Abstract
The US diabetic foot ulcer (DFU) incidence is 3-4% of 22.3 million diagnosed diabetes cases plus 6.3 million undiagnosed, 858 000 cases total. Risk of recurrence after healing is 30% annually. Lower extremity multiple nerve decompression (ND) surgery reduces neuropathic DFU (nDFU) recurrence risk by >80%. Cost effectiveness of hypothetical ND implementation to minimize nDFU recurrence is compared to the current $6.171 billion annual nDFU expense. A literature review identified best estimates of annual incidence, recurrence risk, medical management expense, and noneconomic costs for DFU. Illustrative cost/benefit calculations were performed assuming widespread application of bilateral ND after wound healing to the nDFU problem, using Center for Medicare Services mean expense data of $1143/case for unilateral lower extremity ND. Calculations use conservative, evidence-based cost figures, which are contemporary (2012) or adjusted for inflation. Widespread adoption of ND after nDFU healing could reduce annual DFU occurrences by at least 21% in the third year and 24% by year 5, representing calculated cost savings of $1.296 billion (year 3) to $1.481 billion (year 5). This scenario proffers significant expense reduction and societal benefit, and represents a minimum 1.9× return on the investment cost for surgical treatment. Further large cost savings would require reductions in initial DFU incidence, which ND might achieve by selective application to advanced diabetic sensorimotor polyneuropathy (DSPN). By minimizing the contribution of recurrences to yearly nDFU incidence, ND has potential to reduce by nearly $1 billion the annual cost of DFU treatment in the United States.
Keywords
The economic costs of diabetes mellitus (DM) represent a mammoth $245 billion expense to America and its medical system. An estimated 22.3 million Americans are living with diabetes and at least 6.3 million more are believed to be undiagnosed.1,2 $176 billion is spent annually on direct costs for diabetic care, $80 billion of which are foot related expenses comprising a significant direct and indirect cost to society.1,3 For the diabetic population as a whole, risk of developing a foot ulcer is 3 to 6% annually, a condition frequently requiring hospital admission.4,5 Inpatient care is the largest contributor to cost, predominantly for infections, sepsis or gangrene requiring amputation. Of diabetic amputations, 84% are preceded by an ulcer. 6 Of diabetic foot ulcer (DFU) cases, 50% become infected, 20% of which require major or minor amputation for resolution. 7 Disastrously, 15% of all DFU will require an amputation, 8 an event related to mortality as high as 15% in the first year. 9 Avoidance of primary and recurrent DFU seems an attractive target for minimizing health care expenditures, amputations and costs to society, as shown in Figures 1 and 2.
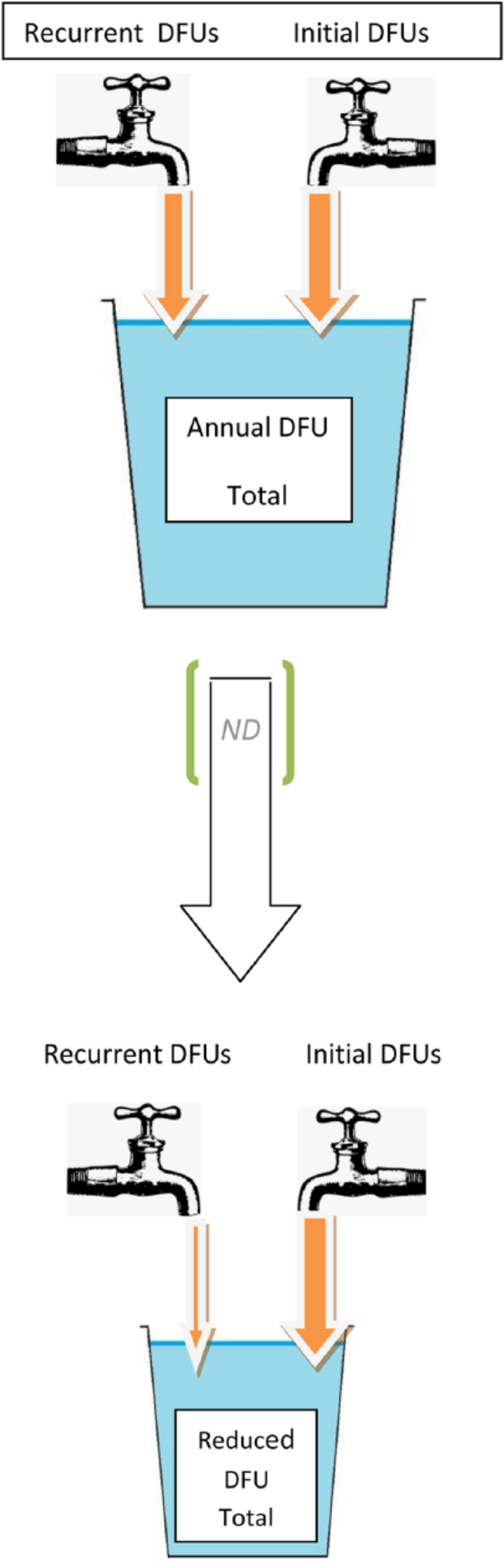
Avoiding recurrent nDFUs with ND will achieve savings by requiring care for fewer ulcers.
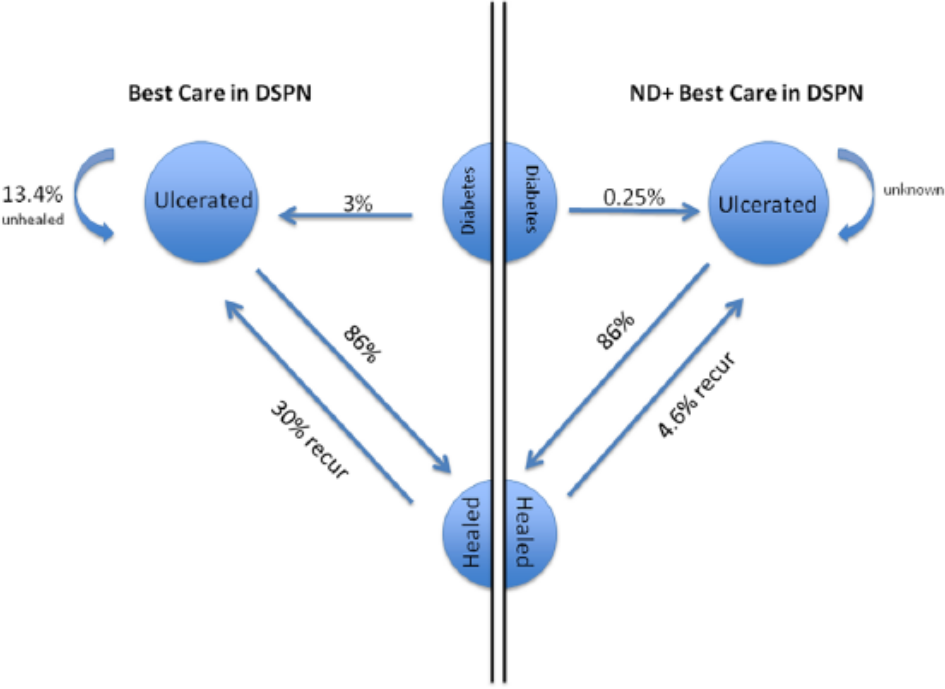
Markov states for diabetes and DFU. Diabetes cases have 3% annual risk of developing a DFU, with healed DFU having 25-30%/year risk of recurrence. ND appears to lower recurrence risk to 4.6% or less. Approximately 13.6% of DFUs remain unhealed despite “best care.” 63 Since no evidence is available on failure to heal a DFU occurring after ND, we assume equivalence.
The Lower Extremity Amputation Prevention program of the U.S. Health Resources and Services Administration promotes the education of physicians and patients on the presence and importance of neuropathy, encouraging patient vigilance, attention to skin care, early recognition of skin breakdown, callus trimming and routine podiatric care for ulcer prevention. Lavery and colleagues have reviewed current treatment recommendations, their relative effectiveness, and the continuing challenges to ulcer prevention. 4 They noted DFU recurrence rates as high as 80% annually; particularly in the presence of vascular compromise. Their review concluded by suggesting that overall DFU incidence could be reduced by over 50% if current treatment guidelines were routinely applied, still leaving at-risk patients a distressing 30% annual risk of ulcer recurrence.
One therapeutic approach to both primary prevention of neuropathic DFU (nDFU) and protection against recurrence was not explored by Lavery and collaborators. This is the outpatient surgical procedure known as external neurolysis, or nerve decompression (ND). ND is a therapy directed at nerve entrapment, a common secondary physical complication of diabetic sensorimotor polyneuropathy (DSPN), 10 targeting the neuropathic origin of the cascade of diabetic foot complications. Nerves enlarge in DSPN due to a sequence of metabolic events.11-15 Entrapment of nerve trunks commonly follows in longstanding diabetes as enlarged nerve trunks must glide and function in unyielding fibro-osseous anatomic tunnels, which are themselves stiffened and contracted by advanced glycation end products in the Maillard reaction. Clinically, these secondary, DSPN-associated entrapments are present in up to 33% of this patient population. 10 Multiple nerve entrapments can generate the classic stocking/glove anesthesia patterns described clinically. 14 Given this nerve/tunnel size mismatch, decompression of entrapped nerve trunks is reasonable to consider for minimizing contributions of secondary entrapment neuropathy to DSPN signs, symptoms and sequelae.16,17 A similar accommodative approach to nerve trunk enlargement, compressions and entrapment has been applied with success for more than 50 years in the case of leprosy neuritis. 16
Since Dellon’s 1988 optimistic enumeration of reasons for treating DSPN symptoms with ND surgery, 17 significant supportive clinical evidence has accumulated. The frequent nerve compression present secondarily in DSPN is identified by Tinel’s percussion test at anatomic entrapment sites. When first described, 18 the indication for ND was relief of pain, although recovery of sensibility was frequently noted as well. Infrequent foot ulceration, foot related hospitalizations, and amputation complications were noted following neurolysis.18,19,25,26-29 Yet scant academic or clinical acceptance of this therapeutic approach exists. 30 Skeptics assert the subjective improvements in pain and sensibility reported post-ND could be merely placebo, bias or natural disease progression effects. 31
Objective evidence would rebut or nullify theoretical objections to ND based on bias and placebo confounders in studies of subjective pain and sensibility changes. There is now significant evidence which uses objective outcome measures providing such perspective. Aszmann et al have prospectively studied DFU occurrence or amputation after unilateral ND in painful DSPN, using patients’ contralateral leg as their own control. 32 Zhang et al reported zero primary ulcerations in the 18 months following bilateral ND in 352 diabetics with advanced DSPN symptoms. 27 The same cohort showed significant recovery of slowed preoperative nerve conduction velocities at 3 months after ND. Evidence of immediate intraoperative recovery of EMG activity during ND has been presented to scientific conferences. 33 Long-term recurrence protection has been reported after ND in nDFU with nerve entrapment, demonstrating >80% recurrence reduction.15,26-29 Such evidence suggests ND has potential for a monumental impact on future DFU incidence, prevalence and associated health care expense and noneconomic costs. 34 This report estimates economic benefits potentially available to society if ND were widely applied to healed nDFU cases to minimize recurrence (Figure 2).
Methods
The ND literature suggests that nDFU recurrences can be reduced from 30% per patient per year(pppy) to 4.6% or less, an 85% reduction.4,15,26,29 This implies that an individual future year’s nDFU tally of recurrences can be greatly reduced, though not abolished, by ND. Our illustration is based on review of DFU and ND literature (see Table 1 and Table 2) published since 1988 using the most recent and conservative estimates of prevalence of diabetes {DM}, 1 incidence of neuropathic DFU{InDFU}, 35 change in nDFU incidence following ND treatment {delta InND}, and treatment cost per nDFU {cost}. Historical rates of primary neuropathic DFU and reported DFU recurrence rates were compared to ND literature. Using SPSS version 22 (IBM, Amonk, NY), a chi-square analysis was performed and reported as odds ratios (OR) with 95% confidence intervals.
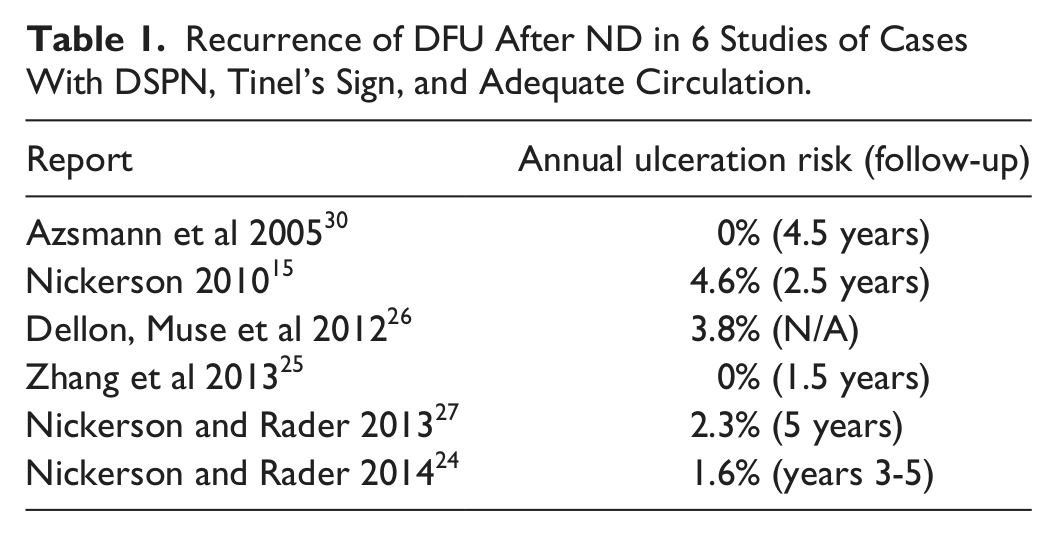
Recurrence of DFU After ND in 6 Studies of Cases With DSPN, Tinel’s Sign, and Adequate Circulation.
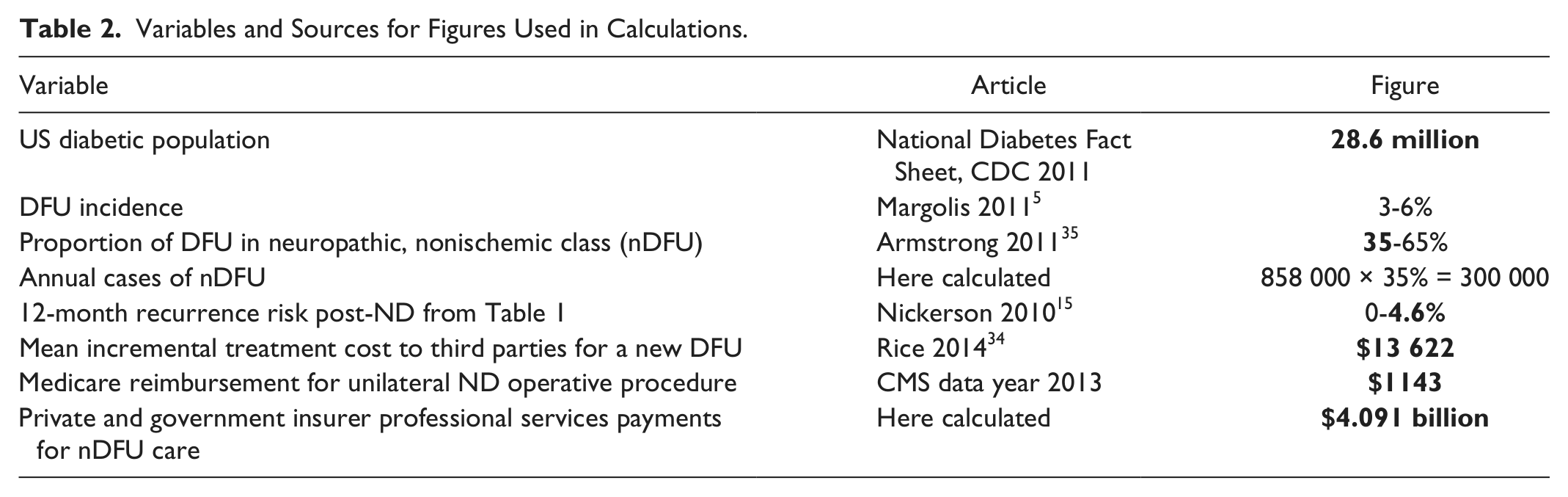
Variables and Sources for Figures Used in Calculations.
Non-third-party-payer direct societal costs identified in Table 3 include patient copay expense, private payments for provider care, neuroactive pain medication expense, and such indirect costs as lost productivity, disability payments, absenteeism, and family care. 34 Additional benefits such as subsequent amputation avoidance, minimization of recalcitrant healing cases and decreased mortality were not included in our calculations. Data used in calculations are in bold in subsequent tables, and quoted costs are in US dollars (2012) or older values adjusted for medical cost inflation.
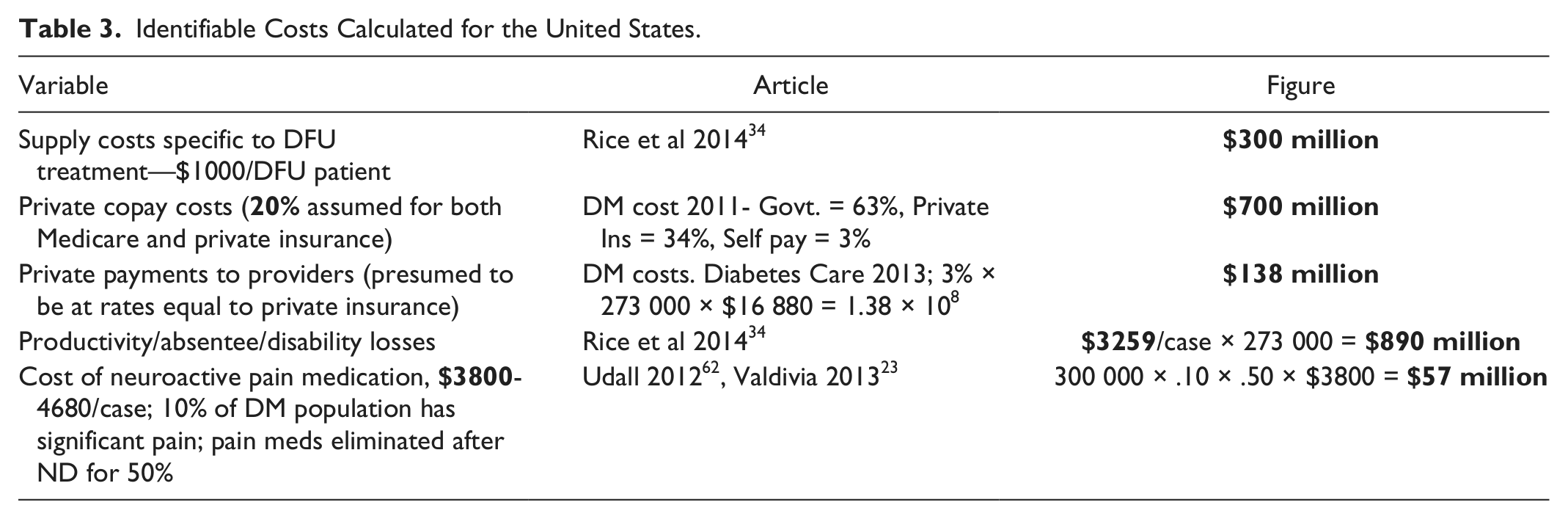
Identifiable Costs Calculated for the United States.
To illustrate the potential for ND to generate cost reductions, Figure 2 and Table 4 display a scenario where ND is applied to a single year’s entire population of nDFUs. Pertinent Markov states are illustrated in Figure 1. Changes in the total of new primary nDFU and recurrences are tracked during the subsequent 3 and 5 year periods. DFU prevalence and incidence are held steady and historical recurrence risk is taken to be 25%, 40%, 60%, and 70% at 1, 2, 3, and 5 years.36,37 The calculation depends on the proportion of first ulcerations to DFU recurrences, data that are not routinely reported. However, 2 articles have described recurrence cohorts in such a way that this can be calculated.38,39 Their recurrence proportions are 40% and 59.3%, from which we presume annual nDFU recurrences to be approximately half. For post-ND recurrence risk we use 4.6% per/patient year, the highest rate in Table 1. 15 Total nDFU numbers generated by historical treatment and recurrence rates are then compared to anticipated results after ND. Recurrent nDFUs calculated in column 3 of Table 4 are the sum of the 4.6% recurrence risk of ND-treated cases times each prior year’s declining intact number of healed DFUs.
Comparison of Post-ND and Historical Ulcer Recurrence Rates.
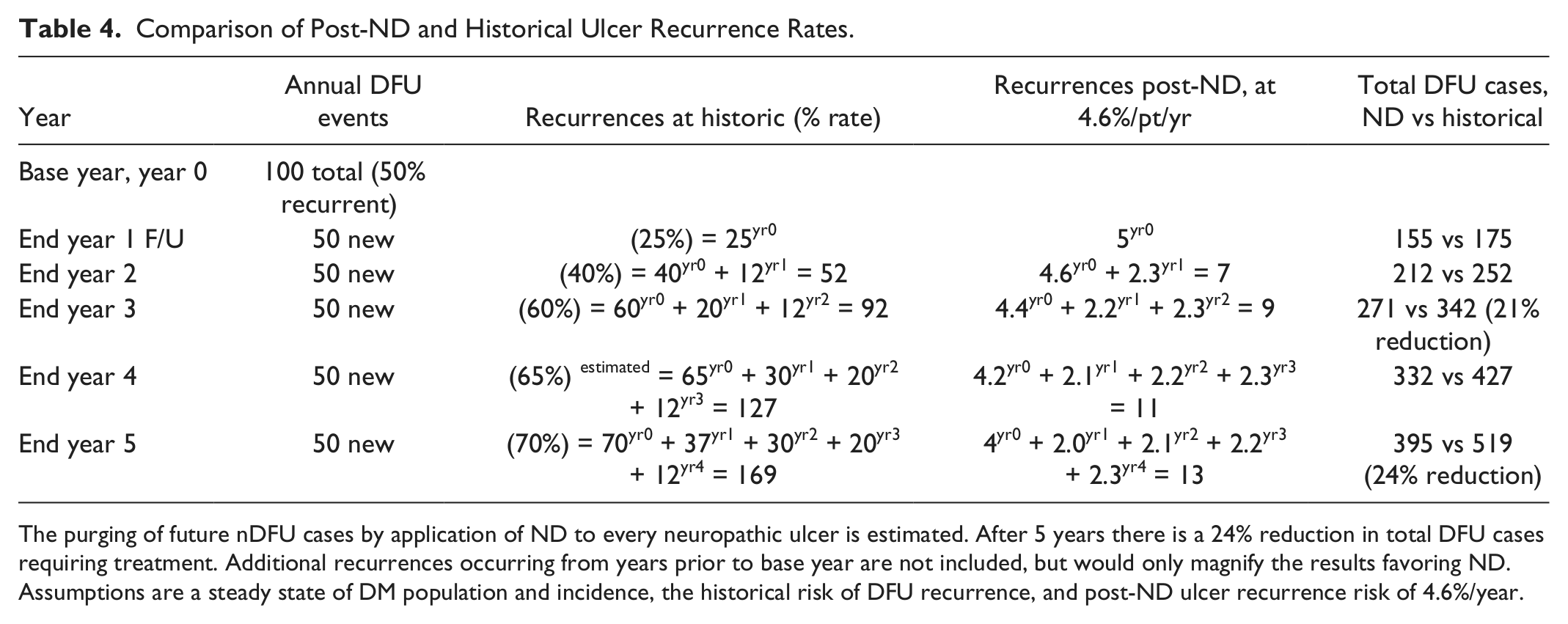
The purging of future nDFU cases by application of ND to every neuropathic ulcer is estimated. After 5 years there is a 24% reduction in total DFU cases requiring treatment. Additional recurrences occurring from years prior to base year are not included, but would only magnify the results favoring ND. Assumptions are a steady state of DM population and incidence, the historical risk of DFU recurrence, and post-ND ulcer recurrence risk of 4.6%/year.
Results
Seven historical reports of DFU recurrence32,40-45 were compared to 3 reports of ND cohorts.15,27,32 The historical reports had varying levels of implementation of “standard” foot care vs insoles or footwear. A weighted average of these historical recurrence rates was calculated (range: 5-58%) resulting in a mean 20.4% (SD = 0.17). This result for nonsurgical standard/experimental care represents a lower recurrence rate than typically reported in the literature. Reports assessed for DFU recurrence included 303 patients who received ND, and 1276 patients who received standard care. Following ND, 4 (1.3%) patients had recurrent ulceration compared to 260 (20%) of the historical care group, a much reduced hazard risk (OR = 0.05, 95% CI = 0.02-0.14).
Only 1 sizeable historical report exists for primary ulcer incidence with standard preventive measures. 46 Twenty-eight of 921 patients had primary ulceration of their feet, while 0/404 patients receiving ND experienced primary ulceration.27,32 Conservative estimation of 1/404 was used for statistical analysis, and ND showed a statistically significant decreased OR for primary ulceration (OR = 0.08, 95% CI = 0.01-0.58).
The expense to third-party insurance payers for nDFU care by medical professionals is as follows:

Additional expenses to society are listed in Table 3 and include supply costs, private payments to providers, 20% copay for insured cases, expense of neuroactive pain drugs like tricyclic antidepressants, gabapentin, and pregabalin, productivity losses, absenteeism, and disability payments. These total $300 + $700 + $138 + $890 + $57 million, respectively. Adding to $4.091 billion gives a grand total of $6.171 billion minimum annual cost to society for nDFU, equaling $20 570 per case cost for 300 000 neuropathic DFUs.
After 3 and 5 years, a 21% and 24% annual reduction in total DFU events could be realized if ND were routinely employed after healing of nDFUs. For year 3, this represents a potential benefit of .21 × $6.171 billion, or $1.296 billion. By year 5, the annual savings reaches 24%, or $1.481 billion. This requires an investment expense of $686 million (300 000 nDFU × $1143/ leg × 2 legs/case), representing a 2.16 × return on investment. Future years would see similar continued savings.
Discussion
Despite the reported success of ND, many question how surgery could be effective in a metabolic disorder such as diabetes. This is really no more curious than the successful surgical approaches to diabetic retinopathy, cardiovascular or peripheral vascular disease, and nephropathy with renal failure. Surgical practitioners do understand that peripheral nerve trunk entrapment is
In this illustration amputation costs are included for the initial DFU treatment course, as they are well recorded in the DFU literature. But amputations forestalled by avoided recurrences in future years are an additional benefit, not estimated here. Nor does our calculation acknowledge the ongoing recurrences recognized in years 1-5 from the ulcers healed in years preceding base year. Additional prospective benefits include a decrease in unhealed or recalcitrant ulcers, decreased expenses for ongoing treatment and infection risk, deferred early mortality from fewer DFU cases at risk of infection and sepsis, an improved patient quality of life and relief of caretaker burden. All of these are suggested in the DFU and ND literature but figures were not well enough quantified to include in our example.
Benefits would accrue to differing parties. Cost reductions and copay amounts would accrue to third-party payers and self-payers. Clinics and institutions might limit DFU care losses. Surgeons would benefit with revenue from ND, offsetting other parties’ savings. Employers, patients, families, and caregivers would share the indirect benefits of reduced disability cost, absenteeism, and retained capability.
Our Table 4 illustration trims the 3-year nDFU recurrence total from 92 to 24 cases, a 76% reduction. But this yields total cost reduction of only 21%, due to continued generation of new primary ulcers from the DSPN reservoir with secondary entrapments. For a greater total cost reduction, primary ulcer incidence must decrease. ND may be useful for this purpose.27,28,32 Zhang’s 352 DSPN patients, with adequate circulation and the highest possible Toronto Clinical Symptom Scores, developed not a single primary ulcer in the 18 months following bilateral ND. This group should have accumulated 16 DFUs (352
An unknown question is how DSPN cases could be selected for prophylaxis of initial DFU by ND. The reports of Yudovsky et al 48 indicate that hyperspectral imaging of oxy/deoxy-hemoglobin ratios can identify, with 90% sensitivity, and 2 months prior to ulceration, those plantar skin areas about to break down in DSPN patients. Non-invasive hyperspectral screening could allow rapid, efficient focused application of prophylactic ND to the specific individuals at particular risk for initial DFU.
Neuroischemic cases offer yet another opportunity for savings. An estimated 50% of DFUs are neuroischemic, 35 and around 65% of such cases will heal with current offloading care. The definition of neuroischemic ulcer varies, from absence of palpable foot pulse, to ankle-brachial index values < 0.9, or toe pressures < 45 mm Hg. Trignano et al show that neuroischemic values of foot trancutaneous pO2 can be reestablished and maintained for a year at > 45 mm Hg in 90% of cases. 49 Currently there is little evidence to guide selection of appropriate neuroischemic candidates. ND might be secondary or supplementary to interventions which improve circulation, but its value for neuroischemic ulceration certainly warrants further investigation.
It is certainly intriguing that ND protects against nDFU recurrence despite unchanged risk factors like plantar pressure concentrations, tight gastro-soleus muscles, lost ankle ROM, past DFU/amputation history, foot deformity, high BMI, and smoking history. The nervous system plays a critical role in tissue homeostasis, a factor particularly important in DSPN complications. 50 Scientific literature links diabetes and vascular phenomena like hyperemic flare, reduced capillary circulation and neuropeptide reductions.51-55 Altered oxygen delivery, fluid filtration, and inflammatory response undoubtedly make the feet of DSPN patients more susceptible to tissue damage. These factors might explain anecdotal reports that ND may be useful in shortening healing time for the recalcitrant 15% of indolent, torpid DFU cases and the 10% which remain unhealed until patient demise.
Surgical site infection, reported by Wukich
56
to be nearly 13% for patients with diabetes and neuropathy, cannot be ignored. Zhang’s prospective study included 560 bilateral procedures on advanced DSPN and healed DFU cases, with >3000 separate incisions, but reported zero surgical site infections.
27
These are significantly different infection rates (
Beyond the favorable economics of ND, indirect benefits can include a better quality of life, caregiver relief, and deferred mortality. The overlying patient-centered goal in DFU care ought to be ulcer-free survival with intact limbs. Traditional treatments provide such survival for only 36 - 45% at 12 months. 57 In contrast, ND produced intact legs and ulcer-free survival in 59% after 5 years, suggesting its superiority as therapy, at least for the painful, Tinel-positive nDFU subgroup. 29
This exercise is limited by a number of assumptions and estimations. Little of the ND literature and few DFU studies58-60 are level 1 evidence-based science. Due to limited quality historical and ND data on primary foot ulceration, stringent meta-analytical techniques were not applied. Comparative statistical figures should be interpreted accordingly. Neurolysis cohorts are primarily patients with painful neuropathy, adequate circulation, and a Tinel sign at anatomic compression points. There is no evidence that painless, Tinel-negative DSPN cases react differently than our illustrative group in objective measures (NCV, EMG, infection risk, DFU incidence, recurrence, or amputation risk) but this issue has not been specifically studied. Available economic data is not sorted by circulatory status, so generalizing overall DFU data to the nDFU could be challenged.
Contributions to continuing recurrences from ulcers initially presenting prior to base year are excluded, but would only magnify the calculated benefit. This number might be around 80 additional recurrences available for ND and recurrence protection if Apelqvist’s risk of 70% by 5 years 35 is applied to the population which provided the 50 recurrences presumed in the base year. Benefits of ND would accumulate as time progresses, to eventually reach a limit, since all possible recurrences would have occurred. Of course, new primary wounds and nonoperated recurrent cases in years’ subsequent to the base year could still benefit.
There is no information available on whether ND might provide enough durable, long lasting recurrence protection to diminish the need for patient education, footwear, shoe inserts, routine podiatric follow-up, or other current “best care” and aftercare recommendations. This could be a fruitful future research area.
The representative illustration of DFU incidence change depends on the proportion of ulcers which are primary versus recurrent, a statistic rarely reported. If the 1:1 ratio on which these calculations are based were incorrect, the projections would need adjustment. Greater benefit would occur if recurrences were a higher percentage, as larger cost reductions would occur. If lower, reductions of initial DFU rates would be necessary to achieve similar results.
Our use of a calculated weighted average yearly recurrence risk (20.4%) one-third lower than Lavery et al’s impression of 30% 4 means the OR calculation may understate the magnitude of protection that ND provides.
It is certain that ND would never be applied to 100% of nDFU cases, due both to withheld patient consent and medical contraindication. Yet the illustration seems persuasive that ND could be an efficient public policy tool to accomplish meaningful reductions in the expense of care for the common and costly DSPN foot complications. Neuropathic DFU then might be seen once again as potentially “cured” rather than only “in remission.” 61
Conclusions
The large and increasing cost of care for diabetic foot problems is determined by the high incidence of DSPN and DFU, large expenses for complication treatment, and the appalling frequency of recurrences. The outpatient surgical procedure of ND at multiple fibro-osseous tunnels for decompression of lower extremity peripheral nerve trunks appears to give significant protection against ulcer recurrence. If confirmed by clinical experience, external neurolysis applied to the 35% of DFU cases thought to be primarily neuropathic, by the third program year could potentially reduce costs by at least $1.296 billion annually. This would require expenditure of $686 million with the cost/benefit ratio being at minimum 1.9:1. Annual savings would reach $1.790 billion by year 5. We suggest that this currently available, effective, but infrequently used therapy could provide $1 billion economic and social benefit annually within 5 years by limiting the complications and cost of DSPN foot problems. With the illustrative calculations presented here, we wish to challenge those skeptical of the ND approach to participate in the design and performance of studies which will either bolster or conclusively terminate the widespread reluctance to acknowledge value in ND for DSPN foot complications.
Footnotes
Abbreviations
DFU, diabetic foot ulcer; DSPN, diabetic sensorimotor polyneuropathy; NCV, nerve conduction velocity; ND, nerve decompression; nDFU, neuropathic DFU; OR, odds ratio.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
