Abstract
Background:
In people with type 1 diabetes (T1D) admitted to hospital, adverse glycemic events (AGE), both hypoglycemia and hyperglycemia, bestow risk for adverse outcomes. Continuous glucose monitoring (CGM) use is increasingly common amongst people with T1D. We investigated AGE frequency in hospital, based on CGM versus point-of-care (POC) blood glucose measures.
Methods:
In this multi-center retrospective analysis of non-critically ill hospitalized adults with T1D who continued wearing their unmasked CGM (FreeStyle Libre 1/2, Dexcom G5/G6, Medtronic Guardian 3) during admission and received standard ward-based POC testing, we compared CGM- and POC-based AGE detection of hypoglycemia (<70 mg/dL) and hyperglycemia (>180 mg/dL).
Results:
In 253 admissions, 127 837 CGM and 5508 POC glucose measures were analyzed, yielding 1391 CGM-detected hyperglycemia AGE and 317 CGM-detected hypoglycemia AGE. For CGM-detected AGE with a concurrent POC AGE evident, CGM detected hyperglycemia a median [interquartile range, IQR] of 70 minutes [22, 166] before POC and at lower glucose concentrations (187 vs 223 mg/dL, P < .0001) and detected hypoglycemia a median [IQR] of 38 minutes [14, 65] before POC and at higher glucose concentrations (67 vs 56 mg/dL, P < .0001). A quarter of CGM-detected AGE were not detected by POC. Only 3% of POC-detected AGE were not detected by CGM.
Conclusions:
Almost all AGE in hospital were detected by CGM, with few detected by POC alone. Compared to POC, CGM detected AGE earlier, with a lesser glycemic extreme, although unmasked CGM use may have influenced these results. Detecting AGE in hospital appears superior with CGM compared to POC glucose alone in people with T1D.
Introduction
Adverse glycemic events (AGEs) in hospital, including hyperglycemia and hypoglycemia, are associated with worse patient outcomes and increased mortality.1,2 Conversely, interventions targeting diabetes care in hospital can improve glycemia and reduce infection.3-5 Although laboratory glucose analyzers are the gold standard method of measuring blood glucose, standard of care for in-hospital blood glucose (BG) monitoring is point-of-care (POC) capillary BG measures via fingerstick testing due to the rapidity and accessibility of results. Typically performed only a few times per day, POC testing has limited ability to provide comprehensive information on glycemic profiles over the course of a hospital admission. Continuous glucose monitoring (CGM) has improved our ability to examine glycemia in detail, with modern devices recording glucose every 1 to 5 minutes.
In recent years, CGM use has significantly increased in many communities, especially amongst people with type 1 diabetes (T1D)6,7 and in countries with reimbursement schemes for diabetes technologies.8-10 Increasingly, people using CGM day-to-day expect to continue using their CGM if admitted to hospital, 11 and the American Diabetes Association Standards of Care support this “if clinically appropriate.” 12 The desire for continued CGM use is unsurprising given the many glycemic and quality of life benefits associated with community use, including reduced hypoglycemia and improved mental health.13-15
The CGM use by health care workers for the purposes of patient management in hospital is not currently approved by the Food and Drug Administration (FDA). However, given the increased quantity of glycemic data available from CGM compared to POC testing, it is anticipated that, if approved, CGM use in hospital will provide health care providers with improved ability to detect AGE and greater opportunity to respond to acute glucose fluctuations.
In Australia, the National Diabetes Services Scheme’s CGM subsidy provides universal access for people with T1D, 10 resulting in a marked increase in CGM use in the community. Amongst people who choose to continue wearing their CGM during hospitalization, there is the opportunity to compare CGM to POC glucose monitoring for AGE detection in hospital. The aim of our study was to investigate if CGM enhanced AGE detection compared to POC testing in adults with T1D admitted to non-critical care hospital wards.
Methods
The CONFIDE-1 study (CONtinuous glucose monitoring For Inpatients with DiabEtes—type 1) is a multi-center retrospective observational study of all adult patients with T1D admitted to hospital whilst wearing their own unmasked CGM devices. The CONFIDE-1 study included participants requiring multi-day acute hospital admissions from July 2020 until December 2023, inclusive, across three health services in Melbourne, Australia (Eastern Health [EH], Melbourne Health [MH], Peninsula Health [PH]), comprising six major metropolitan, inner suburban, and outer suburban hospitals. Results from CONFIDE-1 regarding the accuracy of CGM use in hospital have previously been published. 16 The present analysis investigated AGE frequency and timing by comparing CGM- and POC-glucometrics.
Patients
For this analysis, we included patients who used their own FreeStyle Libre 1 or 2 (Abbott Diabetes Care Inc, VIC, Australia), Dexcom G5 or G6 (Dexcom Inc, San Diego, California), and Medtronic Guardian 3 (Medtronic, Minneapolis, Minnesota) CGM and required elective or emergent acute hospital admissions in non-critical care wards. We included patients with both CGM and POC glucose measures available from their admission. The POC BG measures are performed four times per day (pre-meals and pre-bed) as standard ward practice at each study hospital. As CGM use in hospital has not received regulatory approval, it was expected that routine clinical management was based upon POC, rather than CGM, glucose measures.
We excluded participants admitted under pediatric/obstetric units (to minimize variation in patient case mix between study hospitals), under palliative care/psychiatry/subacute units (minimal POC testing frequency), as well as participants using CGM Reader/Receiver devices (uncertain device date/time accuracy). We also excluded participants receiving medications known to interfere with CGM accuracy according to manufacturer product information: ascorbic acid for Libre 1; ascorbic acid >500 mg/d for Libre 2; hydroxyurea/acetaminophen for Guardian 3 and G5; and hydroxyurea/supratherapeutic acetaminophen for G6.17-20
Materials
The CGM glucose measures were downloaded from each health service’s web-based CGM software accounts (LibreView, Dexcom Clarity, and Medtronic Carelink websites), where they had previously been linked for clinical care purposes. Fingerstick POC BG measures were obtained from hospital medical records, performed using ward glucose meters: Accu-Chek Guide (Roche Diabetes Care, Rotkreuz, Switzerland) at EH, Nova StatStrip (Nova Biomedical, Massachusetts) at MH, or FreeStyle Optium Neo H (Abbott Diabetes Care, Alameda, California) at PH. At each health service, hospital policies mandate daily glucose meter quality control checks by nursing staff.
Glucose measures were included if they occurred during the CGM active period, ie, the time between the first and last CGM glucose measures during the hospital admission, censored at day 14. Participants were included only if ≥70% of expected sensor glucose measures were available during the CGM active period, in accordance with consensus recommendations regarding data sufficiency. 21 Glucose measures obtained during periods of intensive care unit treatment were excluded. Spurious POC measures, according to criteria defined by Weinberg et al, were excluded. 22
Glucose measures exceeding the device’s reportable ranges (40-400 mg/dL for Dexcom and Medtronic Guardian CGM; 40-500 mg/dL for FreeStyle Libre CGM in Australia; 10-600 mg/dL for Nova StatStrip and Accu-Chek Guide POC meters; 20-500 mg/dL for FreeStyle Optium Neo H POC meters) were assigned the device’s relevant lower/upper limit glucose value.18,19,23-27
Glucometric Analyses
Hyperglycemia and hypoglycemia AGE were defined in accordance with consensus guidelines21,28 as follows:
Level 1 hyperglycemia if glucose >180 mg/dL.
Level 2 hyperglycemia if glucose >250 mg/dL.
Level 1 hypoglycemia if glucose <70 mg/dL.
Level 2 hypoglycemia if glucose <54 mg/dL.
A CGM-detected AGE occurred if CGM glucose met glucose threshold criteria for at least 15 minutes and ended if CGM glucose no longer met glucose threshold criteria for at least 15 minutes, as per international consensus guidelines.21,28 A POC-detected AGE occurred if any POC glucose met glucose threshold criteria. Given the limited ability to accurately differentiate between recurrent or persistent POC-detected AGE based upon intermittent POC measures, separate glucometric analyses needed to be conducted from the perspective of CGM-detected events and from the perspective of POC-detected events.
Categorization of Adverse Glycemic Event
Categorization of CGM-detected AGE is presented in Figure 1. A “CGM-only AGE” was defined when there were no POC glucose measures recorded within 60 minutes of the CGM-detected AGE. For CGM-detected AGE that had corresponding POC measures within 60 minutes, these CGM events were further divided into “concordant CGM-POC AGE” (with a corresponding POC-detected AGE within 60 min of the CGM-detected AGE) or “discordant CGM AGE” (when no POC glucose measures recorded within 60 min of the CGM-detected AGE met glucose threshold criteria for POC-detected AGE). The 60-min window was chosen to account for the lower frequency of POC testing, as well as delays in manual entry of results into the medical record.
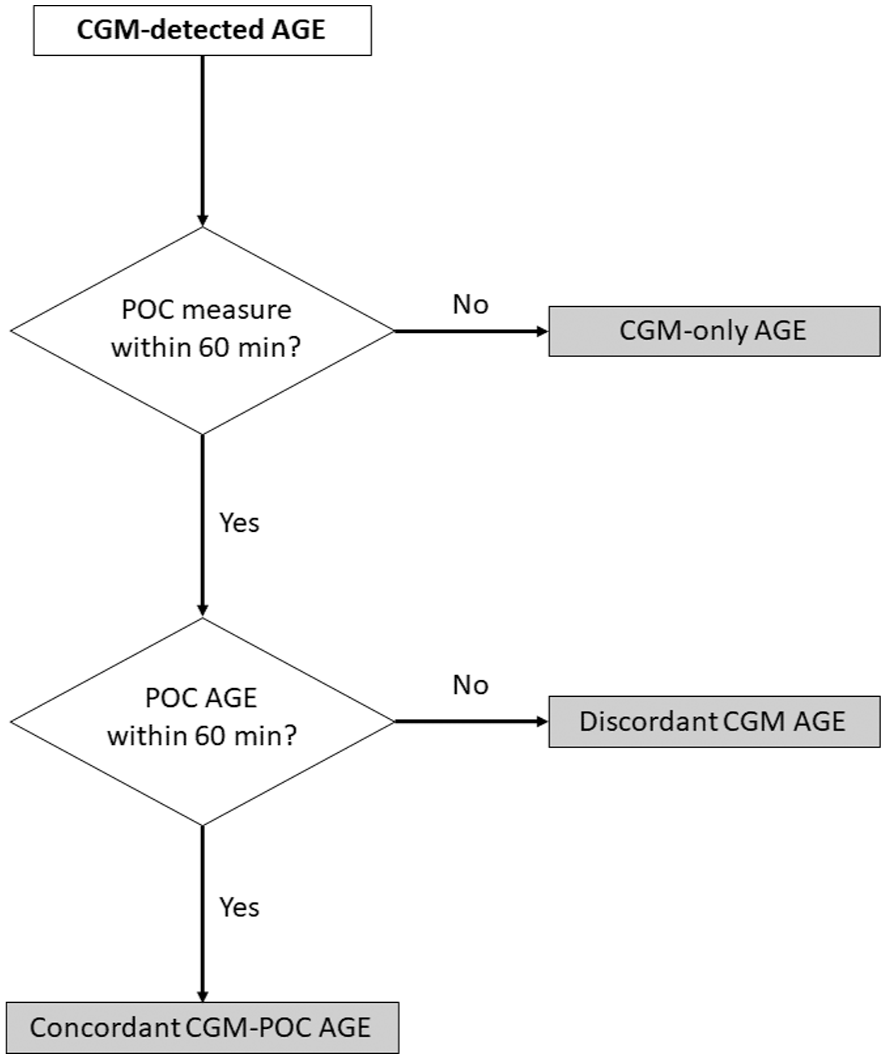
Categorization of continuous glucose monitoring (CGM) detected adverse glycemic events (AGE): “CGM-only AGE” is defined if no POC measure was performed within 60 minutes; “discordant CGM AGE” is defined if a POC measure, but no POC AGE, occurred within 60 minutes; and “concordant CGM-POC AGE” is defined if a POC AGE occurred within 60 minutes.
Concordant CGM-POC events were further subdivided according to whether the CGM event occurred earlier, at the same time as, or later than the POC event. Nocturnal AGE occurred between 00:00 h and 05:59 h, and daytime AGE between 06:00 h and 23:59 h. 28
Among “discordant CGM AGE,” the reason for discordance was determined to be inaccurate POC or CGM glucose measures (where the CGM AGE and the corresponding POC measure occurring closest in time did not meet %20/20 agreement criteria); or discordant but accurate POC and CGM glucose measures (where %20/20 agreement criteria were met, but the corresponding POC measure did not meet AGE glucose threshold criteria). The %20/20 agreement criteria defined agreement if corresponding POC and CGM measures were within 20 mg/dL when reference POC glucose was ≤100 mg/dL, or within 20% when reference POC glucose was >100 mg/dL. 29
Categorization of POC-detected AGE was performed in a similar fashion to CGM-detected AGE.
Statistical Analysis
For baseline and outcome data, continuous variables were reported as mean and standard deviation (SD) if normally distributed, or median and interquartile range (IQR) if non-parametric; number and percentage were reported for categorical variables. Paired and unpaired comparisons of non-parametric variables were performed using the Wilcoxon Signed-Rank test and the Wilcoxon Rank-Sum test, respectively. Two-tailed P < .05 was considered statistically significant. Statistical analyses were performed using base R packages from R version 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria). 30
Ethics Approval
Ethics approval was obtained from each health service’s Office for Research prior to study commencement (EH: QA23-105-102749; MH: QA2023122; PH: SA/103943/PH-2023). An individual patient consent waiver was approved by the relevant human research ethics committee representatives, as data were aggregated in de-identified format, patients had previously provided explicit consent for their health services to access these data, and this analysis would be reasonable usage of the data for quality assessment purposes.
Results
A total of 253 patient admissions (derived from 162 unique patients) were included in this analysis (Table 1), with a median [IQR] age of 45 years [30, 60], admission duration of 3 days [1, 5], and mean (SD) hemoglobin A1c (HbA1c) of 74 mmol/mol (22) or 8.9% (2.0). In total, 127 837 CGM glucose measures (51.5% >180 mg/dL, 24.3% >250 mg/dL, 2.5% <70 mg/dL, 0.5% <54 mg/dL) and 5508 POC glucose measures (49.3% >180 mg/dL, 24.3% >250 mg/dL, 4.7% <70 mg/dL, 1.4% <54 mg/dL) were included for analysis, with a median [IQR] of 319 [129, 602] CGM measures per admission and 8 [4, 16] POC measures per admission.
Study baseline characteristics.
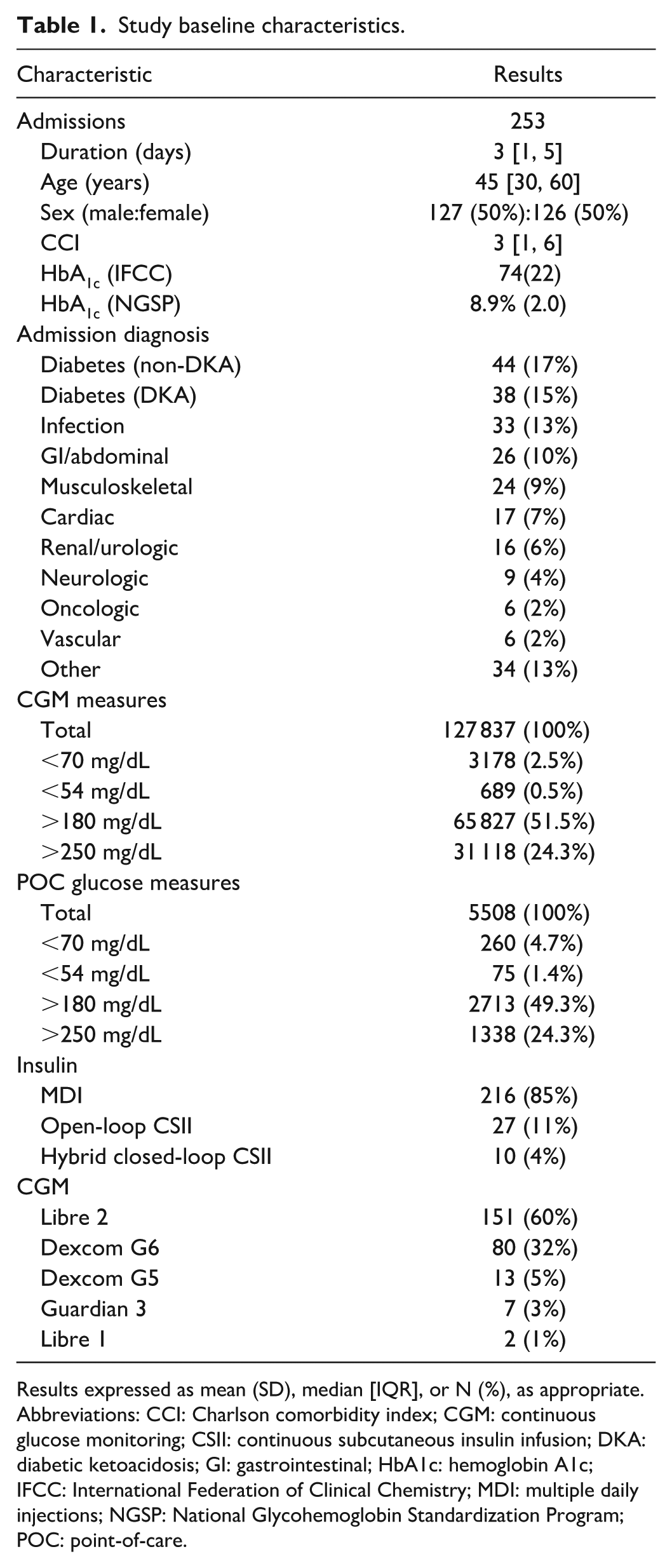
Results expressed as mean (SD), median [IQR], or N (%), as appropriate.
Abbreviations: CCI: Charlson comorbidity index; CGM: continuous glucose monitoring; CSII: continuous subcutaneous insulin infusion; DKA: diabetic ketoacidosis; GI: gastrointestinal; HbA1c: hemoglobin A1c; IFCC: International Federation of Clinical Chemistry; MDI: multiple daily injections; NGSP: National Glycohemoglobin Standardization Program; POC: point-of-care.
Hyperglycemia Continuous Glucose Monitoring-Detected Adverse Glycemic Event
A total of 1391 CGM-detected level 1 hyperglycemia AGE were identified, with a median [IQR] duration of 190 minutes [73, 472] (Table 2). Of these, 806 (58%) were “concordant CGM-POC AGE” (CGM AGE corroborated with an accompanying POC AGE), 264 (19%) were “discordant CGM AGE” (accompanying POC measurements did not detect an AGE), and 321 (23%) were “CGM-only AGE” (no accompanying POC measure within 60 minutes) (Table 2; Supplementary Figure 1A).
Hyperglycemia and hypoglycemia CGM-detected AGE.
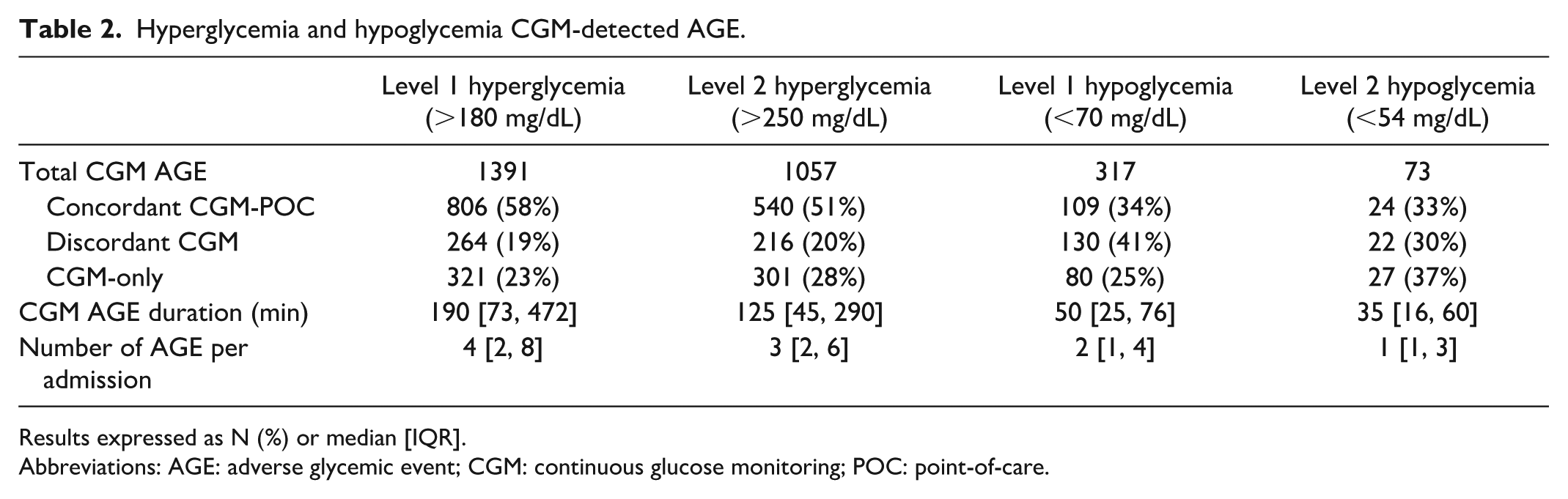
Results expressed as N (%) or median [IQR].
Abbreviations: AGE: adverse glycemic event; CGM: continuous glucose monitoring; POC: point-of-care.
Among level 1 hyperglycemia “concordant CGM-POC AGE,” CGM detected hyperglycemia AGE prior to POC in 712 (88%) cases, and CGM detection was a median [IQR] of 70 minutes [22, 166] earlier than POC (Figure 2). Earlier detection was more evident during the nocturnal period (median [IQR] of 96 minutes [30, 221] earlier) compared to daytime (median [IQR] 68 minutes [20, 158] earlier, P = .007). In addition, at the time of first detection of AGE, CGM glucose was significantly lower than POC glucose (median 187 vs 223 mg/dL, P < .0001), indicating detection at milder levels of adverse glycemia (Figure 3A).
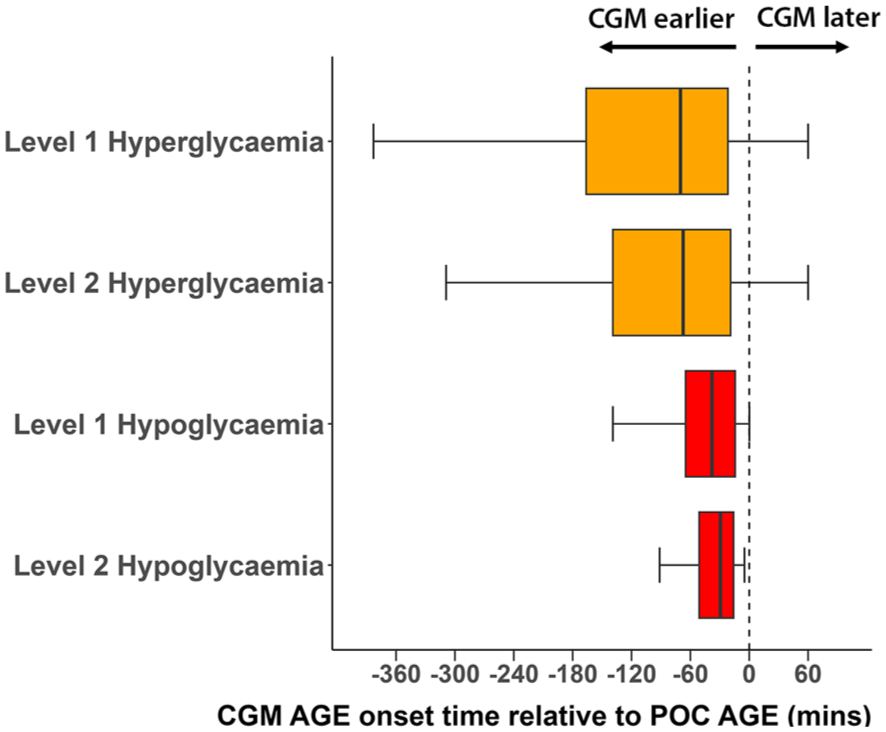
Time difference between adverse glycemic event (AGE) onset as detected by continuous glucose monitoring (CGM) or point-of-care (POC) measures among concordant CGM-POC AGE, for level 1 and level 2 hyperglycemia and hypoglycemia AGE. Negative time values indicate CGM detection occurred earlier than POC detection.
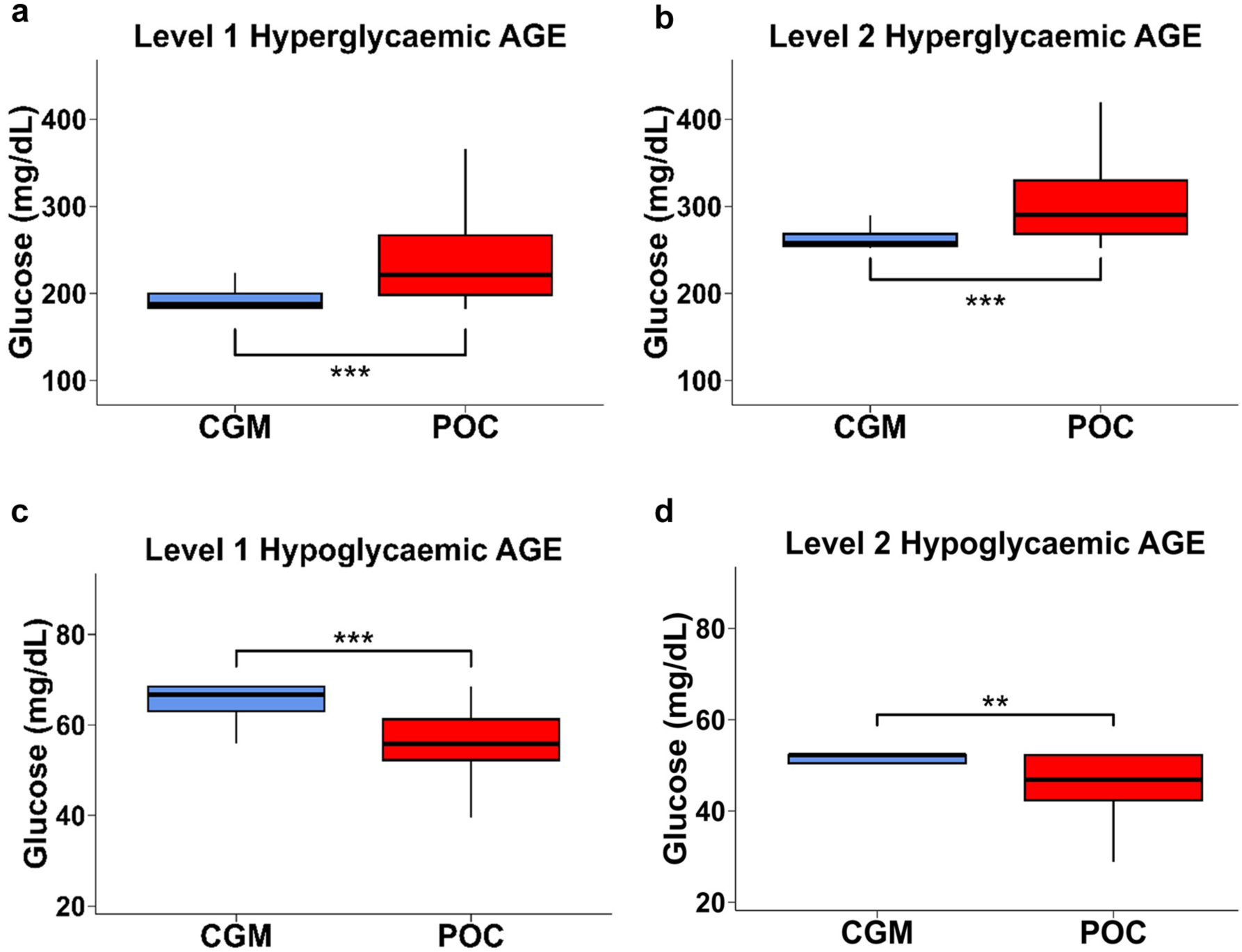
Boxplot of glucose concentrations at onset of hyperglycemia and hypoglycemia adverse glycemic events (AGE) detected by continuous glucose monitoring (CGM) with concordant point-of-care (POC) measures, for level 1 hyperglycemia AGE (Panel A), level 2 hyperglycemia AGE (Panel B), level 1 hypoglycemia AGE (Panel C), and level 2 hypoglycemia AGE (Panel D).
Of the 1057 level 2 hyperglycemia CGM-detected AGE, 51% were “concordant CGM-POC AGE,” 20% were “discordant CGM AGE” and 28% were “CGM-only AGE” (Table 2; Supplementary Figure 2A). Among level 2 hyperglycemic “concordant CGM-POC AGE,” CGM detected hyperglycemia a median [IQR] of 67 minutes [19, 139] earlier than POC (Figure 2) and at a milder degree of hyperglycemia (median 259 vs 293 mg/dL, P < .0001) (Figure 3B).
Hypoglycemia Continuous Glucose Monitoring-Detected Adverse Glycemic Event
A total of 317 CGM-detected level 1 hypoglycemia AGEs were identified, with median [IQR] duration of 50 minutes [25, 76]. Of these, 109 (34%) were “concordant CGM-POC AGE,” 130 (41%) were “discordant CGM AGE,” and 80 (25%) were “CGM-only AGE” (Table 2; Supplementary Figure 3A).
Among level 1 hypoglycemia “concordant CGM-POC AGE,” CGM detected hypoglycemia AGE earlier than POC in 105 (96%) cases; CGM detection was a median [IQR] of 38 minutes [14, 65] earlier than POC (Figure 2). Earlier detection was more evident during the nocturnal period (median [IQR] = 53 minutes [22, 92] earlier) than during the daytime (median [IQR] of 28 minutes [13, 55] earlier), P = .007. In addition, at the time of first detection of AGE, CGM glucose was significantly higher than POC glucose (median 67 vs 56 mg/dL, P < .0001), indicating detection at milder levels of adverse glycemia (Figure 3C).
Findings were similar for level 2 hypoglycemia CGM-detected AGE (Table 2; Supplementary Figure 4A), with CGM detecting AGE a median [IQR] of 29 minutes [16, 51] earlier than POC (Figure 2), and at a milder degree of hypoglycemia (median 52 vs 47 mg/dL, P =.002) (Figure 3D).
For hyperglycemia and hypoglycemia, “concordant CGM-POC AGE,” CGM, and POC glucose concentrations at AGE onset are shown, by hour of day, in Supplementary Figures 5 to 8. Higher POC glucose concentrations occurred during the day reflecting post-prandial hyperglycemia, but hypoglycemia occurred throughout the 24-hour period. The number of CGM AGEs is shown, by hour of day, in Supplementary Figure 9, demonstrating peaks in hyperglycemic AGE post-prandially but a more even distribution of hypoglycemic AGE.
Additional subgroup analyses of hyperglycemia and hypoglycemia CGM-detected AGE according to CGM device are presented in Supplementary Tables 1 (Libre 2) and 2 (Dexcom G6).
Point-of-Care-Detected Adverse Glycemic Event
Amongst POC measures detecting hyperglycemia and hypoglycemia, 3% were “POC-only events” (86/2713 of level 1 hyperglycemia AGE; 35/1338 of level 2 hyperglycemia AGE; 9/260 level 1 hypoglycemia AGE; 2/75 level 2 hypoglycemia AGE [Supplementary Figures 1B-4B]).
Discordant Continuous Glucose Monitoring and Point-of-Care Results
For “discordant CGM AGE,” 54% of level 1 hyperglycemia events (143/264) and 49% of level 1 hypoglycemia events (50/102) exceeded %20/20 agreement limits and were thus deemed due to either inaccurate CGM, inaccurate POC measures, or both. Conversely, 51% of level 1 hypoglycemia events (52/102) and 46% of level 1 hyperglycemia events (121/264) were within %20/20 agreement limits.
Discussion
Our multi-center study compared CGM- and POC-detected AGE in a cohort of non-critically ill adults with T1D admitted to hospital. We observed CGM had greater ability to detect adverse glycemia, which may otherwise have been missed by routine ward-based POC BG monitoring alone. For CGM-detected events, a quarter of Level 1 hyperglycemia and hypoglycemia AGE were detected solely by CGM. In studies of masked CGM use in hospitals among people with predominantly type 2 diabetes (T2D), 50% to 87% of hypoglycemia events were detected solely by CGM.31,32 The lower proportion of hypoglycemia events detected solely by CGM in our study may relate to the use of unmasked real-time CGM, prompting behavioral modification by the person with diabetes and confirmatory POC testing by ward-based health professionals.
In addition, CGM was observed to detect abnormal glucose excursions earlier than routine POC glucose testing. In a small observational study (12 hypoglycemic events in 36 inpatients with T1D/T2D), masked CGM use detected hypoglycemia 3 hours 34 minutes prior to POC. 33 By comparison, in our study, the relatively shorter interval between CGM and POC detection of hypoglycemia may be attributable to the use of unmasked real-time CGM. The earlier detection of hyper- and hypoglycemia AGE by CGM, at lesser degrees of dysglycemia, could provide advanced warning of evolving glycemic excursions and enable the development of proactive interventions to achieve target glycemia in hospital. This is evident in some studies of real-time CGM use in hospital demonstrating shorter hypoglycemia duration (in people with T2D).34,35 The value of CGM for enhanced detection of AGE may be even greater in people with T1D compared to T2D, given greater glycemic lability.
Discordance between CGM and POC measures was evident in our study. In approximately half of “discordant CGM AGE” (both hypoglycemia and hyperglycemia), CGM and POC measures were within %20/20 agreement limits. This suggests glucose measurements by CGM and POC were accurate in these events, and that POC was close to, but did not exceed, the AGE criteria threshold. For the remaining half of “discordant CGM AGE,” where CGM and POC measures exceeded %20/20 agreement limits and therefore did not meet this definition of accuracy, it was not possible retrospectively to confirm which measure (CGM or POC) was more representative of true BG. Manual typographical errors of POC glucose entries in patient electronic hospital records may have contributed to inaccurate POC-detected hypoglycemic events. However, it is also possible CGM device factors contributed, including sensor inaccuracy, the impact of hypotension/hypoxia/hypothermia, transient mechanical compression events, the number of days of sensor wear pre-admission, and sensor lag.36,37
While glycemic events detected by CGM benefit from the existence of consensus definitions,21,28 POC glycemic events in hospital lack standardized definitions, and their single point-in-time measures are not conducive to differentiating between a single continuous AGE or 2 separate AGE with resolution between. Thus, in our analyses, while the occurrence of POC events was numerically higher for some categories (especially hyperglycemic events), this does not necessarily mean that discrete POC events were more common; rather, all POC measures exceeding threshold criteria were counted as separate events, while CGM measures were grouped within discrete event episodes.
An important limitation of our study was the availability of unmasked real-time CGM glucose measures to patients and health care providers, as well as patients’ usual high/low glucose CGM alarms, as these may have affected the incidence of POC events. It is likely that the frequency of POC hypoglycemia might be lower than otherwise expected in our cohort through unmasked CGM use, due to either pre-emptive or reactive self-treatment of hypoglycemia based upon CGM measures without waiting for nursing staff to perform POC testing. Unfortunately, information regarding self-treatment of hypoglycemia was not available, as this was not reliably documented in medical records. Conversely, frequency of POC hyperglycemia may have been increased due to prompting of additional confirmatory POC testing in response to increased availability of glucose measures or alarms from CGM. Nevertheless, these issues only serve to reinforce the potential utility and benefit (and reflect the real-world status) of unmasked CGM use in hospital.38-40 Use of unmasked real-time CGM in people with T1D is likely to become the future reality of mainstream diabetes care in hospital, given people with T1D are increasingly wearing CGM devices. In addition, our study analyzed distinct patient admissions; therefore, patients with multiple admissions may be overrepresented in the present analysis. Finally, it is important to also highlight that our results may not be generalized to people with type 2 diabetes, the elderly, or those who are critically unwell.
To date, randomized controlled trials investigating real-time inpatient CGM use to guide clinical care in non-critical care wards have produced mixed results. In two studies, real-time inpatient CGM use reduced hyperglycemia and mean glucose,34,41 while in other studies, CGM use was not associated with improvements in mean glucose or time-in-range (70-180 mg/dL),35,42,43 although it did reduce hypoglycemia.35,42 Therefore, despite the additional glycemic data provided through real-time CGM use in hospital, optimal strategies to leverage off these detailed glucose observations for improvement in patient outcomes remain to be determined.
Conclusion
In our real-world setting of adults with T1D admitted to hospital, unmasked CGM use was observed to detect a majority of AGE, and at earlier timepoints, compared to POC BG monitoring. Incorporating CGM into the management of inpatients with diabetes could facilitate proactive interventions to reduce AGE. Future studies will investigate the impact of in-hospital CGM use on glycemia, clinical, and patient experience outcomes.
Supplemental Material
sj-docx-1-dst-10.1177_19322968251395088 – Supplemental material for Enhanced Detection of Adverse Glycemic Events in Hospital with Unmasked Continuous Glucose Monitoring Versus Point-of-Care Testing in People with Type 1 Diabetes
Supplemental material, sj-docx-1-dst-10.1177_19322968251395088 for Enhanced Detection of Adverse Glycemic Events in Hospital with Unmasked Continuous Glucose Monitoring Versus Point-of-Care Testing in People with Type 1 Diabetes by Ray Wang, Mervyn Kyi, Brintha Krishnamoorthi, Ailie Connell, Cherie Chiang, Debra Renouf, Rahul Barmanray and Spiros Fourlanos in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to thank each hospital’s diabetes educators for their efforts linking patient CGM devices with hospital CGM software accounts, as well as the Business Intelligence team at The Royal Melbourne Hospital, the Data Analytics and Insights service at Eastern Health, and the Data Analytics and Reporting team at Peninsula Health for their support.
Abbreviations
AGE, adverse glycemic event; BG, blood glucose; CCI, Charlson comorbidity index; CGM, continuous glucose monitoring; CSII, continuous subcutaneous insulin infusion; DKA, diabetic ketoacidosis; EH, Eastern Health; GI, gastrointestinal; HbA1c, hemoglobin A1c; IFCC, International Federation of Clinical Chemistry; IQR, interquartile range; MDI, multiple daily injections; MH, Melbourne Health; NGSP, National Glycohemoglobin Standardization Program; PH, Peninsula Health; POC: point-of-care; SD, standard deviation; T1D, type 1 diabetes; T2D, type 2 diabetes
Author Contributions
R.W., M.K., and S.F. were involved in the conception and design of the study. R.W. and B.K. performed the data acquisition. R.W. performed the statistical analysis and wrote the first draft of the manuscript. M.K., R.B., and S.F. provided the critical revisions to the manuscript. All authors made a significant contribution to finalizing the manuscript and approved the final version of the manuscript. S.F. provided overall study supervision and is the guarantor of this work.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.F. contributes to the advisory panel for Viatris Inc and Pfizer Inc; has received honoraria for speaker fees for AstraZeneca, Boehringer-Ingelheim, Lilly, and Novo Nordisk; has received honoraria for advisory fees for Medtronic, Mylan, Pfizer, and Sanofi; and has received research funding from Dexcom Inc. M.K. has received honoraria for speaker fees for AstraZeneca. R.W. has received research funding from Dexcom Inc.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: R.W. is the recipient of an Australian Commonwealth Government RTP Scholarship, ACADI PhD Grant, Fred Knight Research Scholarship, Rowden White Scholarship, and Gordon P Castles Scholarship.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
