Abstract
Introduction:
Growing research suggests continuous glucose monitoring (CGM) may help improve glycemic outcomes in noninsulin-using people with type 2 diabetes (T2D). The continuous biofeedback from CGM provides considerable opportunities to support personalized behavior changes; however, limited research exists to describe what happens to glycemia in this population when CGM is removed. The purpose of this follow-up study is to evaluate the effects of CGM discontinuation in noninsulin-using people with T2D.
Methods:
The effects of CGM discontinuation were assessed using data from the UNITE study (NCT05928572). Phase 1 of UNITE was a two-month intervention that evaluated the impact of using a nutrition-focused approach during CGM initiation on glycemic measures in people with T2D. In Phase 2, after discontinuing CGM use for four months, blinded CGM data and other measures were collected at Follow-up and compared to data from the post-intervention (Post) period.
Results:
The percent time in range 70 to 180 mg/dL decreased from 77% in the Phase 1 Post period to 60% during the Phase 2 Follow-up period (95% confidence interval [CI] = −22%, −12%; P < .0001). Several additional glycemic metrics also worsened significantly from Post to Follow-up (P < .05). Dietary intake and exercise at Follow-up were not statistically different from Post (P > .05), but physical activity decreased (P = .01).
Conclusion:
In noninsulin-using people with T2D, glycemic measures improved with real-time CGM use, but these improvements deteriorated substantially and significantly when CGM use was discontinued. More research and more sensitive behavioral assessments are needed to better understand which factors and behavior changes may account for the glycemic decline.
Keywords
Introduction
Continuous glucose monitoring (CGM) can improve glycemic outcomes in people with type 1 diabetes and people with type 2 diabetes (T2D) who use insulin. 1 The impact of CGM in people with T2D who do not use insulin has been studied less frequently, but growing data suggest it is likely beneficial in this population as well.2-4 In noninsulin users, the continuous biofeedback from CGM can provide support for personalized behavior changes that lead to improved glycemia (eg, food choices or physical activity).5-7
The UNITE study (Using Nutrition to Improve Time in rangE), was a randomized controlled trial designed to compare the effects of using a nutrition-focused approach (NFA) or a self-directed approach (SDA) during CGM initiation on CGM metrics, dietary intake, and other outcomes in people with T2D not using insulin. 8 During the two-month UNITE intervention, all participants continuously wore a CGM sensor and used the glucose data to help guide behavior changes; they were also asked to keep their diabetes medications stable. The UNITE study showed no difference in the primary outcome of change in percent time in range (time with glucose 70-180 mg/dL [TIR70-180]) between the NFA and SDA arms (P = .14); however, both arms experienced significant within-arm improvements. The TIR70-180 increased from 46% to 71% (+6 hours) in the NFA arm and from 55% to 71% (+4 hours) in the SDA arm (both within-arm changes P < .001).
Given that a 5% (1.2 hour) increase in TIR70-180 is considered clinically beneficial, 9 these improvements were remarkably robust and could play a significant role in improving health outcomes for people with T2D, if sustained. However, the sustainability of the TIR70-180 improvements—if CGM use were discontinued in this population—is unknown. Multiple reasons for discontinuation exist, including limited insurance coverage, barriers to access, or belief that continuous access is unnecessary to maintain behavior changes.
The objective of this follow-up study was to evaluate the effects of CGM discontinuation in people with T2D who do not use insulin. More specifically, the primary focus was to understand the impact of CGM discontinuation on the change in TIR70-180 from the end of the UNITE study intervention to a follow-up four months later.
Methods
Design
The UNITE study was a randomized, two-arm, parallel-group, pilot conducted by the International Diabetes Center in Minneapolis, MN (ClinicalTrials.gov identifier NCT05928572). The UNITE study had two phases: Phase 1 included a 10-day baseline assessment, a 50-day intervention, and a 10-day post-intervention assessment (Post), while Phase 2 included a four-month nonintervention period and a 10-day follow-up assessment (Follow-up) (Figure 1).
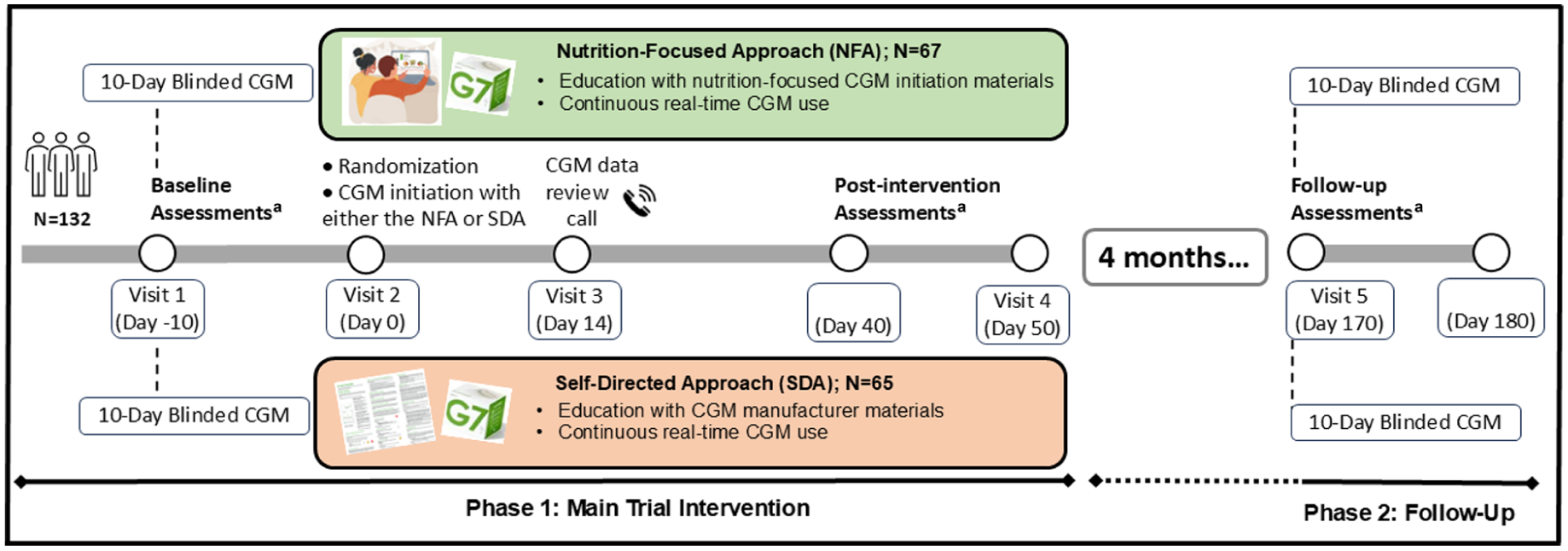
Study overview.
The purpose of Phase 1 was to compare the glycemic effects of using the NFA vs the SDA during CGM initiation in noninsulin-using people with T2D. The purpose of Phase 2 was to evaluate the effects of discontinuing the CGM for four months on glycemia, behaviors, and other outcomes regardless of baseline randomization. In Phase 2, the randomized NFA and SDA arms were consolidated into a single cohort in which all participants discontinued CGM, making the focus of Phase 2 an estimation of within-person change upon CGM discontinuation rather than a between-arm estimation of comparative treatment efficacy (as in Phase 1).
UNITE was conducted in alignment with the ethical standards described in the Helsinki declaration and according to a protocol approved by the HealthPartners Institutional Review Board. All participants provided written informed consent prior to study procedures.
Methodological details for Phase 1 of the UNITE trial are described in the primary outcome manuscript. 8 In brief, key eligibility criteria included: adults with T2D, HbA1c 7% to 10%, stable diabetes medication regimen, no current or planned use of insulin or sulfonylureas, and no recent, current, or planned use of personal CGM. At Baseline, all participants wore a CGM sensor paired with a physically blinded reader (Dexcom G7 series; San Diego, California) for up to 10 days. Once adequate CGM data were confirmed, participants were randomly assigned 1:1 to either the NFA or SDA arms. Participants in both arms were then started on an unblinded Dexcom G7 sensor paired with the G7 application on a personal smartphone. They were instructed to use CGM continuously throughout the intervention period for 50 days. On day 0, all participants received CGM initiation education specific to the NFA or SDA arms; they also completed a virtual CGM data review follow-up call, which was specific to the study arm on day 14. Participants were asked to keep diabetes medications stable throughout the intervention period. From approximately day 40 to 50 (referred to as Post), CGM data were assessed and additional outcomes measured (eg, HbA1c, dietary intake). For Phase 2, from approximately day 50 to day 170, all participants returned to their usual diabetes care providers, they were free to make changes to diabetes medications as needed but were asked to refrain from using CGM until after the follow-up assessments ended. On approximately day 170, participants returned to the clinic for a single visit. They wore another CGM sensor paired with a physically blinded reader for up to 10 days, and they completed several outcome assessments during this time (referred to as Follow-up).
Analytic Cohorts
Two cohorts were defined in the Phase 2 data. The modified intent-to-treat (mITT) cohort included all participants from the Phase 1 analysis who had sufficient CGM data at Follow-up (≥7 days with ≥70% CGM active time), whereas the per-protocol (PP) cohort was limited to those in the mITT cohort who (1) had not used a personal CGM between the Post and Follow-up periods and (2) had not made any changes to their glucose-lowering medication regimen. The PP cohort was selected as the primary analytic cohort in order to derive estimates of glycemic changes after removing CGM support, which were not related to glucose-lowering medication adjustments. However, for completeness, analysis was also performed in the mITT cohort, which reflects the common experience of changes to glucose-lowering medications in people with T2D.
Study Outcomes and Measurements
The primary outcome for Phase 2 was change in CGM-derived TIR70-180 from Post to Follow-up for the combined NFA and SDA arms. During the 10-day outcome assessment windows, all participants were instructed not to make any changes to eating, physical activity, or medication-taking behaviors. The CGM data were assessed using data downloaded from Dexcom Clarity’s cloud-based repository, and efforts were made to follow the best practices described for CGM data use in clinical trials. 10 Exploratory outcomes included changes from Post to Follow-up in: (1) additional CGM-derived metrics, (2) HbA1c, (3) dietary intake parameters including total energy, macro- and micro-nutrient assessment, and total Healthy Eating Index-2015 (HEI) scores 11 using data derived from the National Cancer Institute’s Automated Self-Administered 24-hour recall system (ASA24) 12 and HEI scores calculated with publicly available code, 13 (4) weight and body mass index (BMI), (5) diabetes medication use, (6) patient-reported outcomes and behaviors including diabetes distress measured by the 17-item Diabetes Distress Scale (DDS-17) with total scores and sub-scale scores calculated using established methods, 14 dietary adherence (diet quality) measured by a single validated question, 15 physical activity/exercise assessment based on Toobert et al, 16 and study-specific survey questions to assess general dietary changes and self-efficacy. Finally, change from Baseline to Follow-up in CGM-derived metrics, both overall and by randomized treatment arm (NFA or SDA), were estimated.
Sample Size
Two sample size calculations were made for the two analytic cohorts planned in Phase 2. We estimated 90% of participants from the Phase 1 cohort (90% of 60, or N = 54 per arm) would complete Phase 2 with adequate CGM data at Follow-up and therefore be eligible for the mITT cohort. For the PP cohort, this percentage was down-adjusted to 85% (N = 51 per arm). An analytic sample of N = 54 per arm (mITT) was estimated to provide 80% power (2-sided α = 0.05) to detect an absolute change in pooled study arms of 0.27 standard deviation units of change in CGM-derived TIR70-180 from Post to Follow-up and an absolute change within study arm of 0.39 standard deviations of TIR Post to Follow-up. The actual pooled standard deviation of TIR70-180 post-intervention was 22, meaning the minimum detectable difference in TIR70-180 was 22*0.27 = 5.9% and 22*0.39 = 8.6% change in CGM-derived TIR70-180, respectively. The mITT cohort included N = 50 to 52 per arm, or approximately the projected numbers, and the PP cohort included N = 35 per arm, which is below the projected estimate and mainly due to exclusion based on medication changes.
Statistical Analysis
Participant demographics, clinical measurements, and use of glucose-lowering medications at Baseline, Post, and Follow-up are summarized with descriptive statistics appropriate to the data (eg, mean and SD for continuous variables, and count and percent for categorical variables). The analysis of continuous variables measured at any of the Baseline, Post, and Follow-up timepoints, specifically CGM-derived TIR70-180 and related metrics, utilized general linear mixed models (GLMM) with unstructured means to model repeated measures over time. Data were assumed missing at random; these models utilized all available data at all timepoints and used maximum likelihood-based ignorable methods, which yield valid inference for outcomes missing at random.
The key analytic test for the primary endpoint was a planned contrast estimating change in CGM-derived TIR70-180 from Post to Follow-up with study-arm data pooled, to be estimated in both PP and mITT cohorts. It was expected that TIR70-180 would decline during this time in both cohorts. With respect to the exploratory measures, model-based means and confidence intervals (CIs) were estimated at both Post and Follow-up time points, for both study arms, and for change within study arm. The same strategy was applied to other CGM-derived metrics, dietary intake, and other endpoints. The analysis of binary outcomes measured at Post and Follow-up (eg, % reaching TIR70-180 >70%) utilized a GLMM framework (with logit link and binary error distribution and a random person-specific intercept), and these results are reported as odds ratios (ORs) and ratios of ORs with corresponding CIs to quantify the difference in differences (DIDs) contrasts of interest. To model the change from Baseline to Follow-up, the GLMM framework with unstructured means described above was used, with the addition of a Baseline timepoint and accompanying data.
Analysis of the ASA24-derived total HEI scores mimics the process described in Phase 1, 8 wherein each study arm and timepoint (here, Post and Follow-up) was analyzed separately.
Neither subgroup analyses nor analyses to assess heterogeneity of treatment effects were planned or performed. All statistical testing was done with α = 0.05, and no adjustments for multiple comparisons were made. Analysis was conducted with SAS v9.4 (SAS Institute, Cary, North Carolina).
Results
Participants were recruited from July 2023 to June 2024, and the last participant completed the final follow-up assessment in December 2024. In the Phase 1 intervention, N = 132 participants were randomized to either NFA (N = 67) or SDA (N = 65), and N = 124 were included in the final analytic cohort. In this Phase 2 Follow-up, N = 102 met criteria for the mITT analysis, and N = 70 met criteria for the PP analysis (Figure 2). Thirty-two participants were removed from the mITT analysis due to changes in either class or dose of diabetes medications or use of CGM (N = 2) between the Post and Follow-up assessments. Participant characteristics for both analytical cohorts are presented in Table 1.
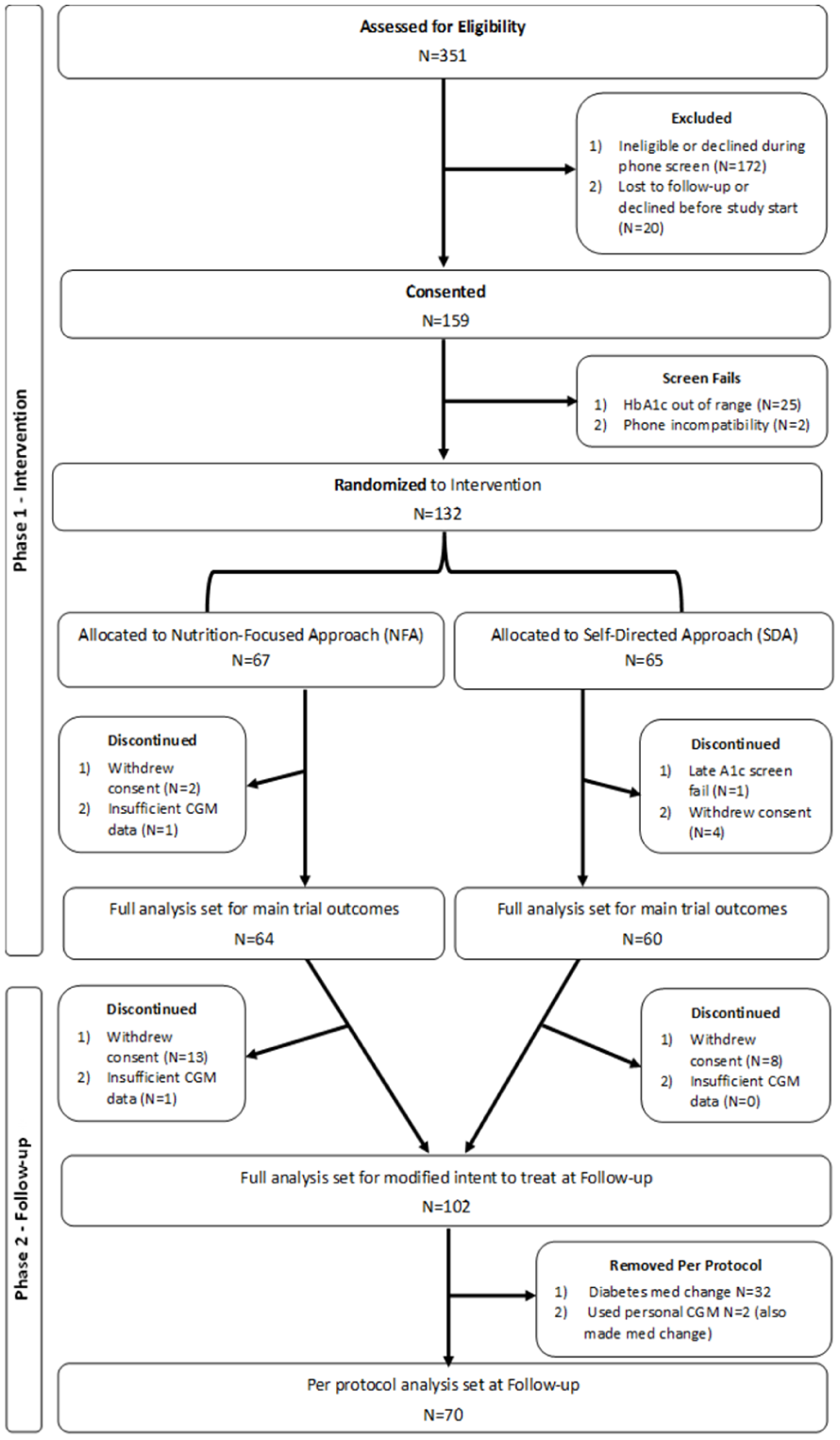
CONSORT diagram.
Participant Characteristics by Analytical Cohort at Baseline.
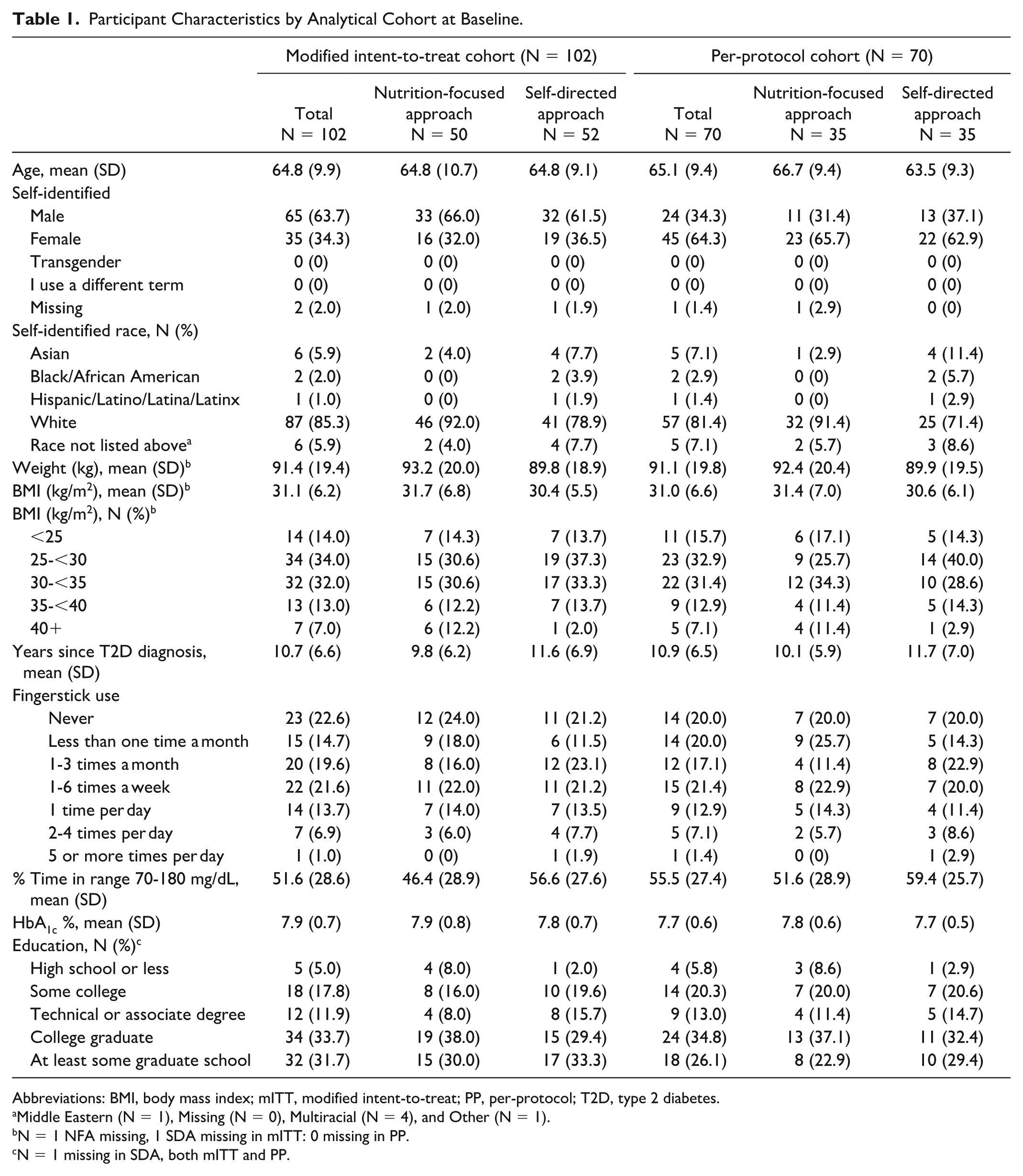
Abbreviations: BMI, body mass index; mITT, modified intent-to-treat; PP, per-protocol; T2D, type 2 diabetes.
Middle Eastern (N = 1), Missing (N = 0), Multiracial (N = 4), and Other (N = 1).
N = 1 NFA missing, 1 SDA missing in mITT: 0 missing in PP.
N = 1 missing in SDA, both mITT and PP.
Glycemic Outcomes
The primary outcome of TIR70-180 decreased from 77% at Post to 60% at Follow-up (−4 hours) for all participants in the PP cohort (−17% [95% CI = −22%, −12%]; P < .0001) (Table 2 and Figure 3). The Post to Follow-up change in TIR70-180 was significant, but attenuated, for participants who added medications and/or used CGM in the mITT cohort (−11%, [−18, −5]; P < .001). Additional glycemic metrics, including time above range >180 mg/dL, time above range >250 mg/dL, mean sensor glucose, coefficient of variation, Glycemic Management Indicator, and HbA1c also increased significantly from Post to Follow-up in both the PP and mITT cohorts (Table 2 and Supplementary Table 1). Likewise, time in tight range 70 to 140 mg/dL decreased and the percent of participants reaching TIR70-180 >70% dropped from 71% in the Post period to 40% in the Follow-up period (all P < .05). Result patterns from Post to Follow-up were nearly identical when looking at glycemic changes within each randomized study arm (data not shown); no differential changes by arm were detected.
Glycemic Outcomes.
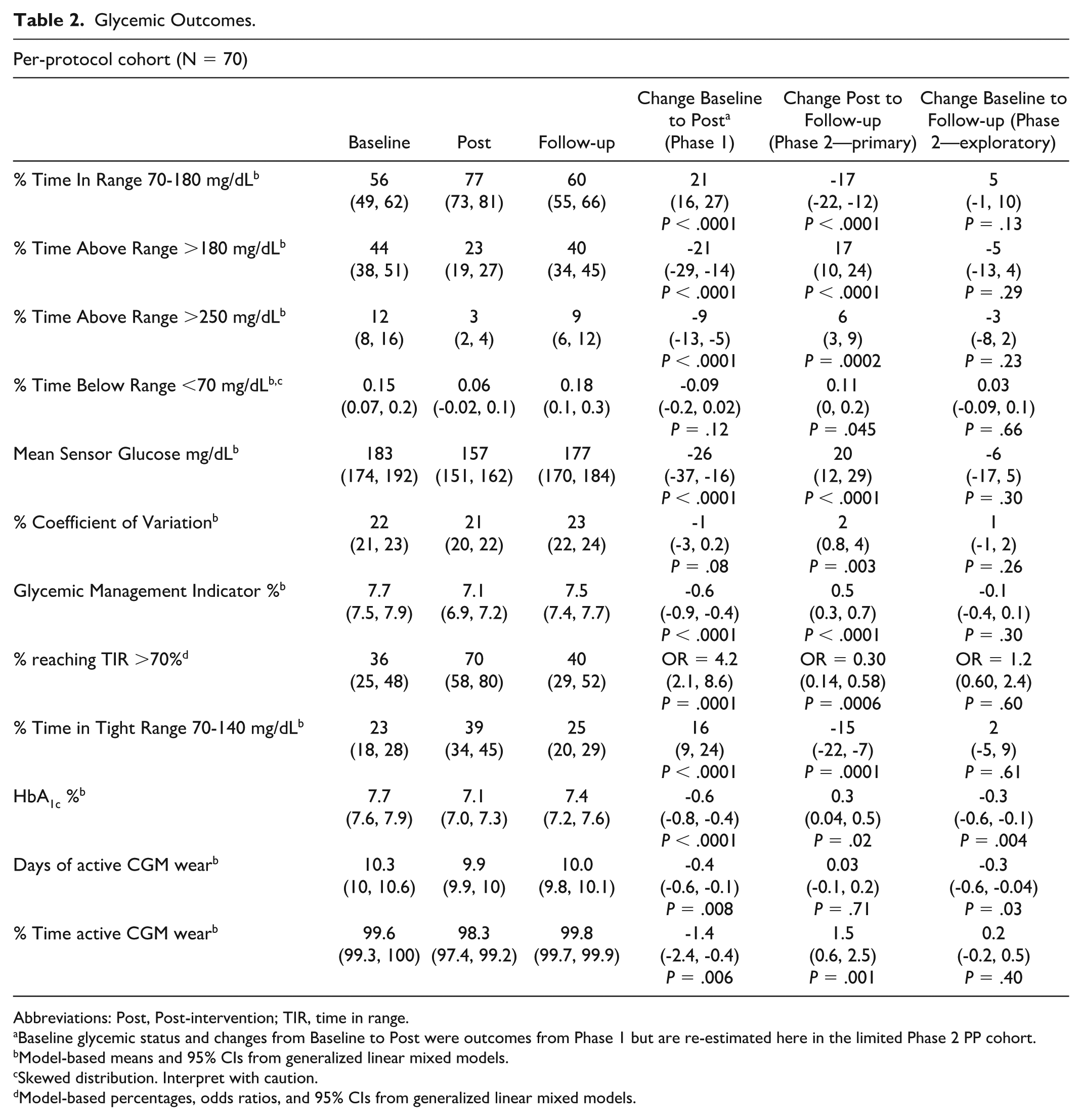
Abbreviations: Post, Post-intervention; TIR, time in range.
Baseline glycemic status and changes from Baseline to Post were outcomes from Phase 1 but are re-estimated here in the limited Phase 2 PP cohort.
Model-based means and 95% CIs from generalized linear mixed models.
Skewed distribution. Interpret with caution.
Model-based percentages, odds ratios, and 95% CIs from generalized linear mixed models.
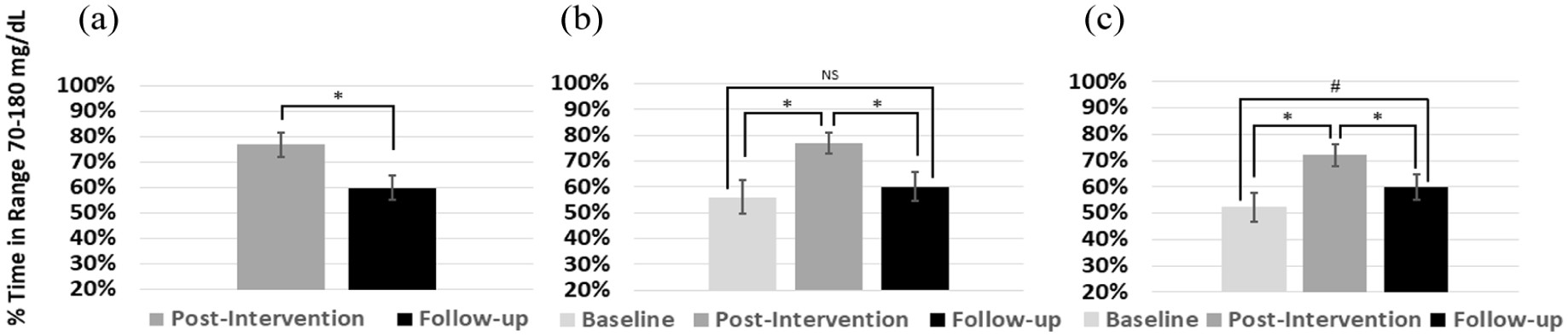
TIR70-180 with and without CGM use. (a) Impact of CGM discontinuation shown as the change in TIR70-180 from Post to Follow-up in the per-protocol cohort (primary outcome). (b) Progression of TIR70-180 from Baseline to Post to Follow-up in the per-protocol cohort (exploratory outcome). (c) Progression of TIR70-180 from Baseline to Post to Follow-up in the modified intent-to-treat cohort (exploratory outcome). NS represents non-significant difference.
To further understand how much of the original glycemic improvements were maintained relative to the pre-intervention Baseline values, differences between the Baseline and Follow-up periods in the pooled cohort were also estimated as an exploratory outcome. In the PP cohort, none of the CGM-derived measures at Follow-up were significantly different from those at Baseline (P > .05), indicating that the positive effects of CGM on glycemic metrics were lost in the four months post-intervention. The one glycemic exception was HbA1c, which despite a significant increase from Post to Follow-up (0.3%, [0.04%, 0.5%], P = .02), was still significantly lower at Follow-up than Baseline (−0.3%, [−0.6%, −0.1%] P = .004). In the mITT cohort, nearly all CGM-derived measures at Follow-up remained improved over those at Baseline (P < .05), indicating that adjusting glucose-lowering medications or continuing CGM use attenuated glycemic deterioration (Supplementary Table 1).
Dietary Intake and Body Weight Outcomes
Dietary intake measured as total energy, macro- and select micro-nutrients did not differ from Post to Follow-up based on analyses of the PP or mITT cohorts, or with respect to either within- or between-arm changes, P < .05 for all comparisons (Table 3; PP only). Total HEI scores were evaluated based only on original study arm assignment; scores did not differ from each other in the Follow-up period based on overlapping confidence intervals (HEI scores at Follow-up [mean (CI)]): NFA: 60 (54, 65) and SDA: 64 (59, 68). The HEI scores also did not change within each arm from Post to Follow-up.
Dietary Intake.
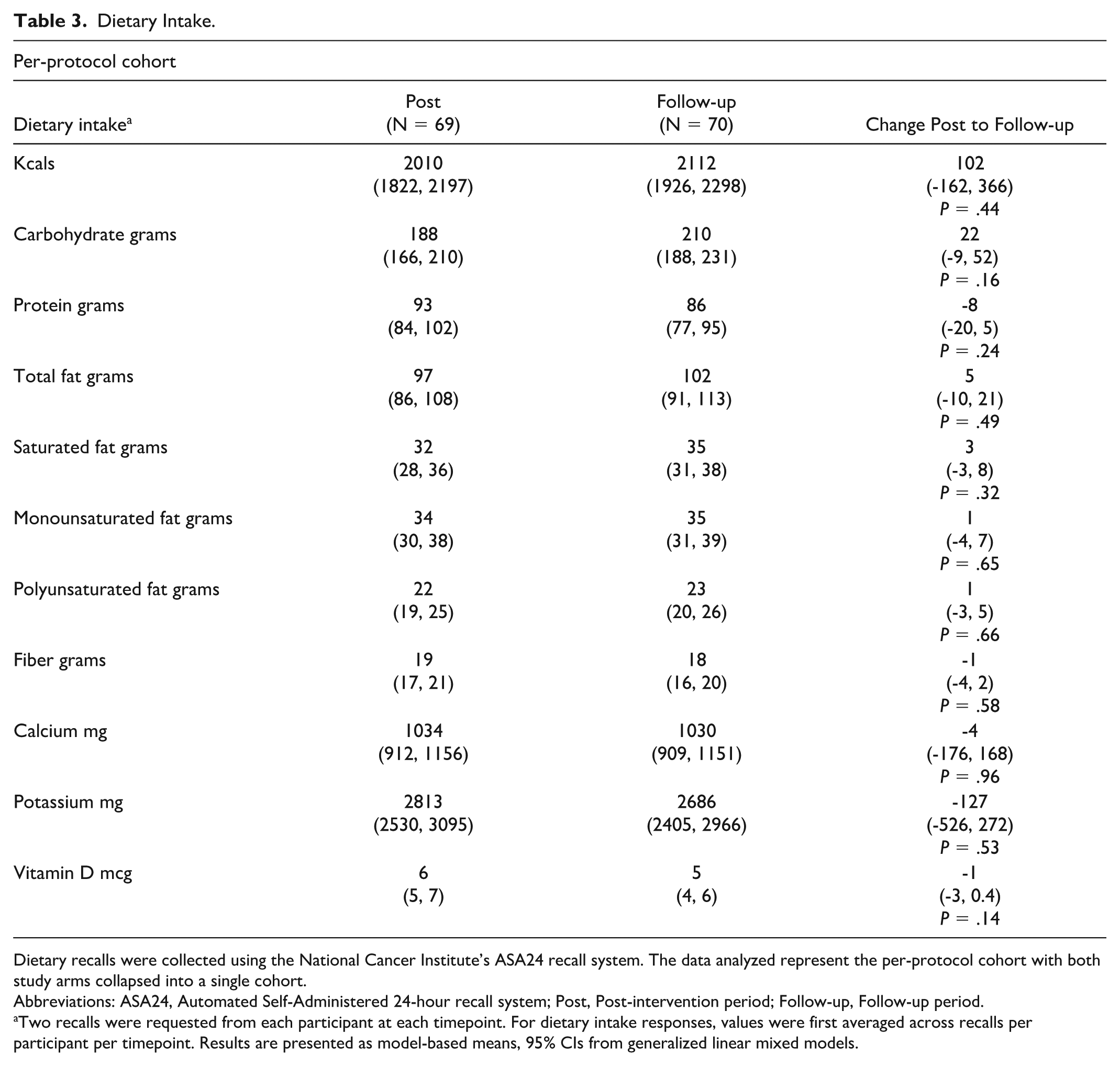
Dietary recalls were collected using the National Cancer Institute’s ASA24 recall system. The data analyzed represent the per-protocol cohort with both study arms collapsed into a single cohort.
Abbreviations: ASA24, Automated Self-Administered 24-hour recall system; Post, Post-intervention period; Follow-up, Follow-up period.
Two recalls were requested from each participant at each timepoint. For dietary intake responses, values were first averaged across recalls per participant per timepoint. Results are presented as model-based means, 95% CIs from generalized linear mixed models.
Body weight and BMI did not differ significantly from Post to Follow-up based on the PP or mITT cohorts (data not shown).
Diabetes Medication Use
Between Post and Follow-up, N = 32 participants (31%) in the mITT cohort added, discontinued, and/or changed a dose of a glucose-lowering medication (Table 4). Of the changes made, most intensified glucose-lowering medication use (N = 23), with slightly more participants adding glucagon-like peptide-1 receptor agonist (GLP-1 RA) therapy than other medications between Post and Follow-up. Of note, GLP-1 RA use in the Post period was slightly lower for UNITE participants than would be expected based on national trends during the same time—about 20% of participants were taking GLP-1 RAs in the Post period and national estimates suggest 26.5% GLP-1 RA use for adults with diabetes, 17 but UNITE study participant use of GLP-1 RAs increased to the national average during Follow-up. No glucose-lowering medication changes were made between Post and Follow-up in the PP cohort.
Glucose-Lowering Medication Use by Count and Class and by Study Arm and Period in mITT Cohort.
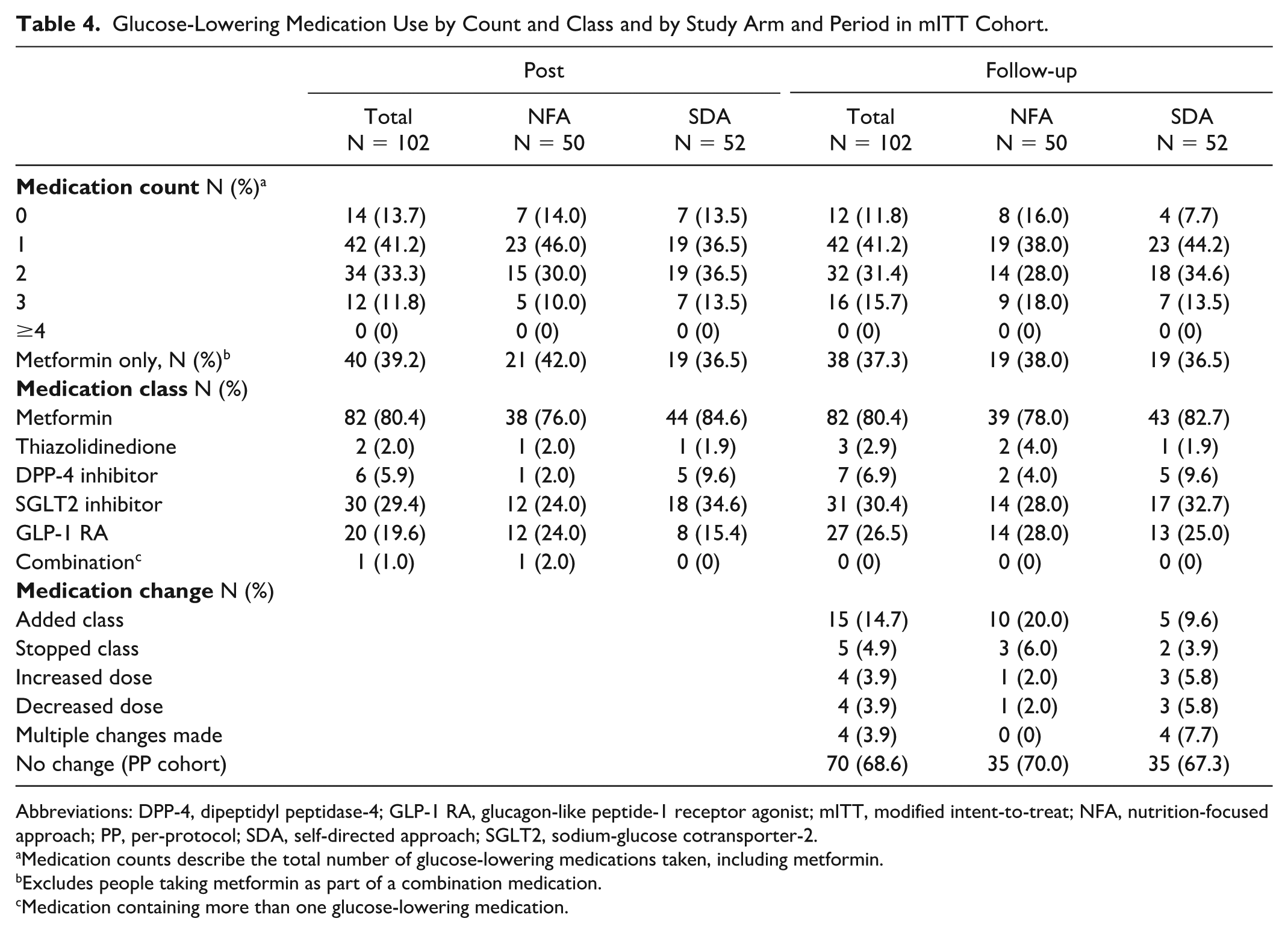
Abbreviations: DPP-4, dipeptidyl peptidase-4; GLP-1 RA, glucagon-like peptide-1 receptor agonist; mITT, modified intent-to-treat; NFA, nutrition-focused approach; PP, per-protocol; SDA, self-directed approach; SGLT2, sodium-glucose cotransporter-2.
Medication counts describe the total number of glucose-lowering medications taken, including metformin.
Excludes people taking metformin as part of a combination medication.
Medication containing more than one glucose-lowering medication.
Patient-Reported Outcomes and Behaviors
Total and sub-scale DDS-17 scores did not differ from Post to Follow-up (data not shown).
Self-reported dietary adherence (diet quality), based on the question, “In general, how healthy is your overall diet?” did not differ from Post to Follow-up overall or within original study arm (all P > .05). However, more than one third of participants in the PP cohort reported changing the amount (31%) or the type (34%) of food they consumed during the Post to Follow-up period. Only N = 21 (30%) participants reported feeling “very confident” that they could “choose foods that help keep glucose in target” during Follow-up (Table 5).
Participant Confidence (Self-Efficacy) With Making Food Choices During Follow-up.
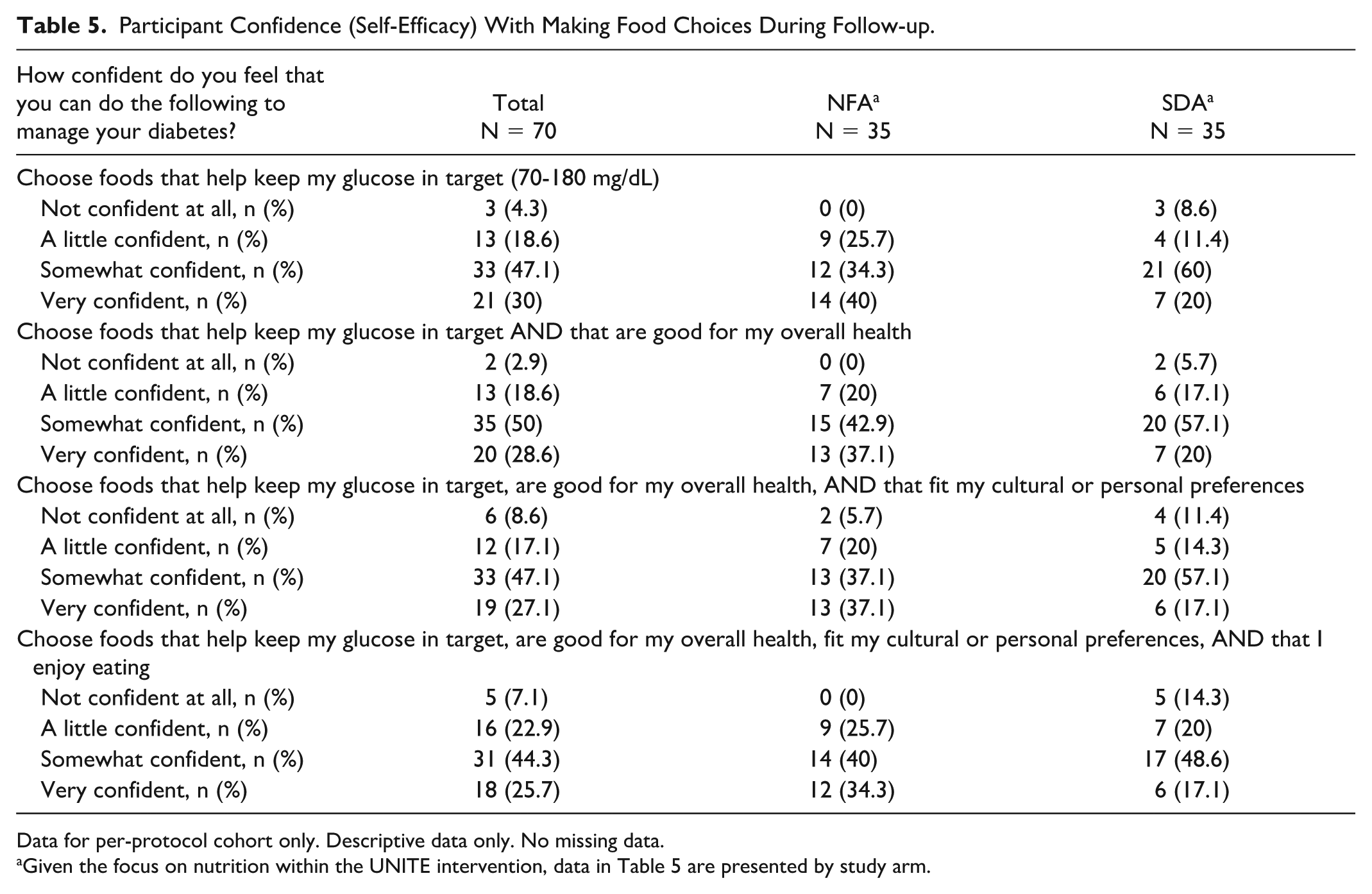
Data for per-protocol cohort only. Descriptive data only. No missing data.
Given the focus on nutrition within the UNITE intervention, data in Table 5 are presented by study arm.
Self-reported seven-day estimates of physical activity and exercise were assessed. The mean number of days of physical activity significantly decreased from Post to Follow-up in both the PP and mITT cohorts (from 4 days to 3 days, P = .01), while the mean number of days of exercise appeared stable between the two periods (~3 days).
Discussion
In Phase 1 of the UNITE study, noninsulin-using adults with T2D experienced significant improvements in TIR70-180 when using real-time CGM as a means to help improve their glycemic status. 8 In this Phase 2 Follow-up, which evaluated the four-month period after the active UNITE intervention ended, TIR70-180 significantly decreased in the PP cohort, from approximately 77% to 60% when CGM use was discontinued. The 17 percentage-point decline in TIR70-180 represents the loss of most of the original TIR70-180 gains; in fact, exploratory analyses confirmed that TIR70-180 at Follow-up was not significantly different from Baseline in the PP cohort. However, the same analysis in the mITT cohort showed that TIR70-180 at Follow-up remained significantly improved relative to Baseline, which is likely explained by the fact that most mITT participants intensified glucose-lowering medications and some also used a personal CGM during Phase 2.
Very few studies have evaluated the glycemic effects of CGM discontinuation in people with T2D, and the ones available report mixed results using primarily HbA1c data. The most robust data come from the MOBILE study, which was a US-based real-time CGM intervention in a diverse sample of people with T2D using basal insulin. 18 MOBILE originally randomized participants to blood glucose monitoring or CGM use for eight months and included a six-month follow-up period where participants were re-randomized to either continue or discontinue CGM. 19 The MOBILE follow-up study estimated that CGM discontinuation resulted in a loss of about 50% of the TIR70-180 improvements observed during the intervention. It is possible that MOBILE study participants may have retained more of their TIR gains than UNITE study participants because of the role CGM played in optimizing basal insulin dosing and encouraging medication adherence during their CGM use. In a smaller study of noninsulin-using people with T2D in Japan, Wada et al 20 reported that the glycemic benefits from using an intermittently scanned CGM for 12 weeks persisted for 12 weeks after CGM use was discontinued, but this was based on HbA1c, which provides an incomplete picture of glycemic changes. In another small study, Moon et al 21 evaluated the effect of intermittent, short-term CGM use in noninsulin-using people with T2D in Korea. In this study, all participants received a single session of lifestyle education based on six days of blinded CGM data; they were then randomized into one of three arms for six months: (1) blood glucose monitoring (control), (2) single week-long session of real-time CGM immediately post-randomization, or (3) two week-long sessions of real-time CGM, one immediately post-randomization and another 12 weeks later. Results showed the HbA1c improvement was not sustained from three to six months in the arm with only one real-time CGM session, but it was maintained in the arm with two real-time CGM sessions if participants also performed ≥1.5 blood glucose tests per day. Unfortunately, among these three studies, two did not assess dietary or lifestyle behavior changes19,20 and one used brief self-care behavior surveys, 21 which makes it difficult to interpret what factors may have changed or contributed to these mixed results.
The UNITE study assessed behavior changes more thoroughly than previous studies; however, even with high-quality dietary assessments and questions to assess physical activity and exercise, the behavioral data offered little to explain the glycemic decline seen when CGM was removed. It is likely that more sensitive dietary and behavioral assessment methods are needed to understand what drives the observed outcomes. For example, the ASA24 dietary recall system used in UNITE was designed to provide an accurate estimate of total energy and macro/micro-nutrient intake, but not to account for nuances in eating behavior that may also influence glycemia, such as the order foods are consumed, 22 specific protein-carbohydrate food pairings, 23 or eating speed. 24 Similarly, questions about exercise and physical activity over seven days may pick up large changes in activity but could miss smaller changes in physical behaviors that may also impact glucose, such as activity time-of-day 25 or proximity of activity relative to a meal. 26 It is plausible that access to the real-time biofeedback from CGM leads users to identify and adopt several small, and potentially even subconscious, lifestyle changes that are not detectable with current methods but which may meaningfully contribute to improved glycemia. As indicated in our study and elsewhere,5,6 efforts are needed to reduce the gap in knowledge explaining how specific CGM-related behavior changes contribute to improved glycemia.
Current guidance recommends insulin-using people with diabetes should use real-time CGM daily; 1 however, guidance on frequency of CGM use for noninsulin-using people with T2D is unclear. Some guidance has proposed that intermittent use of CGM every three months may be sufficient for noninsulin-using people with T2D; 27 however, the currently available research does not seem to support this and rather suggests that continuous access and use could be key for maintaining glycemic benefit. The role of continuous biological feedback to motivate behavior changes has been described, but effective strategies for implementing and using behavior change techniques are still needed. 7
These UNITE study data add to the small body of literature evaluating the impact of CGM discontinuation, but further research is certainly needed; for example, studies specifically designed to identify the optimal duration and interval of CGM use needed to achieve and maintain glycemic goals in a diverse population of noninsulin-using people. Larger, full-scale trials that could allow for mediation analyses to better explain how specific behaviors (or behavior changes) may contribute to glycemia are critical. In addition, research with more robust dietary and behavior assessments with and without CGM use would also be beneficial.
This study had several strengths including high-quality CGM data, careful diabetes medication tracking, and use of several validated patient-reported and behavioral outcome assessments. Limitations to this research include the lack of a comparison arm that continued CGM during follow-up in order to demonstrate the effects of sustained CGM use within a randomized controlled trial design; a highly white, non-Hispanic, male population, which limits generalizability of the findings; and no objective assessment of physical activity (eg, accelerometer).
Conclusions
In noninsulin-using people with T2D, TIR70-180 and other glycemic metrics improved with real-time CGM use, but these improvements deteriorated substantially and significantly after CGM use was stopped. Given the glycemic benefits shown during CGM access and the sharp declines once the CGM was removed, these data further support the use of CGM for noninsulin-using people with T2D. More research on the impact of CGM discontinuation in this population is needed, as are more sensitive behavioral assessments which could better define the dietary and behavior changes that account for glycemic declines.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251404496 – Supplemental material for Effects of Continuous Glucose Monitoring Discontinuation in Adults With Type 2 Diabetes Not Using Insulin
Supplemental material, sj-docx-1-dst-10.1177_19322968251404496 for Effects of Continuous Glucose Monitoring Discontinuation in Adults With Type 2 Diabetes Not Using Insulin by Holly J. Willis, Sally K. Gustafson, Elizabeth Johnson, Meghan M. JaKa and Richard M. Bergenstal in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank the participants who gave their time to this study. The authors also thank the research assistants, lab technicians, and study coordinators who diligently followed every participant. The authors specially thank Kelsea Forrester, MS, RD (study coordinator) for her careful monitoring of the UNITE study data as well as her thoughtful review of this manuscript.
Abbreviations
ASA24, Automated self-administered 24-hour recall; BMI, body mass index; CIs, confidence intervals; CGM, continuous glucose monitoring; CV, coefficient of variation; DDS-17, diabetes distress scale-17; DID, difference in differences; GLMM, generalized linear mixed model; GMI, glucose management indicator; HEI, healthy eating index; mITT, modified intent to treat; NFA, nutrition-focused approach; OR, odds ratio; Post, post-intervention period; PP, per-protocol; SD, standard deviations; SDA, self-directed approach; TAR180, percent time with glucose >180 mg/dL; TAR250, percent time with glucose >250 mg/dL; TIR70-180, percent time with glucose 70-180 mg/dL; TITR70-140, percent time with glucose 70-140 mg/dL T2D, type 2 diabetes; UNITE, Using Nutrition to Improve Time in rangE.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: HJW consulted with and received educational grant funds from Abbott Diabetes Care, she also consulted with Dexcom, and received research grant funding and study supplies from Abbott Diabetes Care and Dexcom during the conduct of this study. HJW received no personal payment, all payments went to the non-profit, HealthPartners Institute. SKG has no conflicts of interest to report. MMJ has no conflicts of interest to report. EJ received educational grant funds from Abbott Diabetes Care and was a speaker for Dexcom and Sequel; EJ received no personal payment, all payments went to the non-profit, HealthPartners Institute. RMB has received research support, has acted as a consultant, or has been on the scientific advisory board for Abbott Diabetes Care, Ascensia, Amgen, CeQur, Dexcom, Eli Lilly, Insulet, Luna Health, Medtronic, Novo Nordisk, Roche-Genentech, Sanofi, Tandem Diabetes Care, Vertex Pharmaceuticals, and Zealand Pharma. RMB’s employer HealthPartners Institute contracts for his services as a research investigator and/or consultant. No personal income from any of these services goes to RMB.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is the result of work originally supported by American Diabetes Association grant #7-22-JDFN-27. Funding for the Phase 2 research presented in this manuscript was provided by Dexcom Inc. and the Park Nicollet Foundation. All glucose monitoring supplies were provided by Dexcom Inc. The funding entities played no role in the study design, execution, results interpretation, or any decisions related to data dissemination.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
