Abstract
Phytochemical investigation of methanolic extract of Limonium leptophyllum (Plumbaginaceae), led to the isolation of 1 new isoflavonoid with a rare 5-membered dihydrofuran ring (
The genus Limonium (Plumbaginaceae family) is known as halophytes and consists of 300 species worldwide, of which 18 are found in Kazakhstan. 1 Species in this genus are also known as statice, caspia, sea-lavender, or marsh-rosemary. The genus is widely distributed in Asia, Africa, Australia, Europe, and North America. More recently, several species of the Limonium genus are used to treat arthritis and fever. 2 This genus possesses anti-inflammatory, antibacterial, 3,4 antiviral, 5 and cytotoxic properties. 3 Previous phytochemical and biological studies on the Limonium genus have shown the presence of different classes of compounds with various biological activities. 6–14 In continuation of our previous work and search for novel plant-derived biological agents, 6,7,15–17 further investigations on the phytochemical constituents of Limonium leptophyllum (Schrenk) O. Kuntze were carried out. Herein, we are reporting the isolation and characterization of 1 new and 8 known compounds from L. leptophyllum.
Compound
1H and 13C NMR Data for Compounds 1 and 2 in CDCl3 (δ Cand δ H in parts per minute; J in Hertz).

NMR, nuclearmagnetic resonance.
Recorded at a500 MHz, b125MHz.

Key HMBC and COSY correlations of compound 1.
The known isolated compounds (Figure 2) were identified as euchrenone b9 (
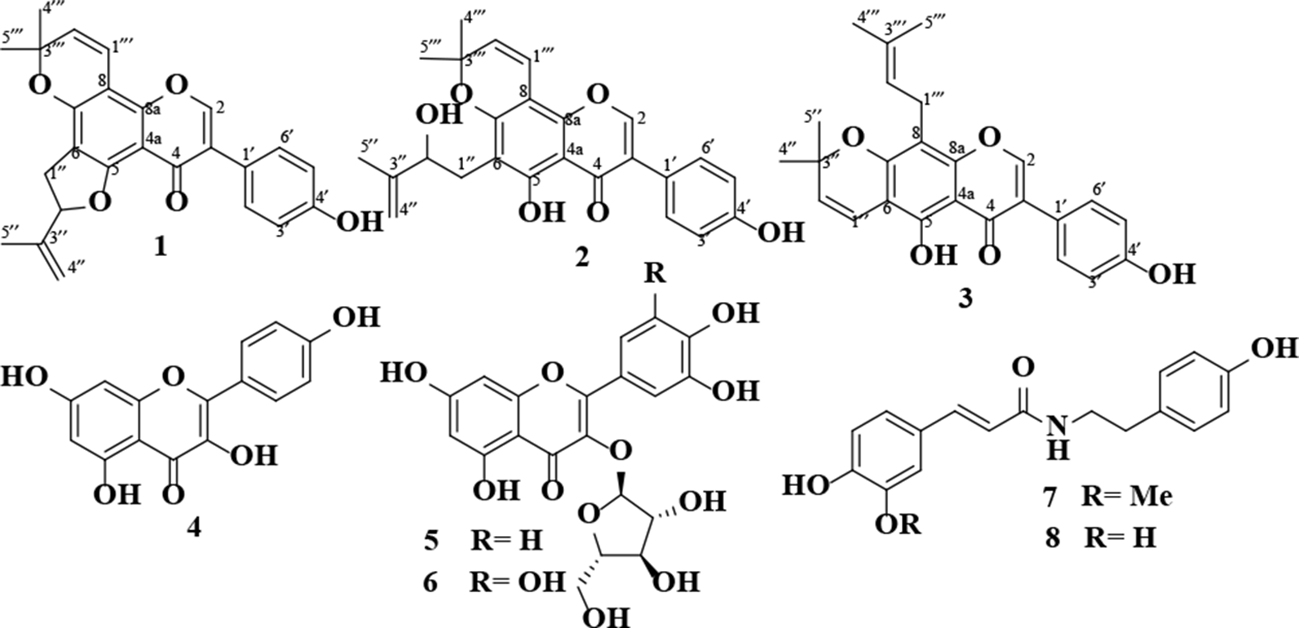
Compounds isolated from L. leptophyllum.
Crude methanolic extract of aerial parts of L. leptophyllum exhibited moderate activity toward endocannabinoid receptors (cannabinoid receptor type 1 [CB1] and CB2). Auriculasin (
Experimental
General
Bruker model AMX 500 NMR and 400 NMR spectrometers operating on a standard pulse system were used to acquire 1H and 13C NMR and 2D spectra. The instruments ran at 500 and 400 MHz for 1H whereas they ran at 125 and 100 MHz for 13C. CDCl3, dimethyl sulfoxide-d 6, and acetone-d 6 were used as NMR solvents, and trimethyl silane was used as an internal standard. Electrospray ionizationmass spectrometric (ESI-MS) data were recorded on Thermo Orbitrap Fusion (Thermo Scientific). Samples were analyzed in the positive mode of ionization and were directly infused at 3 µL/min. Mass was analyzed in Orbitrap (mass error on the instrument <2 ppm). Electrospray ionization-MS data were obtained on a Micromass Q-Tof micromass spectrometer. Fourier transform mass spectrometry-ESI was analyzed on Thermo Orbitrap Fusion (Thermo Scientific). The sample was analyzed in the positive mode of ionization. Thin-layer chromatography was performed on precoated silica gel GF254 plates and column chromatography was performed on silica gel (200-300 mesh) and Sorbadex-LH20 (Sorbent Technologies, Atlanta, GA, USA) with detection provided by UV light (254 and 366 nm) and by spraying with 1% vanillin-H2SO4 reagent followed by heating for 5 to 10 minutes (105°C). Quantitative high-performance liquid chromatography (HPLC) was conducted using an Agilent 1100 HPLC system equipped with a degasser (G1379A), quaternary pump (G13311A), auto sampler (G1313A), column oven (G1316A), and UV-diode array detector (G1315B) controlled by Chemstation software. The analysis was carried out on RP-C18 columns (150 × 4.6 mm; particle size 5 µm; Luna) and (250 × 10.0 mm; particle size 10 µm; Luna) with column oven temperature set at 25°C and using the isocratic system of eluent water (A) and acetonitrile (B) for the separation of the target compounds. Acetonitrile and water solvents were of HPLC grade, where acetic acid was added as a modifier to achieve a final concentration of 0.1% in each solvent. The flow rates of the solvent were 1.0 mL/min for the analytical injections whereas 5 mL/min for the semipreparative ones and the injection volumes were 5.0 and 50 µL for the analytical and semipreparatives, respectively. All the analysis was carried out at wavelengths of 254 and 280 nm with a run time of 40 minutes.
Plant Material
Limonium leptophyllum (Schrenk) O. Kuntze aerial parts were collected in August 2016, from Kyzylorda region, Kazakhstan. The plant material was authenticated by Akhtaeva Nursulu and the specimen voucher (No.0916.) was stored at the department of biology and biotechnology of al-Farabi Kazakh National University.
Extraction and Isolation
Limonium leptophyllum aerial parts (830 g) were extracted using MeOH (3 L × 3 times); the extract was thereafter filtered and concentrated on a rotary evaporator at 40°C yielding 90.3 g of crude methanolic extract. This extract (50 g) was fractionated over HP 20 gel (200 g) column using H2O-methanol (1:9), methanol, acetone, and dichloromethane (DCM) (each 750 mL). Each fraction was evaporated to dryness to give 16.47 g fraction A (H2O:methanol 1:9), 7.87 g fraction B (methanol), 11.25 g fraction C (acetone), and 1.17 g fraction D (DCM).
Fractions C and D were combined and the mixture (E, 12 g) was loaded onto a normal phase silica gel vacuum liquid column (210 g) and eluted using hexane-acetone gradient to yield 12 fractions (E1-E12) and continued with methanol-DCM gradient to yield 10 more fractions (E13-E22). Fraction E5 was identified as β-sitosterol (
Leptoisoflavone a (1)
Yellow solid.
[α]D 22.5: –3.125 (c 0.064, MeOH).
1H and 13C NMR: Table 1.
High-resolution ESI-MS: m/z 403.1532 [M + H]+ (calc. C25H22O5 + H, 403.1545).
Cannabinoid and Opioid Receptor Assays
The affinity of the total extracts, fractions, and isolated compounds toward cannabinoid and opioid receptors was analyzed according to the published method.
24
The crude methanolic extract exhibited moderate activity toward endocannabinoid receptors. Compound
Footnotes
Acknowledgments
The authors thank Dr Jon Parcher for proofreading the manuscript. The authors acknowledge Award Number P20GM104932 from the National Institute of General Medical Sciences for bioassay results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by National Center for Natural Product Research, School of Pharmacy, University of Mississippi, University, MS 38677, USA. This work was supported in part by the USDA Agricultural Research Service Specific Cooperative Agreement No. 58-6060-6-015.
