Abstract
Cudratricusxanthone A (CTXA), a natural bioactive compound extracted from the roots of Cudrania tricuspidata Bureau, is known to possess hepatoprotective, antiproliferative, and anti-inflammatory activities. Inhibition of high-mobility group box 1 (HMGB1) and restoration of endothelial integrity is emerging as an attractive therapeutic strategy in the management of severe sepsis or septic shock. Here, we examined the effects of CTXA on HMGB1-mediated barrier disruptive responses and survival rate in a mouse sepsis model. The anti-inflammatory activities of CTXA were monitored by the effects on lipopolysaccharide- or cecal ligation and puncture (CLP)-mediated release of HMGB1. In addition, treatment with CTXA reduced CLP-induced mouse mortality in vivo. Our results indicate that CTXA could be a candidate for a therapeutic agent for various severe vascular inflammatory diseases via the inhibition of the HMGB1 signaling pathway.
Sepsis is defined as a systematic inflammatory response syndrome caused by infection. 1 Sepsis and severe sepsis (sepsis accompanied by acute organ dysfunction) are leading causes of death in high-income countries and the most common cause of death among critically ill patients in non-coronary intensive care units, 1 despite advances in antibiotic therapy and intensive care. 2,3 In October 2011, the only Food and Drug Administration-approved drug for severe sepsis, Xigris, was withdrawn from the market. 4 As a result, we are now left with no effective preventative or treatment option for severe sepsis. A wide array of proinflammatory cytokines, including tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and interferon-γ, contribute to the pathogenesis of lethal systemic inflammation. 5 For instance, neutralizing antibodies against TNF-α 6 reduce lethality in an animal model of endotoxemic/bacteremic shock. However, the early kinetics of systemic TNF-α accumulation makes its targeting difficult in the clinical setting, 6 which prompted the investigation of other late proinflammatory mediators as potential therapeutic targets for sepsis, such as high-mobility group box-1 protein (HMGB1). HMGB1 can be secreted into the extracellular space through a passive process initiated by necrotic cells, thereby allowing the innate immune system to recognize damaged cells. 7 Elevated serum levels of HMGB1 are measurable in most patients up to 1 week after a diagnosis of sepsis or septic shock, and these levels are correlated with the degree of organ dysfunction. 8,9 As HMGB1 is a late inflammatory mediator of sepsis, it provides a wide therapeutic window for clinical intervention and remains an attractive target for sepsis treatment. 10
Cudrania tricuspidata Bureau (Moraceae), a deciduous tree, widely cultivated in Korea, China, and Japan, has been investigated phytochemically and biologically. 11 The species is a rich source of xanthones and flavonoids, some of which have anticancer, hepatoprotective, anti-inflammatory, and antioxidant activities. 12 Cudrania tricuspidata has been used as a traditional Chinese medicine for the treatment of lumbago, hemoptysis, and contusion. 13 Moreover, the root bark has been used for the treatment of gonorrhea, rheumatism, jaundice, and dysmenorrhea. 14 During our continuing search for natural products that modulate septic responses, we found that cudratricusxanthone A (CTXA) in C. tricuspidata exhibited potent antiseptic effects against HMGB1-mediated septic responses in both cell and animal models. Here, we report, for the first time, the anti-septic effects of CTXA on HMGB1-mediated responses in vitro and in vivo.
HMGB1 is actively released by monocytes and macrophages in response to lipopolysaccharide (LPS), as well as by necrotic cells. 15 -18 HMGB1 levels rise slowly after 8 hours and are correlated with the progression of sepsis. 19 Thus, the effect of CTXA on the LPS-mediated release of HMGB1 by human umbilical vein endothelial cell (HUVEC) cells was studied. As shown in Figure 1(a), CTXA inhibited the release of HMBG1 in a dose-dependent manner, with a minimal effective concentration of 5 µM. To confirm these activities in vivo, CTXA was evaluated for its ability to inhibit HMGB1 release using a mouse model of cecal ligation and puncture (CLP)-induced sepsis. Treatment with CTXA resulted in a marked reduction in HMGB1 release (Figure 1b). The average circulating blood volume for mice is 72 mL/kg. 20 Because the average weight of the used mice was 27 g, and the average blood volume 2 mL, the amount of CTXA (0.147 or 0.294 mg/kg) injected yielded a maximum concentration of 5 or 10 µM in the peripheral blood. In conjugation with this, cell viability assays were performed to probe the toxicity of CTXA in HUVECs after 48 hours. At the concentrations used (up to 50 µM), CTXA did not affect cell viability (Figure 1c). Taken together, these results indicate that CTXA may be a viable early intervention to prevent the release of HMGB1 and progression to severe sepsis and septic shock.
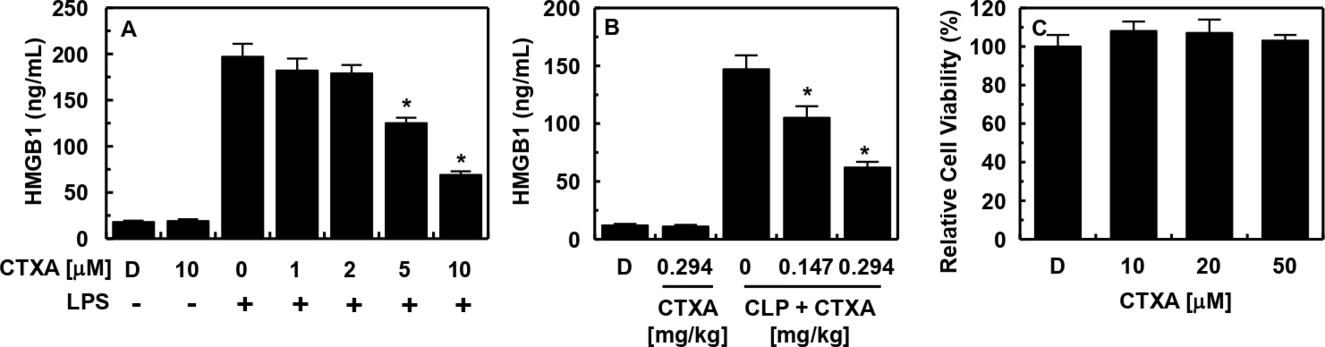
Effects of CTXA on HMGB1 release. (a) HUVECs were treated with the indicated concentrations of CTXA for 6 hours, after stimulation with LPS (100 ng/mL, 16 hours), and HMGB1 release was measured by ELISA. (b) Male C57BL/6 mice that underwent CLP were administered CTXA (intravenously 12 hours after CLP, n = 5). Mice were euthanized 24 hours after CLP. serum HMGB1 levels were measured by ELISA. (c) Effect of CTXA on cellular viability was measured by MTT assay. The results shown are mean ± standard deviation from 3 separate experiments on different days with triplicate wells. D = 0.2% dimethyl sulfoxide is the vehicle control. *P < 0.05 versus LPS alone (a), CLP alone (b). CLP: cecal ligation and puncture; CTXA: cudratricusxanthone A; ELISA: enzyme-linked immunosorbent assay; HMGB1: high-mobility group box-1 protein; HUVEC: human umbilical vein endothelial cells; LPS: lipopolysaccharide.
As LPS and HMGB1 are known to cause cleavage and disruption of the endothelial barrier, 21 a permeability assay was performed using HUVECs to determine the effects of CTXA on the maintenance of barrier integrity. HUVECs were treated with various concentrations of CTXA for 6 hours following the addition of either LPS (100 ng/mL) or HMGB1 (1 µg/mL). The results show that CTXA dose-dependently inhibited LPS- and HMGB1-mediated membrane disruption (Figure 2a, b). The in vivo effects of CTXA on vascular permeability were assessed to corroborate the in vitro results. Figure 2(c) shows that CTXA induced a marked inhibition of the peritoneal dye leakage induced by HMGB1. HMGB1 elicits an inflammatory response through diverse pathways, including MyD88/IRAK/TRAF, which leads to nuclear factor kappa B activation; the Rac1/PI3K pathway; via the activation of ERK 1/2 and p38 mitogen-activated protein kinase (MAPK) downstream of TLR2/4; and the Ras/p38 pathway downstream of RAGE. 22 -24 To determine whether the p38 pathway is affected by CTXA, HUVECs were stimulated by HMGB1 and treated with CTXA. HMGB1 upregulated the expression of phosphorylated p38, which was clearly reduced by treatment with CTXA (Figure 2d). The reduction in HMGB1-induced permeability and p38 activation by CTXA points toward a promising role as antisepsis drugs.
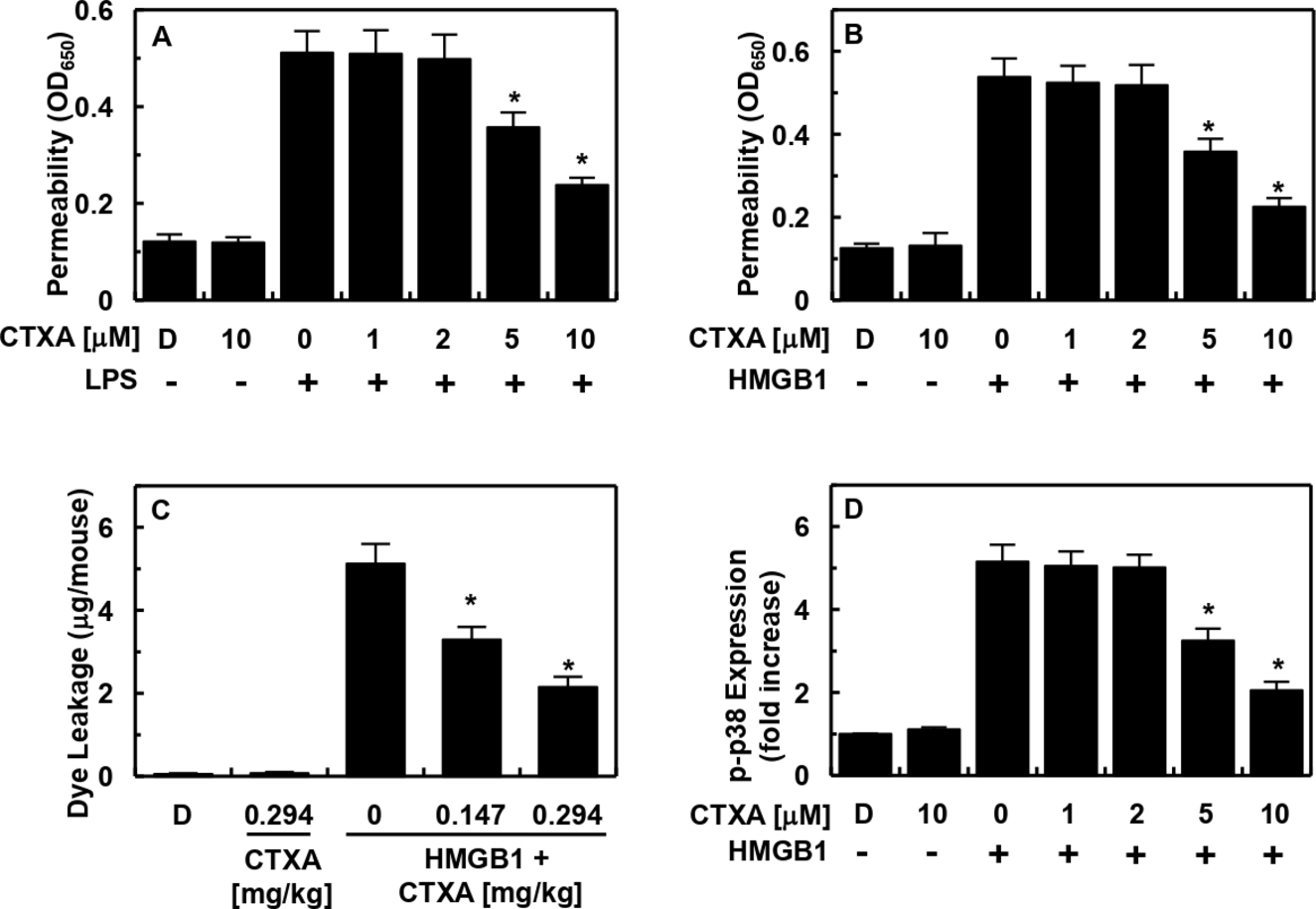
Effects of CTXA on HMGB1-mediated permeability in vitro and in vivo. Effects of treatment with different concentrations of CTXA for 6 hours on barrier disruption caused by either LPS (a, 100 ng/mL, 4 hours) or HMGB1 (b, 1 µg/mL, 16 hours) were monitored by measuring the flux of Evans blue-bound albumin across HUVECs. (c) The effects of CTXA on HMGB1-induced (2 µg/mouse, intravenous) vascular permeability in mice were examined by measuring the amount of Evans blue in peritoneal washings (expressed µg/mouse, n = 5). (d) HUVECs were activated with HMGB1 (1 µg/mL, 16 hours), followed by treatment with different concentrations of CTXA for 6 hours. The effects of CTXA on HMGB1-mediated expression of phospho-p38 were determined by enzyme-linked immunosorbent assay. Results are expressed as the mean ± standard deviation of 3 separate experiments on different days. *P < 0.05 versus LPS (a) or HMGB1 (b, c, d). CTXA: cudratricusxanthone A; HMGB1: high-mobility group box-1 protein; HUVEC: human umbilical vein endothelial cells; LPS: lipopolysaccharide.
High levels of HMGB1 are correlated with the progression of sepsis to severe sepsis, septic shock, and death. CTXA used in the present work showed promising results in preventing the release of HMGB1, and also inhibited its downstream signaling and effects. This prompted us to evaluate the effects of CTXA on the survival rate of mice with sepsis following CLP operation. To determine whether CTXA protects mice from CLP-induced sepsis lethality, CTXA was administered to mice after CLP. A single administration of CTXA (0.147 or 0.294 mg/kg, 12 hours after CLP) did not prevent CLP-induced death (data not shown). Thus, we next administered the same amounts of CTXA twice (once 12 hours after CLP and once 50 hours after CLP). This increased the survival rate from 40% to 60%, according to Kaplan-Meier survival analysis (P < 0.00001, Figure 3). The marked improvement in survival rate achieved by the administration of CTXA suggests that the suppression of HMGB1 release and HMGB1-mediated inflammatory responses is a viable therapeutic strategy for management of sepsis and septic shock.

Effects of CTXA on lethality after CLP. Male C57BL/6 mice (n = 20) were administered CTXA at 0.147 mg/kg (iv □) or at 0.294 mg/kg (iv ■) at 12 and 50 hours after CLP. Animal survival was monitored every 12 hours after CLP for 132 hours. Control CLP mice (●) and sham-operated mice (○) were administered sterile saline (n = 20). Kaplan-Meier survival analysis was used to determine the overall survival rates versus CLP-treated mice. CLP: cecal ligation and puncture; CTXA: cudratricusxanthone A; IV: intravenous.
The present work was undertaken to evaluate the protective effect of CTXA on vascular barrier integrity. Maintenance of endothelial barrier integrity is emerging as an important therapeutic strategy for the treatment of sepsis. Along with its involvement in the progression of sepsis, HMGB1 has also been implicated as a proinflammatory cytokine and as a proangiogenic factor in sustained proinflammatory responses observed in severe vascular inflammatory diseases, such as sepsis and septic shock. 10 The results of this study suggest that the antiseptic effects of CTXA occur through the inhibition of HMGB1 release and HMGB1-mediated barrier disruptive responses.
Collectively, the results of this study demonstrate that CTXA inhibits both LPS- and CLP-mediated release of HMGB1 and HMGB1-mediated barrier disruption by increasing barrier integrity and inhibiting the expression of p38. Further, the barrier protective effects of CTXA were confirmed in a mouse model, in which treatment with CTXA reduced CLP-induced mortality. Our findings indicate that CTXA may be a potential candidate for use in the treatment of severe vascular inflammatory diseases, such as sepsis and septic shock.
Experimental
Reagents
CTXA, bacterial LPS (serotype: 0111:B4, L5293), Evans blue, crystal violet, 2-mercaptoethanol, and antibiotics (penicillin G and streptomycin) were purchased from Sigma (St Louis, MO). Human recombinant HMGB1 was purchased from Abnova (Taipei City, Taiwan), and fetal bovine serum and Vybrant DiD from Invitrogen (Carlsbad, CA).
Cell Culture
Primary HUVECs were obtained from Cambrex Bio Science (Charles City, IA) and maintained using a previously described method. 25,26 HUVECs were used in cell culture at passages 3 to 5.
Animals and Husbandry
Male C57BL/6 mice (6-7 weeks old, 27 g), purchased from Orient Bio Co. (Sungnam, Republic of Korea), were used in this study after a 12-day acclimatization period. Animals (5 per polycarbonate cage) were housed under controlled temperature (20-25°C) and humidity (40%-45% RH), with a 12:12 hour light/dark cycle. Animals received a normal rodent pellet diet and water ad libitum during acclimatization. All animals were treated in accordance with the “Guidelines for the Care and Use of Laboratory Animals” issued by Kyungpook National University (IRB No. KNU 2016-54).
Cecal Ligation and Puncture
To induce sepsis, male mice were anesthetized with Zoletil (tiletamine and zolazepam, 1:1 mixture, 30 mg/kg) and Rompum (xylazine, 10 mg/kg). The CLP-induced sepsis model was prepared as previously described. 27,28 In brief, a 2 cm midline incision was made to expose the cecum and adjoining intestine. The cecum was then tightly ligated with a 3.0 silk suture 5.0 mm from the cecal tip and punctured once using a 22-gage needle to induce high-grade sepsis. 29 The cecum was then gently squeezed to extrude a small amount of feces from the perforation site and returned to the peritoneal cavity. The laparotomy site was then sutured with 4.0 silk. In sham control animals, the cecum was exposed, but not ligated or punctured, and then returned to the abdominal cavity. This protocol was approved by the Animal Care Committee at Kyungpook National University conducting the study (IRB No. KNU 2016-54).
Competitive Enzyme-Linked Immunosorbent Assayfor HMGB1
A competitive enzyme-linked immunosorbent assay (ELISA) was performed, as previously described, to determine the HMGB1 concentrations in cell culture media or mice serum. 26 HUVEC monolayers were treated with LPS (100 ng/mL) for 16 hours, followed by CTXA for 6 hours. Cell culture media were collected for the determination of HMGB1. To perform the ELISA, 96-well flat plastic microtiter plates (Corning, NY) were coated with HMGB1 protein in 20 mM carbonate-bicarbonate buffer (pH 9.6) containing 0.02% sodium azide overnight at 4°C. Plates were then rinsed 3 times in PBS-0.05% Tween 20 (PBS–T) and kept at 4°C. Lyophilized culture media were preincubated with an anti-HMGB1 antibody (Abnova, diluted 1:1000 in PBS-T) in 96-well plastic round microtiter plates for 90 minutes at 37°C, transferred to the precoated plates, and incubated for 30 minutes at room temperature. Plates were then rinsed 3 times in PBS-T, incubated for 90 minutes at room temperature with peroxidase-conjugated anti-rabbit immunoglobulin G antibodies (diluted 1:2 000 in PBS-T, Amersham Pharmacia Biotech), rinsed 3 times with PBS-T, and incubated for 60 minutes at room temperature in the dark with 200 µL of substrate solution (100 µg/mL o-phenylenediamine and 0.003% H2O2). After stopping the reaction with 50 µL of 8 N H2SO4, absorbance was measured at 490 nm.
Cell Viability Assay
MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide) was used as an indicator of cell viability. Cells were grown in 96-well plates at a density of 5 × 103 cells/well. After 24 hours, cells were washed with fresh medium, followed by treatment with CTXA. After a 48-hours incubation period, cells were washed, and 100 µL of MTT (1 mg/mL) was added, followed by incubation for 4 hours. Finally, dimethyl sulfoxide (150 µL) was added to solubilize the formazan salt formed. The amount of formazan salt was determined by measuring the OD at 540 nm using a microplate reader (Tecan Austria GmbH, Austria).
Permeability Assay in Vitro
For the spectrophotometric quantification of endothelial cell permeability in response to increasing concentrations of each compound, the flux of Evans blue-bound albumin across functional cell monolayers was measured using a modified 2-compartment chamber model, as previously described. 18 HUVECs were plated (5 × 104 cells/well) in transwells with a pore size of 3 µm and a diameter of 12 mm for 3 days. Confluent monolayers of HUVECs were treated with either LPS (100 ng/mL) for 4 hours or HMGB1 (1 µg/mL) for 16 hours, followed by treatment with CTXA. Transwell inserts were then washed with PBS (pH 7.4), and growth medium containing 0.5 mL of Evans blue (0.67 mg/mL) and 4% bovine serum albumin (BSA) was added. Fresh growth medium was then added to the lower chamber, and the medium in the upper chamber was replaced with Evans blue/BSA. Ten minutes later, OD in the lower chamber was measured at 650 nm.
Permeability Assay in Vivo
For the in vivo study, male mice were anesthetized with 2% isoflurane (Forane, JW Pharmaceutical, South Korea) in oxygen delivered via a small rodent gas anesthesia machine (RC2, Vetequip, Pleasanton, CA), first in a breathing chamber and then via facemask. Mice were allowed to breath spontaneously during the procedure. Mice were treated with HMGB1 (2 µg/mouse, intravenous [IV]) for 16 hours, followed by treatment with CTXA (0.26 or 0.39 mg/kg, IV). Vascular permeability, expressed as micrograms of dye in the peritoneal cavity/mouse, was determined using a standard curve, as previously described. 26 To assess leukocyte migration, mice were euthanized after 6 hours, and the peritoneal cavities were washed with 5 mL of normal saline. The obtained samples of peritoneal fluids (20 µL) were mixed with 0.38 mL of Turk’s solution (0.01% crystal violet in 3% acetic acid), and the number of leukocytes was counted under a light microscope.
ELISA for Phosphorylated P38 MAPK
The activity of phosphorylated p38 MAPK was quantified according to the manufacturer’s instructions using a commercially available ELISA kit (Cell Signaling Technology, Danvers, MA). Values were measured using an ELISA plate reader (Tecan, Austria GmbH, Austria).
Statistical Analysis
All experiments were performed independently at least 3 times. Values are expressed as means ± standard deviation. The statistical significance of differences between test groups was evaluated using SPSS for Windows, version 16.0 (SPSS, Chicago, IL). Statistical relevance was determined by one-way analysis of variance and Tukey’s post hoc test. P values less than 0.05 were considered to indicate significance.
Footnotes
*
The authors Sumin Yang and Bong-Seon Lee contributed equally to the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant number: HI15C0001). This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No.2018R1A5A2025272).
