the sample solution.
The result was presented into IC50 value, defined as the extract concentration to scavenge 50% of DPPH•. IC50 was calculated from the linear regression equation of the curve: % Inhibition as a function of extract concentrations. Butylated hydroxytolune (BHT), a synthetic antioxidant was used as a positive control. The result corresponds to the mean of three trials.
Ferric Reducing Antioxidant Power (FRAP) Assay
The reducing power of the extract was determined according to the method 20 with some modifications. Briefly, 0.4 mL of sample plant (different concentrations) were mixed with 1 mL of sodium phosphate buffer solution (200 mM, pH 6.6) and 1 mL of potassium ferricyanide (1%) [K3Fe (CN)6] solution. After incubation (50 °C, 20 min), 1 mL trichloroacetic acid (10%) was added to the mixture to stop the reaction and the whole mixture is centrifuged (3000 tours/ mn, 10 min). Afterwards, the supernatant (1 mL) was mixed with distilled water (1 mL) and Fe Cl3 solution (0.2 mL at 0.1%) and left 30 min in obscurity before to measure the absorbance at 700 nm against a blank. The reducing power was determined according the formula: % RP = 100 [(As - Ab)/ Aa]
Where: Ae and Ab are the absorbance of the sample and the blank respectively.
The result was represented by the values of the effective concentrations (EC50) which gave an absorbance of 0.5 for reducing power. Ascorbic acid, a synthetic antioxidant was used as positive control. The result corresponds to the mean of three trials.
Total Antioxidant Capacity (TAC) Assay
This test is based on reducing Mo (VI) by the sample analyte at acid pH to phosphate/Mo (V) green in color and it was done by method. 21 A suitably diluted sample (0.1 mL) was added to 1 mLl of reaction solution (600 Mm sulphuric acid, 28 mM sodium phosphate (Na3PO4), and 4 mM ammonium molybdate). The mixture was then incubated in a boiling water bath for 60 min. Absorbance was measured at 695 nm against a blank tube. Te result was expressed as mg Gallic acid equivalents per gram dry extract (mg GAE/g DE).
Cytotoxicity Assay
The cytotoxicity of Aha extracts (EO or AE) was evaluated against murine macrophage cells (Raw 264.7) maintained in DMEM Medium (Dulbecco's Modified Eagle's Medium) enriched with Serum from Filtered Chicken (SVF) growth factor and 100 U mL−1 antimycotic and antibacterial solution as described previously. 22 Briefly, 2 × 105 cells /mL were incubated in 96 plates at 37 °C and 5% CO2 for 24. The culture medium was then gently removed and replaced with 100 μL of different concentrations of Aha extracts (31.25 to 1000 µg mL−1) and then, plates were incubated for additional 72 h under the same conditions. The supernatant was removed and replaced by 100 μL of MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] solution at 1 mg/mL in Phosphate-Buffered Saline buffer (PBS) then incubated for 4 h at 37 °C. After discarding the supernatant, the resulting MTT crystals were dissolved in dimethyl sulfoxide (DMSO), and the absorbance was measured at 570 nm. The percentage of cytotoxic effect was calculated using the following formula: Inhibition % = [100 – (DOe/ DOt) / times 100]
Where: DOe and DOt refer to the absorbances of the diluted samples and the medium control, respectively.
Cytotoxic effect expressed as CC50 (concentration of the extract capable of reducing 50% of living cells) was resolute from the equation representing the linear dose (extract concentrations) /response (percentage inhibition %) curve. Result was expressed as the average of three trials.
Anti Leishmanial Activity and Selectivity Index
The antileishmanial activity of Aha EO was assessed following established procedure. 22 In brief, different concentrations of Aha EO (ranging from 15.62 to 1000 µg/mL) were introduced into 96-well culture plates containing Roswell Park Memorial Institute (RPMI)-1640 medium. The plates were then incubated for 24 h at 37 °C in 5% CO2 atmosphere, in the presence of 2 × 105 parasites/mL of both L. infantum and L. major promastigotes. Subsequent to 72 h incubation at 26 °C, the viability of the parasites was determined using a colorimetric assay with MTT reagent. Formazan crystals produced by viable parasites were dissolved using pure DMSO, and the optical density was measured at 570 nm using a spectrophotometer (Synergy, Bioteck). IC50 value was calculated by fitting a sigmoidal regression to a dose–response curve using Graph Pad Prism™ (version 6.0 for Windows). Amphotericin B served as the drug control (0 −12.5 μg/mL). All measurements were conducted in triplicate. The selectivity index (SI) was also calculated as the ratio of the toxic concentration macrophage (CC50) to the IC50 parasite).
Antimicrobial Activity
Aha AE (or Aha EO) was tested against three grams (+) (Staphylococcus aureus ATCC 6538, Lysteria monocytogenes ATCC 19195), Enterococcus feacalis ATCC 29212), three gram (–) (Klebsiella pneumoniae ATCC 104725, Salmonella enteritidis DMB560, Echerichia coli ATCC 25922, and one yeast strain (Candida albicans ATCC 10231).
The agar diffusion method was employed. In brief, a few colonies from a 24-h fresh culture on Luria-Bertani (LB) medium were suspended in 10 mL of physiological water. The suspension was agitated using a vortex and then adjusted approximately to 107 CFU /mL turbidity (absorbance 0.5). The bacterial suspension was spread using a sterile cotton swab onto Petri dishes previously prepared with MH (Mueller-Hilton) medium for bacterial or with Sabouraud Dextrose (SD) medium for C. albicans. Afterward, wells were created in the agar into which plant extracts were added. The bottoms of the wells were coated with soft agar (8 g/L) to prevent sample dispersion. After drying, 10 μL of essential oil (equivalent to 10 mg) or 50 μL of extract were added to the wells and allowed to dry before incubation (24 h, 37 °C). 19 The presence of an inhibition zone around the wells indicates antimicrobial activity. The diameters of the inhibition zones were measured. Results are presented as the mean of three values ± standard deviation. The minimal inhibiting concentration (MIC) of the Aha extracts was determined by micro broth dilution assay as described previously. 23 Briefly twice diluted concentrations of the sample (from 31.25 to 2000 µg mL−1) were tested on cell suspensions (107 CFU/mL) in LB or SD medium. The plates are then incubated for 24 h at 30 °C. The extraction solvent (sterile-water) was used as negative control, while tetracycline and nystatin (30 μg/ disk) were used as positive controls for bacteria and yeast respectively.
Bioautography-TCL-guided Detection
Aha AE (7 μL) at an appropriate concentration was applied onto a silica gel plate (silica gel 60 F254 from Merck, Germany, coated with aluminum, layer thickness 0.20 mm) and allowed to dry. Subsequently, development was carried out in a glass chamber pre-saturated with a solvent mixture of butanol/ acetic acid/ water in a ratio of 4: 0.5: 1 (v/v/v). The bands were visualized under UV light at 254 nm and identified by calculating the retention factor (Rf). Fractions exhibiting antibacterial activity were detected using a method adapted from Grzelak et al 24 with some modifications. In brief, the chromatograms obtained from Thin-Layer Chromatography (TLC) were placed in square-shaped Petri dishes, with the silica gel side facing outward, that had been prepared in Advance. The TLC plates were gently covered with a soft nutrient agar (0.8% agar + 2% LB medium) that had been inoculated with a suspension of the strain showing increased sensitivity to Aha AE. The setup was then incubated overnight at 30 °C, and active fractions were identified using MTT (0.5%) reagent. Active fractions converted the MTT reagent into a violet formazan derivative, whereas non-colored zones indicated the presence of inhibition.
Antibacterial Compounds Identification
The TLC technique was performed under the same conditions by depositing 100 μL of Aha AE (100 spots, each 10 μL) onto the plate. After development, the active spots identified by their Rf values were scraped off and subjected to silica gel removal through successive washes with methanol. The recovered supernatant was concentrated by evaporating under a stream of nitrogen at room temperature. The resulting residue was then subjected to the silylation method. In summary, 100 mL of a derivatizing agent BSTFA (NO-bis trimethylsilyl trifluoro acetamide) was mixed with pyridine (100 μL) and the sample. The mixture was incubated in a water bath for 45 min at 70 °C. This procedure converts hydrophilic groups into corresponding silyl groups (-O-TMS), allowing for separation by gas chromatography.
Statistical Analysis
Mean and standard deviation calculation were statistically analyzed by SAS software (Version 8) using the Duncan test performed after analysis of variance (ANOVA).
Results
Essential Oil: Yield and Composition
The essential oil (EO) extracted from A. herba-alba (Aha) exhibited a light yellow color, a highly bitter taste, and a strong, pleasant aroma. The yield of the EO was 0.99% (Table 1).
Extraction Yield, Total Phenolic Content, Flavonoid Content, Tannin Condensed Content, Total Antioxidant Capacity (TAC), DPPH Scavenging Activity (IC50), and Ferric-Reducing Power (EC50) of Artemesia Herba-alba Aerial Parts.

All values are represented as mean ± standard deviation (n = 3), with the exception of yield.
TPC: Total Phenolic Content (mg Gallic Acid Equivalent per g of Dry Extract).
FC: Flavonoid Content (mg Catechin Equivalent per g of Dry Extract).
CTC: Condenced Tannins Content (mg Catechin Equivalent per g of Dry Extract).
GC-MS analysis identified 19 compounds representing 98.93% of the total oil constituents (Table 2, Figure 1). Oxygenated monoterpenes constituted the main fraction at 86.23%. Camphor was the major compound, comprising 51.14% of the total, followed by 1,8-Cineole (19.30%), Camphene (6.90%), Chrysanthenone (5.68%), and endo-Borneol (3.72%).

GC-MS profile of the essential oil from A. herba alba areal parts.
Essential oil Composition (%) of Artemesia Herba- alba Aerial Parts.

RI Lit (a): Retention index according to the literature 7 ; RI Lit(b): Retention index according to the literature (Adam and previous studies on Artemesia species). The bold numbers represent the major compounds.
Extract: Yield and Phytochemical Fraction
An economically promising yield of 14% was obtained with Aha aqueous extract (AE) (Table 1). The contents of total polyphenols (TPC), flavonoids (FC), and condensed tannins (CTC) from Aha AE were 45.08 ± 0.23 mg GAE/g DW, 16.32 ± 0.63 mg CE/g DW, and 8.82 ± 1.03 mg CE/g DW, respectively (Table 1).
Antioxidant Activity
Total antioxidant capacity (TAC) values for the Aha aqueous extract (AE) and essential oil (EO) were 59.14 ± 0.03 mg GAE/g DE and 10.21 ± 0.41 mg GAE/g EO, respectively (Table 1).
DPPH-scavanging activities of the Aha (AE) and the BHT standard were determined to have IC50 values of 88.4 ± 2.21 μg/mL and 10.6 ± 0.12 μg/mL, respectively. Additionally, the DPPH activity of the Aha (EO) showed an IC50 of 4245 ± 1.52 μg/mL, which is significantly lower compared to that of the Aha AE (Table 1).
In our investigation, the antioxidant activity of Aha (AE), assessed using the FRAP method, showed an EC50 value of 1004.32 ± 2.03 μg/mL, significantly higher than the 41.52 ± 0.22 μg/mL observed for the ascorbic acid standard. Additionally, the FRAP activity of Aha (EO) was noteworthy, with an EC50 value of 3124 ± 5.12 μg/mL (Table 1).
Antimicrobial Activity
The antimicrobial effects of Aha (AE) and Aha (EO) are summarized in Table 3. The inhibition zone diameter (IZD) for the EO varied as follows: 11 mm (against S. aureus) to 30 mm (against E. faecalis) for Gram-positive strains, 6 mm (against S. enteritidis) to 12 mm (against E. coli) for Gram-negative strains, and 20 mm for C. albicans. For the AE, the IZD ranged from 10 mm (against L. monocytogenes) to 16 mm (against E. faecalis) for Gram-positive strains, 6 mm (against K. pneumoniae and S. enteritidis) to 8 mm (against E. coli) for Gram-negative strains, and 14 mm for C. albicans. As a reference, Tetracycline and Nystatin at a concentration of 20 µg/mL exhibited inhibitory effects with IZD values of 17 mm and 28 mm, respectively. The lowest minimum inhibitory concentration (MIC) value, 0.0625 mg/mL, was observed for the EO against the E. faecalis strain.
Antimicrobial Activity of Artemesia Herba-alba Aerial Parts.

Agar diffusion method was used. IZD: inhibition zone diameter. MIC: minimum inhibitrice concentration.
(—): no antimicrobial activity. Values represent the mean of duplicate essay.
Cytotoxicity and Antileishmanial Activity
The cytotoxicity and antileishmanial activity results are summarized in Table 4. The aqueous extract (AE) and essential oil (EO) of Aha exhibited CC50 values of 686.52 ± 4.41 µg/mL and 134.3 ± 3.27 µg/mL, respectively.
Antileishmanial and Cytotoxic Activities of A. Herba-alba Aerial Parts.
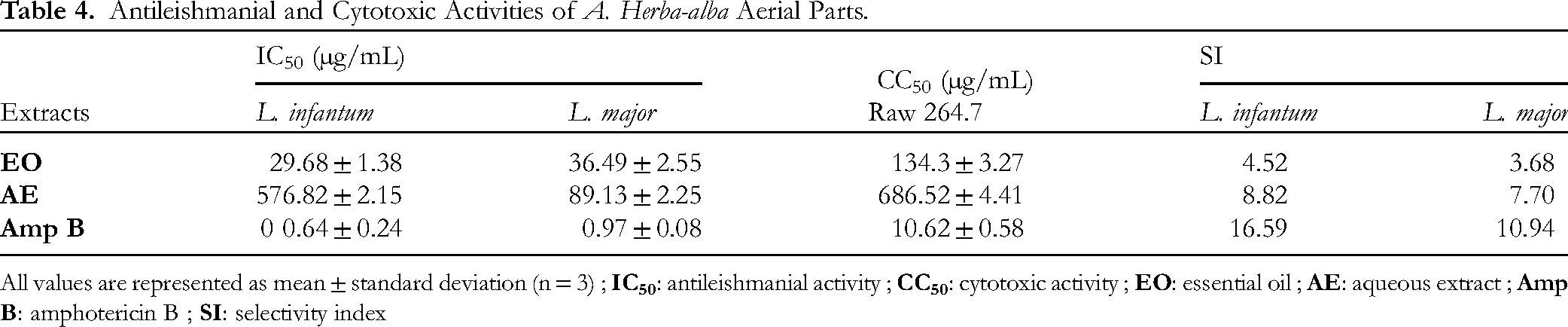
All values are represented as mean ± standard deviation (n = 3) ;
The Aha AE demonstrated dose-dependent antileishmanial activity against both promastigote forms. Specifically, the IC50 values for L. infantum and L. major promastigotes were 76.82 ± 2.15 µg/mL and 89.13 ± 2.25 µg/mL, respectively, with corresponding selectivity index (SI) values of 8.82 and 7.70. Aha EO also exhibits dose-dependent antileishmanial activity against both parasites. The IC50 values were 29.68 ± 1.38 µg/mL for L. infantum and 36.49 ± 2.55 µg/mL for L. major, with corresponding SI values of 4.52 and 3.68, respectively.
Antibacterial Compounds Identification
The TLC chromatogram demonstrating the separation of the Aha aqueous extract (AE) was exposed to the E. fecealis strain, which exhibited the highest sensitivity to the extract (refer to the detailed TLC/Autobiography methodology in the ‘Experimental’ section). This technique resulted in the identification of the suspected band exhibiting anti-E. fecealis activity with a retention factor (Rf) of 0.72 (Figure 2A). Following treatment with MTT reagent solution, the suspected band remained colorless (Figure 2B), confirming the presence of anti-E. fecealis compounds within it. A TLC plate was prepared under identical conditions, using an appropriate quantity of the extract. The silica gel area corresponding to the zone of inhibition was then recovered, washed, and concentrated. The obtained residue was subjected to derivatization (silylation) technique followed by analysis using GC-MS. The resulting chromatogram (Figure 3) revealed two peaks, accounting for 80.73% of the total area: one with an area of 32.8% and a retention time (RT) of 18.395 min, identified as palmitic acid trimethylsilyl ester; and the other with an area of 47.93% and a RT of 23.442 min, identified as oleic acid trimethylsilyl ester (Figure 4). These compounds corresponded to palmitic acid and oleic acid present in the extract.

(A): plate silica TLC profile of A. herba abla aerial part.
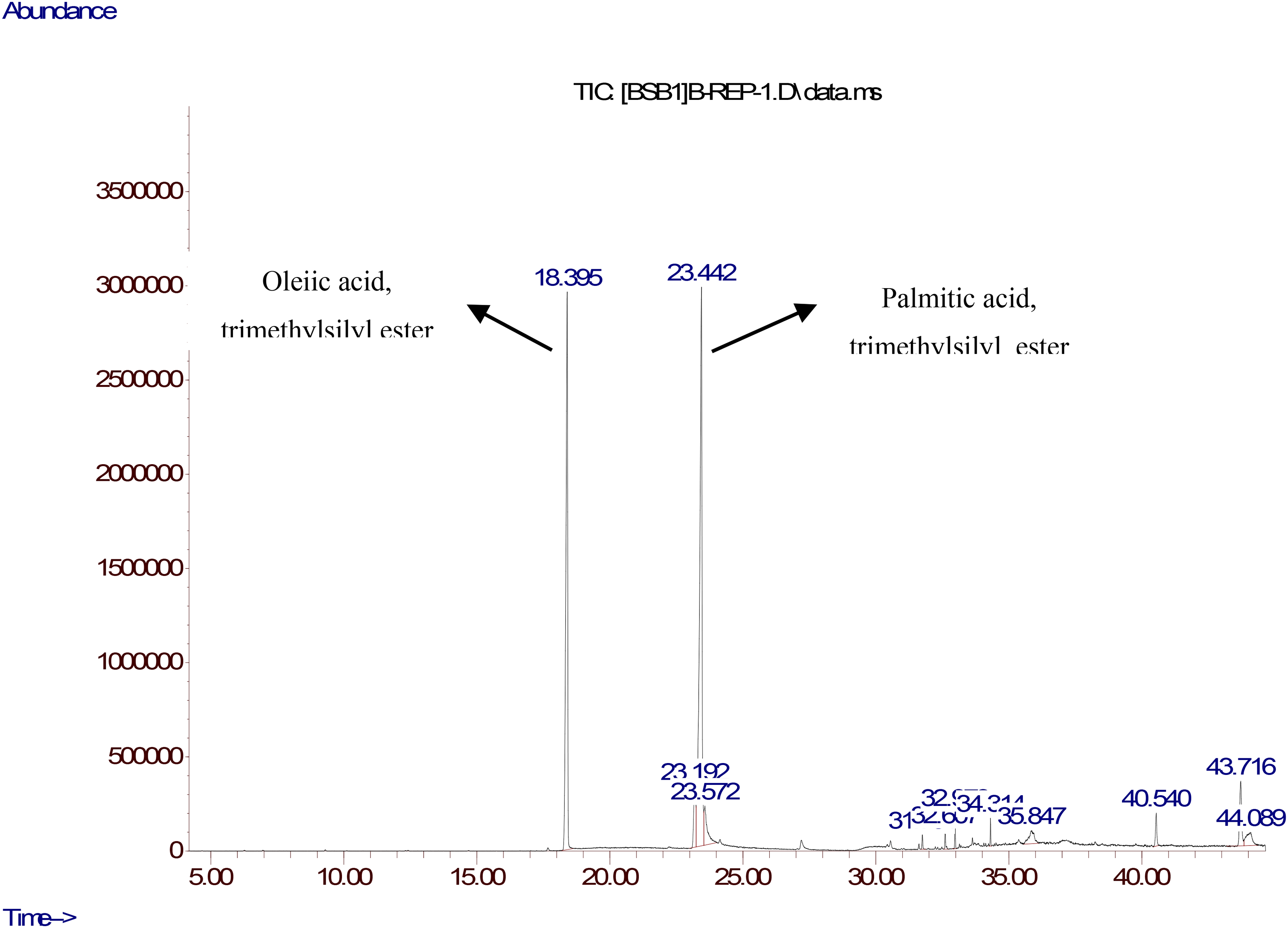
GC-MS profile of the antibacterial fraction detected from A. herba alba areal parts aqueous extract.
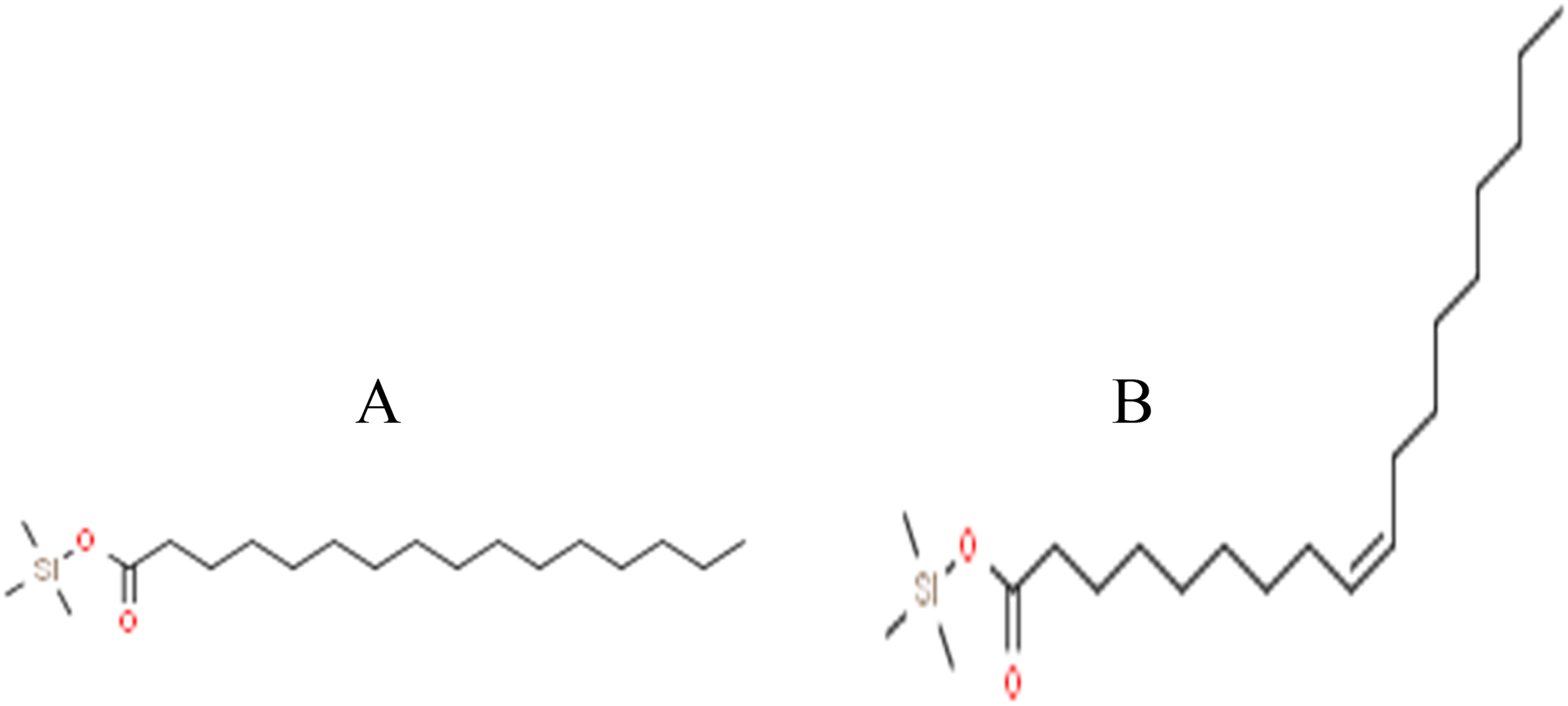
Structures(A) palmitic acid trimethylsilyl, (B) oleic acid trimethylsilyl.
Discussion
Essential Oil Extraction and GC–MS Analysis
The yield of 0.99% obtained for Aha EO is almost the same (0.95%) as that reported for Algerian Aha from the ‘Biskra’ region. 25 In comparison, Aha EOs from other Algerian regions such as ‘Boussaâda’, ‘Djelfa’, and ‘Batna’ were obtained with yields of 0.33%, 15 0.8%, 16 and 0.6% 17 respectively.
The predominance of camphor (51.14%) in Aha EO from the ‘Tébessa’ region contrasts with findings from other studies on Algerian populations of this species. For example, previous research reported cis-chrysanthenyl acetate as the major compound in the ‘Biskra’ region (25.12%), 25 while α-Thujone was dominant in the ‘Boutaleb’ (23.5%), ‘Bougaâ’ (28.1%), ‘Boussaâda’ (27.7%), 2 and ‘Bechar’ (48.7%) regions. 26 Additionally, other studies have identified uncommon compounds in smaller quantities within the Artemisia genus, such as ‘mesitylene’ in A.herba-alba27,28 and A. fragrans, 29 and ‘Psi-Cumene’ in A. absinthium, 30 which are often associated with solvents found in petroleum. The presence of Filifolid A has also been noted in A. fragrans. 29 Our study is the first to reveal the presence of ‘Psi-Cumene’ in the leaves of A. herba-alba, as well as ‘Thymyl angelate’, which has been previously reported in the essential oil of Foeniculum vulgare seeds. 26 However, the EO composition should have been consolidated with the calculation of retention indices (RI), but this was not done due to the lack of standards (n-alkanes) in the laboratory. These results suggest that regional and geographical environmental factors significantly influence the yield and composition of EOs in the same species, A. herba-alba, cultivated within the same country. Specifically, Aha species from the ‘Tébessa’ region demonstrate unique characteristics in both yield and EO composition.
Phytochemical Compounds Analysis
Several studies have demonstrated that the phenolic fraction yield extracted with water is more effective than when using pure solvents.31,32 For instance, Ayad et al reported an extraction yield of 12.14% for Aha using water. 29
Our results for TPC and FC values are lower than those reported for Aha AE collected from the ‘Tiaret’ region in Algeria, where TPC and FC were 52.44 mg GAE/g DW and 31.86 ± 0.8 mg CE/g DW, respectively. 32 Similarly, the TPC reported for Aha AE from southern Tunisia (133.33 mg GAE/g DW) was also higher than ours. 33 However, the TPC value reported for Aha methanolic extract from the ‘Kairouan’ region in Tunisia (27.65 ± 0.08 mg GAE/g DW) 33 was lower than what we observed. In addition, TPC and FC values of 19 mg GAE/g extract and 7.96 mg ER (equivalent rutin)/g extract, respectively, were indicated for A. vulgaris AE. 34 The CTC value reported for the methanolic extract of A. absinthium collected in the ‘Tozeur’ region in Tunisia (11.62 mg EC/g DW) was higher than ours. 35 It is well established that the content and composition of phytochemical constituents vary depending on the plant source, extraction solvent, and analytical method employed.
Antioxidant Activity
Pavithra et al 36 reported total antioxidant capacity (TAC) values of 198, 748, and 5 mg AAE (ascorbic acid equivalent)/g dry extract (DE) for water, methanol, and chloroform extracts of Artemesia. pallen, respectively. For Tunisian A. absinthium extracts, TAC values ranged from 21.38 to 85.24 mg GAE/g DW, depending on the sampling site. 35 In comparison, our study found that the TAC value for Aha EO was 10.21 ± 0.41 mg GAE/g EO, which is lower than the TAC value of 7.299 mg AAE/g EO reported for Artemesia aragonensis EO from Morocco. This suggests that the antioxidant activity of the Aha EO is less potent compared to that of A. aragonensis EO. 37 These differences in TAC values can be attributed to variations in plant species, geographical locations, and extraction methods used.
The DPPH radical scavenging assay is a widely used technique to assess antioxidant activity. The IC50 value reported for the methanolic extract of Tunisian Aha aerial parts (100 μg/mL) 1 was slightly higher compared to our result (88.4 ± 2.21 μg/mL). Additionally, the IC50 value reported for the ethanolic extract of Moroccan A. campestris aerial parts (355 μg/mL) 38 was four times higher than ours. However, the A. vulgaris aqueous extract exhibited the greatest scavenging potential, with an IC50 of 11.4 μg/ML. 32 The DPPH activity of our Aha EO (IC50: 4245 ± 1.52 μg/mL) was significantly lower compared to that of Moroccan Aha EO (1130 μg/mL) but was nearly similar to Tunisian Aha EO (5030 μg/mL). 39
Our results also indicate that the FRAP activity of Aha (EO) was lower than that of the Aha (AE) (Table 1). Comparing our results with those reported in the literature, the EC50 value for the methanolic extract of aerial parts from Tunisian Aha was found to be 372 µmol Fe2+/g. 38 Additionally, the reducing power of the leaf methanol extract from Aha grown in central Lithuania was reported as 60 µmol Trolox Equivalent (TE)/g DW. 40 EC50 values for hydro-methanolic extracts of wormwood from five regions in Tunisia ranged from 654.05 to 1366.45 μg/mL. 35 The EC50 values reported for Aha EO from Tunisia and Morocco were 79 µmol Fe2+/g and 870 μg/mL, respectively. 39
Polyphenols are recognized as the main secondary metabolites with therapeutic potential due to their antioxidant and free radical scavenging properties. They help reduce the risk of degenerative diseases by preventing the oxidation of macromolecules and mitigating oxidative stress. Additionally, the potential antioxidant effects of EOs are attributed to their richness in oxygenated monoterpenes and/or sesquiterpenes, which act as electron donors to scavenge free radicals. This explains the antioxidant activity demonstrated in our study, primarily attributed to the significant fraction of oxygenated monoterpenes content (86.02%). Furthermore, Camphor, known for its exceptional antioxidant activity, 41 constitutes more than half of all the identified constituents in our case (51.14%). Generally, EOs with potential antioxidant activity exhibit a high total quantity of terpenes, regardless of whether they are present in the majority or minority quantities.37,41
Antimicrobial Activity
Our results validate the significant antimicrobial activity of Aha extract and essential oil, aligning with findings from several previous studies.1,15–17,32,37,39,42 Notably, the inhibition zones produced by Aha extract and Aha EO were generally larger against Gram-positive bacterial strains compared to Gram-negative strains. This variation in sensitivity can be attributed to the structural differences between Gram-positive and Gram-negative bacteria. Gram-positive bacteria have a simpler cell wall composed of a single layer of mucopeptides, making them more vulnerable to antimicrobial agents. In contrast, Gram-negative bacteria possess a more complex outer membrane containing lipopolysaccharides and phospholipids, which hinder the diffusion of antimicrobial substances.
The antimicrobial efficacy of Aha EO is likely due to the presence of two major oxygenated terpenoids, camphor and 1,8-cineole, which are known for their antimicrobial properties. Additionally, the synergistic action of other terpenoids present in smaller quantities may also contribute to its overall antimicrobial effectiveness.29,35,43
Moreover, phenolic compounds, particularly flavonoids and phenolic acids, are recognized as the main antimicrobial agents in the extract Aha. Although we did not analyze the chemical composition of the Aha extract in our study, previous research has identified key compounds. Specifically, a study reported the presence of aglycone and glycosylated flavonoids, such as apigenin-6-C-arabinosyl-8-C-glucoside (isoschaftoside) and quercetin-rhamnoside-glucoside, as well as phenolic acids, with 1,4-dicaffeoylquinic acid being the predominant compound in the methanolic extract of Aha aerial parts collected from the ‘Karouane’ region of Tunisia. 1 We had hoped to determine the composition of our Aha extract, as was done for the essential oils (EO), in order to compare it with those of Aha samples collected from different locations. However, this could not be achieved due to the unavailability of the LC-MS instrument in the laboratory.
The significant antimicrobial activities demonstrated by both polar and non-polar extracts in our study highlight the potential applications of Algerian A. herba-alba in the food, pharmaceutical, and cosmetic industries. These findings are particularly relevant to SDG 3, as they suggest opportunities for developing new, effective treatments derived from natural products.
Cytotoxicity and Antileishmanial Activity
Our cytotoxicity results for Aha AE (Table 4) indicate that the extract is considered safe and non-toxic. According to the literature, the cytotoxicity of an extract is categorized based on CC50 values as follows: highly cytotoxic (CC50 < 100 µg/mL), moderately cytotoxic (100 ≤ CC50 ≤ 500 µg/mL), lowly cytotoxic (500 ≤ CC50 ≤ 1000 µg/mL), or non-cytotoxic (CC50 > 1000 µg/mL). 31 In contrast, moderate cytotoxic activities have been reported for the EO and methanolic extract of Aha aerial parts, with CC50 values of 160 µg/mL and 131.5 µg/mL, respectively, against murine macrophage cells.43,44
The potential antileishmanial activity observed could be attributed to the major compound “Camphor,” which constitutes 51.14% of the essential oil and is well-known for its antileishmanial properties.45,46 However, Aloui et al, 43 reported lower leishmanicidal activity (IC50 = 68 μg/mL) against L. infantum promastigotes, which could be related to the moderate proportion of “Camphor” (36.82%) in their study. 43 In contrast, essential oils from Ethiopian A. absinthium, which is also rich in Camphor, demonstrated high effectiveness against both L. aethiopica and L. donovani promastigote forms, with a MIC value of 0.156 µg/mL. 40 This variability in results can be attributed not only to differences in the composition of essential oils but also to the varying sensitivity of the isolated parasite strains. For example, the Camphor-rich essential oil (52.02%) of Artemisia annua leaves exhibited potent leishmanicidal activity against L. donovani, with an IC50 of 14.63 µg/mL. 47 However, major compounds alone are not solely responsible for such activity; minor constituents, even those present in trace amounts, also contribute by enhancing the overall effect through synergistic interactions with other compounds.
Antibacterial Compounds Identification
Fatty acids (FAs) synthesized by plants and algae, play a role in their defense against pathogens including multidrug-resistant bacteria. Several studies have demonstrated the significant impact of saturated fatty acids (SFAs) on both gram-positive and gram-negative bacterial strains. For instance, palmitic acid and stearic acid have exhibited efficacy against Methicillin-resistant S. aureus, B. cereus, S. aureus, and P. aeruginosa.31,48 It has been documented that these two SFAs, when encapsulated in liposome transporters, exert a bactericidal effect against multidrug-resistant S. epidermidis and Vancomycin-resistant E. faecalis. 49 Unsaturated fatty acids (uFAs) have also shown significant antibacterial activity against many pathogens and this property became significally efficacy when they were combined to antibiotic. Selvadoss et al 50 tested oleic acid Liposome's containing antibiotics against 32 strains of multi-drug resistant P. aeruginosa and found that MIC values were up to 4 times lower than those of free antibiotics. This explains why the liposomal oleic acid combination restored the sensitivity of these multi-resistant strains of P. aeruginosa to antibiotics. It has been shown that, unlike extracts, EOs can easily reach the cytoplasmic membrane of bacteria due to their lipophilic nature. As a result, antimicrobial substances disrupt the structure of polysaccharides, fatty acids and phospholipids, causing the bacteria to die.
Conclusion
This study represents the first to evaluate the phytochemical composition and biological activities of the aqueous extract AE) and essential oils (EO) of Algerian Aha collected from the “Bir Elaatar” region in Tebessa. Our investigation revealed that this plant is characterized by significant biological activities attributed to its richness in both polar (polyphenols) and apolar (terpenes) bioactive compounds, wich combat free radicals implicated in serious diseases such as cancer, antibiotic-resistant microbes, and leishmaniasis. Consequently, both the AE and EO of Aha aerial part growing wild in the “Tebessa” Algerian region show promising potential for industrial applications across various sectors: including agri-food, medical, pharmaceutical, and cosmetic.
Footnotes
Acknowledgments
The authors thank DrAbderrazak Smaoui (Laboratory of Extremophile Plants, Center of Biotechnology of Borj Cedria (CBBC) for his help in identifying the plant, and gratefully acknowledge the advice and support provided by the Head of the Bioactive Substances Laboratory, Biotechnology Center of Borj Cedria, BP 901, Hammam Lif, 2050, Tunisia. The present research study was supported by the Tunisian and Algerian Ministries of Higher Education and Scientific Research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
