Abstract
The chemical constituents in the roots of 3 Cremanthodium campanulatum Diels (Asteraceae) samples collected in Yunnan and Sichuan Provinces, China, were chemically investigated. A total of 8 bisabolane-type sesquiterpenoids, 2 of which were new, and a new oplopane-type sesquiterpenoid, along with a new lignan were isolated and their structures elucidated based on spectroscopic analyses. This is the first report on the chemical study of C. campanulatum.
Plants in the Hengduan Mountains area can provide good materials for the study of plant diversity and evolution. We have been studying the inter- and intraspecific diversity of Ligularia species in this area using a combination of chemical and genetic analyses and have demonstrated various modes of diversity in many species. 1,2 The genus Cremanthodium (Asteraceae) is taxonomically very close to Ligularia, growing on high mountains (usually more than 4000 m in altitude) of the Tibetan plateau and adjacent areas. 3 Most Cremanthodium species are small in size 4,5 and it is challenging to collect sufficient amounts for chemical analysis. Hence, chemical studies have been limited to a few species, for example, Cremanthodium ellisii, 6 -10 Cremanthodium discoideum, 11,12 Cremanthodium potaninii, 13 and Cremanthodium pleurocaule. 14 Bisabolane- and oplopane-type sesquiterpenoids, triterpenoids, and other compounds have been isolated from these species. We have investigated chemical constituents of Cremanthodium helianthus, 15 Cremanthodium stenactinium, 16 Cremanthodium rhodocephalum, 17 and Cremanthodium lineare, 18 and bisabolane, oplopane, and eremophilane sesquiterpenoids have been isolated. These results suggested that the root chemicals in Cremanthodium are close to those in Ligularia. As part of our ongoing studies on the interspecific diversity of Cremanthodium species, we focused on Cremanthodium campanulatum Diels. There have been no reports on the chemical composition of this plant. We report here that C. campanulatum growing in Yunnan and Sichuan Provinces produces highly oxygenated bisabolane sesquiterpenoids bearing an epoxide in the cyclohexane ring as major constituents.
The roots of 3 samples collected in Yunnan and Sichuan Provinces (Table 1) were dried and extracted with EtOAc. The extracts were subjected to silica gel column chromatography and high performance liquid chromatography (HPLC) to isolate 2 new,
Localities of 3 Samples and Isolated Compounds.

Asterisks denote new compounds and underlines the major constituents.
aBorder between Jiulong and Kangding counties, Sichuan.
bXiangcheng County, Sichuan.
cShangrila County, Yunnan.
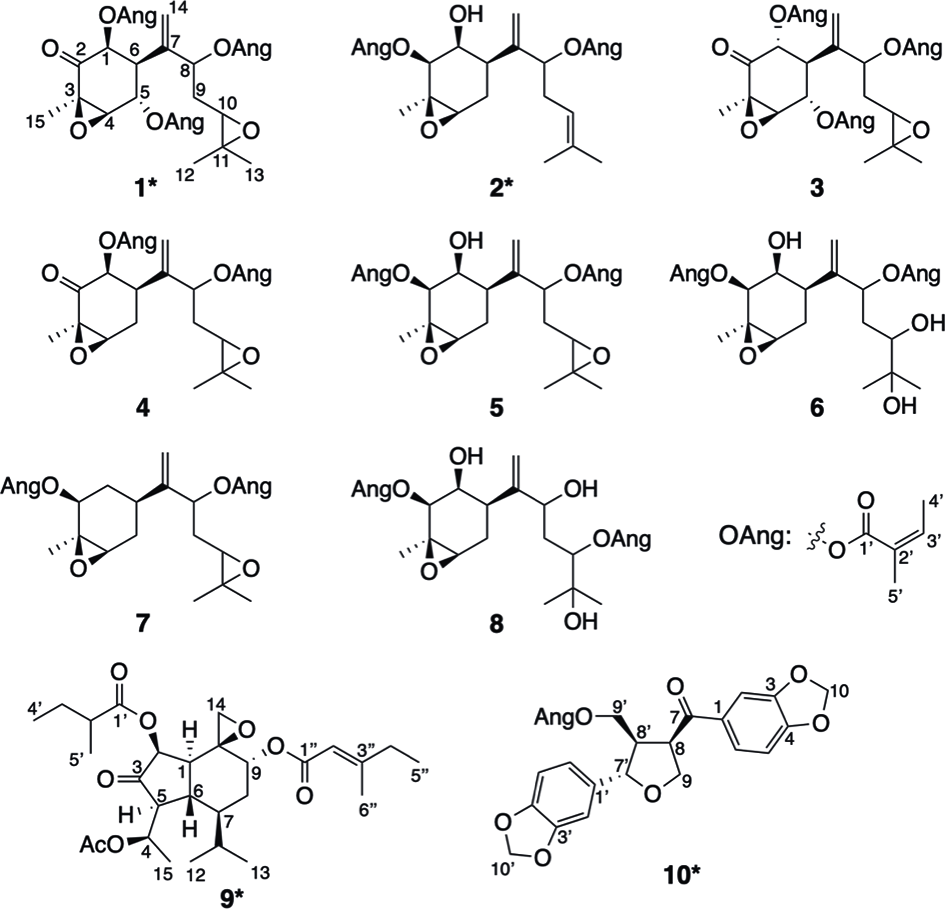
Compounds isolated from Cremanthodium campanulatum (asterisks denote new compounds).
The molecular formula of compound

Major 2D correlations detected for compound 1.
1H and 13C NMR Data of Compounds 1 and 2 (in C6D6).

Compound
The relative configuration of the cyclohexanone ring in
The molecular formula of compound

Major 2D correlations detected for compound 2.
The absolute configuration of the 6-membered ring in bisabolanes was determined as depicted in the formula by the positive Cotton effect (θ: +13 000) at 298 nm in the electronic circular dichroism (ECD) spectrum of compound
The molecular formula of compound

Major 2D correlations for compound 9.
1H and 13C NMR Data of Compounds 9 and 10 (in C6D6).
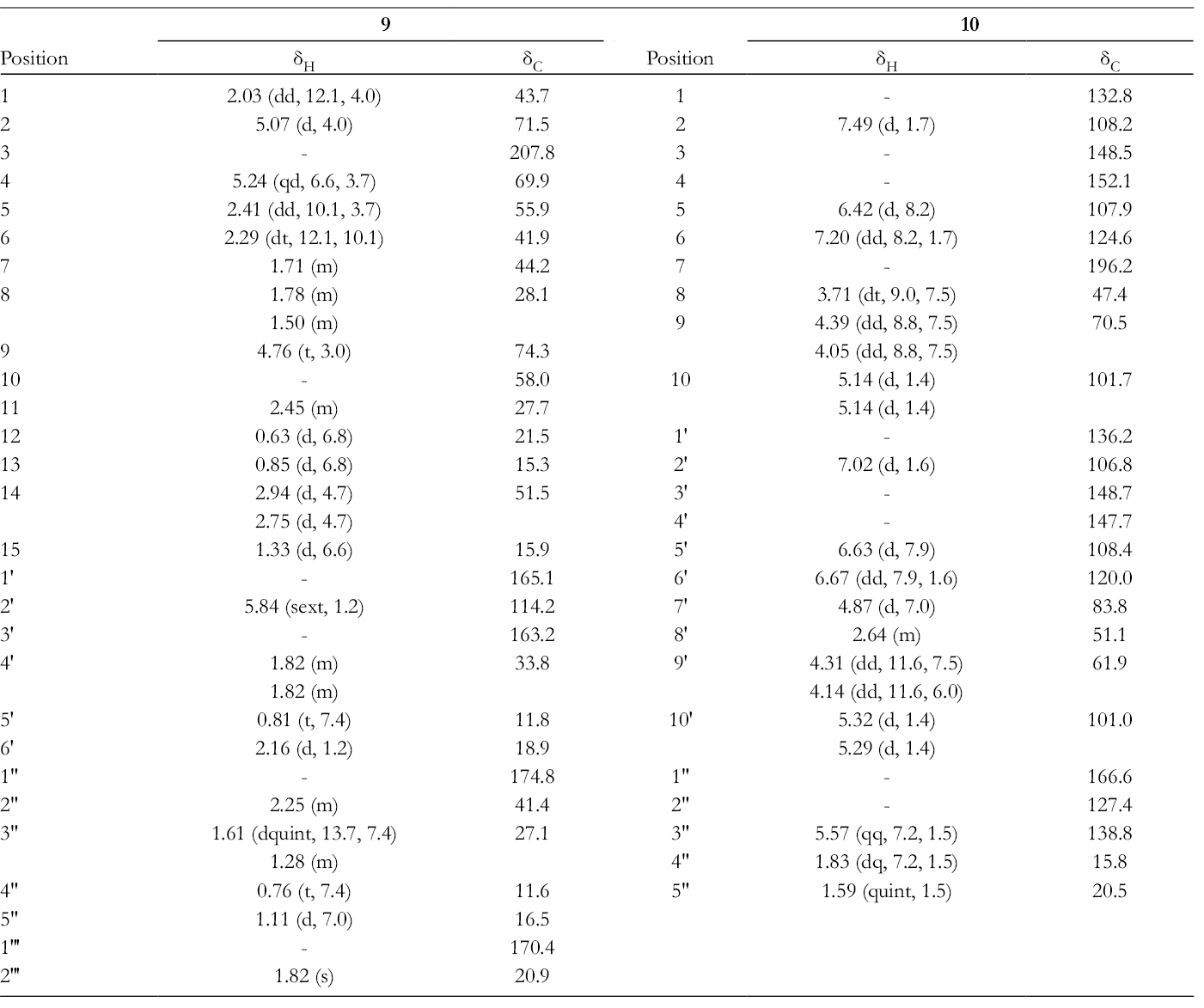
Coupling constants J 1,6 (12.1 Hz), J 5,6 (10.1 Hz), and J 6,7 (10.1 Hz) and NOEs between H-1/H-7 and H-5/H-7 suggested that the ring junction for rings A and B was trans, and that H-1, H-5, and H-7 were all on the same side. The 2-methylbutanoyloxy group at C-2 was determined to be β-orientation by detecting the NOE between H-1 and H-2. The acetate group should be at the C-4 (δC 69.9) position, because the NOE between H-4 and the acetyl methyl group was detected. Therefore, the 3-methylpent-2-enoyloxy group was indicated to be attached at C-9 (δC 74.3). The proton at C-9 resonated at δ 4.76 as a triplet indicating the equatorial nature of H-9. The NOEs between H-9/(H-8α and H-8β) and H-9/H-14 (δH2.75) were detected. Therefore, the 3-methylpent-2-enoyloxy group at C-9 was determined to be in the α-orientation from these observations (Figure 4). The configuration at C-4 of other oplopane compounds had been reported to be R by X-ray analysis, and their NMR data were used to establish its stereogenic center. 21 The proton resonating at δH 5.24 (qd, J = 6.6 and 3.7 Hz, H-4) was in good agreement with the previously reported oplopanes suggesting that C-4 was in an R configuration. 21 The stereochemistry of an epoxide at C-10 was deduced to be as shown in Figure 4, because NOEs between H-14a (δH 2.94)/H-2 and H-14b (δH2.75)/H-9 were observed. If the configuration at C-10 was the opposite, the NOEs as described above would not be observed. Oplopane sesquiterpenoids reported so far have a double bond at C-10/C-14 and those having an epoxide here were quite rare. 22,23 The absolute configuration was established as depicted in the formula from the negative Cotton effect (θ: –6400) at 320 nm in its ECD spectrum (in EtOH). 21,24-27
The spectroscopic data of compound

Major 2D correlations for compound 10.
Other compounds,
Finally, the cytotoxicities of compounds
Cytotoxicities of Compounds 1 to 8 [IC50 (µM)].

Experimental
General
Specific rotations and ECD spectra were measured on a JASCO P-1030 and a JASCO J-725 auto-recording polarimeter; IR spectra, on a SHIMADZU FT/IR-8400S spectrophotometer; 1H and 13C NMR spectra, on a Varian 500MR (500 and 125 MHz, respectively) spectrometer. Mass spectra, including high-resolution scans, were recorded on a JEOL JMS-700 MStation. Chemcopak Nucleosil 50-5 (4.6 × 250 mm) with a solvent system of hexane-ethyl acetate was used for HPLC (JASCO pump system). Silica gel BW-127ZH (100-270 mesh, Fuji Silysia) was used for column chromatography. Silica gel 60 F254 plates (Merck) were used for thin layer chromatography (TLC).
Plant Materials
Samples 1 to 3 were collected in August 2007 and 2009 at different locations in Sichuan and Yunnan Provinces (Table 1) and were identified by X.G., one of the authors. Voucher specimen numbers of samples 1 to 3 are 2007-56, 2007-120, and 2008-12, respectively (Kunming Institute of Botany).
Isolation of Compounds
The roots of each sample were cut into pieces and extracted with EtOAc. Compounds were separated using silica-gel column chromatography (hexane-EtOAc) and HPLC (hexane-EtOAc) to isolate each compound.
The roots of sample 1 (2007-56) (dry weight 1.8 g) produced an extract (32.2 mg). From this extract, compounds
The roots of sample 2 (2007-120) (dry weight 3.4 g) produced an extract (50.5 mg). From this extract, compounds
The roots of sample 3 (2008-12) (dry weight 3.6 g) produced an extract (89.5 mg). From this extract, compounds
1β,5α,8-Trisangeloyloxy-3β,4β,10,11-bisepoxybisabol-7(14)-en-2-one (1)
Fourier transform infrared spectroscopy (FT-IR) (KBr): 1718, 1647 cm−1.
MS chemical ionization (CI): m/z = 545 [M+H+], 445, 375, 345, 245, 83 (100).
HRMS-CI: m/z 545.2744 [M+H+], calcd for C30H41O9: 545.2750.
CD [θ] (nm) (EtOH): +7400 (245), –9800 (228), +360 (220).
1H and 13C NMR data in Table 2.
2β,8-Bisangeloyloxy-3β,4β,10,11-bisepoxybisabol-7(14)-en-1β-ol (2)
Compound deteriorated before measuring the specific rotation and 13C NMR.
FT-IR (KBr): 3491, 1713, 1647 cm−1.
MS-CI m/z = 432 [M+], 363, 333, 263, 233, 83 (100).
HRMS-CI: m/z 432.2502 [M+], calcd for C25H36O6: 432.2511.
1H NMR data in Table 2.
4-Acetoxy-10,14-epoxy-2β-(2-methylbutanoyl)oxy-9α-(E-3-methylpent-2-enoyl)oxyoplopan-3-one (9)
FT-IR (KBr): 1757, 1730, 1647 cm−1.
MS (CI): m/z = 507 [M+H+] (100), 447, 333, 231, 97.
HRMS-CI: m/z 507.2966 [M+H+], calcd for C28H43O8: 507.2958.
CD [θ] (nm) (EtOH): –6400 (320), +3300 (245), –5800 (230), +150 (215), –11 000 (206).
1H and 13C NMR data in Table 3.
9′-Angeloyloxy-3,4,3′,4′-bismethylenedioxy-9,7′-epoxylignan-7-one (10)
FT-IR (KBr): 3491, 1713, 987 cm−1.
MS (CI): m/z = 453 [M+H +], 435, 353 (100), 335, 203, 176, 149.
HRMS-CI: m/z 453.1554 [M+H +], calcd for C25H25O8: 453.1549.
1H and 13C NMR data in Table 3.
Cell Lines and Cell Culture
Human cervical cancer cell line HeLa and human promyelocytic leukemia HL-60 were obtained from the RIKEN BioResource Center (Tokyo, Japan). These cell lines were cultured in the minimum essential medium (MEM) (Gibco) supplemented with 10% fetal bovine serum (FBS) (v/v) (Nichirei Bioscience, Japan) and 1% penicillin-streptomycin at 37°C in a humidified atmosphere of 5% CO2 (v/v).
Cell Viability Assay
Cells were cultured in 96-well plates for 12 hours. The cells were then treated with compounds
Footnotes
Acknowledgments
We thank Prof Ryo Hanai, Rikkyo University, and Dr Takayuki Kawahara, Forestry and Forest Products Research Institute, for their helpful suggestions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by a Grant-in-Aid for Scientific Research from JSPS (21404009, 25303010 and No. 16K18897), the Japan–China Scientific Cooperation Program from JSPS and NSFC, and the Strategic Research Foundation Grant-aided Projects for Private Universities from the Ministry of Education, Culture, Sports, Science, and Technology, Japan.
