Abstract
Bedbugs are blood-feeding arthropods that cause anemia in humans upon heavy feeding. Control measures are unsuccessful due to resistance to development, environmental pollution, and cost. There is a need to explore natural herbal-based phytochemicals to combat bedbugs. In this study, compounds from Comiphora africana resin were sequentially extracted, fractionated, identified, and evaluated against bedbugs as individual compounds and in combinations. The chemical constituents of the most active fraction were identified using Gas Chromatography-Mass Spectrometry. Results showed that the dichloromethane crude extract had the highest mean repellency (98.5% with an LC50 of 4.96 mg/L after 24 and 72 h of exposure time, respectively), similar to the positive control (neocidol). Column chromatographic separation of the dichloromethane extract yielded 9 fractions where “
Introduction
Bedbugs (Cimex lectularius Linnaeus) are a menace in many homes, hotels, public transport, and institutions, especially in developing countries. Bedbugs are blood-feeding ecto-parasites mainly obtaining their blood meals from humans, chickens, bats, and other domestic animals.1,2 Frequent and heavy feeding on hosts can result in significant blood loss, eventually leading to anemia. 3 They also impact negatively on people's health by causing severe physical and mental problems (skin rashes, allergic symptoms, and disruptions of sleep) resulting in negative social and economic effects. 4 Control measures for bedbugs by using insecticides have become futile due to their resistance to a number of conventional insecticides, which are also harmful to the environment, particularly dichlorodiphenyltrichloroethane (DDT). 5 Despite the growing concern about the negative environmental effects of synthetic insecticides, these control methods are costly. 5 Biological methods have shown some substantial success in combating bedbugs. Such methods include the use of insect pathogenic fungi, entomopathogenic bacteria, and oil formulations of biopesticide residuals. Spore formulations of Metarhizium anisopliae, 6 Aspergillus tubingensis, and Trichoderma harzianum 7 fungi showed high and rapid mortality against tropical bedbugs. However, there is a problem of specificity, the process is slow, and complete eradication is impossible and costly.6,7
This study sought to exploit the use of natural products as an alternative tool for control of bedbugs and other related arthropods, as they are believed to be environmentally friendly. Along the Kenyan coastal region, crude resin from the Commiphora africana A. Rich. tree is traditionally used by local people in the management of arthropods such as lice, bedbugs, fleas, and ticks. In this study, the use of C. africana resin which is normally considered as a waste after tree logging was exploited for maximum benefit. 8 In this study, we hypothesized that; (i) C. africana resin contains polar and non-polar phytochemicals that are active against bedbugs, and (ii) blending of active compounds identified from the highly potent fraction will lead to a synergistic effect on the responses of bedbugs.
In the current study, C. africana resin was sequentially extracted, and fractionated, the compounds in the most active fraction identified, and the extracts/fractions/selected blends evaluated for repellency and toxicity against bedbugs under laboratory conditions. This is the first study conducted to identify bioactive principles of C. africana resin and evaluate their insecticidal activity against bedbugs, which have become a major public health and medical hazard in Kenya. This study will lay a solid background in the development of an effective, cheap, safe, and sustainable bedbug control interventions by formulating an eco-friendly insecticide as an alternative to conventional synthetic insecticides.
Results and Discussion
Mean Repellency of Bedbugs on Exposure to C. africana Resin Crude Extracts
The repellency of bedbugs on treatment with C. africana resin extracts was evaluated and their means are summarized in Table 1. Generally, all the resin crude extracts exhibited significantly (P < .05) higher mean repellency against bedbugs as compared to the acetone solvent (blank control) (Table 1). The dichloromethane resin extract showed significantly (P < .05) higher mean repellency than the hexane, ethyl acetate, methanol, and water crude extracts (Table 1). The hexane and dichloromethane resin extracts exhibited higher mean repellencies of 98.5% and 100.0%, respectively, at 12 h exposure against bedbugs than the ethyl acetate (82.5%), methanol (75.0%), and water (72.5%) resin extracts (Table 1) due to the presence of high levels of nonpolar and mid-polar compounds which possess potential and beneficial biological properties, such as insecticidal and fungicidal, as previously reported by Ahmed et al. 9 At the 30-minute period of exposure of the bedbugs to the treatments, neocidol (positive control) demonstrated significantly (P < .05) higher mean repellency as compared to the hexane, dichloromethane, ethyl acetate, methanol, and water resin crude extracts. However, after 1, 6, and 12 h exposure of the bedbugs to the treatments, the dichloromethane resin extract had a significantly (P < .05) higher mean repellency than neocidol (Table 1). In addition, the hexane extract equally showed significantly (P < .05) higher mean repellency than neocidol after 12 h of exposure (Table 1). All the bedbugs were repelled (100%) on exposure to the dichloromethane extract and neocidol after 12 h exposure. Generally, the mean repellencies of bedbugs on exposure to the treatments were time-dependent. For instance, the mean repellency of bedbugs when exposed to all the extracts for 72 h was significantly (P < .05) higher than those exposed for 24 or 48 h (Table 1). A similar trend was observed by Loko et al 10 where Dinoderus porcellus mortality on yam chips mixed with powders varied with the dosage, the plant tested, and the exposure time.
Mean Repellency of C. africana Resin Crude Extracts in Acetone Against Bedbugs.
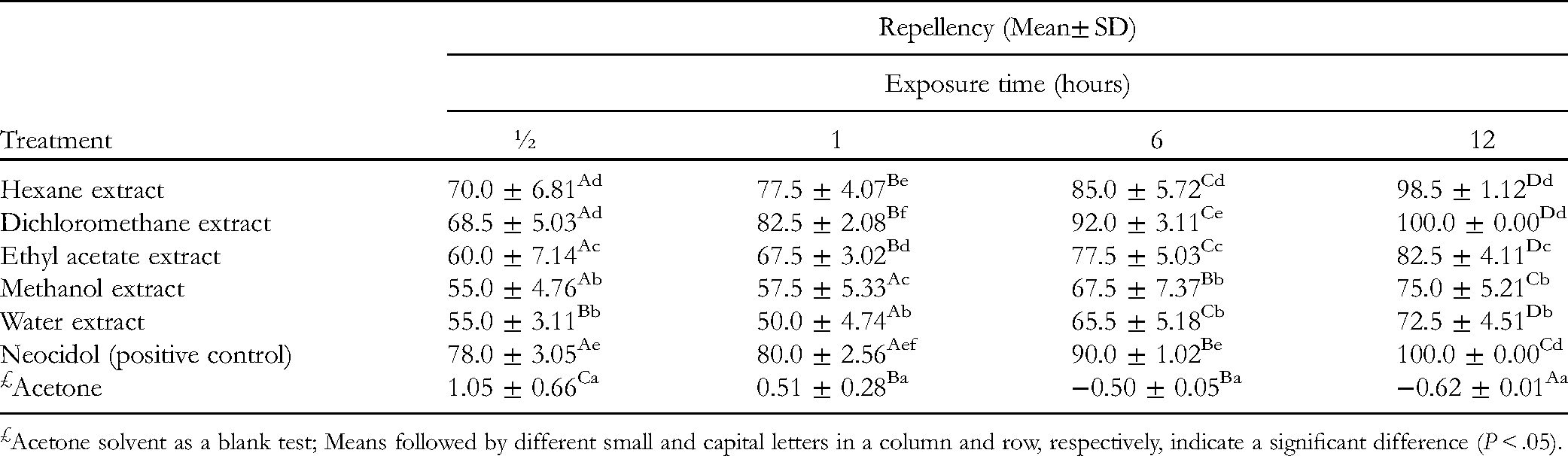
Acetone solvent as a blank test; Means followed by different small and capital letters in a column and row, respectively, indicate a significant difference (P < .05).
The mean repellencies of bedbugs to the dichloromethane resin extract were observed to be dose dependent (Figure 1) and increased with an increase in concentration to 1.25% (v/v), which was optimal (Figure 1). Notably, the mean repellency of bedbugs to 1.25% (v/v) dichloromethane resin extract was significantly (P < .05) higher than that of neocidol (Figure 1). In another study carried out by Swai, 11 the repellency activity of C. swynnertonii exudate against ticks depended on both the concentration of the extract and post-exposure time.

Mean repellency of bedbugs to different concentrations of dichloromethane crude extract in acetone. The error bars SD for percentage repellency.
Toxicity of C. africana Resin Crude Extracts Against Bedbugs
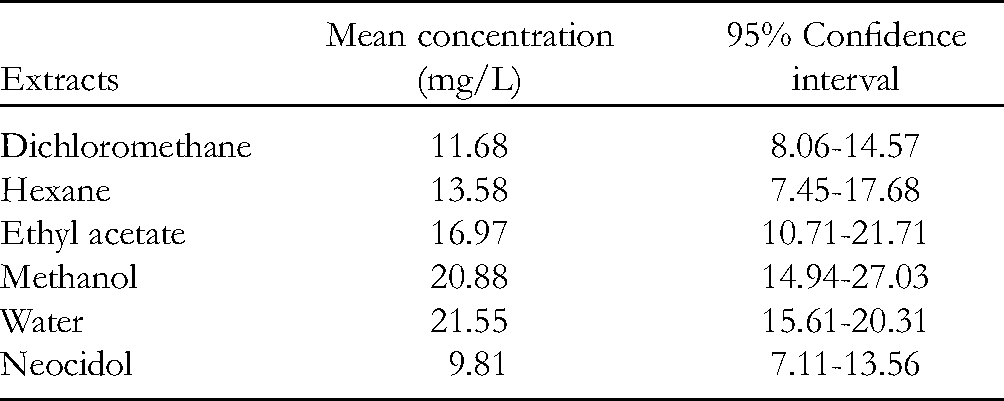
From the dose-response analyses on mortality of bedbugs on exposure to resin crude extracts at different concentrations, the LC50 values of the extracts after 24 h exposure were calculated; these are summarized in Table 2. The mean LC50 values of the extracts were significantly (P < .05) decreased with an increase in exposure time. For instance, the LC50 of the CH2Cl2 resin extract at 24, 48, and 72 h exposure were 11.48, 9.12, and 4.96 mg/L, respectively. This observation agrees with that of Ahmad et al, 12 where toxicity to adult pests (Tribolium castaneum and Rhyzopertha dominica) found in the stored grains increased with increasing extract concentrations of clove flower buds, chinaberry fruits, dill seeds, cumin seed, Datura stramonium, and Lantana camara leaves.
Mean LC50 of C. africana Resin Crude Extracts Against Bedbugs After 24 h Exposure.
The mortality of bedbugs on treatment with the dichloromethane crude extract of C. africana resin was evaluated at different concentrations with a 24-h exposure time, and the results are summarized in Figure 2. The 1.0% (w/v) extract was optimal in killing all the bedbugs (100% mortality), similar to that of neocidol. The bedbugs exhibited dose-dependent mortality when exposed to the dichloromethane resin extract.

Mean percentage mortality of bedbugs on treatment with dichloromethane crude extract of C. africana resin at varying concentrations. The error bars indicate SD for percentage mortality.
Mean Repellency of Bedbugs on Exposure to Chromatographic Fractions of the Dichloromethane Resin Extract
The repellency of bedbugs on exposure to the 9 obtained fractions of the dichloromethane resin extract (
Mean Repellency of Bedbugs on Treatment With Selected C. africana Dichloromethane Fractions.

Elution solvent: % hexane in ethyl acetate
Acetone solvent
Neocidol (a known bedbug pesticide-positive control).
Means followed by different small and capital letters in a column and row, respectively, indicate a significant difference (P < .05).
The repellencies of bedbugs on treatment with the fractions increased with an increase in time of exposure due to the saturation of the bedbug odor receptors with the test compounds.
14
For instance, the repellency of fraction
Toxicity of Chromatographic Fractions of Dichloromethane Resin Extract to Bedbugs
Exposure of bedbugs to the dichloromethane resin crude extract and its fraction
LC50 of Fractions of C. africana Dichloromethane Resin Extracts After 24 h of Exposure.

Means followed by different small letters in a column indicate a significant difference (P < .05).
Chemical Composition of Fraction FR7
The most bioactive oily fraction
Constituents of Fraction FR7 Identified by GC-MS.

Major constituents identified from fraction
Citronellyl formate (racemate mixture) isolated from Pelargonium graveolens essential oil was previously observed to kill various fungal strains (eg, Mucor mucedo and Aspergillus species) in a dose dependent manner. 19 Naphthalene and most of its derivatives, including 1,2-methoxynaphthalene derivatives, showed high toxicity levels to Coptotermes formosanus (Formosan subterranean termite) where the substitution of the naphthalene moiety significantly altered the toxicity, speed of action, route of penetration, volatility, and consequently the residual activity. 20 Triazine derived herbicides (eg, atrazine, simazine, cyanazine, and hexazinone) were also previously reported to be capable of potentiating the toxicity of organophosphate insecticides. In most cases, it was observed that 100 µg/L of triazine was necessary to significantly increase organophosphate insecticide toxicity, and higher concentrations of triazine caused a greater degree of potentiation. 21
Repellency of Selected Individual Compounds from Fraction FR7
Selected constituents of the most active fraction (FR7) of the CH2Cl2 resin extract identified from the GC-MS were evaluated for repellency against bedbugs and the results are summarized in Table 6. Generally, all the selected compounds showed significantly (P < .05) higher mean repellency in comparison to that of the acetone solvent (Table 6). However, these compounds elicited significantly (P < .05) lower repellency than neocidol. Of all the constituents, cedrol had a significantly (P < .05) higher mean repellency (Table 6). It was also observed that there was an increase in mean repellency of bedbugs with an increase in exposure time. For example, exposure of bedbugs to cedrol for half an hour and 12 h showed mean repellency of 64.0 ± 5.1% and 83.00 ± 3.4%, respectively (Table 6).
Repellency of Bedbugs on Treatment with Selected Constituents of Fraction FR7.

Acetone solvent as a blank test; Means followed by different small and capital letters in a column and row, respectively indicate a significant difference (p < 0.05).
Cedrol is also known for its toxicity and repellency to several insects of human importance including cockroaches, beetles, clothes moths, ants, 18 and common bedbugs. 21 This result also agrees with a previous study by Flor-Weiler et al 22 who reported 100% repellency with 50% cedrol against red imported fire ants and ticks.22,23 The mechanism of action of essential oils in insects remains unclear, but some studies have indicated that these compounds may be neurotoxic, 24 interfere with growth 25 and maturation, 26 and reduce reproductive capacity. 27
The selected compounds of the FR7 fraction showed varying bedbug mortality levels. All the selected compounds showed lower mortality as compared to neocidol, indicated by the significantly (P < .05) higher LC50 values (Table 7). Among the tested compounds, cedrol showed the highest mortality, and n-hexadecanoic acid the lowest, with mean LC50 values of 27.43 and 44.01 mg/L, respectively, after 24 h exposure (Table 7). It was observed that the mortality of bedbugs on exposure to the selected compounds increased with increases in exposure time, as indicated by the decreasing LC50 values (Table 7). The results agreed with the study conducted by Wachira et al 28 where the larvicidal activity of 6 plant extracts from Tithonia diversifolia and Ricinus communis against the malaria vector Anopheles gambiae increased with the exposure time. The results also agreed with a report by Ayalew. 29 In his studies, the insecticidal activity of Lantana camara oil extract against maize grain weevils was observed to increase with exposure time. The study by Bezuneh et al 30 on Prosopis juliflora leaf extract against Macrotermes spp (Isoptera: Termitidae) showed that the mortality of termites increased with increased concentration and time of exposure.
Mean LC50 Values of Selected Constituents of FR7 After 24 h Exposure.

Means followed by different small letters in a column indicate significant difference (P < .05).
Mean Repellency of Selected Blends From Constituents of the Most Potent Fraction FR7
The repellency of bedbugs to selected blends was evaluated at a concentration of 1.25% (v/v), and the results are summarized in Table 8. As expected, the mean repellencies of the blends were significantly (P < .05) higher than that of acetone (Table 8). Generally, the mean repellencies of bedbugs on treatment with the various blends were significantly (P < .05) higher than their corresponding individual constituents, as indicated in Tables 6 and 8, respectively. A six-component blend comprised of 9-octadecenoic acid-ethyl ester, octadecadien-1-ol, citronellyl formate, cedrol, n-hexadecanoic acid, and 1,2-dihydro-6-methoxy-naphthalene showed significantly (P < .05) higher mean repellencies than any other tested blend. However, the mean repellency of the six-component blends was not significantly (p > 0.05) different from that of a two-component blend composed of citronellyl formate and cedrol (Table 8). Moreover, these 2 blends indicated similar mean repellencies to neocidol after half an hour of exposure (Table 8). It was also observed that in each of the blends tested, the mean repellencies of bedbugs significantly (P < .05) increased with an increase in exposure time (Table 8).
Mean Repellency of Selected Blends from Constituents of Fraction FR7.

The compound ID numbers 1, 2, 3, 4, 5, and 6 represent; hexadecanoic acid, 9-octadecenoic acid ethyl ester, citronellyl formate, octadecadien-1-ol, cedrol, and 1,2-dihydro-6-methoxynaphthalene, respectively.
£Acetone solvent; Means followed by different small and capital letters in a column and row, respectively indicate a significant difference (P < .05).
The higher mean repellency of bedbugs on exposure to the blends as compared to individual compounds is attributed to the additive/synergistic repellency effect of the active constituents. This is attributed to the fact that different classes of compounds have different active functional groups which bind to different odor receptors with the same phenotype and thus an increased repellency effect. This was also observed by Wachira et al 31 where a four-component blend comprised of delta-nonalactone, heptanoic acid, 4-methylguaiacol, and geranyl acetone reported significantly higher repellency effects against tsetse flies as compared to any other 2 or 3 component blends, and individual constituent compounds. From subtractive bioassays, delta-nonalactone was reported to have the highest contribution in the blend in repelling tsetse flies.
Mean Toxicity of Selected Blends From Constituents of the Most Potent Fraction (FR7)
The toxicities of identified individual compounds and selected blends from fraction FR7 were evaluated against bedbugs in varying concentrations and the results are summarized in Table 9. As expected, the mortality of bedbugs on treatment with the various blends was significantly (P < .05) higher than their corresponding individual constituent compounds, indicated by significantly lower LC50 values (Tables 7 and 9). Generally, the mortality of bedbugs increased with an increase in exposure time to the blends (Table 9). All the selected blends showed significantly (P < .05) lower bedbug mortality than neocidol, which is indicated by their significantly (P < .05) higher LC50 values as compared to that of neocidol (Table 9). The six-constituent blend (comprising 9-octadecenoic acid-ethyl ester, octadecadien-1-ol, citronellyl formate, cedrol, n-hexadecanoic acid, and 1,2-dihydro-6-methoxy-naphthalene) showed significantly (P < .05) higher mean mortality than all the other selected two-component blends, as indicated by its significantly (P < .05) lower LC50 value (15.06 mg/L at 24 h exposure, Table 9). Among the two-component blends tested, that of citronellyl formate and cedrol (LC50 = 25.74 mg/L at 24 h exposure) showed significantly (P < .05) higher bedbug mortality than any other two-component blend, as indicated by its significantly (P < .05) lower LC50 value (Table 9). The higher mean mortality of the blends than the individual compounds is a result of additive or synergistic effects of each bioactive compound in the blend. Cedrol showed higher insecticidal activities than any other individual constituent tested, and that it had the highest contribution to the overall response of the selected blends (Tables 7 and 8). The compounds identified in the oily fraction FR7 are in the same classes as those previously reported by Rattan 24 and Pavela. 32 Products that constitute a number of essential oils rich in monoterpenes, sesquiterpenes, and closely related secondary metabolites tend to be potential insecticides against insect pests such as bedbugs. The potential insecticidal activity of the overall blends was therefore attributed to the synergistic effects of the essential oils in the blends.
LC50 of Selected Blends from Constituents of Fraction FR7 After 24 h Exposure to Bedbugs.

The compound ID numbers 1, 2, 3, 4, 5, and 6 represent; hexadecanoic acid, 9-octadecenoic acid ethyl ester, citronellyl formate, octadecadien-1-ol, cedrol, and 1,2-dihydro-6-methoxynaphthalene, respectively. In each blend, the constituent compounds were mixed in equal proportions; Means followed by different small letters in a column indicate a significant difference (P < .05).
Conclusion
Results from this study have established that C. africana resin has high potency in repellency and mortality of bedbugs. The CH2Cl2 extract, fractions, and identified compounds can be applied in bedbug management and control upon further research on toxicology to humans and the entire environment. Considering that bedbugs have developed resistance against most insecticides and are, therefore, resurgent, a bio-pesticide product from this extract can be developed and used as an alternative in combating bedbugs. The identified compounds of the most potent CH2Cl2 extract of C. africana resin consisted of alkenes, alcohols, carboxylic acids, and esters. The order of abundance was 1,2-dihydro-6-methoxy-naphthalene > cedrol, 9-octadecenoic acid ethyl ester > n-hexadecanoic acid > methyl-10-trans12-cis-octadecadienoate > citronellyl formate. Among these identified compounds, cedrol had the highest repellency and mortality against bedbugs. There was an observed synergistic repellency and mortality effect of bedbugs on exposure to various selected blends as compared to their respective individual identified compounds. A six-constituent blend comprising 9-octadecenoic acid-ethyl ester, octadecadien-1-ol, citronellyl formate, cedrol, n-hexadecanoic acid, and 1,2-dihydro-6-methoxy-naphthalene reported the highest repellency and mortality on exposure to bedbugs than any other combination.
Materials and Methods
Adult bedbugs (C. lectularius) of mixed sexes were collected from selected infested hostels of Kenyatta University where there were no chemical treatments for a period of more than 3 months. They were carefully handpicked from the bed crevices and beddings, and kept in glass containers, each covered with wire gauze that effectively prevented their escape, but provided good aeration. Small pieces of sponge were put in the container to retain warmth and provide harborage where bedbugs aggregated and hid. They were taken to the Department of Zoology (Kenyatta University) for rearing and multiplication. C. lectularius confirmation was carried out by a taxonomist in the National Museums of Kenya where a sample was deposited for reference and assigned a voucher number (NMK/Siz/2018/IC-011).
Rearing of Bedbugs
Bedbug rearing was according to a protocol of Araujo et al 33 where a cube cage (45 × 45 × 45 cm) made of glass was designed and partitioned into 2 equal sections. Five mice were introduced into one section and 100 bedbugs in the other. Various strips of cotton cloths were put across the cage and taped in place to ensure that bedbugs could move freely and feed on the hosts adequately. The cage containing both bedbugs and mice was housed in a carton that was left open on the top side. Laboratory conditions were maintained at 28 ± 2°C and relative humidity of 78 ± 3%. Pieces of sponge and wood with sufficient cracks were used for bedbug harborage, and also provided sites for the rearing of eggs and development into adults. Bellies of mice were shaved to facilitate easy feeding of bedbugs. Adult bedbugs were removed for bioassay 7 weeks from the time they were introduced into the cage.
Solvent Extraction of C. africana Resins
One kilogram (1 kg) of dried C. africana resin was sequentially extracted in order of increasing polarity of the organic solvent (hexane, dichloromethane, ethyl acetate, and finally methanol), each for 72 h. This was followed by filtering through a muslin cloth of pore size 0.7 mm, followed by a Whatman filter paper No 1. Excess solvent from the filtrate was removed with a vacuum rotary evaporator. A total aqueous extract of 1 kg C. africana resin was also prepared and the solvent was removed by lyophilization. The crude extracts obtained were evaluated for repellency and toxicity against C. lectularius. The crude extract that demonstrated promising activity was subjected to column chromatography.
Column Fractionation of Dichloromethane Crude Extract
A 100 g of the most active crude extract was subjected to column separation (100 cm long by 6 cm internal diameter) using Kieselgel silica gel 60G (70 −230 mesh). Silica gel (450 g) was transferred to a 1-L beaker and 700 mL hexane was slowly added while stirring with a glass rod to make a slurry which was slowly packed in the column. Excess hexane was drained out of the column until its level was slightly above that of the stationary phase.
A 100 g of dichloromethane crude extract was thoroughly mixed with 100 g silica gel with a mortar and pestle. This powdered crude sample was carefully packed on top of the slurry in the column ready for separation. The sample was first eluted with hexane, then a slow gradient of hexane-ethyl acetate, and finally with ethyl acetate. The polarity of the solvent was further increased by adding 10% methanol in ethyl acetate so as to elute the most polar compounds from the column, as described by Zhang et al. 34 Fractions with similar profiles were pooled together, concentrated in vacuo using a rotary evaporator at 45 °C, and stored at 4 °C until further analysis.
Identification of Compounds From the Most Active Dichloromethane Fraction
Oil fractions from the column were evaluated on bedbug responses. From the assays, compounds present in the most active fraction were identified by Gas Chromatography - Mass Spectrometry (GC-MS) (Shimadzu QP 2010-SE, Japan) by comparing their retention indices (RIs) and the obtained mass spectra with those of the authentic standards from the NIST library. 35 Ultrapure helium was used as the carrier gas at 1 mL/min in a column (A BPX5: 30 m long; 0.25 mm internal diameter; and 0.25 µm film thickness). The GC program was: 50 ˚C (1 min); 5 ˚C/min to 250 ˚C (9 min) with a total run-time of 50 min. The constituents of the most active fraction were sourced from Sigma-Aldrich (Tsuschem, Germany) as > 98% analar grade.
Bioassay Tests
Repellency of Bedbugs on Treatment With C. africana Resin Extracts and Compounds
The crude extracts, oil fractions, standards, blends, identified compounds from the most active fraction, and positive and negative control materials were evaluated for repellency with the aim of identifying potentially effective repellents against bedbugs. Glass petri dishes of 90 mm diameter and 15 mm depth were used to evaluate the repellency of all the extracts and individual constituents as described by Ojianwuna et al. 36 A filter paper was folded in the middle to make 2 halves; one half was treated with each extract/compound/blend in acetone while the other half was left untreated (acetone solvent alone). The treated filter paper was introduced into a clean petri dish and the open end was covered with a fine mesh fabric to prevent the bedbugs from escaping and to provide aeration. Ten bedbugs were introduced in the middle and their movements were observed after ½, 1, 2, 6, and 24 h. After the exposure time had lapsed, the choices made by the bedbugs, and the numbers in each half (treated and untreated) were recorded. These assays were carried out at room temperature (25 ± 2 °C) with a relative humidity of 75 ± 2%. Percentage repellency was calculated as (C–T)/(C + T)*100 (where C and T represent the number of bedbugs in the untreated and treated halves of the filter paper, respectively).
Bedbug Mortality/Toxicity Test
Bedbug mortality counts were performed using the method described by Ojianwuna et al 36 where circular filter papers (Whatman No. 2) with a diameter of 90 mm were treated by spraying 2 mL of resin crude extracts with varying concentrations (0.0, 0.25, 0.50, 0.75, 1.0, 1.25, and 1.40% v/v) in acetone. The filter papers were air-dried for 30 min at room temperature and thereafter transferred into a clean petri dish. Ten bedbugs were introduced separately into the dishes with the treatments, control solvent (acetone), and neocidol (positive control). Mortality rates were recorded at 24, 48, and 72 h after exposure. A similar procedure was carried out for the fractions, constituent pure compounds, and blends, in triplicates. Every bioassay was conducted at room temperature (25 ± 2 °C) and relative humidity of 75 ± 2%. To avoid any spread of bedbugs or infestation, proper personal protective equipment (PPEs) was put on and muslin cloth was used to cover the petri dishes immediately after bedbug introduction. The number of dead bedbugs was counted and mortality was determined as a ratio of D/T (where D and T represent the number of dead bedbugs and the total number of bedbugs introduced, respectively), expressed as a percentage.
Data Analysis
Repellency and toxicity data were rank-transformed and subjected to Analysis of Variance (ANOVA) and the means separated by Student Newman-Keuls’ post-hoc analysis test using SPSS version 20 (Chicago, IL, USA) software. The average bedbug mortality data were subjected to probit analysis for dose-response analysis to estimate the LC50 of the crude extracts, fractions, standards, and blends at a 95% confidence limit. Values with P < .05 were considered statistically significant.
Footnotes
Acknowledgments
We appreciate the support received from Martin Murigi (Jomo Kenyatta University of Agriculture and Technology) in Gas Chromatography-Mass Spectrometry analysis; and Mr Protus Omondi (Biochemistry Department of Kenyatta University), Mr Eliud Muhoro and Mr Richard Odera from the Department of Plant Sciences of Kenyatta University for technical support in bioassay tests, bedbug rearing, and data analysis.
Compliance With Ethical Standards
The study followed the guidelines of animal care and husbandry as stated by the IUCAC committee of Kenyatta University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Kenya Forestry Research Institute (grant no. 2018/2019FY).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
