Abstract
Background:
continuous glucose monitoring systems (CGMs) play an important role in the management of T1D, but their accuracy may reduce during rapid glucose excursions. The aim of study was to assess the accuracy of recent rt-CGMs available in Italy, in subjects with T1D during 2 sessions of physical activity: moderate continuous (CON) and interval exercise (IE).
Method:
we recruited 22 patients with T1D, on CSII associated or integrated with a CGM, to which a second different sensor was applied. Data recorded by CGMs were compared with the corresponding plasma glucose (PG) values, measured every 5 minutes with the glucose analyzer. To assess the accuracy of the CGMs, we evaluated the Sensor Bias (SB), the Mean Absolute Relative Difference (MARD) and the Clarke error grid (CEG).
Results:
a total of 2355 plasma-sensor glucose paired points were collected. Both average plasma and interstitial glucose concentrations did not significantly differ during CON and IE. During CON: 1. PG change at the end of exercise was greater than during IE (
Conclusions:
the exercise affects the accuracy of currently available CGMs, especially during CON, suggesting, in this circumstance, the need to maintain blood glucose in a “prudent” range, above that generally recommended. Further studies are needed to investigate additional types of activities.
Introduction
The continuous glucose monitoring systems (CGMs) constitute a valid therapeutic tool to optimize glycemic control in patients with type 1 diabetes (T1D). Many studies have documented their benefits1-4 defining their important role in open loop systems and as base for closed loop control.5,6 These devices are made up of 3 components: a sensor, a transmitter. and a receiver. 7 The sensor, positioned in the subcutaneous tissue, continuously measures the interstitial glucose concentration and transforms it into an electrical signal, which, after processing by specific algorithms, will correspond to a blood glucose value. 7 In order to optimally perform their functions, these devices must have good accuracy, which means the sensor data should be as close as possible to the reference, represented by plasma glucose or self-monitoring of blood glucose. This crucial aspect, usually quantified by the Mean Absolute Relative Difference (MARD), 8 is used to categorize sensors’ performance. In particular, a MARD cut off of 10% has been proposed as a requisite for direct use of CGM in insulin dosage decisions. 9 The CGM sensors’ performance studies have generally been carried out in basal resting conditions, where a balance in glucose levels between plasma and interstitial compartments is expected. 10 However, this is not always true in real life, and only a few studies have assessed the accuracy of CGM during conditions in which the blood glucose steady state is perturbed. One of these conditions is typically during and after physical activity,11,12 in which rapidly occurring glycemic changes may be recorded by sensors with a significant lag-time. 13 In addition, other factors can potentially influence the accuracy of CGM during exercise: changes in tissue pH and O2 concentration,14,15 increased skin temperature, 16 glucose uptake by muscles, 17 and even the mechanical forces 11 to which the sensor is exposed.17,18 Interestingly, as different exercise types may produce different effects on glucose levels, there are potential exercise type-dependent differences in the performance of glucose sensors. However, available information on this issue is limited and results are conflicting.19-21 The aim of our study was to assess the accuracy of several CGMs, all those currently available in Italy, in patients with T1D during 2 different sessions of physical activity: moderate continuous (CON) aerobic exercise and interval exercise (IE).
Research Design and Methods
Study Design and Participants
Patients aged ≥ 18 years with T1D for at least 1 year, on continuous subcutaneous insulin infusion (CSII) for at least 6 months associated or integrated with a CGM, regularly engaged in physical activity (at least 150 minutes of physical activity per week) and with HbA1c ≤ 69.4 mmol/mol (8.5%) were invited to participate in the study. Exclusion criteria included cardiovascular events (ischemic heart disease or stroke) in the last 6 months, severe hypoglycemia within 2 weeks prior to the study, taking acetaminophen in the 2 weeks prior to the study. Recruitment was done at the University Hospital of Verona between July 2018 and November 2019. Study protocol was approved by the local Ethical Committee and, after a detailed explanation of the aims, procedures and risks of the study, all participants signed an informed consent. All subjects underwent a complete medical examination and resting heart rate was measured using a heart rate monitor (Polar Electro, Kempele, Finland). Heart Rate Reserve (HRR) was calculated by the Karvonen 22 formula. Participants were assigned, in a random order, to both a 30’ CON session, at 60% of HRR, and to a 30’ IE session, in which 2 minutes at 45% HRR were alternated with 1 minute at 75% HRR. The 2 exercise sessions were carried out in the afternoon, at least 2 days apart. Exercise sessions were performed on a cycle ergometer (Ergoselect, COSMED, Roma, Italia) under continuous heart rate monitoring and with the supervision of exercise specialists in order to constantly maintain the HR target required by the protocol. Each exercise session was preceded by a 45 minute of rest, to start the exercise in a steady state condition and was followed by a recovery phase of 45 minutes.
All patients were already using a glucose sensor and were skillful in CGM use. A second different sensor, based on the availability, was applied in each patient at least 24 h before the first session, according to the instructions provided by the manufacturer, to reduce the impact of inter-individual differences in the comparison between their performances. The sensors used in this protocol were the following, which represent all the CGM’s models currently available in Italy: Dexcom G5 (Dexcom, San Diego, CA), Dexcom G6 (Dexcom, San Diego, CA), Eversense (Senseonics, Germantown, MD), Guardian Sensor (Medtronic, Dublin, Ireland), Enlite (Medtronic, Dublin, Ireland). All CGMs were placed on the arm.
All patients were instructed to perform calibrations when required by the 2 sensors, using the same glucometer, already habitually used at home. In addition, patients were allowed to perform their habitual physical activity and they were instructed to avoid unusual exertion the day before the study sessions. Moreover, all patients were instructed to ingest the same amount of carbohydrates at lunch before each exercise session and to administer the insulin dose, calculated according to the pre-meal blood glucose, the individual insulin-to-carbohydrate ratio (ICR) and the insulin sensitivity factor (ISF). Patients were told to have lunch at 12 am and to arrive at the clinic at around 1 pm. Each patient was invited to reduce the prandial bolus of the meal before physical activity by a certain percentage, according to the glycaemia before the meal and the usual response to physical activity. They were also instructed to adapt, when necessary, the basal insulin infusion rate and/or to eat additional carbohydrates, in order to avoid either hypoglycaemias or marked hyperglycaemias before the sessions. Upon arrival at the clinic, an intravenous catheter was inserted in the antecubital vein of the patient’s forearm, to collect blood samples for plasma glucose and lactate assays. As a condition for starting each study session, blood glucose at the beginning of the sessions had to be between 100 mg/dl (5,56 mmol/l) and 250 mg/dl (13,89 mmol/l). During the 45 min observation period before starting physical exercise no significant deviations were observed.
Study Measurements
Plasma glucose was assessed every 5 minutes with a glucose analyzer (YSI 2300 STAT Plus Glucose and Lactate Analyzer; YSI Inc., Yellow Springs, Ohio, USA), using the glucose-oxidase method. Lactate concentration was measured every 15 minutes in the resting phases, before and after physical activity, and every 10 minutes during the exercise sessions, using the YSI 2300 STAT Plus Glucose and Lactate Analyzer. Interstitial glucose was automatically recorded by each sensor. The timings of the blood samples and of the sensor readings were synchronized. At the end of both physical activity sessions, all data were downloaded using dedicated software for each CGM (CareLink for Medtronic Guardian, Clarity/Diasend for Dexcom G5 and Dexcom G6, Eversense DMS for Eversense).
Calculations
Plasma glucose was considered as the reference in order to assess the accuracy of the sensors. The following parameters were calculated to evaluate the performance of each sensor during the different types of physical exercise:
- Sensor Bias (SB): the difference between interstitial glucose and plasma glucose. It describes the direction of the error and has a positive sign when the sensor overestimates the plasma glucose value and a negative sign when it underestimates it;
- Mean Absolute Relative Difference (MARD): the average of the relative errors, expressed as a percentage, between the glucose values measured by the CGM system and the corresponding reference plasma glucose values;
- Clarke Error Grid (CEG): 23 the plasma glucose values measured by the glucose analyzer, on the x axis, plotted against the values generated by the CGM, on the y axis. The diagonal represents the perfect concordance between the 2 measurements, while the data that are positioned above and below it represent an overestimation and an underestimation of the blood glucose value, respectively. The CEG quantifies graphically the potential clinical impact of the performance of systems used for glucose measurements. In particular, the different areas into which the CEG is divided are: (A) where error is less than 20%; (B) where error is greater than 20% but still not requires corrective actions by the patient; (C) where the error can lead the patient to an excessive correction of glycemic values; (D) where the error can prevent to detect dangerous blood glucose values; (E) where the error can cause a therapeutic intervention opposite to what would be necessary.
Statistical Analysis
Data were presented as mean ± SD for continuous variables and as number of cases or percentage for the categorical variables. Parametric variables were compared by T-test or ANOVA and non-parametric variables were analyzed by Mann-Whitney, Kruskal-Wallis, and Wilcoxon test, as appropriate. A
Results
Patients Characteristics
Clinical and anthropometric characteristics of all patients (M/F = 13/9) are shown in Table 1. Mean (±SD) age was 39.5 ± 9.7 years and mean HbA1c was 55.6 ± 7.8 mmol/mol (7.2 ± 0.9%). According to the study protocol, all patients were using an insulin pump associated or integrated with CGM: in particular, 9 subjects routinely wore a CGM Dexcom G5, 3 subjects a CGM Dexcom G6, 4 subjects a CGM Guardian Sensor, and 6 subjects an Eversense CGM. In all patients, a second, different sensor was applied, in addition to the already used device. The Enlite sensor was applied only as a second sensor, because it was not routinely used by any of the study participants.
Baseline Characteristics of Participants (
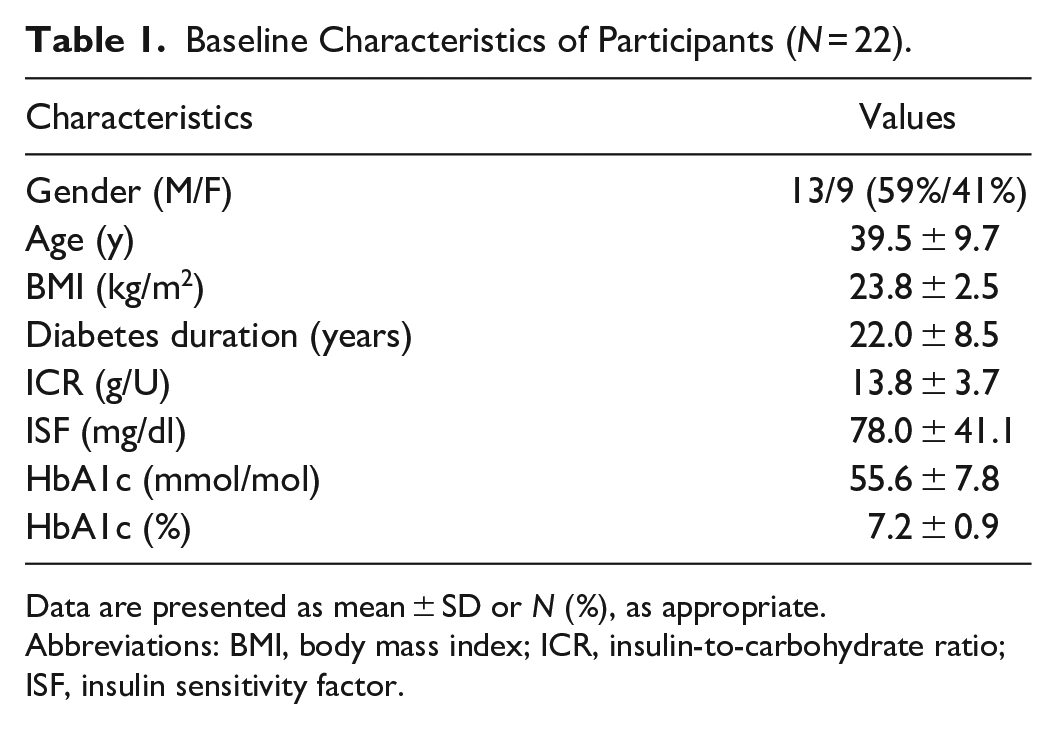
Data are presented as mean ± SD or
Abbreviations: BMI, body mass index; ICR, insulin-to-carbohydrate ratio; ISF, insulin sensitivity factor.
A total of 40 comparisons were obtained in CON sessions, because 2 Enlite readings, 1 Guardian reading and 1 Dexcom G6 reading were missing due to technical reasons; a total of 39 comparisons were obtained during IE sessions for the same reason. In particular, 3 Enlite readings, 1 Guardian reading and 1 Dexcom G6 reading were missing. As a whole, a total of 2355 plasma-sensor glucose paired points were collected.
Table 2 summarizes the nutritional and insulin therapy parameters of patients before the 2 exercise sessions, the mean values of the amount of carbohydrates eaten at lunch, the pre-meal insulin bolus administered, the basal insulin infusion rate during the sessions, and the percentage of changes in the pre-meal insulin bolus and in the basal insulin infusion rate adopted in order to limit the risk of exercise-induced hypoglycemia, as compared to what was usually done in resting conditions by these patients.
Mean Nutritional and Insulin Therapy Parameters of Patients Before the 2 Physical Activity Sessions.
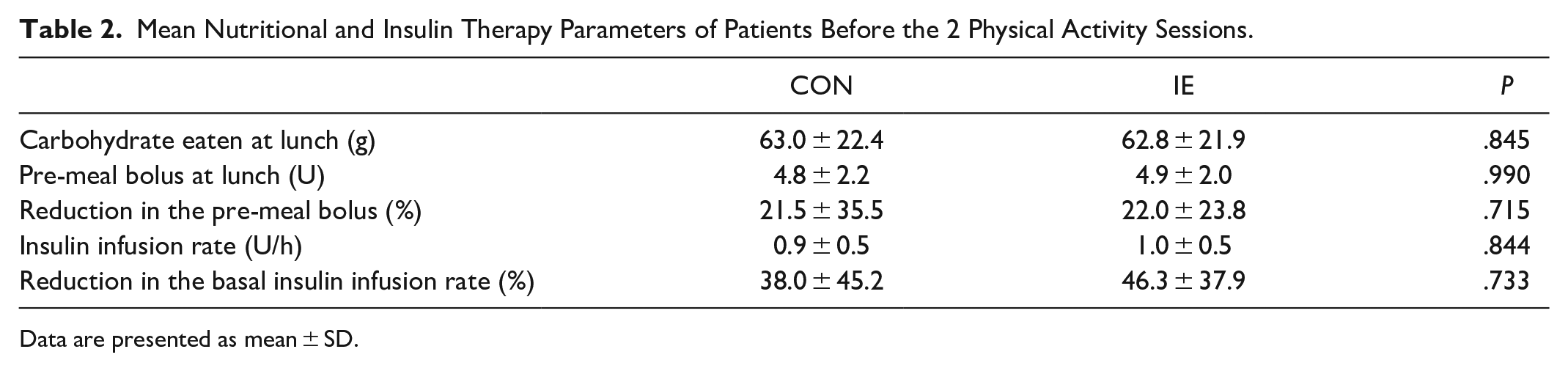
Data are presented as mean ± SD.
Lactate and Glucose and Curves During the Exercise Sessions
Plasma lactate concentrations were similar before the 2 exercise sessions and, increased to a greater extent during IE as compared to CON (mean during the 30 minutes of activity respectively 1.90 ± 0.70 vs 1.53 ± 0.64 mmol/l,
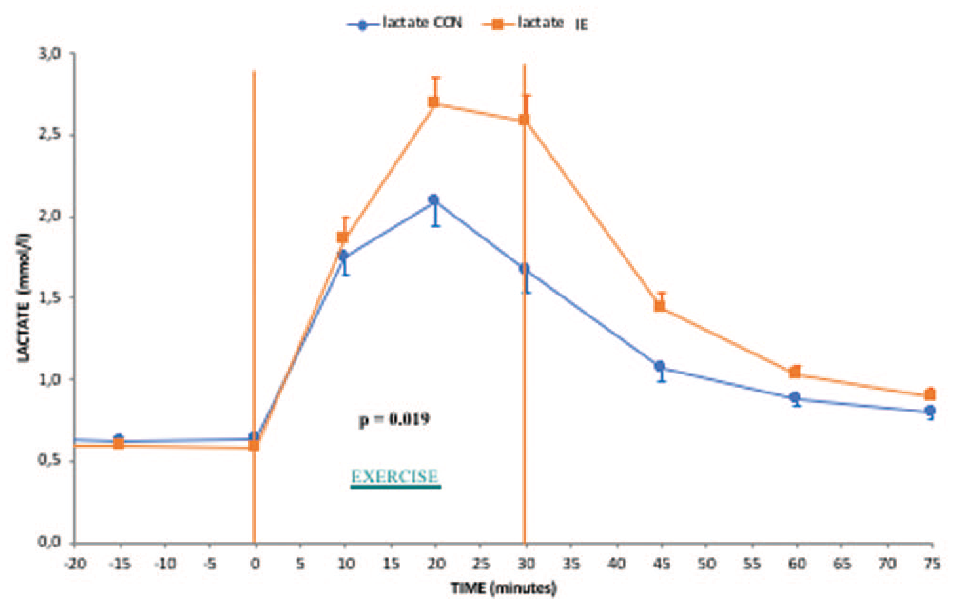
Plasma lactate behaviors in the 2 different exercise conditions investigated.
The behaviors of plasma and interstitial glucose during the CON or IE sessions are shown in Figure 2. Plasma glucose levels at the start of CON exercise was 179 ± 52 mg/dl (9.9 ± 2.9 mmol/l) and at the start of IE exercise was 172 ± 45 mg/dl (9.5 ± 2.5 mmol/l). CGM readings showed significantly higher (interstitial) glucose levels as compared to the corresponding blood glucose concentrations throughout both sessions of exercise (mean values during CON: 190 ± 55 mg/dl or 10.55 ± 3.05 mmol/l vs 152 ± 44 mg/dl or 8.44 ± 2.44 mmol/l; during IE: 174 ± 50 mg/dl or 9.66 ± 2.78 mmol/l vs 157 ± 46 mg/dl or 8.71 ± 2.55 mmol/l, respectively, both
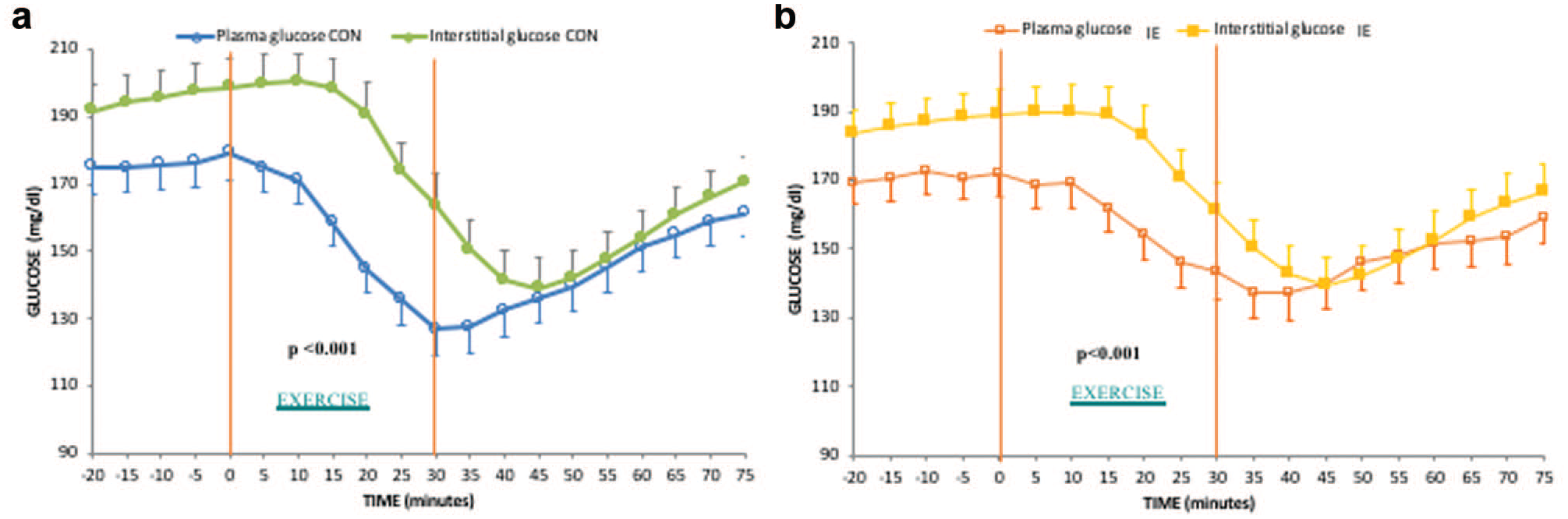
Plasma and interstitial glucose changes in the exercise sessions. (a) During CON. (b) During IE.
The comparisons between plasma and interstitial glucose for each CGM, during the CON and IE sessions, was detailed in supplemental material (S1),
CGM Accuracy During Each Exercise Session and Rest
The MARD values of each sensor calculated during CON were between 27.8% and 44.8%, while during IE they were between 15.8% and 27.5%, without statistically significant differences between each sensor type (Table 3). Therefore, in subsequent analyses all sensors were considered together and an overall MARD value, during CON and during IE, was calculated. During CON exercise, the sensors overestimated plasma glucose more than during IE exercise, as shown by both SB values (38.5 ± 27.1 mg/dl or 2.14 ± 1.5 mmol/l vs 17.2 ± 21.3 mg/dl or 0.95 ± 1.18 mmol/l,
MARD Values of Each Sensor, Calculated During CON and During IE.
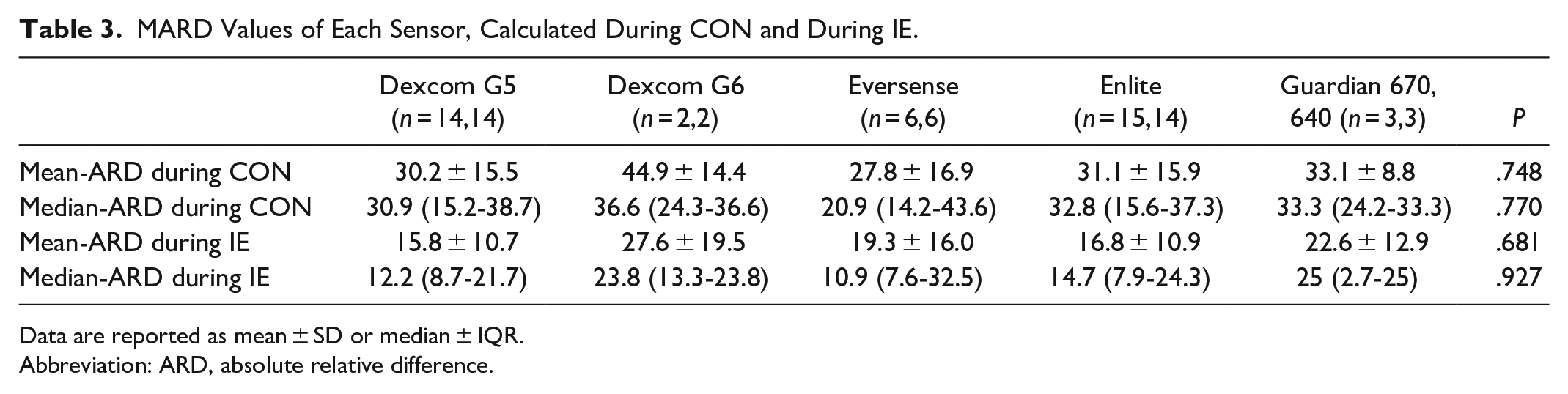
Data are reported as mean ± SD or median ± IQR.
Abbreviation: ARD, absolute relative difference.
Performance of All rt-CGMs, Considered Together, During Continuous (CON) Versus Interval Exercise (IE) and In The Recovery Period After Each Exercise Sessions.

Data are reported as mean ± SD or median ± IQR.
Abbreviation: ARD, absolute relative difference.
A head-to-head comparison of the accuracy of CGMs was reported in supplemental material (S2).
In classifying the performance according to the Clarke Error Grid, no differences were found between the 2 exercise sessions considering the pooled data of all CGMs in the combined metric A+B zones: 97.1% during CON and 98.1% during IE (Figure 3). However, separating the data from zone A and zone B, the two exercises showed statistically significant differences in data distribution (in CON A: 31.9%, B:65.2%; in IE A: 64.8%, B:33.3%;
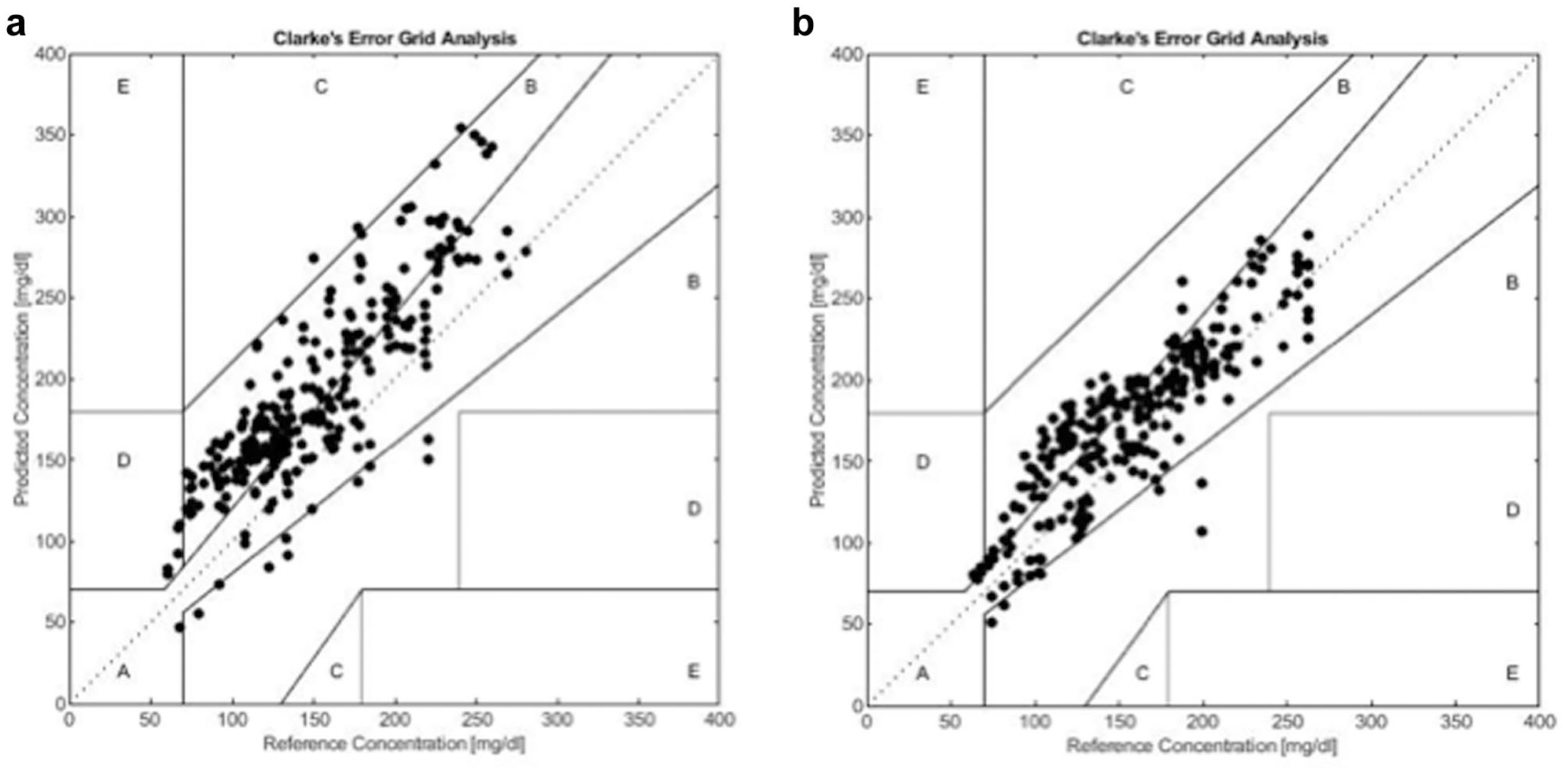
Clarke Error Grid analysis for all assessed CGMs, comparing plasma glucose versus the respective sensor values during CON (a) and during IE (b).
Discussion
This study assessed the performance of several CGM systems during 2 different exercise sessions, continuous exercise and interval exercise. As expected, reduction of plasma glucose levels was greater during continuous exercise than during interval exercise. Throughout the study, interstitial glucose overestimated the corresponding plasma glucose values and the accuracy of the sensors worsened during exercise, to a greater extent during continuous than during interval exercise, as shown by both the sensor bias and MARD values. Nevertheless, the CEG, considering the combined metric A+B zone, that is commonly used in the literature19-21,24,25 to define the potential clinical impact of the performance of systems used for glucose monitoring, showed that the clinical impact of sensor accuracy may be similar during the 2 types of exercises, also suggesting sufficient reliability of these instruments even during exercise. Available data about accuracy of sensors during exercise are limited and results are heterogeneous. To the best of our knowledge, this study is the first that assessed SB and MARD values of several recent CGMs, all those currently available in Italy, during both continuous and interval exercise sessions. Four previous studies evaluated the accuracy of older generation CGM sensors comparing different types of physical activity. Biagi et al 20 assessed the performance of the Medtronic Paradigm Enlite 2, in a small sample of 6 patients with diabetes, each of whom wore 2 sensors and carried out 2 types of exercise, aerobic exercise and weightlifting. In this study, an impairment in the MARD value during exercise, as compared to rest, was reported during aerobic exercise, whereas no impact on accuracy was found during anaerobic exercise. CEG showed nearly 100% of values in the A + B zone for both exercises. Moser et al 21 assessed the performance of Medtronic EnliteTM in a small sample of 7 patients with diabetes who carried out continuous and interval exercises on a bicycle at 3 intensity levels (low, moderate, high). A worse accuracy was found during continuous exercise than during interval training, except for the pair of moderate intensity exercises. CEG showed more than 96% of values in the A+B zone for both continuous and interval exercises, with the exception of the high intensity continuous exercise, in which performance was less (78% of the values in A+B zone). Bally et al 19 assessed the performance of Dexcom G4 in ten patients with diabetes who performed 2 types of cycling exercises, moderate continuous and high-intensity interval exercise. In this study the MARD parameter was used to evaluate the accuracy and no significant differences were found between the 2 types of exercise. CEG showed, for both exercises, 100% of values in A+B zone. The only study in which 2 different sensors were compared was carried out by Taleb et al 24 In this study the performance of Dexcom G4 platinum and Medtronic Paradigm Veo enlite system were assessed in 17 patients with diabetes who carried out 2 types of cycling exercise, continuous and interval exercise. The authors concluded that the 2 devices showed similar performances (assessed as MARD and SB) in both rest and exercise conditions, and that accuracy was less during exercise, without differences between the 2 different exercises. CEG showed more than 97% of values in the A+B zone for both exercises. Other studies have only made a comparison between exercise and non-exercise, reporting that during exercise sensors may have a lower accuracy.26-28 The performance of Dexcom G6 was recently tested during 3 different types of physical activity, aerobic, resistance and interval exercise, in a sample of 24 patients with DM1. In this study SMBG was used as a reference for the calculation of Median ARD, which did not differ significantly between the different exercise types, as calculated in the period from the start to the end of the exercise sessions. 25
There are some interesting points in our study that can be highlighted. First, we have confirmed that continuous and interval exercise may have different acute effects on blood glucose, probably due to the expected differences in adaptive hormonal responses. 29 Second, we have assessed the performances of 5 different, currently available sensors, reporting similar results during 2 different types of exercise, although for some of these CGMs the number of comparisons was limited. However, during CON all sensors showed worse accuracy and the overall MARD was about 30%. It is important to underline that the MARD values that are usually reported for these instruments refer to basal resting conditions, where a balance in glucose levels between plasma and interstitial compartments is expected. 10 Possibly, additional metrics should be defined to better evaluate the reliability of these instruments in conditions in which the blood glucose steady state is perturbed.
Third, despite the significant differences in the MARD values during the 2 types of exercise, from a clinical point of view, the combined parameter A+B zone in CEG has shown that these recent CGMs maintain good clinical reliability, even during exercise, without differences from the 2 types of exercises. However, separating the data from zone A and B, the distribution of points differed between CON and IE, being greater in zone B during CON than during IE, which suggests a clinically significant difference between the 2 exercises. By definition, the zone B includes errors greater than 20% but that still not requires corrective actions by the patient. Our results showed that SB during CON was almost 40 mg/dl. This difference may actually be not clinically significant in a blood glucose range “safe” for the patient. However, it may expose the patient to a risk for blood glucose values closer to euglycemia.
These considerations are corroborated by results of the Bland-Altman analysis. Interestingly, applying the standards ISO 15197:2013 (as defined for glucometers) to these data, during CON only 35% of the values would fall within the proposed performance limits of ± 15%. As a whole, when CGMs are used for glucose monitoring, these data suggest that it may be prudent to avoid starting exercise, especially continuous exercise, with glucose levels close to the normal range, that is, at the lower limits of the target glycemic range that is recommended in order to start physical activity, 90-250 mg/dl, 30 and this caution should then be maintained during prolonged exercise.
There are several strengths in our study: (1) The gold standard plasma glucose was used as the reference for assessing the performance of CGMs; (2). Each participant wore 2 different sensors, reducing the impact of inter-individual differences and obtaining 2355 pairs of plasma-sensor glucose points; (3) Exercise sessions have been carefully standardized and supervised, in order to reduce inter-subject variability and obtain more reproducible data; (4) Recent types of sensors have been assessed, about which there are few or no data regarding their performance during physical activity. There are also some limitations in our study given that only moderate intensity exercises were explored, the glycemic range of hypoglycemia has not been investigated and comparisons for some CGMs were numerically limited. In conclusion: physical exercise affects the accuracy of CGMs and this phenomenon is greater during continuous exercise than during interval exercise, likely due to a larger acute glycemic variation in the former condition. This result suggests the need to maintain blood glucose in a “prudent” range, above that generally recommended, when blood glucose is monitored through currently available CGMs, especially during continuous exercise. Further studies are needed to investigate the effect of additional types of activities, characterized by different intensity and duration.
Supplemental Material
sj-pdf-1-dst-10.1177_19322968211023522 – Supplemental material for Accuracy of CGM Systems During Continuous and Interval Exercise in Adults with Type 1 Diabetes
Supplemental material, sj-pdf-1-dst-10.1177_19322968211023522 for Accuracy of CGM Systems During Continuous and Interval Exercise in Adults with Type 1 Diabetes by G. Da Prato, S. Pasquini, E. Rinaldi, T. Lucianer, S. Donà, L. Santi, C. Negri, E. Bonora, P. Moghetti and M. Trombetta in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
None
Author’s Note
The material in this manuscript has been previously presented as an oral communication at a scientific conference: 28° SID online national congress, December 2-5, 2020.
Abbreviations
T1D, Type 1 Diabetes, CGMs, Continuous Glucose Monitoring Systems, CSII, Continuous Subcutaneous Insulin Infusion, HRR, Heart Rate Reserve, CON, Continuous Aerobic Exercise, IE, Interval Exercise, ICR, Insulin-to-Carbohydrate Ratio, ISF, Insulin Sensitivity Factor, SB, Sensor Bias, MARD, Mean Absolute Relative Difference, CEG, Clarke Error Grid, PG, Plasma Glucose.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
