Abstract
A new δ-truxinic acid sucrose diester and a new ε-truxillic acid sucrose diester (named trigohonbanosides E and F) were isolated from the leaves of Trigonostemon honbaensis. Their chemical structures were determined by extensive analysis of their HR-ESI-MS and NMR spectra. At a concentration of 20 µM, trigohonbanosides E and F exhibited weak inhibitory effects on NO production in LPS-activated RAW264.7 cells with inhibitory percentages of 22.7% ± 1.1% and 18.5% ± 1.4%, respectively.
The chemical constituents of Trigonostemon species have attracted the attention of pharmaceutical chemists because of their unique structure backbones and interesting biological activities. 1 -4 To date, over 200 compounds have been identified from Trigonostemon species; diterpenoids, β-carboline alkaloids, and phenolics are the major and most important constituents. 1 A number of these compounds have been reported to have potent anti-viral, insecticidal, and cytotoxic activities. 2 Several of these bioactive compounds and their analogs have been synthesized, such as daphnane diterpenoid orthoesters and β-carboline alkaloids. 5 -7 T. honbaensis Tagane & Yahara is recently recorded as a new and endemic species of Vietnam. Our previous chemical investigation of this plant identified four new sucrose diesters of β-truxinic acid derivatives. 8 Sucrose diesters of truxinic acid derivatives are rarely found in natural sources. A few have been previously isolated from Imperata cylindrica, Coix lachryma-jobi, Bidens parviflora, and oat grains. 9 -12 Derivatives of truxinic acid and its isomer, truxillic acid, are naturally occurring compounds formed by [2 + 2] cyclodimerization of cinnamic acid derivatives. Their structures are interesting because of containing a cyclobutane ring and a variety of stereoisomers. 13,14 In our continuing study, this paper describes the isolation from the leaves of T. honbaensis and structure elucidation of a new stereoisomer of a δ-truxinic acid derivative, and a new ε-truxillic acid derivative (Figure 1). The anti-inflammatory activity of the isolated compounds was evaluated by determining the inhibition of NO production in LPS-stimulated RAW264.7 cells.
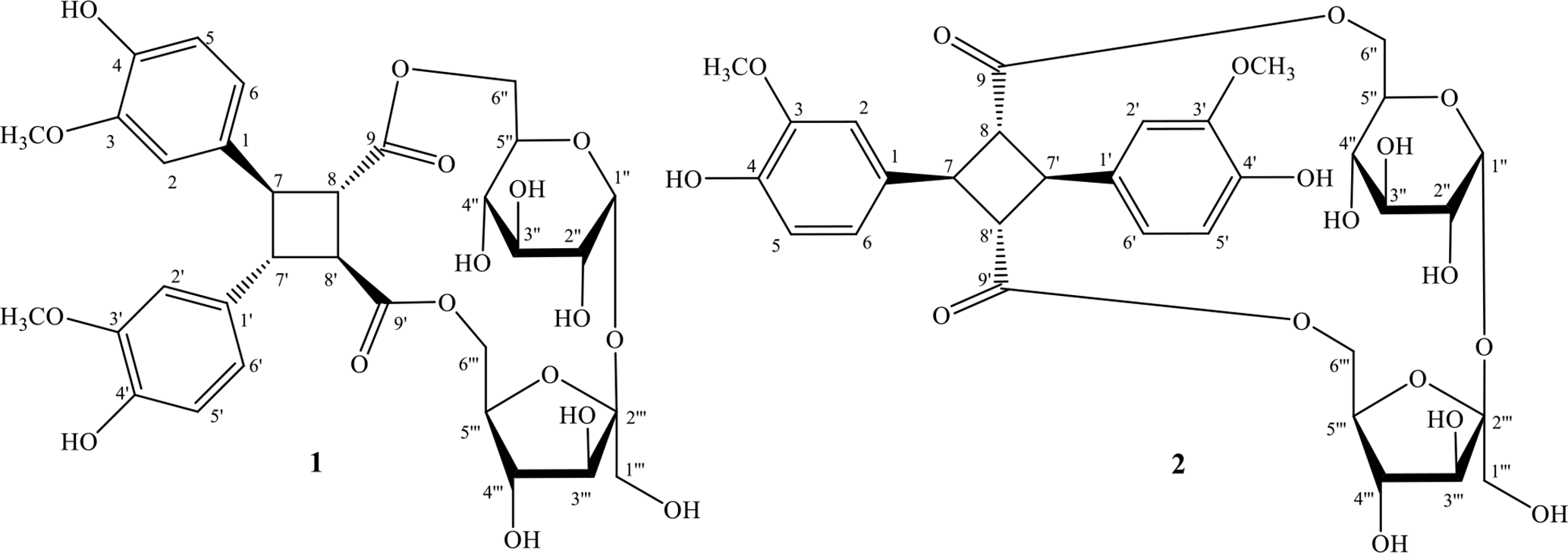
Chemical structures of compounds 1 and 2.
Results and Discussion
Compound

Important HMBC, COSY and NOESY correlations of compounds 1 and 2.
Compound
1H NMR and 13C NMR Spectroscopic Data for Compounds 1 and 2 in Deuterated Methanol.
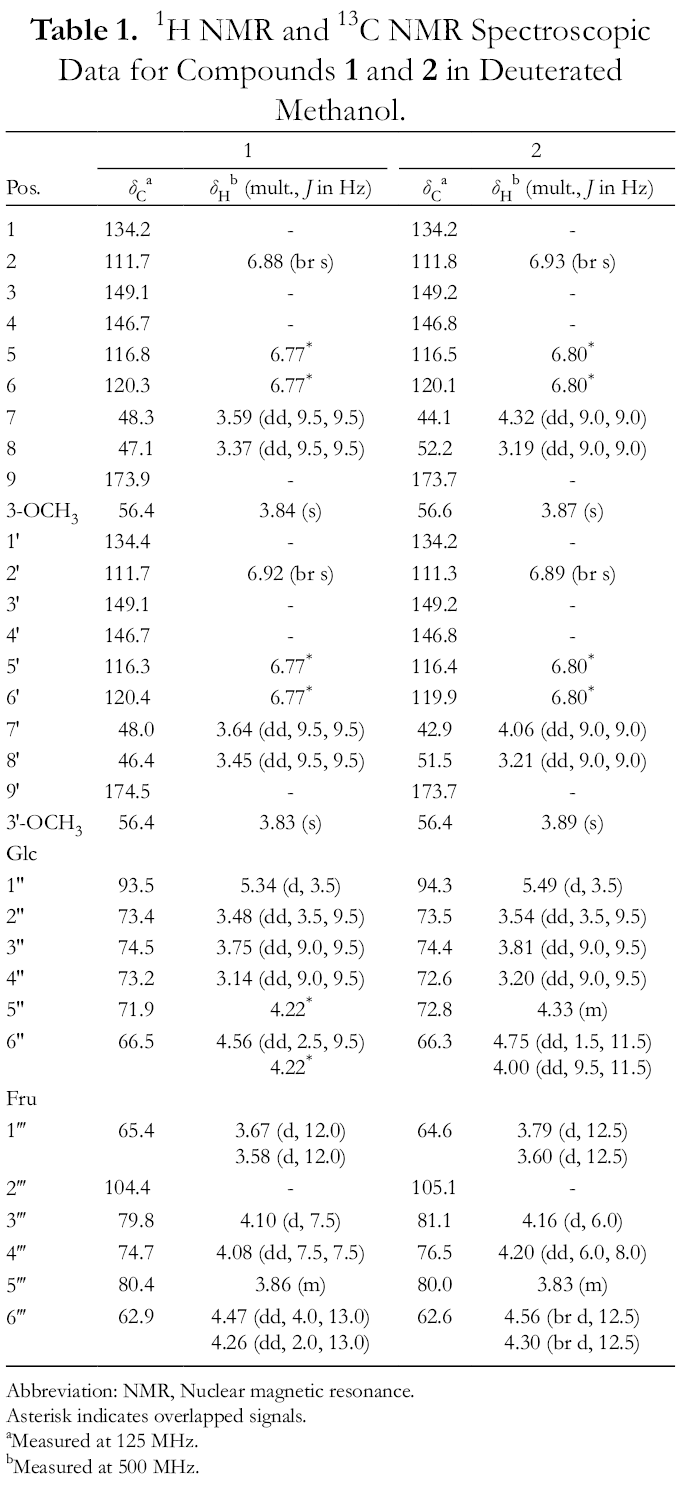
Abbreviation: NMR, Nuclear magnetic resonance.
Asterisk indicates overlapped signals.
aMeasured at 125 MHz.
bMeasured at 500 MHz.
In the plant kingdom, truxinic acid and truxillic acid are generated by cyclodimerization of cinnamic acid. Truxinic acid is formed by head-to-head [2 + 2] cyclodimerization of cinnamic acid, while truxillic acid is obtained by head-to-tail [2 + 2] cyclodimerization.
14
Therefore, compounds
Material and Methods
General Experimental Procedures
Optical rotation was recorded on a Jasco P-2000 polarimeter. HR-ESI-MS were acquired on an Agilent 6530 Accurate Mass Q-TOF system, and NMR spectra on a Bruker Avance III 500 MHz spectrometer. Column chromatography was performed using either silica gel or reversed phase (C-18) resins as adsorbent. Thin layer chromatography was carried out on pre-coated silica gel 60 F254 and/or RP-18 F254S plates. Compounds were visualized under UV irradiation (254 nm and 365 nm) and by spraying with H2SO4 solution (5%), followed by heating with a heat gun.
Plant Material
The leaves of Trigonostemon honbaensis Tagane & Yahara were collected at Nui Chua National Park, Ninh Thuan Province, Vietnam in December 2018. Its scientific name was identified by one of the authors, Prof. Ninh Khac Ban. A voucher specimen (No. NCCT-P79) is kept at the Institute of Marine Biochemistry, Vietnam Academy of Science and Technology.
Extraction and Isolation
The leaves of Trigonostemon honbaensis (dried powder, 4 kg) were ultrasonically extracted with methanol at room temperature, 3 times (each 10 L MeOH, 60 minutes). After removal of the solvent under vacuum, the methanol extract (450 g) was suspended in distilled water (3.0 L) and successively partitioned with dichloromethane and ethyl acetate to give dichloromethane (80.7 g), ethyl acetate (4.1 g), and water-soluble portions. The water layer was separated on a Diaion HP-20 column, eluting with methanol/water (1/3, 1/1, 3/1, 1/0, stepwise each 1.5 L, v/v) to give 4 fractions (TH1-TH4). Fraction TH2 was chromatographed on a reversed phase C18 (RP-18) column, eluting with methanol/water (1/1, v/v, 2 L) to yield 6 fractions (TH2A- TH2F). Fraction TH2D was then separated on a Sephadex LH-20 column, eluting with methanol/water (1/1, v/v, 1 L) to give four fractions (TH2D1-TH2D4). Fraction TH2D2 was purified by preparative HPLC using a J'Sphere ODS-H80 column (20 × 250 mm, 4 µm) and an isocratic mobile phase of acetonitrile (20%) in water to give compounds
Trigohonbanoside E (1)
White amorphous powder,
Trigohonbanoside F (2)
White amorphous powder,
Alkaline Hydrolysis
Refer to Supplemental Material.
Nitric Oxide Assay
Refer to Supplemental Material.
Conclusions
Two new sucrose diesters of δ-truxinic acid and ε-truxillic acid derivatives (named trigohonbanosides E and F) were isolated from the leaves of Trigonostemon honbaensis. At a concentration of 20 µM, trigohonbanosides E and F exhibited weak inhibitory effects on NO production in LPS-activated RAW264.7 cells with inhibitory percentages of 22.7% ± 1.1% and 18.5% ± 1.4%, respectively.
Supplemental Material
Supplementary Material 1 - Supplemental material for New Truxinic and Truxillic Acid Sucrose Diesters From the Leaves of Trigonostemon honbaensis
Supplemental material, Supplementary Material 1, for New Truxinic and Truxillic Acid Sucrose Diesters From the Leaves of Trigonostemon honbaensis by Ninh Khac Ban, Bui Huu Tai, Vu Kim Thu and Phan Van Kiem in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number: 104.01-2018.300.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
