Abstract
Preparation of amides by biocatalyzed aminolysis reactions has greatly increased because these processes play an important role in the preparation of some pharmaceutical products and their intermediates. A highly efficient synthesis of malonic (
Dicarboxylic acid esters and their derivatives possess a myriad of applications in different academic and industrial sectors; they are important starting materials in the manufacture of polyamides and of di- and polyesters. Dicarboxylic acid monoamides are interesting synthons and intermediates for the preparation of biologically active amino acids, lactams, and other fine and pharmaceutical compounds by ammonolysis and aminolysis reactions. 1 In addition, the selective transformation of a unique functional group in a molecule presenting more complex functionality and avoiding side reactions is one of the most important tasks in the preparation of many compounds. 2
The development of sustainable and efficient methods for reaching chiral building blocks has become imperative for contemporary chemistry over the past decade. Biocatalysis and biotransformations are the most attractive routes to obtain a range of bioactive molecules using clean, easy, and eco-friendly procedures, and are the most targeted by the fine-chemical industry. 3
Enzymes can catalyze chemo-, regio-, and stereoselective reactions of building blocks and achieve the desymmetrization of diacids, diesters, and other symmetric compounds by modification of one functional group that eliminates one or more elements of symmetry of the substrate.
4
Particularly, the aminolysis of diesters is a process of great utility for the synthesis of nitrogenated organic compounds; in some cases, this process is useful for the preparation of mono amide esters by means of the desymmetrization of diesters, which are difficult to obtain by chemical procedures under benign conditions (Figure 1). In this context, the purpose of this work was to study the chemo-/regioselectivity of the lipase aminolysis desymmetrization of the diesters diethyl malonate (

Synthesis of monoamides (5) and (6) by desymmetrization lipase catalyzed aminolysis of diesters diethyl malonate (
Initially, the enzymatic aminolysis of diethyl malonate (
Lipase Catalyzed Synthesis of Monoamides From Diethyl Malonate (
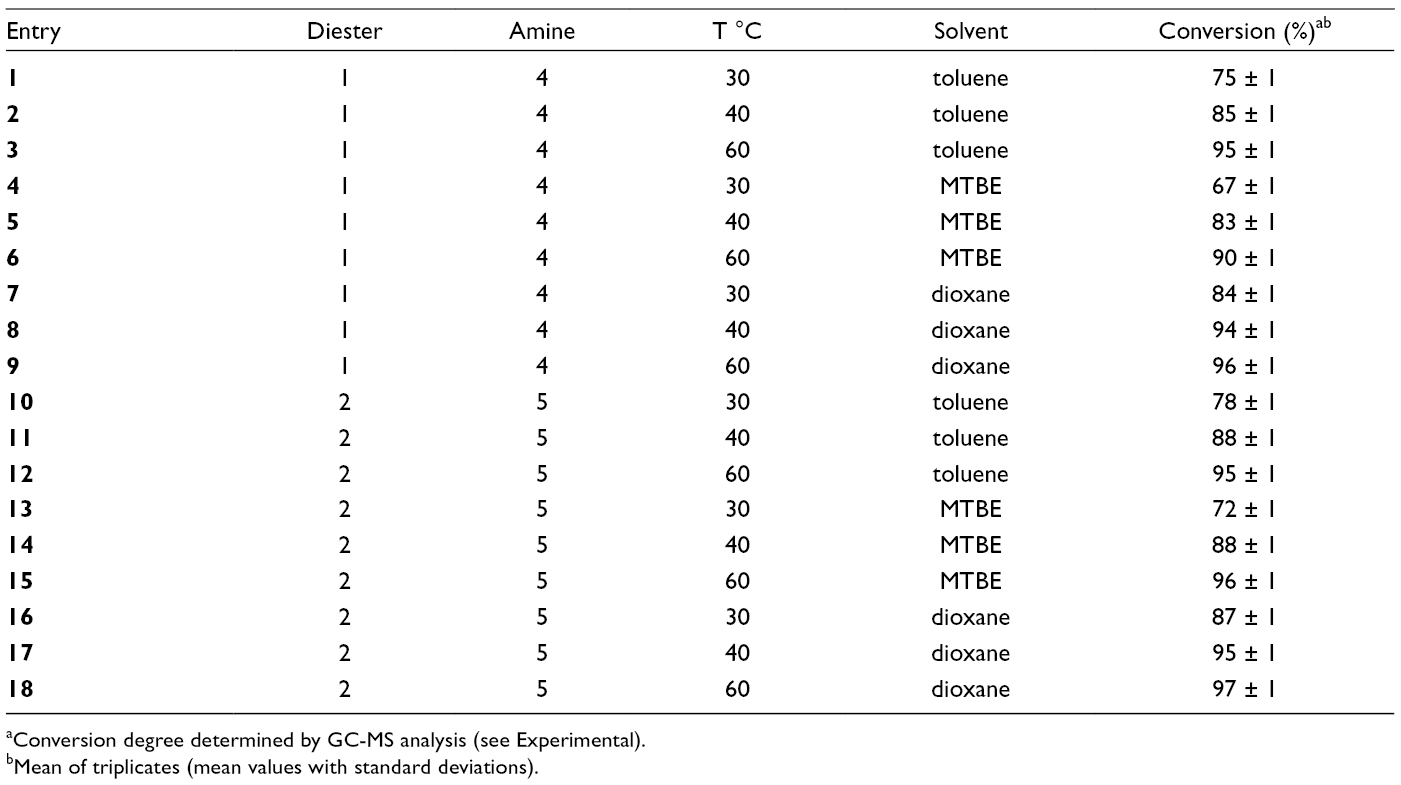
aConversion degree determined by GC-MS analysis (see Experimental).
bMean of triplicates (mean values with standard deviations).
As can be seen in Table 1, the mono amides (
Besides being an environmentally friendly and an economical alternative to traditional chemical synthesis, CaL-B as a biocatalyst has many important advantages and added benefits: mild reaction conditions, high chemo- and regio-specificity, and also catalyzes of enantioselective transformations of racemic mixtures of acids, esters, and amines yielding optically active amidoesters with excellent enantiomeric excess. In a previous study,
7
enantioselectivity was not observed when several racemic 2-hydroxy acid ester aminolysis reactions were tested by analysis of the corresponding Mosher’s derivatives; virtually only racemic 2-hydroxy amides were obtained. These results were in sharp contrast to the high enantioselectivity reported for a similar aminolysis process of regioisomeric racemic 3-hydroxy esters.
8
Because of these different results displayed by CaL-B, with a great differencehz. in the enantioselectivity of amidation between a racemic (R,S)-β-hydroxy ester and a racemic (R,S)-α-hydroxy ester as substrates leading to the corresponding amides, the study of the aminolysis reaction of the racemic (R/S) hydroxysuccinic acid methyl ester (or (R/S)-malic acid methyl ester) (

Monoamide (
Regioselective Monoamide Synthesis by CaL-B Catalyzed Aminolysis of Diesters (R,S)-Diethyl Malonate (

aConversion degree determined by GC-MS analysis (see Experimental).
bMean of triplicates (mean values with standard deviations).
After the aminolysis reaction of diester dimethyl malonate (
In conclusion, in this study C. antarctica B lipase catalyzed the aminolysis reaction of diesters (
Experimental
Lipases and Chemicals
Chemical reagents, solvents, recombinant CAL-B lipase (Novozyme SP-435, 7000 propyl laureate units/g solid enzyme), lipases from Candida rugosa, Rhizomucor miehei, Carica papaya, and Pseudomonas cepacia were all purchased from Sigma-Aldrich (Sigma-Aldrich Química S.A. de C.V., Mexico). Analytical grade organic solvents were dried by refluxing with 3 Å molecular sieves, distilled, and stored over 3 Å molecular sieves. Flash chromatography was performed using silica gel 60 (230-240 mesh).
CAL-B Catalyzed Mono Amide Ester 5, 6, and 8 Synthesis
A suspension was prepared, under nitrogen atmosphere, of the corresponding diesters (
Gas Chromatography/Mass Spectrometry Analysis of Mono Amide Esters
Biocatalyzed reactions were monitored by gas chromatography-mass spectrometry on a Clarus 580 S GC coupled to a Clarus SQ 8 S MS (Perkin Elmer, Mexico). Samples were analyzed in an Elite 5 ms fused-silica capillary column (poly(dimethylsiloxane), 30 m × 0.32 mm i.d. × 0.25 µm film thickness (Perkin Elmer,USA). Carrier gas was helium, 1.5 mL/min constant flow; 250°C injector temperature; 1:30 split ratio; at several temperature gradients; 230°C ion source temperature; 250°C transfer line temperature; 70 eV ionization energy. Electron ionization mass spectra were acquired over the mass range of 35-400 amu.
1H NMR Analysis
1H NMR spectra, recorded in CDCl3 on an Agilent-Varian, Mercury Plus spectrometer at 300 MHz are reported as parts per million (ppm) downfield from tetramethylsilane internal standard.
Ethyl 3-(Benzylamino)-3-Oxopropanoate (5)
IRνmax/cm-1: 3447 (NH2), 1731 (C = O ester), 1651 (C = O amide).
1H NMR (CDCl3) δH: 1.24 (t, 3 H, CH3), 3.64 (s, 2 H, CH2), 4.21 (q, 2 H, CH2), 4.32 (s, 2 H, CH2), 6.63 (br s, 1 H, NH), 7.27-7.39 (m, 5 H, Ar).
Ethyl 4-(Benzylamino)-4-Oxobutanoate (6)
IRνmax/cm-1:3441 (NH2), 1733 (C = O ester), 1650 (C = O amide).
1H NMR (CDCl3) δH: 1.10 (t, 3 H, CH3), 2.51 (t, 2 H, CH2), 2.62 (t, 2 H, CH2), 4.10 (t, 3 H, CH3), 6.63 (br s, 1 H, NH), 7.24-7.32 (m, 5 H, Ar).
(R,S)-Methyl 4-(Benzylamino)-3-Hydroxy-4-Oxobutanoate (8)
IRνmax/cm-1: 3351 (NH2), 1730 (C = O ester), 1650 (C = O amide). (CDCl3) δH: 2.7 (q, 1 H, CH), 3.0 (q, 1 H, CH), 3.7 (s, 3 H, CH3), 4.07 (br s, 1 H, NH), 4.50 (m, 1 H, CH), 7.20-7.45 (m, 5 H, Ar)
Footnotes
Acknowledgement
The authors thank Patricia M. Hayward-Jones M Sc and Dulce Ma. Barradas-Dermitz M Sc for the critical reading of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by National Institute of Technology of Mexico-Veracruz Institute of Technology (Tecnológico Nacional de México-Instituto Tecnológico de Veracruz).
