Abstract
This report concerns the identification of steroid constituents of a Far-Eastern glass sponge Aulosaccus sp. (the Sea of Okhotsk), belonging to one of the least chemically investigated group of sponges. Steroid mixtures, isolated from the extract of Aulosaccus sp., were fractionated by high-performance liquid chromatography on normal-phase and/or reversed-phase columns, and the obtained fractions were analyzed by nuclear magnetic resonance spectroscopy and gas chromatography–mass spectrometry. In result, 64 compounds, including 32 3-keto derivatives of sterols with saturated, Δ7-, Δ8(14)-, Δ4-, and Δ4,6-tetracyclic systems and 32 sterols (stanols, 4α-methyl-stanols, Δ5 -, Δ7-, 4α-methyl-Δ8(14)-sterols), were identified. Most of these steroids were not found in glass sponges earlier. The structures of 2 new steroids, 24-propyl-5α-cholest-24(28)Z-en-3-one and 24-nor-cholest-4-en-3-one, were elucidated. Sterol esters were not found. 3-Keto derivatives of stanols were major components in the steroids of Aulosaccus sp. It is probable that association of Aulosaccus sp. with bacteria, capable of oxidizing sterols, led to the transformation of a significant part of sponge sterols into their 3-keto derivatives.
Most sponges (phylum Porifera) are sessile colonial filter-feeding animals that feed on bacterioplankton and phytoplankton. These invertebrates are distributed in 4 classes: demosponges (Demospongiae), calcareous (Calcarea), homoscleromorph (Homoscleromorpha), and glass (Hexactinellida) sponges. Glass sponges may be considered as one of the least chemically investigated class of Porifera.
Different sterols are common in sponges, but 3-keto derivatives of sterols are less frequently found in these animals. 1 –3 A single comparative study of steroids from glass sponges 4 showed that hexactinellids, in general, contained Δ5-sterols, stanols, 3-keto derivatives of stanols, and Δ4-3-ketosteroids in varying proportions. In these invertebrates, sterols, as a rule, were presented at higher concentrations than their 3-keto derivatives, but sometimes these compounds were found in comparable concentrations. 4 In contrast, we observed an unusually high content of 3-keto derivatives of sterols in the steroids isolated from a Far-Eastern glass sponge Aulosaccus sp. (the Sea of Okhotsk). Although the maximum number of the 3-ketosteroids, reported earlier to occur in a hexactinellid sponge, was 4, 4 preliminary gas chromatography–mass spectrometry (GC–MS) employed by us for the tentative characterization of the 3-keto derivatives of sterols of Aulosaccus sp. showed many more components. This prompted us to obtain detailed information on the steroid composition of this sponge. In addition, steroids of glass sponges from the North-West Pacific have not been investigated previously.
The extractable material of Aulosaccus sp. was separated by column chromatography on Sephadex LH-20 and silica gel. Then, most of the obtained mixtures, containing steroids, were fractionated by high-performance liquid chromatography (HPLC) on normal-phase and/or reversed-phase columns. The obtained fractions were analyzed by nuclear magnetic resonance (NMR) spectroscopy and GC–MS (electron impact ionization). As a result, 64 compounds, namely, 32 3-keto derivatives of sterols (including the 2 new ones) and 32 sterols, were identified (Figure 1).

Steroids from Aulosaccus sp.
The least polar fraction I (Table 1) was a major steroid fraction isolated from Aulosaccus sp. A part of fraction I was separated by HPLC in the reversed-phase mode (RP-HPLC). In result, several subfractions, containing compounds
Compositions of Four Steroid Fractions Isolated from Aulosaccus sp.
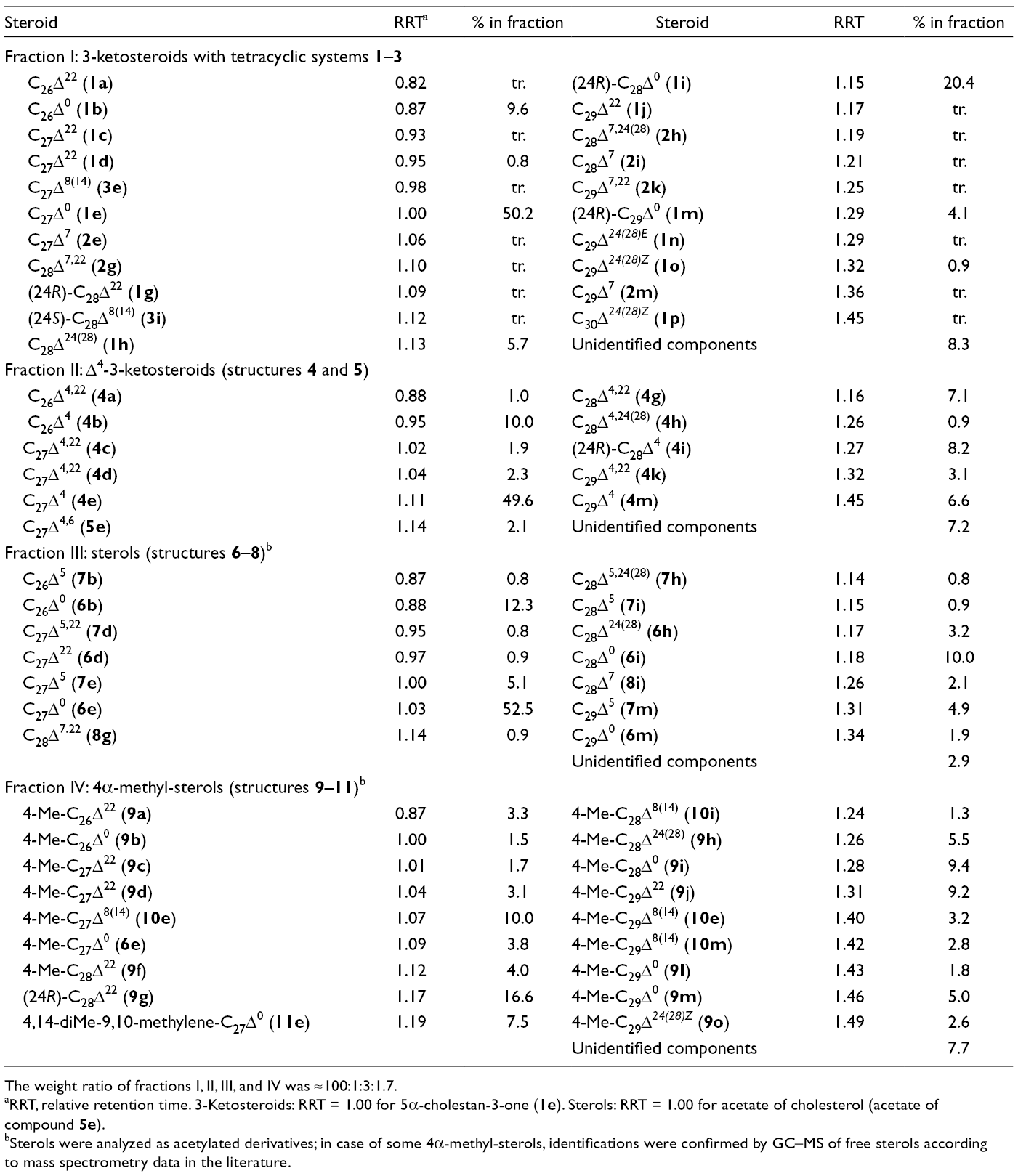
The weight ratio of fractions I, II, III, and IV was ≈100:1:3:1.7.
RRT, relative retention time. 3-Ketosteroids: RRT = 1.00 for 5α-cholestan-3-one (
Sterols were analyzed as acetylated derivatives; in case of some 4α-methyl-sterols, identifications were confirmed by GC–MS of free sterols according to mass spectrometry data in the literature.
3-Keto derivatives of stanols (
Among minor monounsaturated 3-keto derivatives of sterols (fraction I), an unknown compound
Earlier, 3-ketosteroids
In fraction I, 3-keto derivatives of Δ7- and Δ8(14)-sterols were present in trace amounts. In the mass spectra of these compounds, typical fragmentation patterns were observed according to reference spectra and GC–MS data described in Iida et al
8
and Smith and Brooks.
9
In addition, the 1H NMR spectra of subfractions, containing Δ7-components (
Fraction II (Table 1), consisting of Δ4-3-ketosteroids
1H (700 MHz, CDCl3) and 13C (176 MHz, CDCl3) NMR Spectra a of 24-Nor-3-Ketosteroids 1b and 4b.

All signals were assigned based on 1H‒1H COSY, HSQC, and HMBC experiments.
13C NMR spectrum was obtained through CH correlations in HSQC and HMBC experiments; δC of C-3 was not determined due to the paucity of compound
The mass spectrum of
Fraction III (Table 1), containing major sterols of Aulosaccus sp., was obtained using repeated column chromatography on silica gel. This fraction, analogous to fraction I, was rich in saturated components, especially in C27 compound. Namely, a high content (76.7%) of saturated sterols, including stanols
It was reported that steroids of the glass sponge Aulosaccus cf. mitsukuri (Galapagos) included stanols (54%), Δ5-sterols (4%), and 3-keto derivatives of stanols (42%). 4 Similarly, stanols found in the present study were major sterols of a related Aulosaccus sp. In addition, both the sponges of the genus Aulosaccus contained significant amounts of steroid ketones. However, the total amount of 3-keto derivatives of sterols in Aulosaccus sp. was more than 20 times higher than the total amount of its sterols (Table 1). We have observed a similar trend in our previous study on steroids from some sponges. Namely, an Antarctic demosponge Haliclona sp. was shown to contain Δ4-3-ketosteroids and Δ5-sterol esters although this invertebrate was characterized by the almost total absence of free sterols (only cholesterol was found in trace amount). 41 We tried to find sterol esters in Aulosaccus sp., but our attempt was unsuccessful. Indeed, the characteristic signals (including multiplet of esterified НС-3 group) of these compounds were not observed in the 1Н NMR spectrum of the least polar fraction, which has been expected to contain sterol esters. Probably, as a result of the intensive oxidation of sterol precursors, 3-ketosteroids became dominant in the steroids of Aulosaccus sp. whereas biosynthesis of sterol esters was blocked. Perhaps, this metabolic shift induced the substitution of sterols by their 3-keto derivatives in sponge membranes. It seems to be interesting that, in the 1Н NMR spectra of cholesterol and 5α-cholestan-3-one, the virtually indistinguishable chemical shifts of Н3С-18 (δ Н 0.681 and 0.678, respectively) and Н3С-19 (δ Н 1.009 and 1.005, respectively) may imply a certain similarity in the molecular features of these two compounds.
Our earlier study 46 indicated that Aulosaccus sp. contained significant amounts of fatty acids of microbial origin. Namely, the occurrence of some branched-chain, cyclopropane-containing fatty acids and their monoenoic precursors in Aulosaccus sp. suggested that this sponge was associated with mycobacteria or related microbes. 46 Notably, the bacteria of this group (phylum Actinobacteria) are not only known as sponge specific microorganisms, 47 but these microbes are also potent steroid degraders, particularly active in the catabolism of sterols. 48 The proposed oxidation of sterols in Aulosaccus sp. is reminiscent of oxidation by cholesterol oxidase or NAD(P)-dependent dehydrogenase, produced by a variety of Actinobacteria including Mycobacterium (see review 48 and references cited herein). These enzymes transform Δ5-sterols into 3-keto-4-enes that is considered as the first step in the sterol catabolism pathway. In addition, it was shown that cholesterol oxidase not only transformed Δ5-sterols, but also catalyzed the transformations of stanols, Δ7- and Δ8(14)-sterols into corresponding 3-ketosteroids, 9 including those found in the present study. Therefore, we suggested that association of Aulosaccus sp. with bacteria, capable of oxidizing sterols, led to the transformation of the significant part of sponge sterols into their 3-keto derivatives. Similarly, the presence of 3-ketosteroids in different sponges (as a rule, in species from cold waters of polar or temperate latitudes and in deep-water species) may be the result of the bacterial degradation of sterols. For example, such degradation may explain the presence of biologically active, hydroxylated Δ4-3-ketosteroids in a deep-water Alaska demosponge 49 because some bacteria can not only oxidize C-3 in the sterol but also introduce a hydroxy group into the sterol side chain followed by deeper transformations of steroid structure. 48
To explain the co-occurrence of Δ5-sterols, stanols, 3-keto derivatives of stanols, and Δ4-3-ketosteroids in glass sponges, a stereoselective route of Δ5-sterol conversion into 5α-stanols was proposed, where the steroid ketones were intermediates in the stanol formation. 4 In contrast to this proposal, we suggest that the 3-keto derivatives of sterols are products of catabolism rather than biosynthesis of sponge sterols. However, further studies of marine sponge-associated bacteria are needed to confirm or reject our suggestion.
Experimental
General Experimental Procedures
1H NMR, 1Н− 1Н-COSY, DEPT, HSQC, and HMBC spectra (CDCl3) were recorded on Bruker Avance III HD 500 and Bruker Avance III 700 spectrometers at 500 and 700 MHz. 13C NMR spectra (CDCl3) were recorded on Bruker Avance III 700 spectrometer at 176 MHz. GC analyses were done on an Agilent 6850 Series GC System chromatograph (Agilent, Germany) equipped with an HP-1 (Agilent technology, Inc., USA) capillary column (30 m × 0.32 mm), the carrier gas was helium (flow rate 1.7 mL/min), the detector temperature was 300°C. GC–MS analyses were carried out on a Hewlett Packard HP6890 GC System (Hewlett Packard Company, USA) with an HP-5MS (J&W Scientific, USA) capillary column (30.0 m × 0.25 mm), helium as the carrier gas, and 70 eV ionizing potential. The GC–MS analyses of acetylated sterols, free sterols and their 3-keto derivatives were done using the injector temperature of 270°C and the temperature program 100°C (1 min)–20 °C/min–270°C (30 min). Column chromatography was performed using Sephadex LH-20 (GE Healthcare, Sweden) and silica gel (50/100 µm, Sorbpolimer, Russia). HPLC separations were performed using a Du Pont Series 8800 Instrument (Du Pont Company, USA) with a RIDK-102 refractometer (Laboratorni pristroje Praha, Czechoslovakia). An Agilent ZORBAX SB-C18 column (4.6 × 150 mm; Agilent Technologies, USA) and an Altex UltrasphereTM-Si column (10 × 250 mm; Beckman Instruments, Inc., USA) were used in the HPLC separations.
Animal Material
The sponge sample of the genus Aulosaccus (phylum Porifera, class Hexactinellida, order Lyssacinosida, family Rossellidae), 930 g (wet weight), was collected in July 2011 by dredging (with a small Sigsbee trawl) from 505 m depth near Iturup Island (the Sea of Okhotsk, Kuril Islands, 45°01′5″ N, 147°00′3″ E) during a cruise onboard the r/v “Academik Oparin.” The species was identified by Dr A.L. Drozdov (National Scientific Center of Marine Biology, Far-Eastern Branch of RAS, Vladivostok, Russia). A voucher specimen (041-030) is on deposit in the collection of G.B. Elyakov Pacific Institute of Bioorganic Chemistry, Vladivostok, Russia.
Extraction and Isolation
The collected sponge (930 g, wet weight) was frozen, stored at −15°C, then cut and extracted with EtOH at room temperature. A mixture, containing cerebrosides, fatty acids, and steroids (895 mg), was isolated from the EtOH extract as described in our previous report.
50
This mixture was subjected to column chromatography (SiO2:hexane/ethyl acetate, 10:1 → 5:1). The least polar fraction (79 mg: phthalates, minor 3-ketosteroids), a sum of 3-ketosteroids
24-Propyl-5α-Cholest-24(28)Z-En-3-One (1p)
1H NMR (700 MHz, CDCl3): 0.68 (3H, s, H3C-18), 0.73 (1H, m, H-9), 0.94 (3H, d, J = 6.5 Hz, H3C-21), 0.94 (3H, t, J = 7.5 Hz, H3C-30), 0.97 (3H, d, J = 7.0 Hz, H3C-26, 27), 0.94 (3H, t, J = 7.5 Hz, H3C-30), 0.92‒2.05 (m, CH and CH2 pool), 2.02 (2H, m, H2C-29), 2.08 (1H, m, H-4a), 2.26 (1H, “pseudotriplet,” J = 14.5 Hz, H-4b), 2.29 (1H, m, H-2b), 2.38 (1H, m, H-2a), 2.80 (1H, m, H-25), 5.01 (1H, t, J = 7.0 Hz, H-28).
GC–MS: m/z (%) = 426 [M]+ (6), 411 (2), 314 (100), 299 (27), 271 (16), 258 (9), 245 (7), 231 (19), 217 (7).
24-Nor-Cholest-4-En-3-One (4b)
IR (CDCl3): 1661 (C = O) cm−1.
1H and 13C NMR (CDCl3): Table 2.
GC–MS: m/z (%) = 370 [M]+ (46), 355 (13), 328 (21), 313 (10), 299 (3), 285 (10), 271 (10), 247 (29), 229 (56), 149 (21), 147 (24), 124 (100).
Footnotes
Acknowledgments
The authors express their gratitude to Drs O.P. Moiseenko and L.P. Ponomarenko for their kind help in the GC–MS and GC analyses. The study was carried out on the equipment of the Collective Facilities Center "The Far-Eastern Center for Structural Molecular Research (NMR/MS) PIBOC FEB RAS".
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by Grant No. 17-04-00034 A from the Russian Foundation for Basic Research.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
