Abstract
Background:
Foot temperature monitoring can prevent diabetic foot ulceration (DFU). This technical report determined the validity of the Feetsee device (InvCam) in measuring temperature differences between feet compared with current reference standard: FlirA615, with Food and Drug Administration-approved TotalVision software (RefCam).
Methods:
Measurements were taken in 107 participants (mean age: 48 years; 49% female) across three groups: people with diabetes and active foot ulcer (DFU, n = 38; 57 ± 12.2 years), diabetes without ulcers (diabetes mellitus DM, n = 35; 51.5 ± 16.3 years), and healthy young controls without diabetes (C, n = 34; 34.2 ± 10.7 years).
Results:
Strong agreement was shown in foot temperature measurements between the devices with no systematic bias, an intraclass correlation coefficient of 0.95 and typical error of 0.31 deg.
Conclusions:
The InvCam system demonstrated validity for foot temperature measurement compared with the current reference standard and offers the advantage over current systems of being non-contact, eliminating the risk of infection and cross-contamination.
Introduction
The development of diabetic foot ulcers (DFU) is associated with an increased foot temperature prior to tissue breakdown.1,2 Foot temperature monitoring is known to be an effective strategy for DFU prevention. 3 - 5
Previous studies3,4 showing the efficacy for DFU prevention have used digital temperature probes and while effective, only measure discrete points/areas on the foot and risk missing potential regions of concern. Testing of thermistor sensors embedded in Siren socks (San Francisco) in a thermostatic water-bath demonstrated an accuracy of ±0.2°C compared with a reference standard across six sensors located at common ulcer sites. 6
Other systems include temperature “mats” (Podimetrics SmartMat, Somerville) where patients stands on the digital mat. This system has been shown as effective for identifying differences between feet with a sensitivity ranging between 50% and 97% and specificity between 43% and 81%. 7
Smartphone-based IR modules offer affordability and portability; however, their performance varies significantly. While earlier studies reported ICCs of 0.981 to 0.987 for a FLIR One attachment compared with a high-end camera, 8 a more recent study highlights differences in temporal and spatial temperature stability. 9 Non-contact infrared thermography systems, such as FLIR-based handheld cameras, Meditherm systems, and various research-grade IRT platforms, face notable limitations, including image quality and measurement accuracy being affected by camera-to-foot distance, viewing angle, ambient reflections and inconsistent patient positioning, impacting reproducibility and sensitivity.
A non-contact, whole-foot measurement device represents a major step-forward for clinical assessment of the diabetic foot and DFU prevention, removing infection risk associated with contact procedures, and comprehensive coverage of the whole-foot area. This system incorporates a dedicated stand to standardize foot placement, fixed imaging geometry to minimize variability and asymmetry-based analysis algorithm—thereby enhancing sensitivity to clinically meaningful temperature differences.
Here, we test the validity of a new method for non-contact temperature measurement of the whole foot, the Feetsee device (InvCam) against the acknowledged “reference standard” thermal camera FlirA615, with FDA approved TotalVision software (RefCam).
We hypothesize that the InvCam system provides valid measurements of inter-foot temperature asymmetry in comparison to the reference standard RefCam, across participants groups.
Materials and Methods
Study Participants
This cross-sectional study included measurements taken from 107 participants (48.6% female overall) at Vilnius University Teaching Hospital “Santaros Klinikos.” Participants were recruited into one of three groups: (1) active diabetic foot ulcer (DFU); (2) diabetes, no foot ulcer (DM); and (3) healthy young controls without diabetes (C). The DFU group were older (mean age 57 ± 12.2 years) compared with the DM (51.5 ± 16.3 years) and young controls (34.2 ± 10.7 years). Compared with the DM group, DFU participants had a longer diabetes duration and higher prevalence of peripheral neuropathy (Table 1). No other inclusion/exclusion criteria were applied.
Study Participants Characteristics.
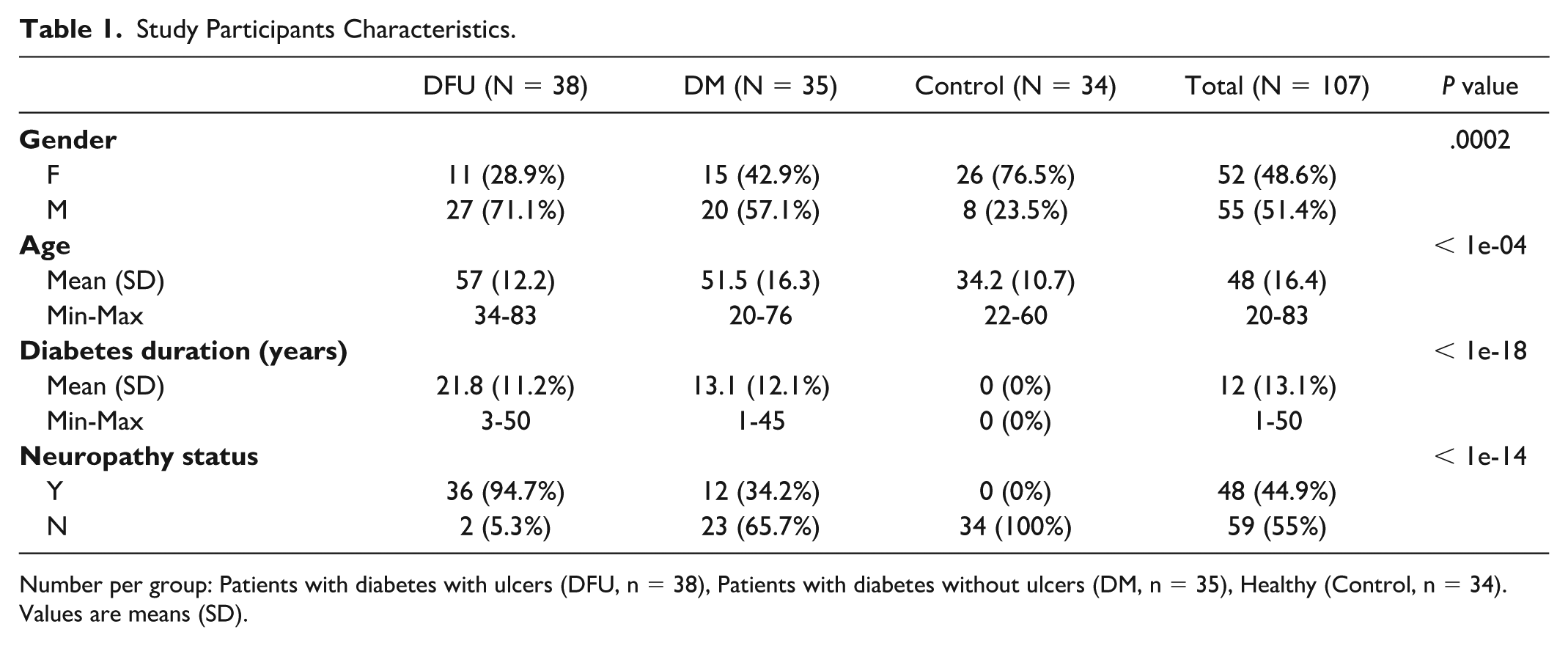
Number per group: Patients with diabetes with ulcers (DFU, n = 38), Patients with diabetes without ulcers (DM, n = 35), Healthy (Control, n = 34). Values are means (SD).
Participants provided written informed consent. The study protocol was approved by the Vilnius Regional Biomedical Research Ethics Committee (No. 2019/12-1172-660) and is registered at ClinicalTrials.gov (NCT05222490).
Experimental Set-up
The experimental set-up for both systems is shown in Figure 1. Two thermographic devices have been used to acquire thermograms: InvCam (Feetsee camera together with Feetsee diagnostic software model SW v1.01, Diabetis JSC, Vilnius, Lithuania; Figure 1b) and RefCam (Teledyne FLIR, Inc., Wilsonville, Oregon, with TotalVision medical thermography software v3.1.1.0 Med-Hot Thermal Imaging, Inc., Lakeland, Florida; Figure 1a). Table 2 highlights the differences in the level of resolution and other key parameters between the cameras.

Thermal measurement experimental set-up with (a) RefCam and (b) InvCam device.
Key Parameters for Both Cameras Used in the Study.

The InvCam was used as a self-check device placed on an accessory stand and RefCam was mounted on a tripod as shown in Figure 1. The distance between the feet and camera was ~30 cm for InvCam and ~45 cm for RefCam because of a narrower Field of View for RefCam (see appendix for calculations).
During measurement, feet were captured so that they covered ~100px in foot length and ~40px in feet width on the InvCam camera, and ~400px in foot length and ~160px in feet width on the RefCam. Therefore, the foot area on the InvCam device covered 100*40*0.75=3000px, and 400*160*0.75=48000px on RefCam. All pixels are used to evaluate the foot, since the convexity does not affect the results of the thermal imagery. Every pixel in the thermal sensor corresponds to the foot cell of approximate size 3 × 3 mm on InvCam and 0.75 × 0.75 mm on RefCam. Physical units were estimated according to the camera’s Field of View and distances to the object.
Temperature Measurement Method With the InvCam
InvCam is a non-contact whole-foot thermography system designed for diabetic foot monitoring. It captures high-resolution plantar thermal images in less than five seconds, combined with optical imaging for automated contour detection. Standardized foot positioning is ensured using a dedicated stand, while fixed imaging geometry minimizes measurement variability. The device outputs a standardized temperature map, enabling inter-foot asymmetry analysis, a validated predictor of DFU risk. InvCam algorithm tests in a laboratory setting demonstrated 95.8% accuracy for foot outline detection and 94.3% accuracy for hot spot detection. 10
This work was designed to test temperature asymmetry zones between feet using a temperature map of foot shape divided into 750 cells. A cell size 1/100 of foot height was used approximately corresponding to 2.5 mm, or 4 pixels for a typical thermal foot image. Values of pixels falling into one cell are averaged. For each foot, we calculated the temperature difference by subtracting the values of the temperature map of one foot from the corresponding values on the opposite foot.
Statistical Methods
One-way ANOVA with Tukey post hoc test was used to test differences in demographics between participant groups (DFU, DM, and controls). To test the agreement between devices, differences were compared within each subgroup (DFU, DM, and C) using intraclass correlation coefficient (ICC 2,1) and the Bland and Altman Limits of Agreement. Typical error temperature values were calculated between the devices according to formula:
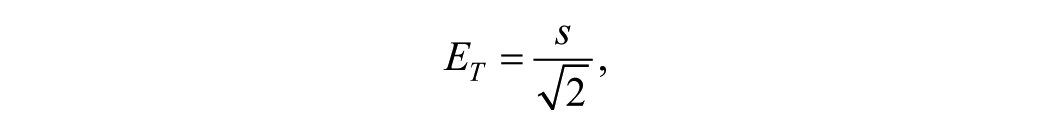
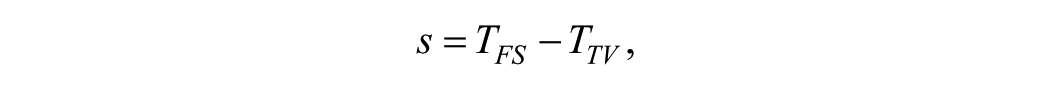
where
Results
Absolute foot temperature measured with the two devices across the three groups of 107 participants are shown in Table 3. Typical error values for temperatures between the two devices were 0.26 deg for controls, 0.34 deg DFU group and 0.33 deg DM group (0.31 deg for the overall combined sample).
Temperature Values (°C) per Foot and per Device.
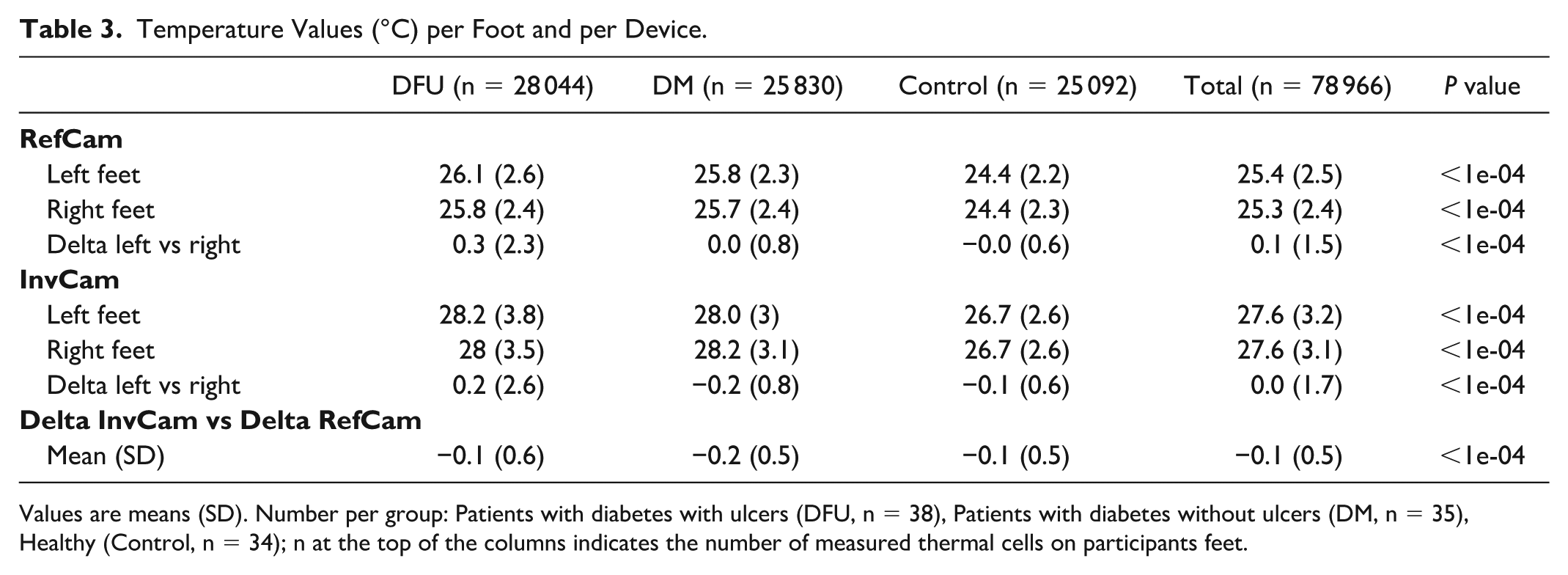
Values are means (SD). Number per group: Patients with diabetes with ulcers (DFU, n = 38), Patients with diabetes without ulcers (DM, n = 35), Healthy (Control, n = 34); n at the top of the columns indicates the number of measured thermal cells on participants feet.
The delta temperature values between the two feet with each device are shown across the three groups in Figure 2. There is a much larger spread of values in the DFU group. Importantly, delta temperature values for each group are clustered around the line of identify, indicating close agreement between the two devices regardless of group assignment.
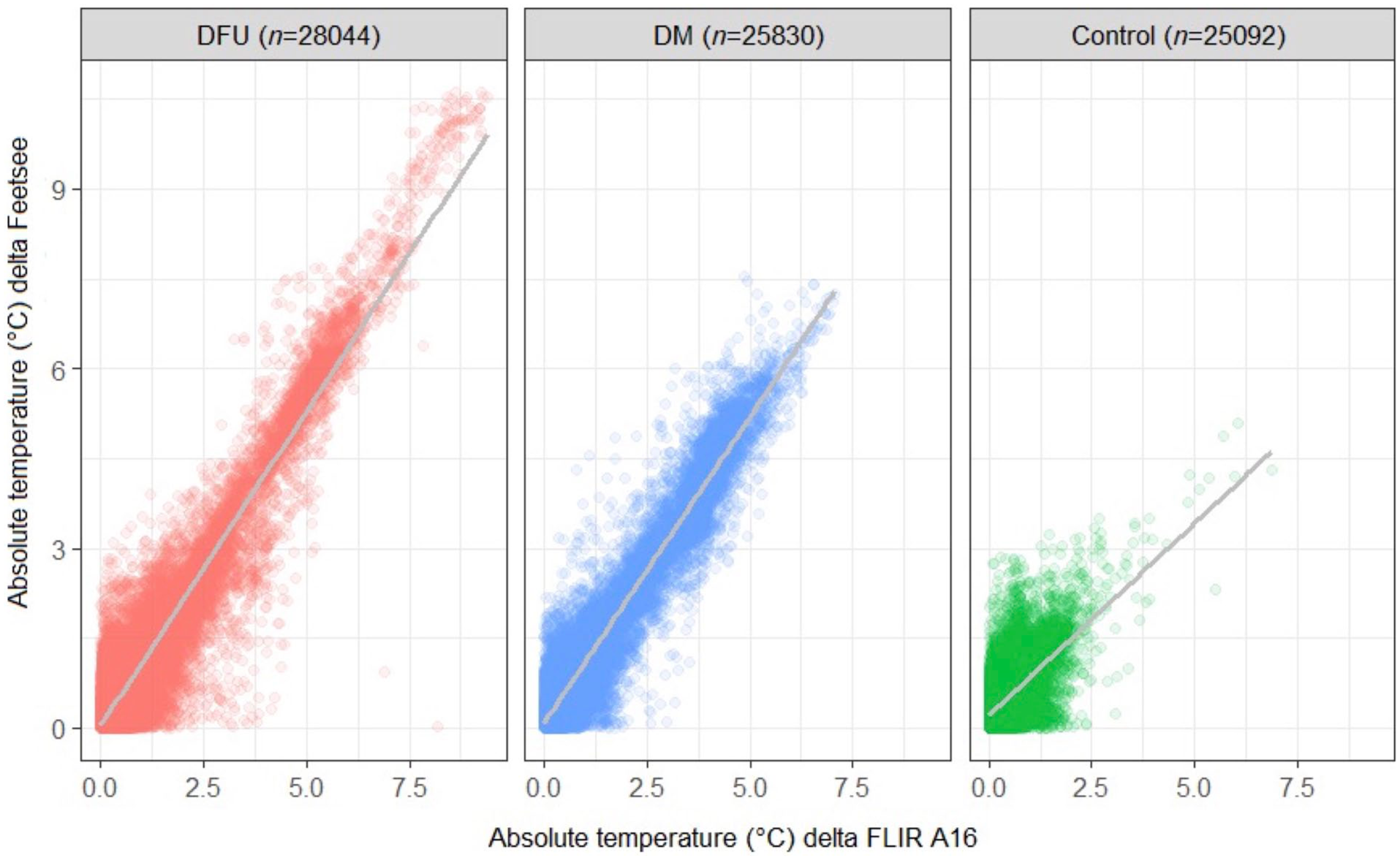
Scatter plot of absolute temperature (°C) for delta InvCam versus delta RefCam. Patients with diabetes with ulcers (DFU, n = 38), patients with diabetes without ulcers (DM, n = 35), Healthy (Control, n = 34); n at the top of each figure bar indicates the number of measured thermal cells on participant’s feet.
Figure 3 shows the Bland-Altman plot for delta temperature values between the two feet measured with the two devices across all three groups. The Bland-Altman plot shows the values clustered around the zero line, indicating good agreement between the two methods. This good level of agreement is supported by an intraclass correlation coefficient of 0.95 (95% CI: 0.95-0.96) between the two devices. The Bland-Altman plot shows little evidence of systematic bias.
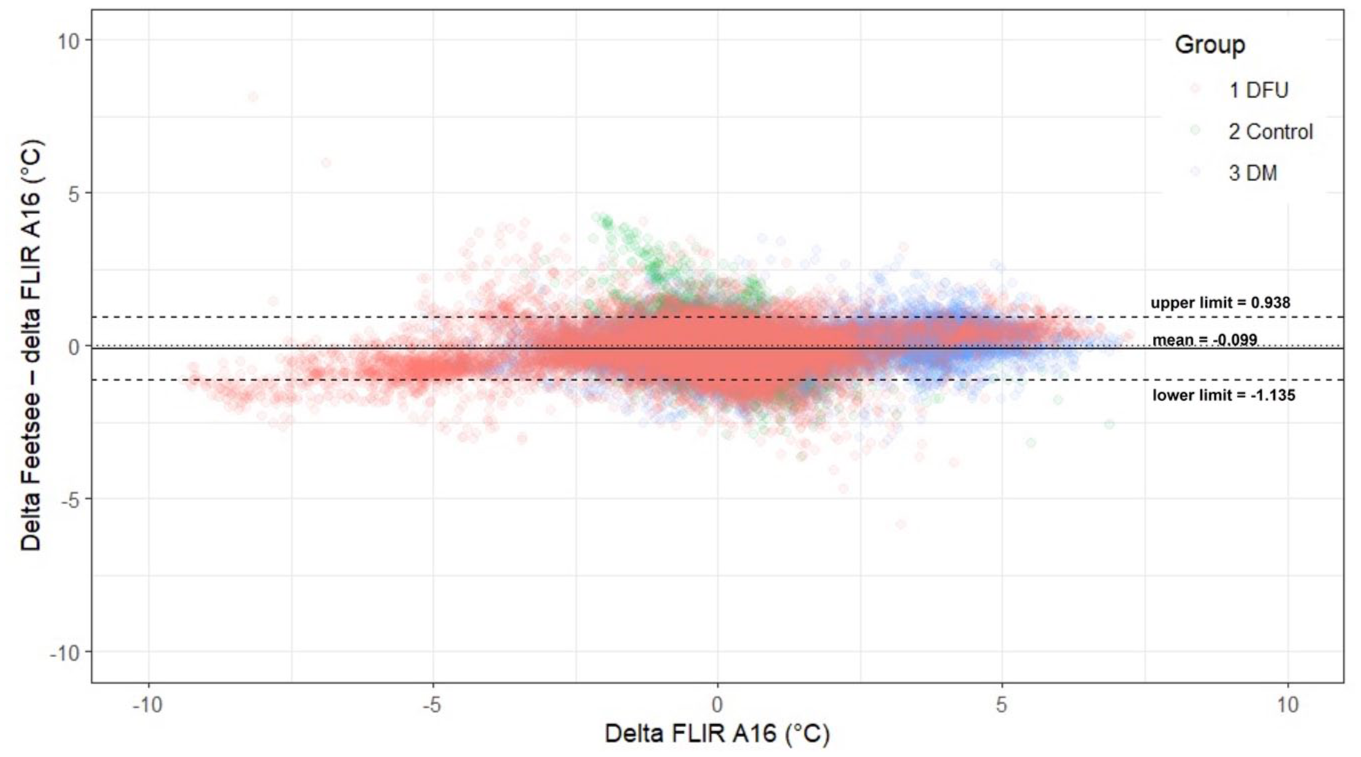
The Bland-Altman plot suggests a good level of agreement between the InvCam and RefCam devices. Red dots indicate DFU (n = 38), blue dots indicate DM (n = 35), and green dots indicate Controls (n = 34).
Discussion
This study has demonstrated the validity of the InvCam system for accurately measuring differences in foot temperature between feet in people with diabetes with and without active foot ulcers. This is supported by an intraclass correlation coefficient of 0.95 and a typical error of 0.31 deg compared with measurements taken using the current reference standard thermal camera, RefCam.
Although the reference RefCam system has a higher spatial resolution than InvCam, this is unlikely to meaningfully affect the results. InvCam’s resolution is sufficient to detect clinically relevant temperature asymmetries across 750 foot cells and the analysis focused on delta values between corresponding regions of the left-right foot. Therefore, validity metrics presented are believed to reflect genuine device performance.
Figure 3 shows that there was no systematic bias in the temperature measurements with the InvCam camera as data points are clustered around the line of identify. The range of delta temperature values was much larger in those with active foot ulcers compared with those without active ulcers and controls, reflecting the increase in temperature that occurs with development of diabetic foot ulcers (Figure 2).
The present study confirms the InvCam system as a valid and clinically viable tool for detecting temperature asymmetries between feet in individuals with diabetes—differences that may serve as early indicators of diabetic foot ulcers—by providing measurements that closely align with the current reference standard. The InvCam system offers significant practical advantages compared with other available technologies3-6 and addresses long-standing limitations of traditional thermographic and temperature monitoring approaches including being non-contact and with measurements covering the whole-foot area. The contactless design eliminates infection risk and any potential cross-contamination between patients, simplifying procedures by negating disinfectant procedures. Unlike smartphone-based IR cameras, InvCam ensures standardized foot positioning and consistent imaging quality, minimizing user-related variability. This innovation allows earlier and more precise detection of localized temperature changes, potentially improving preventive interventions for diabetic foot ulcers. Consequently, InvCam opens opportunities for routine home monitoring, empowering patients and caregivers to detect early signs of foot complications without needing specialized skills or equipment.
Despite promising results, this study has several limitations. First, the study focused on temperature asymmetries rather than longitudinal monitoring of ulcer development, so DFU predictive value could not be assessed. Second, as with many other systems, environmental factors including room temperature and patient activity prior to measurement may influence readings. Third, this study was conducted in a controlled clinical setting, while performance in home environments may differ, requiring further validation. Future research will explore longitudinal deployment, automated alert systems, and integration into remote diabetic foot care pathways. In particular, studies evaluating InvCam’s ability to predict DFU development will be essential. By enabling proactive, accessible, and hygienic monitoring, InvCam has the potential to become a cornerstone of DFU prevention strategies.
Footnotes
Appendix
Determination of the distance of the camera from the foot for the two different camera systems was based on the following calculation: A large male foot can reach ~30 cm in length and ~12 cm in width, thus the foot area may be calculated as 30*12*0.75=270 cm2, where 0.75 is a ratio that takes into account the influence of the roundness of the foot on the rectangular foot area (estimated from our previously accumulated data).
Acknowledgements
None.
Abbreviations
DFU, diabetic foot ulcer; DM, diabetes mellitus; FDA, Food and Drug Administration; InvCam, FeetSee camera; RefCam, FlirA615.
Declaration of Conflicting Interests
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Lithuanian authors received funding from Diabetis JSC to conduct the research study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Diabetis JSC, Vilnius, Lithuania.
