Abstract
Background:
We used real-world electronic health record (EHR) data to examine HbA1c levels among children and adults with type 1 diabetes (T1D) who are classified as continuous glucose monitor (CGM) users after T1D diagnosis and switch to self-monitoring of blood glucose (SMBG) during follow-up, versus people who opt for SMBG after T1D diagnosis and switch to CGM during follow-up visits.
Methods:
We conducted an observational, case-crossover study using electronic medical record (EMR) data from the T1D Exchange Quality Improvement Collaborative. The primary outcome in this study was HbA1c. Baseline HbA1c levels were taken at the index date, corresponding to initial device classification, and compared with HbA1c value recorded at the clinic visit following device switch.
Results:
Of all patients classified in the SMBG group, 7,706 switched to CGM use within the 5-year study time frame, and 5,123 of all initial CGM users switched to SMBG within the study time frame and were included in this analysis. At baseline, median (interquartile range [IQR]) HbA1c for SMBG use was 8.1 (2.4), whereas postcrossover to CGM use, there was a decline in median (IQR) levels to 7.7 (1.9) (P < .001). For baseline CGM users, median (IQR) HbA1c levels were 7.9 (2.0), and postcrossover to SMBG, median (IQR) HbA1c levels increased to 8.0 (2.9) (P < .001).
Conclusion:
We found that people who switched to CGM use had significantly improved HbA1c levels compared to those who switched to glucose monitoring with SMBG.
Keywords
Introduction
Evidence from numerous clinical research studies has established that among people with type 1 diabetes (T1D), the use of continuous glucose monitors (CGMs) for diabetes management is superior to self-monitoring of blood glucose (SMBG) with regard to glycemic outcomes.1 -4 With the use of diabetes technology, including recent-generation real-time CGM devices and intermittently scanned CGM devices, people with T1D have lower risk of hypoglycemia and more people can achieve American Diabetes Association recommended hemoglobin A1c (HbA1c) targets. 5 Continuous glucose monitor use has also been shown to reduce adverse diabetes-related outcomes for vulnerable populations, including young children, as well as people of minority race/ethnicity 6 -10 Data show that glycemic management is more challenging for children and adolescents due to cognitive and physical developmental issues, as well as among older adults and people of Black and Hispanic race/ethnicity owing to various of socioeconomic disparities; however, the use of CGMs improves HbA1c levels and diabetes-related clinical outcomes in these vulnerable populations.11 -13
While several randomized clinical trials have now reported the benefit of CGM over SMBG for improved glucose outcomes, there is a limited evidence from observational crossover studies demonstrating how glycemic levels are affected when SMBG patients switch to CGM use, or CGM users switch to SMBG. In this study, we use real-world electronic health record (EHR) data to examine HbA1c levels among children and adults with T1D who are classified as CGM users after T1D diagnosis and switch to SMBG during follow-up, versus people who opt to monitor glucose with SMBG after T1D diagnosis and switch to CGM during follow-up visits. We also examine the change in HbA1c after device switch across race/ethnicity groups.
Methods
Study Design
We conducted an observational, case-crossover study using EHR data from 20 clinics within the T1D Exchange Quality Improvement Collaborative (T1DX-QI Collaborative, a multicenter network of 50 endocrinology clinics across the United States). 14 The T1DX-QI database has been extensively described;14 -16 in brief, diabetes centers contribute over 120 data elements from the EHR to a central platform which is shared amongst collaborators for quality improvement and population health management.
The aim of this study is to compare the effectiveness of CGM use to SMBG on glycemic outcomes. This study used data collected between 2017 to 2021. Pediatric and adult persons with T1D duration of at least 1 year and had complete information on CGM use or SMBG use at baseline and follow-up visits, as well as corresponding HbA1c values were included in this study (Figure 1: Study design).
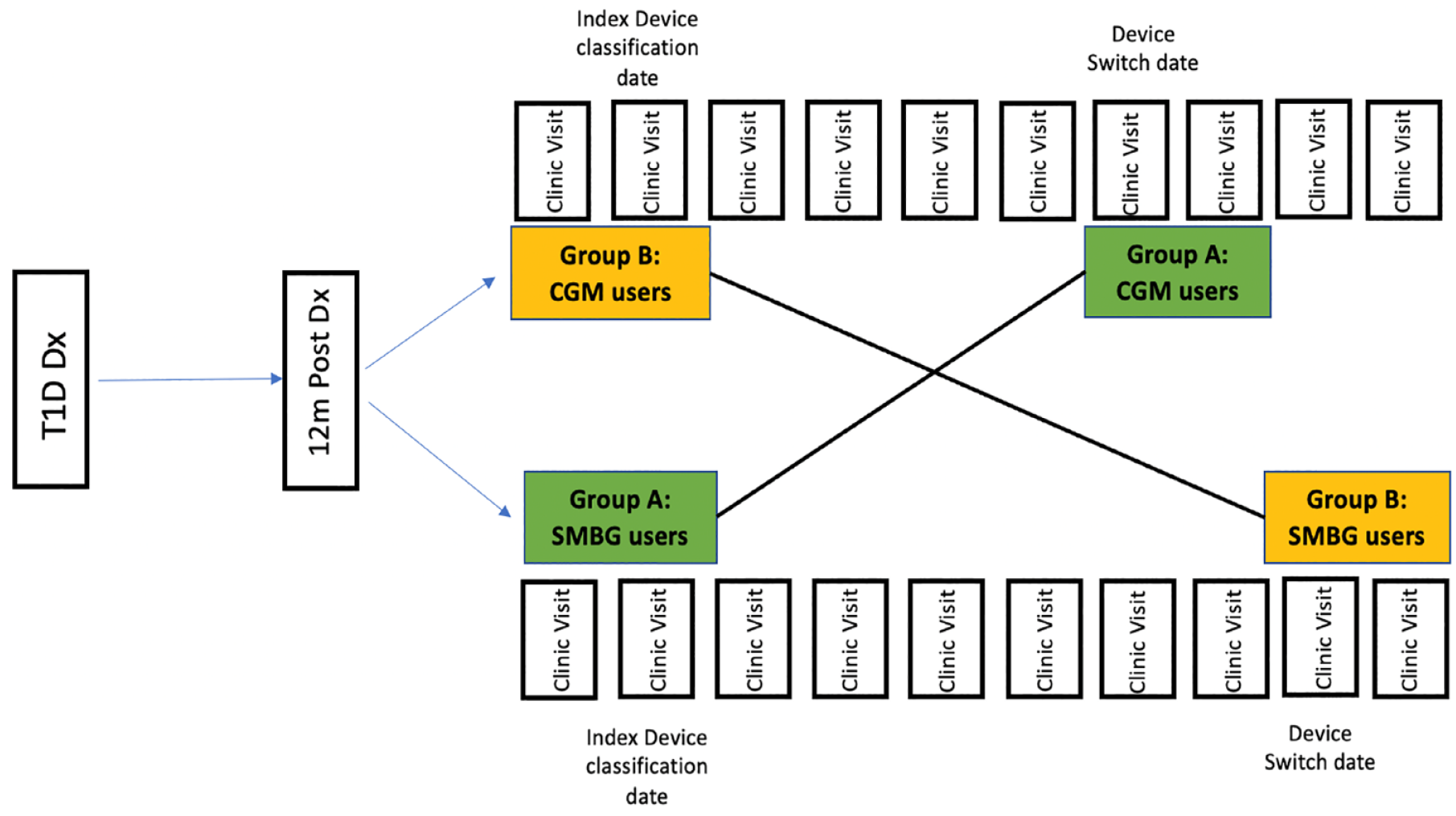
Study design.
Device Classification at Baseline
The index date for glucose monitoring device classification was defined as a patient’s first encounter date after 1 year of T1D diagnosis, with an EHR record indicating CGM use or SMBG use, along with one preceding clinic visit within 6 months with EHR evidence confirming use of the same device between 2017 and 2021. Specifically, T1D patients with an EHR record of SMBG at index date, with additional evidence of SMBG (record of blood glucose meter (BGM) company or model with no evidence of CGM use) from an earlier clinic visit within 6 months prior, were classified as the “SMBG group” (Group A), whereas those with an EHR evidence of CGM use at index date, with an additional record of CGM use from a preceding clinic visit were classified as “CGM group” (Group B). Continuous glucose monitor users included all real-time CGM device users, while patients who reported using an intermittent scanning CGM device or hybrid closed-loop system (either Tandem t: slim X2, Medtronic 670G or Medtronic 770G) were excluded from this analysis.
Crossover Groups
After CGM or SMBG classification at index date, patients were tracked within the January 2017 to May 2021 time frame, and each follow-up clinic visit was checked for device use status. If at a subsequent visit within the follow-up time period, patients in the SMBG group (Group A) report change in glucose monitoring from SMBG to CGM, the date of that visit is taken as date of device switch. For these patients, if a subsequent visit within 6 months postdevice switch date provides confirmatory evidence of device switch, those patients are classified under crossover groups, and the corresponding HbA1c value within 30 days of this confirmatory date is taken as the postcrossover HbA1c value. Similarly, if CGM users (Group B) switch to SMBG at a clinic encounter during follow-up, they are classified under the SMBG group if there is an additional confirmation of SMBG at a subsequent clinic visit within 6 months. While it is possible that patients may switch device back and forth multiple times, only the first switch was evaluated in this study.
Primary Outcome
The primary outcome in this study was HbA1c. We compared baseline HbA1c levels taken at the index date, corresponding to initial device classification for Group A and Group B, relative to the HbA1c value recorded at the clinic visit following device switch.
Secondary Outcomes
Secondary outcomes included in this study were self-reported diabetic ketoacidosis (DKA) and severe hypoglycemia (SH) at index visit and postcrossover visit. Diabetic ketoacidosis events were defined as any patient-reported inpatient or ambulatory DKA event recorded in the EHR from their most recent clinic visit after device switch. Severe hypoglycemia events were defined as any patient-reported inpatient or ambulatory SH event recorded in the EHR from their most recent clinic visit after device switch. Data collection for this project was approved as exempt by the Western Institutional Review Board.
Covariates
Covariates of interest included sociodemographic variables of age, sex, race/ethnicity, insurance status, and information on insulin use including total insulin dose and insulin regimen. Age (in years) was taken at baseline, i.e., corresponding to the index device classification date and was analyzed as a continuous variable. Sex was analyzed as a categorical variable (male/female), whereas for race/ethnicity patients were classified into White, Black, Hispanic, Other groups. Baseline insurance status was categorized into Private (Employee based), Public (Medicare/Medicaid), and Other. Total insulin daily dose (U/kg/day) was recorded in the EHR at baseline. Patients with a record of insulin pump use were classified under “insulin pump,” while those reporting multiple daily injection use were classified under “MDI” category at baseline.
Statistical Analysis
To determine the distribution of patient attributes and clinical outcomes among T1D people in the SMBG or CGM groups at baseline, we performed a descriptive analysis reporting mean and standard deviation for continuous variables and frequency and percentage for categorical variables. Differences between the two groups in the descriptive analysis were assessed using chi-square (for categorical variables), Kruskal-Wallis test (for continuous variables) as applicable. Change in mean HbA1c levels was examined across both groups before and after device switch (Figure 1). We used linear mixed models to determine the difference in mean HbA1c values after adjusting for potential confounders of age, sex, race/ethnicity, insurance status, and total daily insulin.
Subgroup analysis: We also performed an additional analysis using the same cohort of people in Groups A and B stratified by race/ethnicity groups, pediatric (<18 years) and adult (>18 years) populations, as well as restricting the population to those who had an elevated HbA1c of >9% to determine the impact of CGM use on patients with elevated glucose levels. All analyses were performed using R Software (version 4.0.2).
Results
A total of 38,231 people, aged >2 years with T1D diagnosis for at least 1 year, were analyzed using the EHR database. Of the eligible population, 18,169 were classified as SMBG users, whereas 17,819 were grouped under CGM users. Of all patients classified in the SMBG group, 7,706 switched to CGM use within the 5-year study time frame and 5,123 of all initial CGM users switched to SMBG within the study time frame (Figure 2) and were included in this analysis.
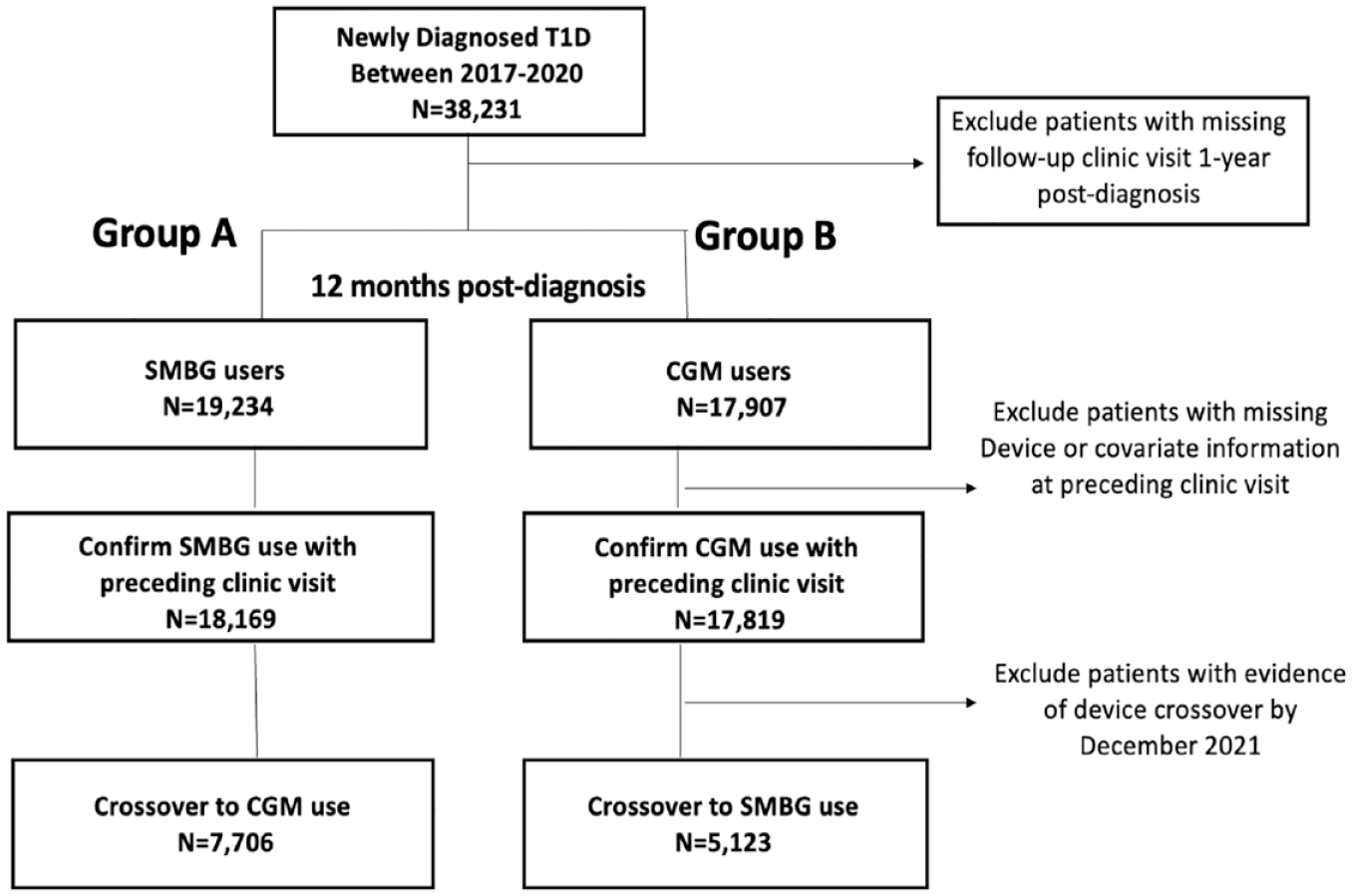
Study flow diagram.
At baseline, Group A (SMBG group who switched to CGM) and Group B (CGM users who switched to SMBG) were comparable with mean (SD) age of 19.3 (13) was 18.7 (11) years, respectively. The two groups were also comparable with respect to sex, race/ethnicity, insurance status, and total daily insulin dose recorded at baseline. Mean (SD) duration of follow-up (in years) until crossover was 0.9 (1.2) for Group A and 2.0 (2.4) for group B. While DKA events reported by patients at baseline were similar in Group A and B (% DKA events in Group A: 3% vs. Group B: 4%; P = .21), self-reported SH events were slightly higher in those using CGM at baseline (Group A: 4% vs. Group B: 7%; P < .001) (Table 1).
Comparison of Baseline Patient Characteristics and Clinical Outcomes Among SMBG Group and CGM Users at Baseline.
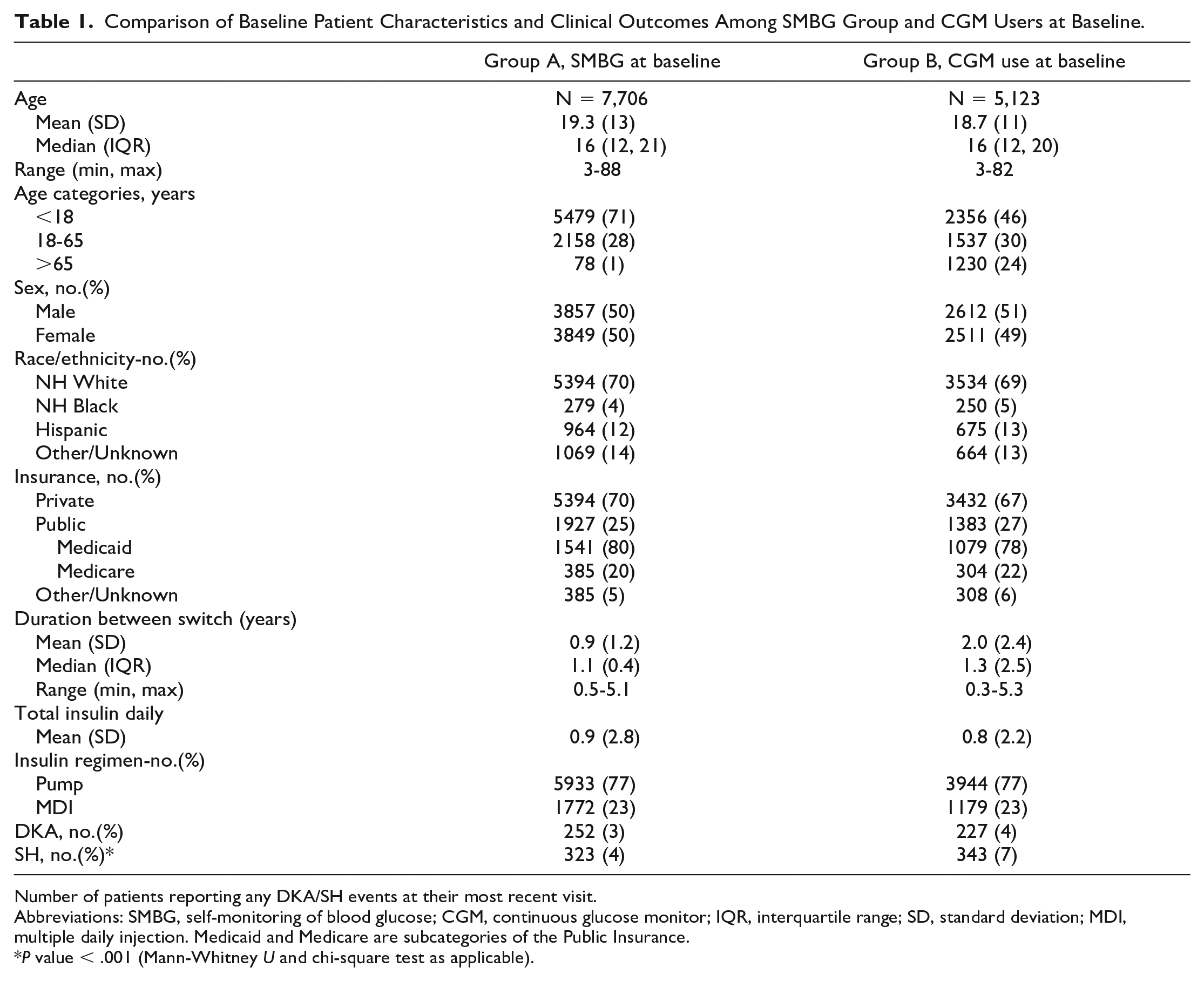
Number of patients reporting any DKA/SH events at their most recent visit.
Abbreviations: SMBG, self-monitoring of blood glucose; CGM, continuous glucose monitor; IQR, interquartile range; SD, standard deviation; MDI, multiple daily injection. Medicaid and Medicare are subcategories of the Public Insurance.
P value < .001 (Mann-Whitney U and chi-square test as applicable).
At baseline, mean (SD) HbA1c for Group A was 8.6 (2.1), whereas postcrossover to CGM use there was a decline in mean (SD) levels to 8.0 (1.7) (P < .001). For Group B, mean (SD) HbA1c levels were 8.3 (2.1) and postcrossover to SMBG, mean (SD) HbA1c levels increased to 8.4 (1.9) (P < .001) (Figure 3). We used a linear mixed model to examine the effect of device group on HbA1c levels. After adjusting for potential confounders including age, sex, race/ethnicity, and insurance status (Table 2), adjusted mean HbA1c (95% CI) for Group A and Group B was 7.9 (7.6,8.1) and 8.3 (8.0,8.6) (P < .001), respectively.
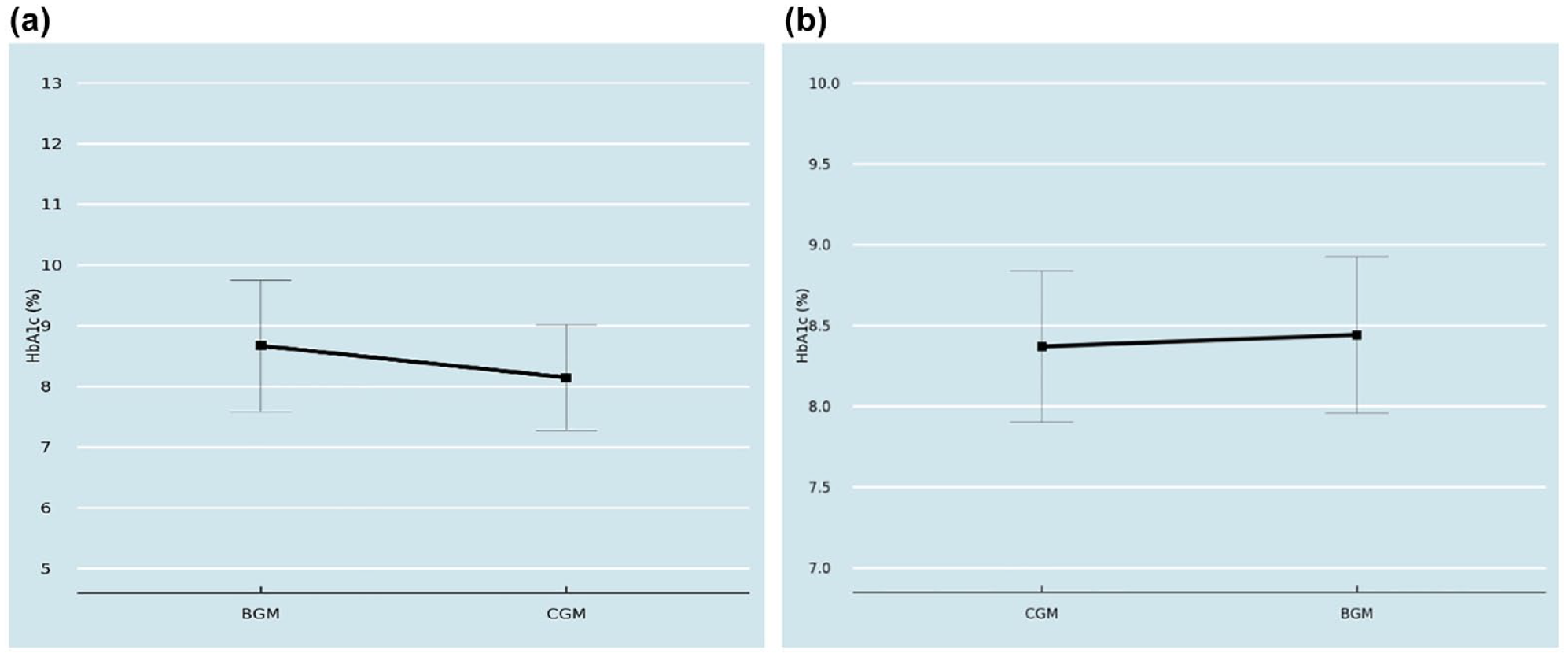
Mean HbA1c levels after switch from (a) SMBG to CGM (Group A) and (b) CGM to SMBG or BGM (Group B).
HbA1c Levels Among People With T1D at Baseline and Post Device Switch.

Abbreviations: SMBG, self-monitoring of blood glucose; CGM, continuous glucose monitor; IQR, interquartile range; SD, standard deviation.
P value < .001 within-group comparison of before and after switch A1c levels.
In addition, we stratified the two groups by race/ethnicity and found that in Group A, while all racial/ethnic groups showed an improvement in HbA1c levels after crossover to CGM, people of Black and Hispanic race/ethnicity had relatively higher HbA1c levels relative to their White peers (mean (SD): 9.1 (2.2) and 8.2 (2.3) vs. 7.6 (2.0), respectively; P value < .001). Similarly, all racial/ethnic groups in Group B showed elevated HbA1c levels after crossover to SMBG; however, this increase was more notable in the Black and Hispanic populations (mean (SD): 10.1 (1.9) and 9.2 (2.0) vs 8.1 (1.9); P value < .001) (Figure 4).
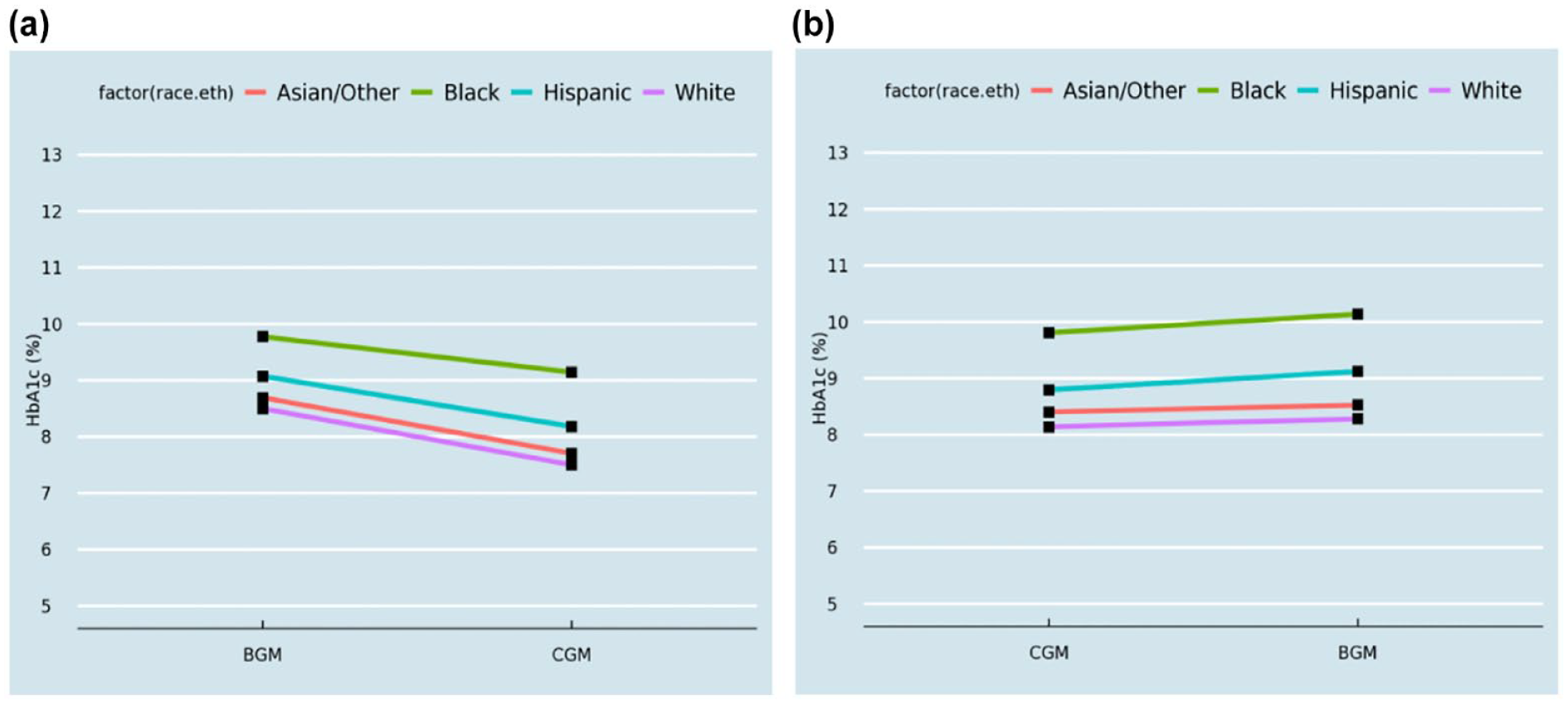
Mean HbA1c levels after switch from (a) SMBG to CGM and (b) CGM to SMBG or BGM across race/ethnicity.
On stratifying Groups A and B by pediatric and adult patients, we found that both children and adults showed an improvement in HbA1c levels after crossover to CGM (mean (SD) A1c: 8.1 (1.0) and 8.0 (1.9), respectively), whereas a small increase in HbA1c was observed among people who switched from CGM use at baseline (Mean(SD) A1c: 8.3 (0.5) and 8.4 (0.9)) to self-monitoring blood glucose (mean (SD) A1c: 8.5 (0.5) and 8.5 (1.0), respectively (P < .001) (Table 3).
Mean (SD) A1c Levels Among Pediatric and Adult People With T1D at Baseline and Post Device Switch.

Abbreviations: SMBG, self-monitoring of blood glucose; CGM, continuous glucose monitor.
P value<.001 within-group comparison of before and after switch A1c levels.
Finally, in additional subgroup analysis, we examined the high-risk sub-population within Group A (i.e., those having a baseline HbA1c > 9%) and found that the difference between baseline and post-crossover to CGM use mean HbA1c levels was −2.1% (P < .001) (Figure 5).
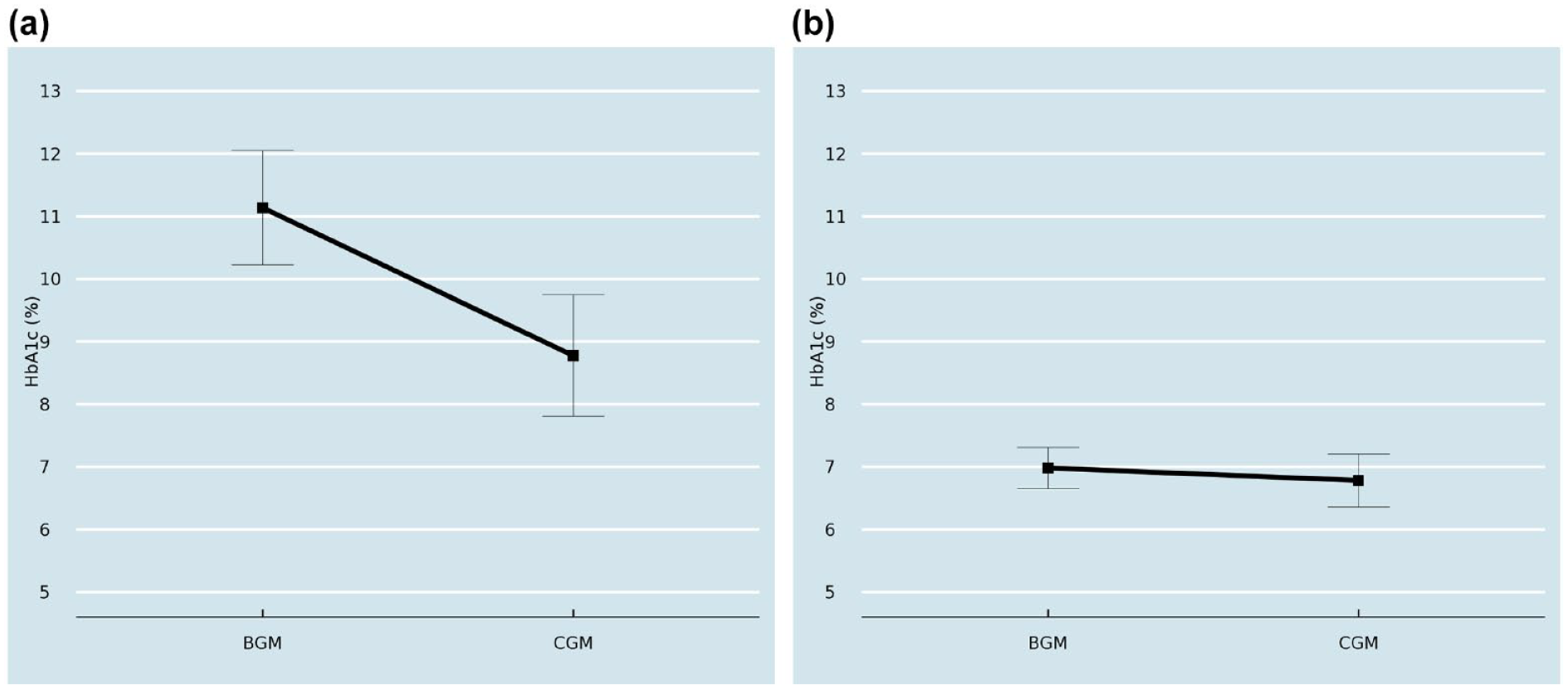
Subgroup analysis showing Mean A1c levels after switch from SMBG (or BGM) to CGM among people with (a) HbA1c >9 and (b) HbA1c <7 at index visit.
Discussion
In this study, we used EHR data from real-world clinical settings to determine the impact of glycemic levels among SMBG and CGM users after switching to CGM and SMBG, respectively. We found that people who switched to CGM use had significantly improved HbA1c levels compared to those who switched to glucose monitoring with SMBG.
The findings of improved HbA1c, as well as CGM-derived glycemic metrics, after initiation of real-time CGM use have been well established by various randomized controlled trials as well as observational studies.2,17,18 The DIAMOND study, a Randomized Controlled Trial, demonstrated a significantly greater decrease in HbA1c level during 24 weeks with CGM versus routine care with adjusted treatment-group difference in mean change from baseline was −0.6% (95% CI, −0.8% to −0.3%; P < .001). 2 Similarly, the GOLD trial, Open-label crossover randomized clinical trial, showed that Mean HbA1c was 7.92% (63 mmol/mol) during continuous glucose monitoring use and 8.35% (68 mmol/mol) during conventional monitoring (mean difference, −0.43% [95% CI, −0.57% to −0.29%] or −4.7 [−6.3 to −3.1 mmol/mol]; P < .001). 3 Our findings agree with these previous studies, confirming that even in uncontrolled, real-world settings, CGM use is associated with improved glycemia among people with T1D.
In this study, we also examined the eligible study population stratified by race/ethnicity groups. Our findings showed that while those who self-monitor blood glucose show significantly improved glycemic outcomes after crossing over to CGM use across each racial/ethnic groups, there remain marked disparities between minority Black and Hispanic racial/ethnic groups relative to their White peers. There are several studies that document racial disparities in diabetes treatment and their impact on adverse diabetes outcomes, including HbA1c levels, DKA, and SH.6 -10 Findings from the T1DX-QI collaborative have also shown disparities in health outcomes despite access to diabetes technology and propose actionable steps to address inequities in health care delivery by engaging diverse stakeholders.19,20 Our findings agree with these studies and suggest that while continuous glucose monitoring improves glycemic outcomes for all vulnerable groups, it is imperative to facilitate access and education to promote CGM adoption and continued use by all patient groups.
Finally, in a subpopulation analysis, we also found that high risk patients, people with elevated HbA1c levels (HbA1c>9%) at baseline who are more likely to experience adverse outcomes, when switched from glucose monitoring to real-time CGM use, showed a significant improvement in their HbA1c levels.
This study had certain limitations. First, the method of glucose monitoring, that is, the use of a CGM device or conventional SMBG, was self-reported by patients at their routine follow-up clinic visit. Second, the study population may be subject to selection bias as we required eligible patients to be included if they had complete data on HbA1c and device use over the longitudinal period; this also means that these patients would consistently have accessed healthcare at their respective clinic for the entire study duration, without changing care. Third, for CGM device use in this study, we do not include intermittently scanning device or flash CGM users, and are therefore not able to investigate the impact of all CGM devices on glycemic outcomes. However, findings from a previous clinical trial have reported superior outcomes from real-time CGM users relative to flash CGM use.21 -23 We also did not exclude pump users on rtCGM, which may also potentially affect outcomes. Fourth, blood glucose levels in diabetes can be impacted by various of other clinical and socio-cultural factors. While we have accounted for a number of sociodemographic confounders in this study, including age, race/ethnicity and insurance status, other important determinants for improved health outcomes, such as comorbidities, income or education status and physical activity, were not accounted for in this study. Fifth, as this study aimed to compare outcomes among rtCGM users versus SMBG, we were unable to investigate the impact of intermittently scanned CGM users on glycemic outcomes. Finally, we do not know the reason why a person switched from CGM to SMBG or vice versa. There are multiple reasons why someone might switch from CGM to SMBG, including changing insurance coverage, discomfort, dissatisfaction, etc. and it is unclear if switch-related outcomes vary by reason for switching. Qualitative methods might be helpful in future studies to understand reasons for switching methods.
Strengths of this study include analysis based on a large T1D study population from real-world clinical settings representative of endocrinology practices across the United States. This EHR database has been curated to consistently collect reliable information on device use and diabetes related outcomes at each follow-up clinic visit through an extensive validation process.
In conclusion, this study provides real-world evidence of the benefit of continuous glucose monitoring for all people with TID, including those across racial/ethnicity groups. This suggests the need for increasing efforts to make CGM devices accessible to patients, as advocated under the ADA standards of care guidelines, 24 and innovations in care delivery to help patients continue to use CGM devices over time.
Footnotes
Abbreviations
EHR, electronic health record; CGM, continuous glucose monitor; HbA1c, hemoglobin A1c; SMBG, self-monitoring of blood glucose; T1D, type 1 diabetes; T1DX-QI, T1D exchange quality improvement collaborative.
Author Contributions
NN developed concept, drafted manuscripts, analyzed data. OE, GN provided critical revisions, edits and discussion points. All authors reviewed, edited and approved final version.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NN is PI on Dexcom and Vertex funded research projects through her organization T1D Exchange. GN is employed at Dexcom. OE is PI on Dexcom, MannKind, Medtronic, Vertex, Abbott funded research projects through his organization T1D Exchange. OE is an advisor for Medtronic Diabetes and Vertex Pharmaceuticals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Project supported by Dexcom and Helmsley Charitable Trust
