Abstract
Some species of
In the last decades, intestinal nematode infections have been treated with benzimidazole drugs, especially albendazole. However, recent research reports evince an increase of the resistance to the commonly used anthelmintic drugs, leading to lower levels of efficacy. 1 Consequently, alternative treatments based on natural products are being developed to control these infections. 2,3
Some species of
Microemulsions (MEs) are colloidal dispersions, with a homogeneous appearance, used in the formulation of oily products. MEs are constituted by an aqueous phase, an oily phase, and a mixture of emulsifiers. A co-emulsifier is usually added in order to improve the ME stability. 14,15 Since the ME systems are composed of specific amounts of those components—water, oil, and surfactant—, it is useful to represent them in ternary diagrams; in case a co-surfactant is included in the mixture, pseudoternary diagrams are used (Figure 1a). MEs may form 3 different microstructures: oil-in-water (O/W), water-in-oil (W/O), and bicontinuous, depending on the molecular arrangement. In O/W and W/O structures MEs have a small droplet size—between 20 and 200 nm—; however, droplets may not be clearly found in bicontinuous structures. 14,16 All these structures can be characterized by their conductivity. 17,18

(
D-optimal mixture designs are commonly used in the formulation of MEs. These methodologies estimate the effect of the components on the formulation properties, such as droplet size. 19,20 D-optimal mixture designs allow optimization of the composition of an ME formulation in order to enhance the oral bioavailability of the AAE. The main aim of the present study was to design a novel ME formulation of a thujones-free AAE to improve its solubility and subsequently, to enhance its oral bioavailability and nematocidal activity.
In previous studies, it was shown that a surfactant mixture (SM) composed of Tween® 80: propylene glycol (1.5:1) was able to dissolve a high amount of AAE and water, so this combination of surfactants was selected for further research. The components of the mixture design were the SM, set to a range of 48% to 82% w/w, and distilled water (W), and AAE, which were set to within ranges of 4% to 80% w/w each (Figure 1a). According to these ranges, the software Design-Expert® assessed randomly 16 experiments or combinations of the 3 components (W, AAE, and SM) adding up to 100% w/w, to fit a quadratic model (Table 1).
Composition and Results of Experiments in the D-Optimal Mixture Design.

Each experiment was prepared by stirring the components until the mixture had a homogeneous appearance, and then analyzed for droplet size (D32) and conductivity. The software was used to develop and evaluate the experimental designs, and to assess further the equation of the model and its quality parameters. The optimization of the final formulation was based on droplet size.
Conductivity
The conductivity results of the 16 ME formulations ranged from 10.68 to 69.82 mS/cm (Table 1). Linear, quadratic, special cubic, and cubic models were fitted to the responses. The quadratic model was found to be the most suitable mathematical model, as it has the lowest PRESS, and a reasonable agreement between predicted
Summary of the Statistical Analysis of the Conductivity and the D32 Responses.

PRESS: predicted residual error sum of squares.
The model equation was assessed and the coefficients are shown in Table 3. The magnitude of coefficients indicates the positive or negative contribution of the components to the system conductivity. 23 The resulting equation evidenced that the presence of W has a high influence on conductivity, because of the nonionic nature of the other components. SM and AAE do not improve the ionic movement in the system; therefore, they have a low or even negative influence on conductivity. As can be seen in Figure 1b, conductivity increased with increasing water-content.
Coefficients in Conductivity and Lg(D32) Models.

Droplet Size
The mean droplet size, expressed as Sauter mean diameter (
Droplet size was highly influenced by the proportions of AAE and specially water content. It could be explained because of the high surfactant content in the experiments; surfactant molecules are organized in layers surrounding the droplets. Hence, when the content of the other components (W and AAE) increases, the droplets may break down, being surrounded by a thin layer of surfactant molecules. 26
According to the droplet size responses, the components of the ME were optimized using the desirability function. 27,28 SM and AAE content were set to be maximized and the content of W was minimized. The optimized solution with a desirability value of 0.7670, was 66.40% w/w of the SM, 29.35% w/w of AAE and 4.25% w/w of W, which was similar to the composition of experiments 2 and 3, already included in the design. The predicted response value was 1.1836 nm (95% CI 0.71-1.99), comparable with the D32 values measured in experiments 2 and 3, 0.87 nm and 1.04 nm, respectively.
In Vitro Dissolution Test
The behavior of the optimized ME was analyzed in a gastric medium. The optimized ME (4.1 g) was dispersed in artificial gastric fluid (250 mL) prepared according to USP 32, simulating fasting conditions.
29
A rotating paddle apparatus was set at 100 rpm, samples (5 mL) were taken at predetermined times (15 and 30 minutes) and their droplet sizes were analyzed. The droplet size distribution of the samples was similar at 15 and 30 minutes and the average droplet size was 11.69 nm.
Ex Vivo Nematocidal Efficacy Against T. spiralis
AAE was dissolved in up to 0.1% w/w dimethyl sulfoxide (DMSO):Tween® 80 (98:2). A dilution of the previously optimized ME formulation in Hank’s balanced salt solution (HBSS) was also prepared in order to obtain a similar concentration of oil (0.1% w/w). Dispersions of the solvents (placebo solution) and ME surfactants (placebo ME) in HBSS were also prepared.
One mL of each formulation was poured into each well of a 24-well plate and mixed with 1 mL of a dispersion of
The results showed no difference between the results from control and placebo groups, and between placebo solution and placebo ME groups (
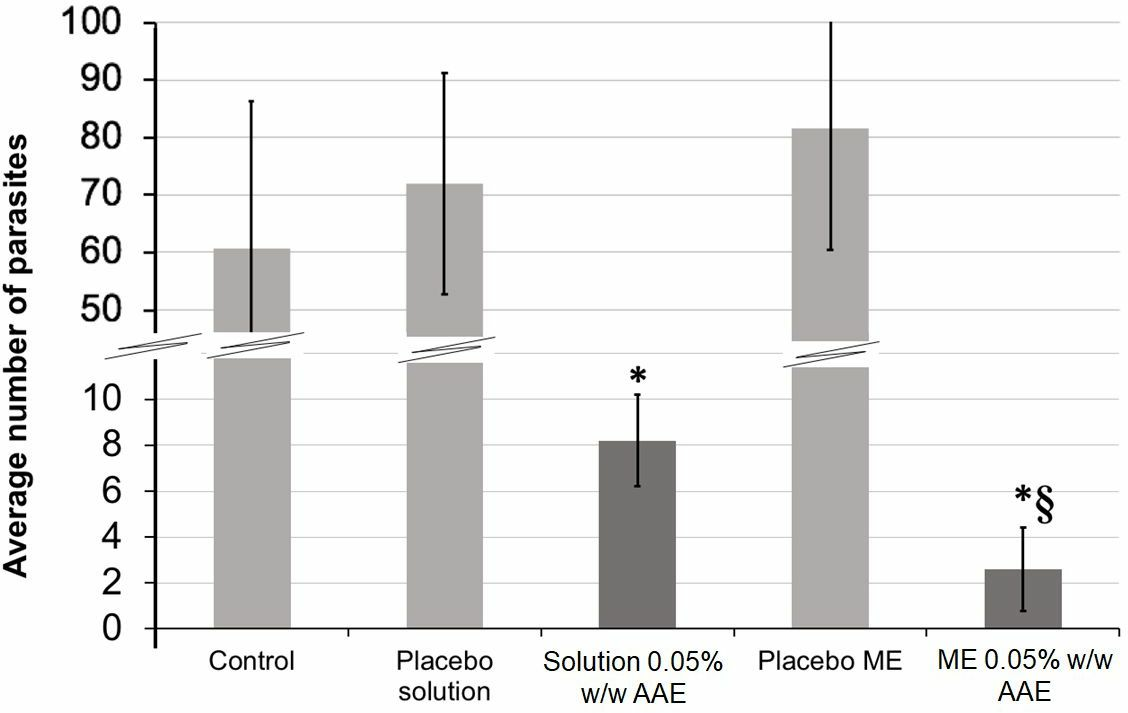
Average number of adult parasites per group ±95% confidence interval (
The AAE solution showed a similar efficacy as that in previous studies. 12 However, the optimized ME is constituted of less-toxic surfactants (Tween® 80 and propylene glycol) than other solvents like DMSO. 31 In addition, a higher amount of AAE is able to dissolve because of those surfactants, as the optimized formulation includes 29.35% w/w of AAE.
This study shows a methodology to formulate a natural extract in a ME system. The optimized ME system is composed of 66.40% w/w of the SM, 29.35% w/w of AAE, and 4.25% w/w of W. It is a W/O system, with the lowest particle size and a low conductivity (around 10 mS/cm). However, its behavior in an aqueous medium showed that the ME is able to disperse in less than 15 minutes, forming 10 nm size droplets. The optimized ME is able to dissolve a higher amount of oil than the AAE solution, resulting in an improved efficacy of the extract against
Experimental
Materials
The AAE was kindly provided by ICA (Madrid, Spain). Polysorbate 80 (Tween® 80), DMSO, and propylene glycol were purchased from Acofarma Distributions, S.A. (Madrid, Spain), and were of pharmaceutical quality. Hydrochloric acid 37% was obtained from Panreac Quimica S.A.U. (Barcelona, Spain). HBSS was prepared by dissolving 8 g/L NaCl, 0.04 g/L KCl, 0.2 g/L MgSO4 7 H2O, 0.06 g/L KH2(PO4)3, 0.5 g/L Na2H(PO4)3, 9 g/L glucose, 0.05 g/L penicillin G, and 0.05 g/L streptomycin in double-distilled water. The solution was vacuum-filtered (pore size 0.22 µm). All the components of HBSS medium and porcine pepsin (P-7000; specific activity of 800-2500 U/mg) were purchased from Sigma Aldrich Quimica, S.A. (Madrid, Spain).
Methods
Droplet size and conductivity were analyzed using Zetasizer NanoZS equipment (Malvern Instruments, Malvern, UK). Dissolution tests were performed using a rotating paddle apparatus described in the European Pharmacopeia (Erweka DT80, Erweka-Gomensoro, Madrid, Spain). Design-Expert® version 7 (Stat-Ease Inc, Minneapolis, MN, USA) was used to develop the mixture design and to analyze the results.
Footnotes
Acknowledgments
The authors wish to thank Dr A González-Coloma (ICA) for providing the AAE and Prof Dr AI Olives (Department of Chemistry in Pharmaceutical Sciences, Faculty of Pharmacy, Complutense University of Madrid) for providing the equipment necessary to perform the conductivity measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed no financial support for the research, authorship, and/or publication of this article.
