Abstract

In clinical settings, diabetes-related foot ulcers (DFUs) have traditionally been measured using the ruler and probe 1 ; however, this method has poor inter- and intra-rater reliability between clinicians. 2 We aimed to evaluate the reliability of the ARANZ SilhouetteStar device (ARANZMedical Ltd, Merivale, Christchurch, New Zealand) in comparison to the traditional ruler and probe method in patients with DFU in a real-world clinical setting.
A prospective study was conducted at the Multidisciplinary Diabetes Foot Ulcer (MDFU) and podiatry clinics at Fiona Stanley Hospital, Perth, Western Australia over a 5-month period from July to December 2021. This study was approved as a quality improvement activity, as wound measurements constitute current standard clinical practice. Inclusion criteria included over 18 years of age, diabetes, and an active DFU. Written informed consent was obtained prior to participation.
During each participant’s routine clinic visit, measurements were taken by three investigators using the ARANZ SilhouetteStar device (including wound length, width, depth and surface area) with a blinded measurement interval of at least ten minutes, and a repeat measurement taken by the principal investigator. Using the ruler and probe, measurements of length (maximum diameter), width (perpendicular diameter), and maximum depth were obtained. Based on these measurements, surface area was calculated using the elliptical wound measurement method 1 and compared against the ARANZ SilhouetteStar values.
Analyses were performed using R. 3 Deming correlation coefficients were undertaken for each dimensional parameter comparing ARANZ SilhouetteStar with ruler and probe technique. Intraclass correlation coefficient (ICC) was used to ascertain the variations in measurements and presented as 95% confidence interval (CI95). A two-way random-effects model with absolute agreement was applied for these analyses.
There were 62 patients with a total of 80 measurable ulcers; 81% of participants were male and 85.7% had type 2 diabetes. The wounds were located on the plantar forefoot (82.5%), plantar hindfoot (10%), and dorsum of the foot (7.5%). According to the WIfI clinical risk class, 4 ulcers were considered very low, low, medium, and high risk of amputation at 1 year in 81%, 10%, 8%, and 2%, respectively.
The ICCs were reported as follows 5 : the intraobserver reliability (ICC [CI95]) of the ARANZ SilhouetteStar was excellent for area (0.996 [0.993-0.997]), length (0.998 [0.997-0.999]), and width (0.995 [0.992-0.997]) and good for depth (0.871 [0.805-0.915]). Inter-rater reliability of 3D wound camera was excellent for area (0.994 [0.991-0.996]), length (0.996 [0.994-0.997]), and width (0.990 [0.986-0.994]) and good for depth (0.853 [0.796-0.898]). Deming regression and Bland Altman Plots were performed to assess the relationship between dimensional parameters for ARANZ SilhouetteStar and ruler and probe measurements (Figure 1).
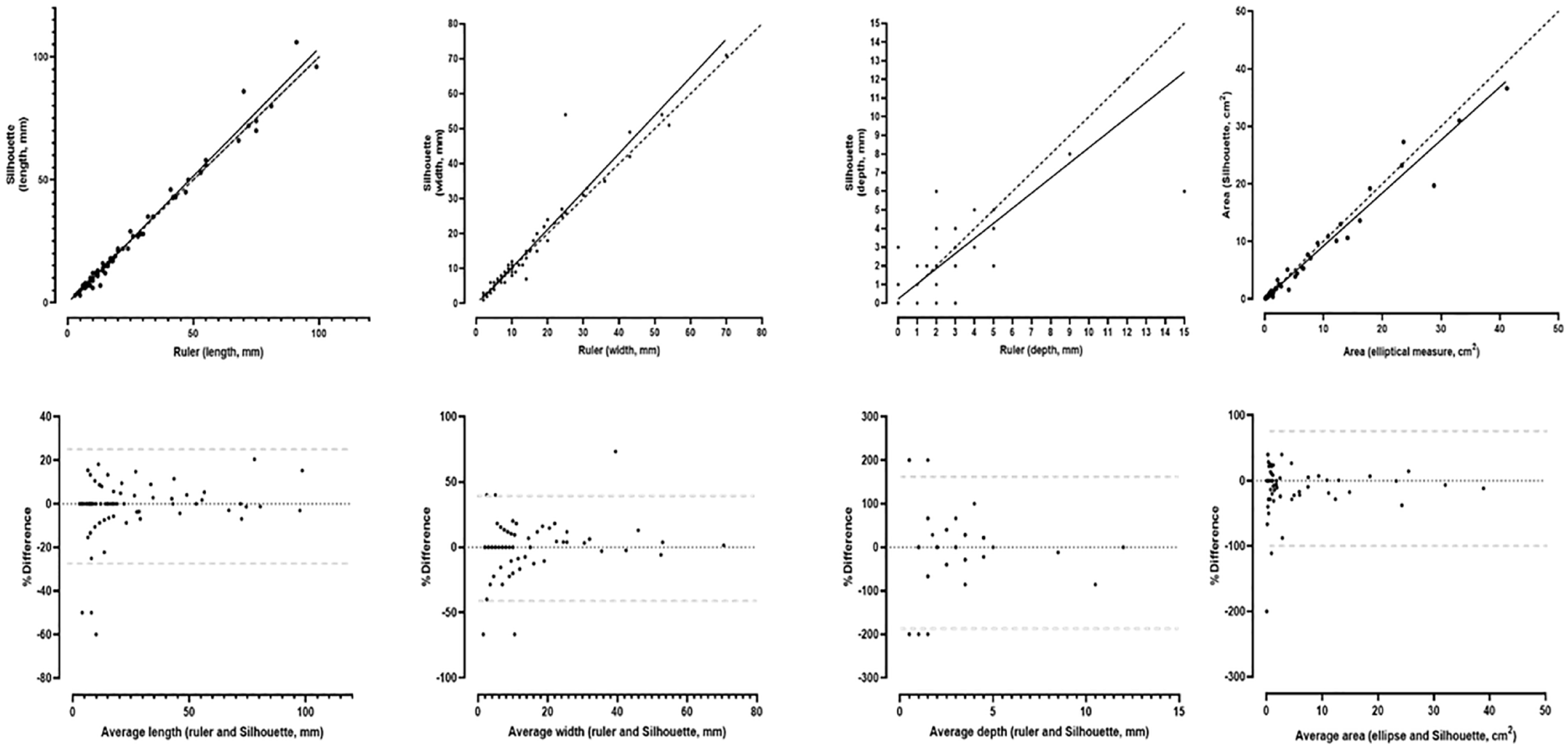
Correlation between ruler and SilhouetteStar. Correlation between ruler and Silhouette measurements from left; length, width, depth, and area. Line of identity (black, dashed line) and Deming regression line (black solid line). Bland-Altman plots are shown (95% confidence interval of bias estimates, dashed gray lines).
Based on our results, the ARANZ Silhouette Star was found to have excellent intra- and inter-observer reliability; however, depth measurement was a potential limitation and may require confirmation with a probe test. This study demonstrates that the ARANZ SilhouetteStar wound camera and software provides contactless, reliable measurements and accurate digital tracking of wound healing progress 6 with a high degree of accuracy, even in a real-world setting with a high rotation of clinical staff.
Footnotes
Abbreviations
3D, three-dimensional; CI95, confidence interval; DFU, diabetes-related foot ulceration; ICC, intraclass correlation coefficient; MDFU, Multidisciplinary Diabetes Foot Ulcer.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: G. Tan was supported with funded research time to help complete this project by the SMHS Allied Health Building Research Capacity Initiative. E.J. Hamilton was supported by a Raine Clinician Research Fellowship by the Raine Foundation and WA Health.
