Abstract
This research presents complete data about phytochemical analysis and biological activities of a new dietary supplement for commercial use “Oligo Grapes” (OG), and its individual constituents: lyophilized pomace (LP), clay supplemented with red wine extract, lyophilized pomace ethanolic extract (PE), and red wine extract. OG, as a commercially available food supplement, has not been previously tested for its biological activity. Now we want to present new data about its phytochemical screening, antioxidant and antibacterial activities, and anticancer properties with respect to its cytotoxicity and effects on redox status in colon cancer cell lines. The tested extracts expressed strong antibacterial activity against Proteus mirabilis and Proteus mirabilis ATCC 12453, where the synergy of contents inside the supplement demonstrated a higher influence on the bacteria than its separately tested constituents. Among the investigated extracts, PE, as the extract with the highest phenolics concentration, had remarkable cytotoxic activity on HCT-116 and SW-480 colon cancer cells. Also, the treatments modulated redox status in the investigated cancer cells, by inducing oxidative and nitrosative stress, which could be one of the preferred mechanisms of the anticancer action. Based on the achieved antimicrobial and anticancer properties, there is a need for producing different food supplements and nutritional products originating from grapes. In this respect, the food supplement OG and its health benefits deserve scientific attention and further research.
Grapes, their extracts and constituents, and different grape products are known for their positive effects on people’s health, as well as their numerous biological activities. 1 Grapes contain various nutrient elements, such as vitamins, minerals, and numerous other phytochemicals. Among these, the most important are phenolic compounds, which mainly include anthocyanins, flavanols, flavonoids, and phenolic acids; anthocyanins are the main polyphenolics in red grapes. Their biological activities and human health benefits were already reported. 2 -4
For the last two decades, the connection between phenolic compounds and activity against pathogens has been a frequently investigated topic. The number of proven effects of flavonoids against a wide range of microorganisms is increasing. For example, proanthocyanidins from different berries inhibit uropathogenic Escherichia coli and oxacillin-resistant Staphylococcus aureus. 5 The activities of winery by products against foodborne and oral pathogenic bacteria, as well as fungi are related to their composition (flavonoid content). Grape phytonutrients are generally accepted as safe and proved to be good for treating a wide range of microbial-induced diseases. 6,7
Colorectal cancer is globally widespread and among the three most frequent cancers, with more than a million detected cases per year and with a high metastatic rate. 8 There is an increased interest in cancer prevention and potential treatment, especially in the knowledge that dietary habits might help in reducing the risk of the occurrence and progression of the disease. 9 Considering that cancer is a multifactorial disease, in addition to multiple genetic alterations, 10 an oxidative stress has a major role in the etiology of cancer. Cancer cells express a high level of reactive oxygen and nitrogen species (ROS and RNS), thus redox status is usually altered, and the antioxidant defense is attenuated. 11 Reactive species are aggressive free radicals produced in cells and their overproduction can lead to oxidative damage and development of various diseases, including cancer. 12 Numerous studies indicate that antioxidative properties of various plants lead to positive therapeutic effects, such as neutralizing oxidative stress by free radical scavenging. 13,14 Many studies confirm that grape products possess significant anticancer activity. 15 Also, antioxidative properties of grape and its products have been widely studied and generally related to antiproliferative effects on human colon cancer. 1,16,17 Therefore, the subject of this research is a dietary supplement for commercial use, “Oligo Grapes” (OG), which is a mixture of phenolic compounds (approved by Ministry of Health, Republic of Serbia, No515/04/5682/2013/10) obtained from Bionys Plus (Krnjevo, Serbia), as well as individual constituents of OG: lyophilized pomace (LP), clay supplemented with red wine extract (CW), lyophilized pomace ethanolic extract (PE), and red wine extract (WE). These products are specific in relation to their chemical composition, geographic position, various ecological factors, and extraction procedure, but their biological activities have not been investigated until now. Our research presents full data about the phenolic content of the new supplement OG and its constituents, as well as their biological activities. In accordance with these, the aim of this research was to determine the phenolics, flavonoids, and proanthocyanidins contents as potential bioactive substances in OG and its constituents, their DPPH radical scavenging activity, antimicrobial activity, as well as cytotoxic effects and redox status in human colon cancer cells (HCT-116 and SW-480), treated by these products.
Total phenolic, flavonoid, and condensed tannin contents were determined in OG and all its components (Table 1). The amounts of secondary metabolites were higher in OG, PE, and WE compared to the other two. The literature data for pomace and red wine confirm this finding, 18,19 while OG was examined for the first time. The correlation between antioxidant activity and the amounts of total phenols, flavonoids, and condensed tannins was complete, which was in accordance with other data. 19 The red wine used for production had 637.5 mg/L total phenols, 89.2 mg/L of which were flavonoids, and 630.9 mg/L proanthocyanidins. These results can be connected with the positive antimicrobial capacity of OG and its constituents. The DPPH assay showed that OG and WE had moderate antioxidant activity in vitro, while PE had very strong antioxidant activity (Table 1).
Phytochemical Characteristics of OG and its Components.
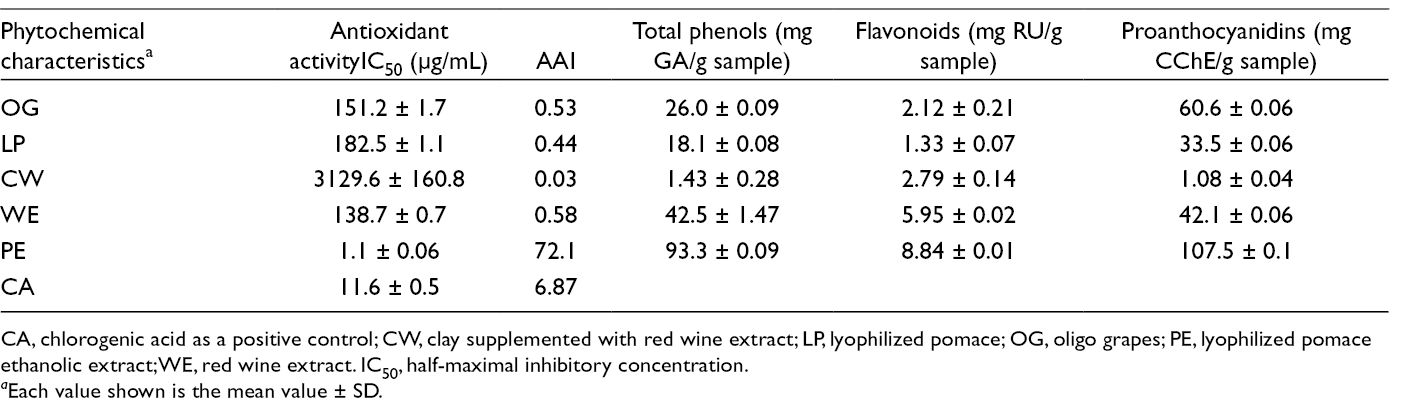
CA, chlorogenic acid as a positive control; CW, clay supplemented with red wine extract; LP, lyophilized pomace; OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract. IC50, half-maximal inhibitory concentration.
aEach value shown is the mean value ± SD.
Antimicrobial activity of OG and its constituents was determined by a microdilution method, and the results are presented in Tables S1 and S2, Supplementary Material. Fifteen strains of bacteria (7 Gram-positive and 8 Gram-negative) and 4 strains of yeast were used; 4 of the microorganisms were probiotics. OG supplement demonstrated a higher influence on bacteria in comparison to its constituents. The influence on yeasts was in all cases constant; it is important that no influence was found on probiotic Saccharomyces boulardii, while some activity against Candida albicans and Rhodotorula mucilaginosa was confirmed 10 (Tables S1 and S2 in Supplementary Material). Depending on processing conditions, grape pomace supercritical extracts showed the influence on C. albicans ATCC 14053. 7 It was reported that the antimicrobial effect of grape skin extracts was better on Gram-negative than Gram-positive bacteria, which was contrary to our data. 20 The potential of different red wines was investigated and the correlation between the contents, DPPH scavenging activity, and antimicrobial activity was determined. 21 In our research, PE (Table S2 in Supplementary Material) had a comparable value of total phenols, with the ethanolic pomace extracts from Pinot Noir and Pinot Meunier in the research of other authors. 22 With Gram-negative bacteria the influence of OG was prominent against Proteus mirabilis and Proteus mirabilis ATCC 12453, and in the range with the control antibiotic for the first mentioned strain. For constituents of this supplement, the activity against Proteus mirabilis and Proteus mirabilis ATCC 12453 was noticed with LP and PE, but was lower in comparison to OG (Tables S1 and S2 in Supplementary Material). This indicated the synergistic potential of the mixture contained in OG. A weaker influence was noticed on Pseudomonas aeruginosa and Pseudomonas aeruginosa ATCC 27853 by OG and PE. The results of other authors include minimum inhibitory concentrations (MICs) for E. coli ATCC 25922 and Pseudomonas aeruginosa ATCC 27853 at 1000 µg/mL. 23 As previously stated, we did not notice any significant influence on these bacterial strains. All the tested samples showed suppressing potential on Gram-positive bacteria. The most prominent influence was that of OG on the strains of Bacillus cereus and Bifidobacterium animalis subsp. lactis; it was in the same range as that of the control antibiotic (Table S1 in Supplementary Material). WE and PE acted against all the Gram-positive bacteria used; the former extract acted noticeably against bacilli, and the latter against bacilli and Staphylococcus aureus and Staphylococcus aureus ATCC 25923 (Table S2 in Supplementary Material). The results for Staphylococcus aureus strains are in accordance with the measured MICs for the NCTC strain of Staphylococcus aureus. 22 The pomace extracts obtained with 80% acetone were also examined. 24 Total phenol content was lower than in our investigation. The extracts tested by Xu et al did not influence E. coli and Salmonella typhimurium strains used, but the MICs for Staphylococcus aureus ATCC 29213 were from 40.6 to 250 µg/mL (MBC 75 – >250). 24 In our research, PE was the most prominent among the substances tested on Staphylococcus aureus strains (Table S2 in Supplementary Material). The lowest MIC reported by Oliveira et al 23 on Staphylococcus aureus ATCC 25923 was almost the same as in our research, but obtained with PE. Bacillus cereus is known to be a food contaminant; it was shown that wine consumed with food could inhibit this bacterial species and its spore germination in simulated gastrointestinal conditions. 25 In our investigation, all substances inhibited Bacillus cereus, especially OG (Table S1 in Supplementary Material), thus confirming that this supplement could be consumed with food. From 1993 to 1998, 12% of all food-borne disease outbreaks (with identified agent) in the Netherlands were caused by Bacillus cereus. 26
Cytotoxic Effects of OG, PE, and WE on HCT-116 and SW-480 Cell Lines, After 24 and 72 hours.

OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract. IC50, half-maximal inhibitory concentration.
Many studies by different research groups concluded that grape-based products are an excellent source of anticancer activity. 9 Different compounds, including phenolics, like flavonoids and proanthocyanidins, present in grape extract and products also were investigated. 1,4 The dietary supplement OG and its constituents with the highest antimicrobial activity (PE and WE) have been tested for their cytotoxic activity on HCT-116 and SW-480 colon cancer cell lines (Figure S1 in Supplementary Material). The results showed that all tested extracts decreased HCT-116 cell viability, generally in a dose-dependent manner, compared with the untreated (control) cells. The longer exposure time induced higher cell sensitivity and PE showed the best inhibitory effect on HCT-116 cell growth, compared with the other extracts. Decreasing of SW-480 cell viability was observed in all investigated treatments, but the highest applied concentrations of extract induced cell recovery after 24 and 72 hours. According to the results, the tested extracts demonstrate a cell-selective effect, namely, SW-480 cells were less sensitive to the treatments compared with the HCT-116 cell line. Based on the obtained inhibition of cell viabilities, the cytotoxic effects of the tested extracts were expressed by half-maximal inhibitory concentration (IC50) values. The American National Cancer Institute has the criterion for cytotoxic activity for the crude extracts of IC50 <30 µg/mL. 27 According to this criterion, PE showed remarkable activity on HCT-116 cells after 24 hours of treatment, while the other extracts were not cytotoxic to the investigated cell lines (Table 2). Although the other tested extracts did not show significant cytotoxicity, they certainly can be used for further investigations as a natural supplement in adjuvant therapy of cancer. The combination of drugs with dietary supplements can improve the efficiency of treatment and simultaneously reduce the side effects of chemotherapy. 28,29 Each grape extract or product showed different activity, which depends on the presence of biologically active components, including phenols such as flavonoids and protoanthocyanidins, which are the most active constituents in grape. These compounds are the best-known group of plant secondary metabolites with a large number of pharmacological activities, including their ability to inhibit cancer cell growth and their cytotoxic activity on different cell lines. 30,31 Our results showed correlations between phenol and flavonoid concentrations and the obtained cytotoxic effects of the investigated extracts. PE has the greatest phenol and flavonoid concentration and achieves the best cytotoxic effect, and OG has the lowest concentration and the weakest cytotoxicity. Much literature data confirm the correlation between phenol and flavonoid concentration and biological activity. 19,32 Also, anticancer effects of anthocyanins, present in the tested extracts, on colon cancer cells were observed, where they inhibit cell survival pathways and induce apoptosis. 9

Effects of OG, PE, and WE on O2.- concentration (related to the number of viable cells) in HCT-116 (a) and SW-480 cells (b), after 24 and 72 hours. Results are presented as means ± SE of 3 independent experiments. *P < 0.05 compared with values for control cells. OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract.
In addition to the investigation of in vitro antioxidant properties of OG, WE, and PE by DPPH assay, colon cancer cell lines were used as a live model system. The presence of superoxide anion radical, as a natural byproduct of normal metabolism of oxygen, is the representative evidence of oxidative stress in cells. The presented results show that all tested extracts increased O2.- concentration in HCT-116 cells after 24 hours, thus suggesting their prooxidative effects (Figure 1a). After a longer time of exposure (72 hours), OG and WE reduced O2.- concentration and acted as antioxidants (with the exception of the highest doses), while PE showed a prooxidative effect. The tested extracts, in general, induced slight antioxidative effects in SW-480 cells after 24 hours, while prooxidative effects were more evident after 72 hours, especially for WE (Figure 1b). The obtained parameters of redox status indicate different sensitivity of the two colon cancer cell lines to the investigated treatments. Namely, the changes in О2 . - concentration were less pronounced in SW-480 cells compared to HCT-116, indicating that SW-480 cells were more resistant to the impact of the investigated extract. Also, it is evident that PE caused more distinct increase in О2 . - concentration compared to the other extracts, which mediated the cytotoxic activity of this treatment (Figure S1 in Supplementary Material, Table 2). The cytotoxic activity as a result of О2 . - increase was documented by other authors. 33 OG and WE had lower prooxidant activity compared to PE after 24 hours, antioxidative effects in the lowest doses, while after 72 hours (Figure 1) they had no cytotoxic effect (Table 2).
The nitrite contents, as indicators of NO generation in aerobic conditions, were also determined in HCT-116 and SW-480 cells treated with OG, PE, and WE (Figure 2). The results show that the investigated extracts increased nitrite concentrations in HCT-116 cells, generally in a time-dependent manner, whereas PE induced the highest production of nitrites (Figure 2a). The treatments also induced a higher level of nitrites in SW-480 cells compared to the control, a better activity after 72 hours compared to 24 hours, when WE showed the highest effect (Figure 2b). Generally, the tested extracts increased nitrite levels, indicating nitrosative stress in the treated colon cancer cell lines, with a noticeably higher nitrite generation in SW-480 cells. The production of NO resulted mainly from arginine by nitric oxide-dependent synthases. Protein expression of inducible nitric oxide synthase (iNOS) was detected by immunofluorescence staining. The lower basal iNOS protein expression occurred in control HCT-116 cells compared to SW-480 (Figure 3). The increased iNOS expression was observed in the treated HCT-116 cells compared to the untreated control (Figure 3), while WE induced the highest iNOS expression. These data correlate with the obtained nitrite concentrations (Figure 2a). In SW-480 cells, only the OG treatment increased iNOS protein expression, in comparison with control cells (Figure 3). Based on these results, high nitrite contents in the treated SW-480 cells were generated endogenously by iNOS or some other ways that generated NO and RNS after exogenous induction. Nitric oxide can be produced independently of the L-arginine-NOS pathway, in a different reaction of nitrite reduction by a variety of enzymes and proteins, and under the influence of numerous NOS-independent factors. 34,35 The high nitrite level in human cells is often a consequence of consuming fruits and vegetables with a high level of nitrite and nitrate 36 ; grape may be an exogenous source for high production of nitrites. In view on this, possible explanations for our findings are the higher production of nitrite in biotransformation processes of the investigated grape products into cells, and consequently high production of RNS, as well as the specificity of the cell line in relation to its gene and protein expression. Also, it has been shown that grape seed extract can induce increase in NO level. 37

Effects of OG, PE, and WE on nitrite concentration (related to the number of viable cells) in HCT-116 (a) and SW-480 cells (b), after 24 and 72 hours. Results are presented as means ± SE of 3 independent experiments. *P < 0.05 compared with values for control cells. OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract.

Protein expression of iNOS in HCT-116 and SW-480 control and cells treated with OG, PE, and WE, after 24 hours. The images were taken using fluorescence microscopy at 60× magnification. Nuclei were stained blue (DAPI color); iNOS was stained green (second antibody Alexa 488). OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract; iNOS, inducible nitric oxide synthase.
Reduced glutathione is a tripeptide with many biological roles, including protective, against oxidative stress in cells. 38 The glutathione (GSH) content, as a first line of antioxidative defense in cells, was also measured in control and treated colon cancer cells (Figure 4). The treatments with OG, PE, and WE increased the concentration of GSH in HCT-116 cells, compared to the control, generally in a dose- and time-dependent manner (Figure 4a). The examined extracts either did not change or slightly decreased GSH concentration in SW-480 cells after 24 hours of treatment. However, after 72 hours of treatments they induced a slight increase in the GSH level compared to the control, except for PE (Figure 4b). The concentration of GSH was evidently higher in the HCT-116 cell line, being more sensitive than the SW-480 cells. The increased GSH level and antioxidant capacities were observed in the treated cells, as a response to oxidative stress. The tendency for the increase in GSH content in the treated cells suggests that phenols from plant extracts can enhance the antioxidant status by increasing the activity of GSH biosynthetic enzymes and the GSH level, 38 as a consequence of reactive metabolite production, such as О2 . - and nitrites. 39 The changes in GSH content typically occur as a consequence of antitumor compound effects in different cell lines. 40 For example, flavonoids can act as regulators of GSH content by stimulation of transcription of the gene involved in GSH synthesis. 41

Effects of OG, PE, and WE on GSH concentration (related to the number of viable cells) in HCT-116 (a) and SW-480 cells (b), after 24 and 72 hours. Results are presented as means ± SE of 3 independent experiments. *P < 0.05 compared with values for control cells. GSH, glutathione; OG, oligo grapes; PE, lyophilized pomace ethanolic extract; WE, red wine extract.
Generally, based on the results of redox status parameters in colon cancer cells, we can conclude that the investigated extracts induce oxidative stress by increasing O2 . - and GSH content, and nitrosative stress by increasing the nitrite concentration. The dual role of ROS and RNS in cancer cells (antineoplastic and proneoplastic) is well known. It depends on the type of tumor, cellular redox status, concentration, and duration of exposure of tumor cells to ROS and NO. 42 Many chemotherapeutic agents increase ROS and RNS production as a mechanism of action of their cytotoxic and proapoptotic activity on cancer cells. 43 Based on this mechanism some of the NO donors were used in variety of anticancer attempts. 44
The previously obtained results for the new dietary supplement “Oligo grape” have shown benefits for human health for which this supplement is formulated, but this research also confirmed some other activities. The ability of OG and its constituents to affect some biological activities has not been examined so far. Phytochemical analyses included the contents of phenolic compounds and their antioxidative activity by using DPPH method, which provided moderate results. Considering the pathogenicity of Gram-negative bacteria, the tested extracts expressed strong activity against Proteus mirabilis and Proteus mirabilis ATCC 12453, which suggests that the product can be a good addition when curing the diseases caused by these bacteria. The tested extracts demonstrated a cell-selective effect on colon cancer cell viability, where HCT-116 cells were more sensitive to the treatments compared to the SW-480 cell line. Of the investigated products, PE showed cytotoxic activity on HCT-116 cells, with the IC50 value of 29.32 µg/mL, which indicates that PE is a remarkable anticancer substance of natural origin. At the same time, PE is the extract with the highest concentration of phenolics compared to OG and WE. Generally, the obtained cytotoxic activity of the investigated treatments depended on their phenol content. The obtained results indicate that the OG supplement and its constituents have an ability to modulate the redox status of the colon cancer cells. OG and its constituents induce oxidative and nitrosative stress, by increasing O2 . -, nitrite, and GSH concentrations in the treated cells, which may be one of the preferred mechanisms of their anticancer action. Based on the investigated activities and obtained results, as well as the results of many other authors, there is a need for producing different food supplements originating from grapes. Therefore, the supplement OG deserves further research and promotion as a potent nutrient and healthy food.
Experimental
General
The supplement: OG (dietary supplement, No515/04/5682/2013/10, Ministry of Health of Serbia) and its constituents (WE, LP, and CW) were obtained from Bionys Plus (Krnjevo, Serbia).
Preparation of Extracts
More details can be found in Supplementary Material.
Phytochemical Analysis
Samples were analyzed by the Folin-Ciocalteu’s 45 and aluminum chloride 46 methods, both already described, 47 and the butanol-HCl method. 48 The total phenolic content was expressed as milligrams of gallic acid equivalents per gram of test substance (mg GAE/g of substance); the concentrations of flavonoids as milligrams of rutin equivalent per gram of test substance (mg of RU/g of substance); and the concentrations of condensed tannins (proanthocyanidins) as milligrams of cyanidin chloride equivalent per gram of test substance (mg of CChE/g of substance). The ability of the substances to scavenge DPPH free radicals was assessed by using the method described in Takao et al, 49 along with antioxidant activity index, 50 and the model previously described in Vasić et al. 47 Chlorogenic acid was used as a positive control.
Antimicrobial Activity
Test Microorganisms
Antimicrobial activity was tested against 19 microorganisms including 15 strains of bacteria (E. coli ATCC 25922, Staphylococcus aureus ATCC 25923, Pseudomonas aeruginosa ATCC 27853, Proteus mirabilis ATCC 12453, Bacillus subtilis IP 5832, Bacillus subtilis ATCC 6633, and isolates E. coli, Staphylococcus aureus, Pseudomonas aeruginosa, Proteus mirabilis, Salmonella enterica, Salmonella typhimurium, Bacillus cereus, Bifidobacterium animalis subsp. lactis, and Lactobacillus plantarum) and 4 strains of yeast: Candida albicans ATCC 10231, C. albicans (clinical isolate), Rhodotorula mucilaginosa, and Saccharomyces boulardii. The Institute of Public Health, Kragujevac provided all clinical isolates. The Microbiology Laboratory, Faculty of Science, University of Kragujevac provided a collection of the other microorganisms.
Suspension Preparation
The initial suspensions were adjusted to 0.5 McFarland’s standard. 51 For bacteria, the suspensions contain about 108 colony-forming units (CFU)/mL and for yeast 106 CFU/mL. Then, 1:100 dilutions of initial suspensions were prepared in sterile 0.85% saline.
Microdilution Method
By using the previously described 47 microdilution method with resazurin, 52 the MIC and minimum microbicidal concentration were established. The positive controls were tetracycline and amphotericin B, dissolved in nutrient liquid medium.
Cell Preparation
Human colon cancer cell lines (HCT-116 and SW-480) were obtained from the American Type Culture Collection. Cells were cultivated according to standard protocols. 29 More details about preparation and treatments are found in Supplementary Material.
Cell Viability Assay (MTT Assay)
Cytotoxic effects of OG, PE, and WE were determined by MTT assay; the protocol was previously described in detail. 53
Analysis of Redox Status Parameters in the Cancer Cell Lines
The concentration of superoxide anion radical (O2 . -) in the samples was determined by NBT assay; concentrations of nitrites – NO2 - (indicator of the nitric oxide — NO level) and GSH content were previously described in detail. 54
Protein expression of iNOS in HCT-116 and SW-480 cells was detected by an immunofluorescence staining protocol. 54 Cells were visualized by a Nikon inverted fluorescent microscope (Ti-Eclipse) at 600× magnification.
The results for all redox status parameters (O2 . -, nitrites, and GSH) were presented as nmol/mL in relation to the number of viable cells (based on data from MTT test).
Statistical Analysis
More details are found in Supplementary Material.
Supplemental Material
Supplementary data - Supplemental material for Phytochemical Evaluation, Antimicrobial and Anticancer Properties of New “Oligo Grapes” Supplement
Supplemental material, Supplementary data, for Phytochemical Evaluation, Antimicrobial and Anticancer Properties of New “Oligo Grapes” Supplement by Milena Milutinović, Sava Vasić, Ana Obradović, Aida Zuher, Milena Jovanović, Milijana Radovanović, Ljiljana Čomić, and Snežana Marković in Natural Product Communications
Footnotes
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (project No. III41010). The authors thanks to Bionys Plus, Krnjevo, Serbia for donating supplement and related components for testing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
