Abstract
Background:
Complexity of glucose regulation in persons with type 1 diabetes (PWDs) necessitates increased automation of insulin delivery (AID). This study aimed to analyze real-world data over 12 months from PWDs who started using the MiniMed 780G (MM780G) advanced hybrid closed-loop (aHCL) AID system at the Diabeter clinic, focusing on glucometrics and clinical outcomes.
Methods:
Persons with type 1 diabetes switching to the MM780G system were included. Clinical data (e.g. HbA1c, previous modality) was collected from Diabeter’s electronic health records and glucometrics (time in range [TIR], time in tight range [TITR], time above range [TAR], time below range [TBR], glucose management indicator [GMI]) from CareLink Personal for a 12-month post-initiation period of the MM780G system. Outcomes were age-stratified, and the MM780G system was compared with previous use of older systems (MM640G and MM670G). Longitudinal changes in glucometrics were also evaluated.
Results:
A total of 481 PWDs were included, with 219 having prior pump/sensor system data and 334 having monthly longitudinal data. After MM780G initiation, HbA1c decreased from 7.6 to 7.1% (P < .0001) and the percentage of PWDs with HbA1c <7% increased from 30% to 50%. Glucose management indicator and TIR remained stable with mean GMI of 6.9% and TIR >70% over 12 months. Age-stratified analysis showed consistent improvements of glycemic control across all age groups, with older participants achieving better outcomes. Participants using recommended system settings achieved better glycemic outcomes, reaching TIR up to 77% and TTIR up to 55%.
Conclusions:
Use of MM780G system results in significant and sustained glycemic improvements, consistent across age groups and irrespective of previous treatment modalities.
Introduction
Most persons with type 1 diabetes (PWDs) do not reach advised glycemic goals. 1 During the last 20 years, continuous subcutaneous insulin infusion (CSII), without or with intermittent scanning (is) or real-time (rt) continuous glucose monitoring (CGM) has yielded improved outcomes compared with multiple daily injections (MDI). 2 Introduction of CGM (both rt-CGM and is-CGM) has strongly improved glucose regulation and outcomes. However, despite these improvements, most PWDs do not reach the internationally established targets for glucose regulation.3,4 The complexity of glucose regulation requires constant adaptations that only automated insulin delivery (AID) can offer. 5 Various AID systems are commercially available. Hybrid closed-loop (HCL) and advanced HCL (aHCL) AID systems are a temporary step between traditional open-loop treatment and fully closed-loop (FCL) systems. 6
Multiple studies have shown that the use of AID systems is safe and yields improved glycemic outcomes,7 -19 psychosocial outcomes,20 -28 PWD satisfaction,29,30 and sleep,31 -33 also in specific PWD groups (e.g. pregnancy,34 -36 very young children,37 -40 elderly, 25 PWDs with high risk for hypoglycemia,41,42 and Muslim PWDs during Ramadan).43,44 A low glucose target (GT) and a high percentage of time spent in automode have been shown to be associated with a higher time in range (TIR). 45 Older age, shorter diabetes duration and shorter active insulin time (AIT, or duration of insulin action [DIA]) have been shown to be predictors of optimal glucose control when using AID systems, 19 whereas younger age and longer diabetes duration have been associated with less improvement, underscoring the importance of ongoing support and re-education among younger PWDs. 17 Over the past two decades, real-world evidence (RWE) studies have rapidly advanced, especially in diabetes care, and are essential in complementing randomized controlled trials (RCTs) to answer complex treatment-related questions. 46 With digital data becoming more comprehensive, RWE will increasingly support physicians, regulators, and PWDs in making informed therapy decisions, despite concerns about the lack of standardization of the term RWE and the varying quality of studies as a result.47,48 However, of the type 1 diabetes (T1D) RWE studies on AID, many only report glucometrics and only a minority of studies investigate possible associations between AID-generated glucometrics and clinical parameters, such as HbA1c and previous treatment modalities.12,17,27,49 -53 Of these, inclusion numbers were relatively low and data were mostly collected retrospectively.
The aim was to analyze a 12-month span of real-world prospectively collected data from PWDs who started the MiniMed 780G (MM780G) system in the “Diabeter” diabetes clinic in the Netherlands. The enriched data set allowed not only focusing on glucometrics but also included clinical data such as HbA1c and previous treatment modalities.
Methods
Study Population
Persons with T1D from our diabeter clinics (five locations in the Netherlands) entitled to change their pump system after four years of use (end of warranty period) and who chose an MM780G system were included. In addition, PWDs who wanted to switch from MDI to an MM780G system were included. Owing to the real-world design, there were no strict inclusion criteria for age and diabetes duration. Choices on treatment modality were made in a shared decision-making process between PWDs and their health care professional (HCP) team. Persons with T1D who continued on an open-loop combination (MDI or CSII) or waited for an alternative AID system (which was limited to the Tandem system with the Control-IQ algorithm during this study period) were excluded. For those subanalyses that specifically compare the MM780G with previous modes of CSII insulin administration, the analysis was restricted to previous users of MM670G and MM640G as comparators. Only PWDs who consented the use of both their HCL system data from CareLink Personal (MiniMed‘s data management system) and their clinical data from the Diabeter electronic health records (EHRs) were included. Data from a 12-month baseline period and from a 12-month follow-up period since the initiation of automode on the MM780G system were collected and analyzed (Figure 1). Allmost all participants were using rapid insulin analogs (95%), with 5% using ultra-rapid insulin analogs.
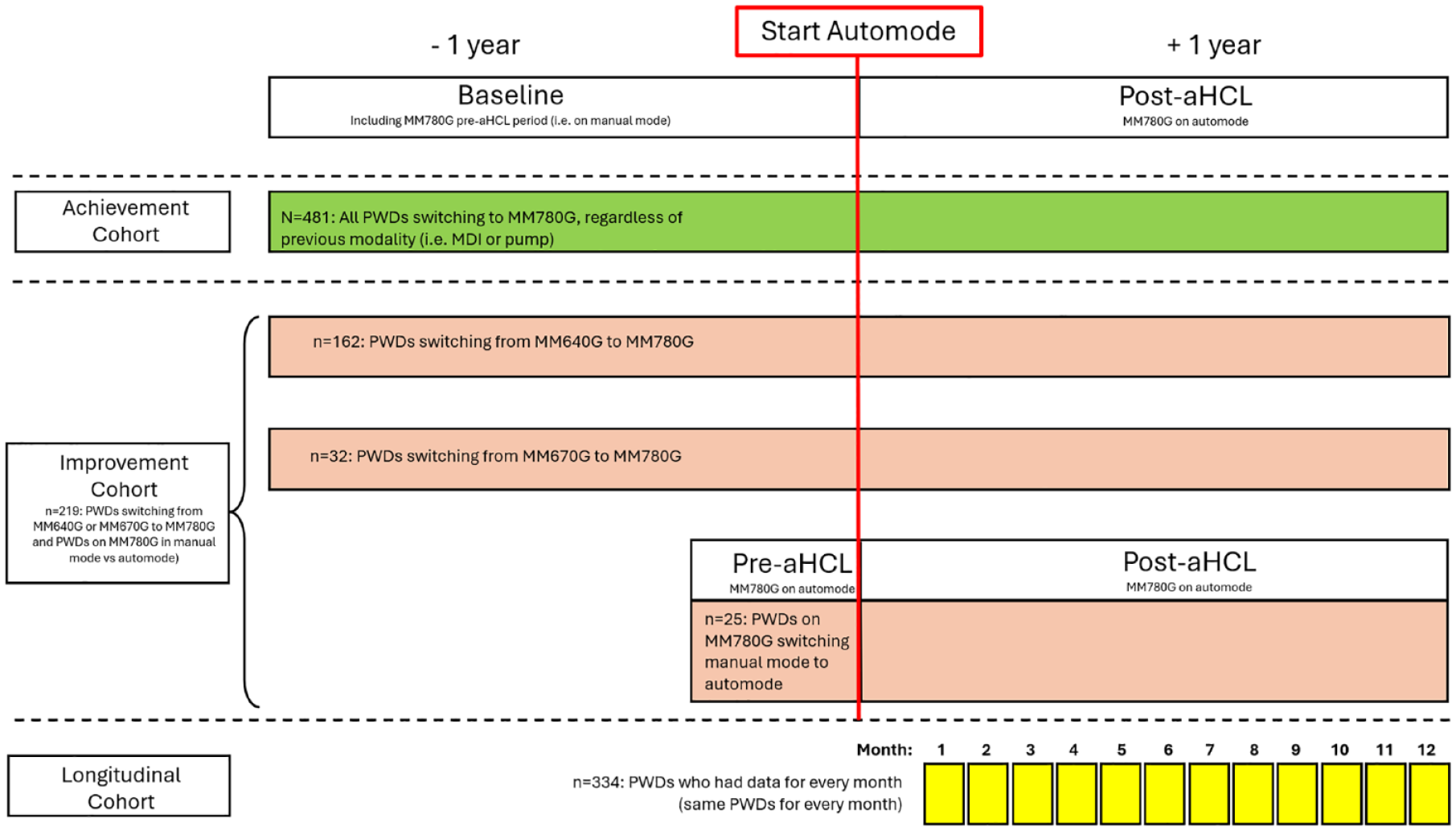
Flowchart of study.
Study Setup
Figure 1 shows the setup of this observational cohort study. Post-aHCL glucometric achievements were determined for the complete cohort (“Achievement cohort”), because all included participants changed to the MM780G AID system. However, baseline/post-aHCL glucometrics comparisons could not be made for the Achievement cohort, because it also included PWDs who did not have baseline CGM data available (for instance, because they were changing from MDI + self-monitored blood glucose monitoring or from CGM sensors for which data were not accessible). One-year use of the MM640G system or the MM670G system prior to switching to MM780G use in automode was compared with one-year use of MM780G in automode to assess improvements of AID systems over previous HCL systems (“Improvement subcohort”). Longitudinal changes in glucometric parameters were assessed among PWDs who had data availalable for each of the first 12 months post-aHCL (“Longitudinal subcohort”). The three cohorts are also explained in Figure 1.
Baseline data of variables comprised the last value before switching from the previous treatment modality to initiation of automode of the MM780G system, looking back a maximum of 12 months. “Pre-AHCL” was defined as the run-in period on the MM780G system in manual mode. This period was included in the baseline period, except for the comparison between being on manual mode versus automode when using the MM780G system. “Post-aHCL” was defined as the period after starting automode on the MM780G system.
Data Collection
Continuous-glucose-monitoring-based data (e.g. insulin delivery data from pump and glucose readings from sensor) of both the MM780G system and of previously used systems (MM640G and MM670G) from users under treatment at Diabeter Netherlands were extracted from CareLink Personal (Medtronic MiniMed™). In the course of their treatment, these data had been uploaded to CareLink Personal software by PWDs or their guardians. Glucometrics between August 1, 2020, and September 1, 2023 (MM780G), and between January 1, 2019, and August 22, 2022 (MM640G or MM670G), were extracted of PWDs who had at least one baseline HbA1c value and one post-aHCL HbA1c value available during the study period and ≥10 days of sensor glucose (SG) data after initiating aHCL (“post-aHCL”). 4 Glucometrics included time above targeted glucose range (TAR, >250 mg/dL [>13.9.0 mmol/L] and >180 mg/dL [10 mmol/L]), time in targeted glucose range (TIR, 70-180 mg/dL [3.9-10 mmol/L]), time in tight glucose range (TITR, 70–140 mg/dL [3.9–7.8 mmol/L]), time below targeted glucose range (TBR, <70 mg/dL [3.9 mmol/L] and <54 mg/dL [<3.0 mmol/L]), and glucose management indicator (GMI, calculated from CGM values). Carelink data were supplemented with clinical data from Diabeter’s EHR system (Vcare). Baseline values used for the analyses of HbA1c, weight, height, and body mass index (BMI) were the last values available from EHRs in the one-year baseline period. Post-aHCL values were the last values available in the one-year follow-up. Data were only used and aggregated from users who provided consent for use of their Carelink and EHR data in accordance with data privacy regulations in the Netherlands.
Statistical Analysis
The data used in this study can be classified into two groups: device-collected data and nondevice data. In the first group, we analyzed SG values, insulin doses, and all other data automatically collected from the device and extracted from the CareLink system. The selection of these data was based on international consensus recommendations for CGM data, specifically cases with ≥10 days of SG data during the observation period. 4 As these data are automatically collected every night and the system is used more than 90% of the time, there were no missing data. Most of the data in this study fell into this group, so there are no concerns about missing data for most of the study. The second group of data consists of EHR data, including HbA1c, weight, and height results. As a real-life study, these data do present cases of missing data. Most of these missing data cases relate to restrictions during the COVID-19 period, which prevented participants from attending in-person clinic visits. As these restrictions applied to all participants, we do not expect systematic selection bias in these results. Therefore, all analyses were conducted using all available data without imputation for missing data.
Where possible baseline and post-aHCL characteristics of the complete cohort were summarized with descriptive statistics (i.e. mean and standard deviation [SD] of age, sex, weight, length, BMI, HbA1c). For PWDs aged 1 to 21 years, BMI z-scores were obtained from reference tables. 54
Post-aHCL glucometrics were averages over the one-year post-aHCL period for the Achievement and Improvement cohorts, and over each post-aHCL month for the Longitudinal cohort. The Achievement and Longitudinal cohorts were also stratified for age (<7 years, 7-13 years, 14-21 years, 22-50 years, and >50 years). For the Longitudinal cohort, it should also be noted that for HbA1c, weight and height, each month comprised different and much smaller groups of participants because for these parameters, not all participants had values available for each month, preventing fair comparisons between months. Differences between baseline and the post-aHCL one-year period were performed using the paired samples t-test.
Results
In total, 481 PWDs had ≥10 days of MM780G SG data and at least one baseline HbA1c value and one post-aHCL HbA1c value available during the study period. From this Achievement cohort, n = 219 PWDs had ≥10 days of SG data with a prior HCL system (MM640G or MM670G: the Improvement subcohort) or with MM780G on manual mode, and n = 334 PWDs had ≥10 days of SG data in each of the first 12 months post-aHCL (the Longitudinal subcohort). Table 1 presents demographic, anthropomorphic characteristics, and treatment modalities of all three cohorts and shows they were comparable. Most participants (74%) were using a pump and sensor before they switched to the MM780G system. About 20% of participants were using MDI + sensor when they switched. A few participants were using MDI or a pump without a sensor.
Baseline Characteristics of Full Cohort (Achievement Cohort) and 2 Subcohorts (Improvement Cohort and Longitudinal Cohort).
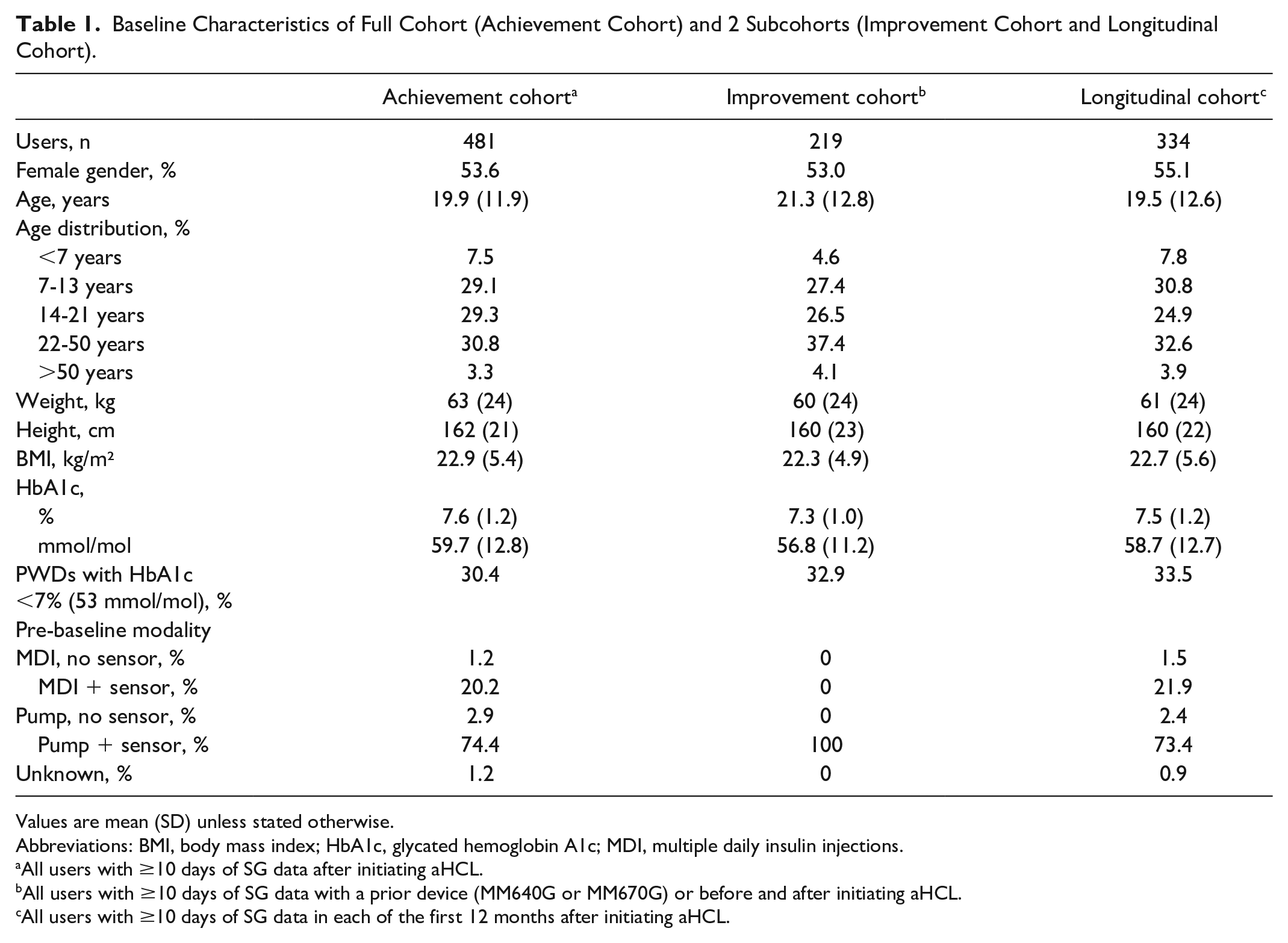
Values are mean (SD) unless stated otherwise.
Abbreviations: BMI, body mass index; HbA1c, glycated hemoglobin A1c; MDI, multiple daily insulin injections.
All users with ≥10 days of SG data after initiating aHCL.
All users with ≥10 days of SG data with a prior device (MM640G or MM670G) or before and after initiating aHCL.
All users with ≥10 days of SG data in each of the first 12 months after initiating aHCL.
Achievement Cohort
In the Achievement cohort (N=481), mean (SD) post-aHCL HbA1c was 0.5 (0.9)% lower than baseline, going from 7.6±1.2% to 7.1±1.0% (P < .0001) (Table 2) and the percentage of PWDs with HbA1c <7% (53 mmol/mol) increased from 30% to 50% (Figure 2). Post-aHCL HbA1c and GMI values were comparable. About two thirds of participants had a GMI <7% and/or TIR >70% post-aHCL. Over half of the participants had a TIR >70%, TBR <4%, and GMI <7% post-aHCL. The overall mean (SD) TITR post-aHCL was 50.3 (9.5)%, with mean TITR of 44 (6) and 55 (9)% in those <7 years and >50 years, respectively. When stratified for age, all age groups showed a decrease in HbA1c after switching from previous modality to the MM780G system (all P < .0001)(Table 2). Both at baseline and post-aHCL, the age categories up to 22 years had a higher HbA1c than the older age categories. Post-aHCL categories up to 22 years showed lower TIR and higher TBR and TAR than the older categories. For the <7 years age group mean (SD) pre-aHCL and post-aHCL BMI z-scores were both 1.0 (1.0). For the 7 to 13 years age group pre-aHCL and post-aHCL BMI z-scores were 0.2 (0.9) and 0.3 (0.9). For the 14 to 21 years age group, pre-aHCL and post-aHCL BMI z-scores were 0.5 (0.9) and 0.6 (1.0), respectively. For 22 to 50 years age group, pre-aHCL and post-aHCL BMI values were 26.4 (4.7) and 26.9 (4.8), respectively. For the BMI and >50 years pre-aHCL and post-aHCL BMI values were 28.0 (4.0) and 27.8 (4.3), respectively.
Baseline Versus Post-aHCL for Achievement Cohort, Stratified for Age Categories.
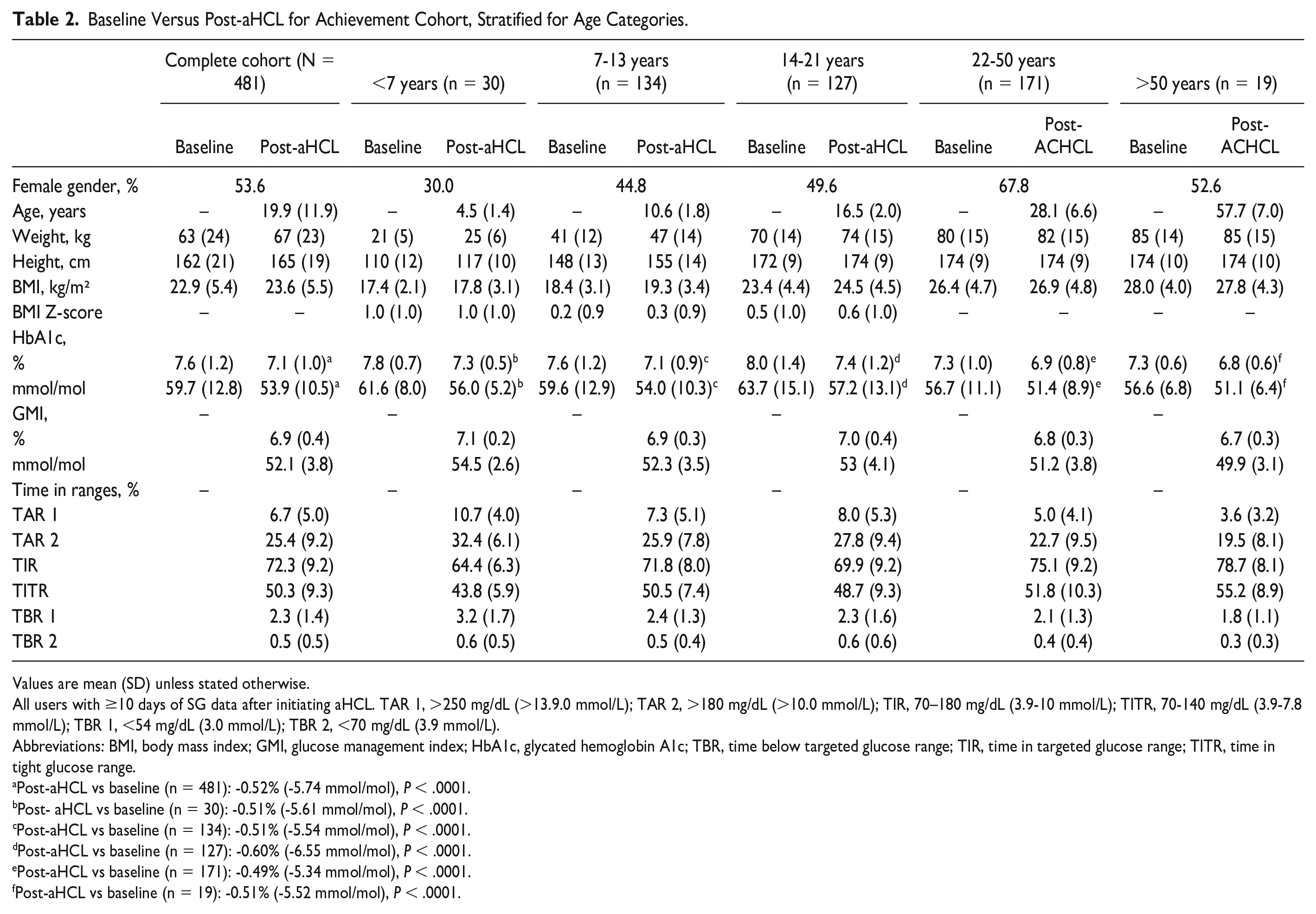
Values are mean (SD) unless stated otherwise.
All users with ≥10 days of SG data after initiating aHCL. TAR 1, >250 mg/dL (>13.9.0 mmol/L); TAR 2, >180 mg/dL (>10.0 mmol/L); TIR, 70–180 mg/dL (3.9-10 mmol/L); TITR, 70-140 mg/dL (3.9-7.8 mmol/L); TBR 1, <54 mg/dL (3.0 mmol/L); TBR 2, <70 mg/dL (3.9 mmol/L).
Abbreviations: BMI, body mass index; GMI, glucose management index; HbA1c, glycated hemoglobin A1c; TBR, time below targeted glucose range; TIR, time in targeted glucose range; TITR, time in tight glucose range.
Post-aHCL vs baseline (n = 481): -0.52% (-5.74 mmol/mol), P < .0001.
Post- aHCL vs baseline (n = 30): -0.51% (-5.61 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 134): -0.51% (-5.54 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 127): -0.60% (-6.55 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 171): -0.49% (-5.34 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 19): -0.51% (-5.52 mmol/mol), P < .0001.
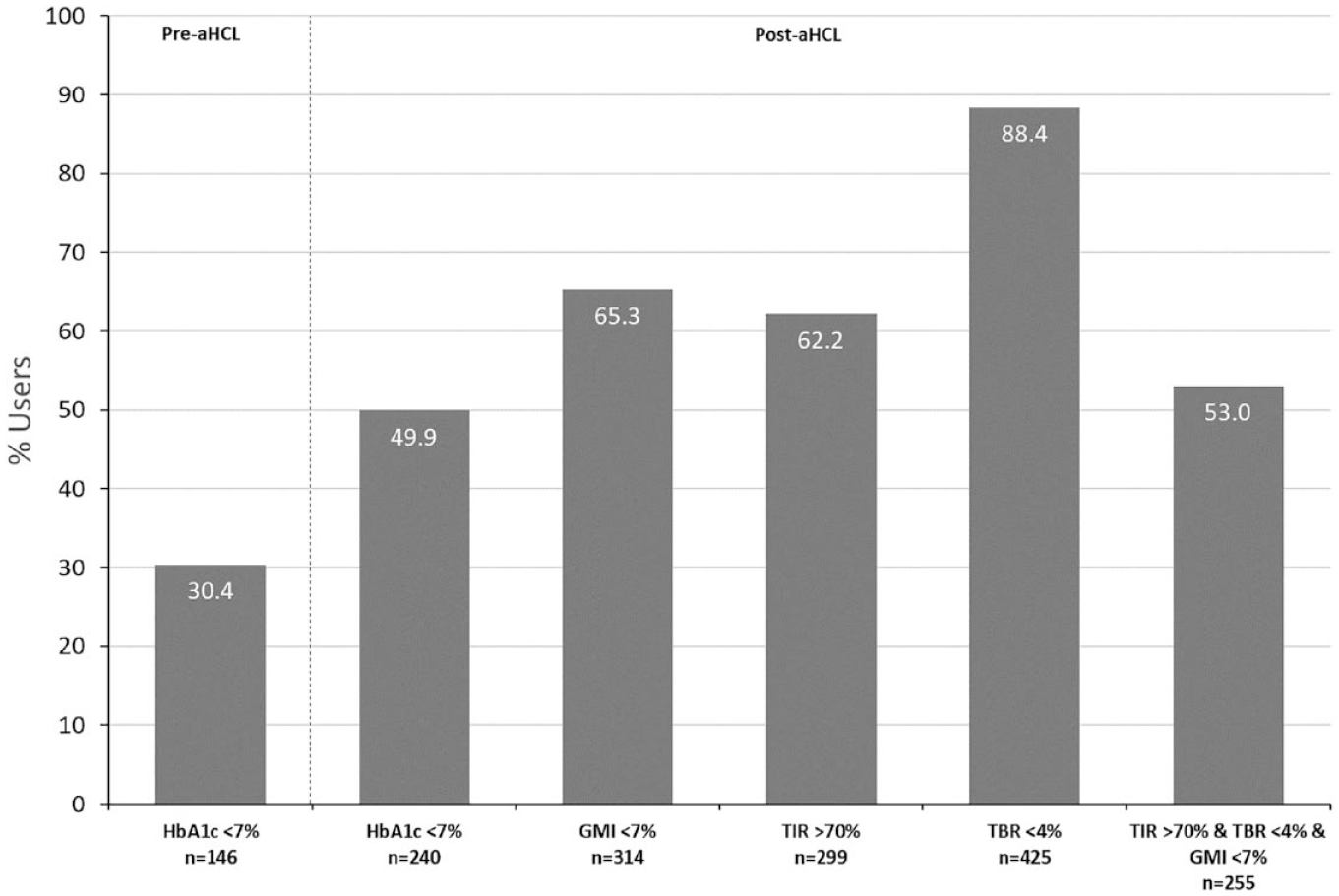
MM780G users achieving treatment goals (achievement cohort, N = 481).
Only 45 (9%) participants used the recommended settings (GT of 100 mg/dL [5.5 mmol/L] and AIT of 2 hours, both used >95% of the time). This group showed better glycemic outcomes (Supplemental Table 1): instead of half of participants having a TIR >70%/TBR <4%/GMI <7%, this applied to 76% of participants using optimal settings.
Improvement Cohort
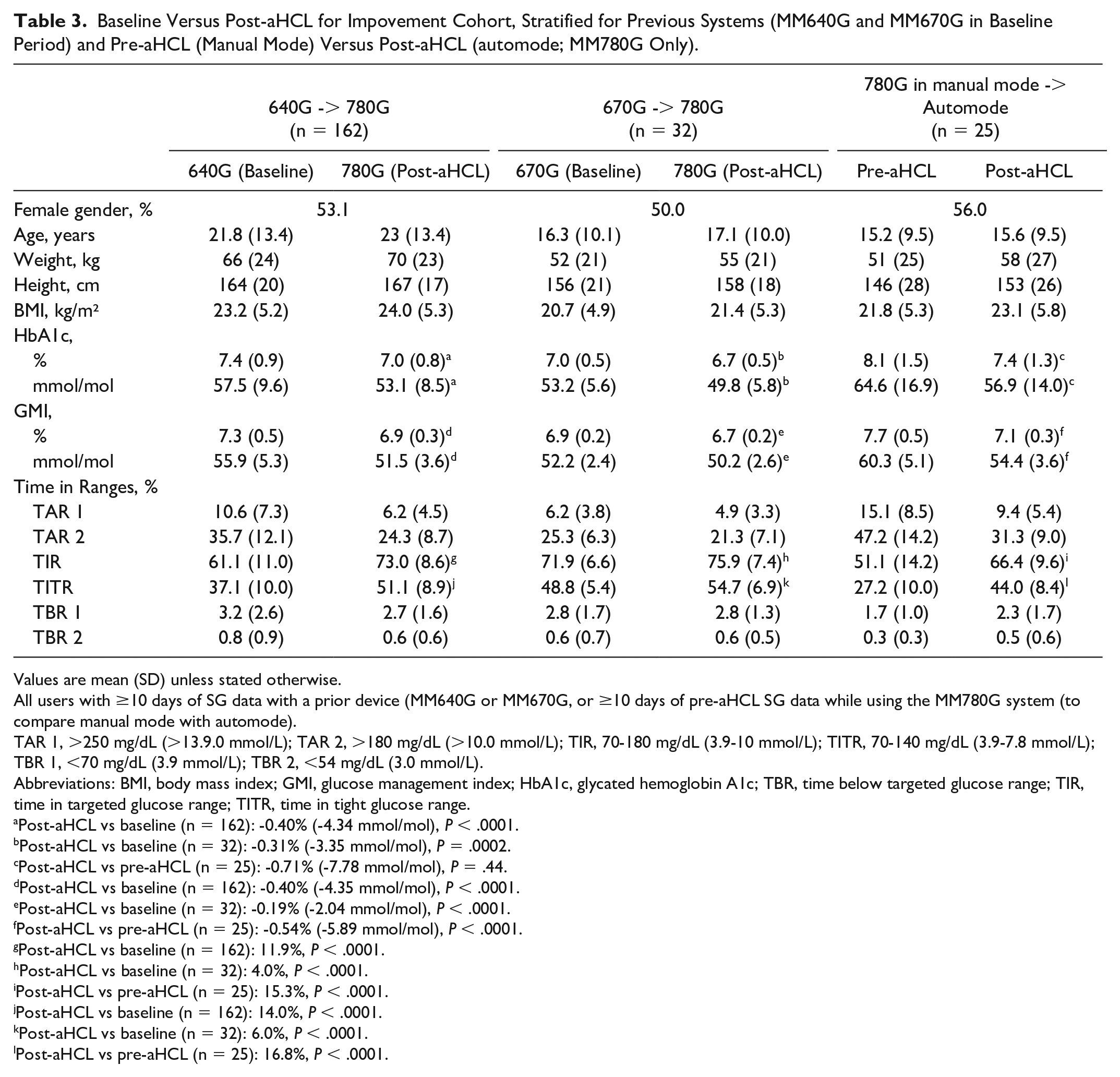
Compared with previous system use, HbA1c and GMI decreased after switching to the MM780G AID system, both when stratified for previous use of an HCL system (MM640G or MM670G) and for MM780G in manual mode vs MM780G in automode use (Table 3). Time in targeted glucose Range increased when participants switched from an MM640G HCL system to an MM780G AID system (61% to 73%, P < .0001) or from an MM670G HCL system to an MM780G AID system (72% to 76%, P < .0001). Time In Tight glucose Range also increased (MM640G to MM780G: 37% to 51%, P < .0001; MM670G to MM780G: 49% to 55%, P < .0001). Both TIR and TITR also increased after switching from manual mode to automode when using the MM780G system (TIR: 51% to 66%, P < .0001; TITR: 27% to 44%, P < .0001).
Baseline Versus Post-aHCL for Impovement Cohort, Stratified for Previous Systems (MM640G and MM670G in Baseline Period) and Pre-aHCL (Manual Mode) Versus Post-aHCL (automode; MM780G Only).
Values are mean (SD) unless stated otherwise.
All users with ≥10 days of SG data with a prior device (MM640G or MM670G, or ≥10 days of pre-aHCL SG data while using the MM780G system (to compare manual mode with automode).
TAR 1, >250 mg/dL (>13.9.0 mmol/L); TAR 2, >180 mg/dL (>10.0 mmol/L); TIR, 70-180 mg/dL (3.9-10 mmol/L); TITR, 70-140 mg/dL (3.9-7.8 mmol/L); TBR 1, <70 mg/dL (3.9 mmol/L); TBR 2, <54 mg/dL (3.0 mmol/L).
Abbreviations: BMI, body mass index; GMI, glucose management index; HbA1c, glycated hemoglobin A1c; TBR, time below targeted glucose range; TIR, time in targeted glucose range; TITR, time in tight glucose range.
Post-aHCL vs baseline (n = 162): -0.40% (-4.34 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 32): -0.31% (-3.35 mmol/mol), P = .0002.
Post-aHCL vs pre-aHCL (n = 25): -0.71% (-7.78 mmol/mol), P = .44.
Post-aHCL vs baseline (n = 162): -0.40% (-4.35 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 32): -0.19% (-2.04 mmol/mol), P < .0001.
Post-aHCL vs pre-aHCL (n = 25): -0.54% (-5.89 mmol/mol), P < .0001.
Post-aHCL vs baseline (n = 162): 11.9%, P < .0001.
Post-aHCL vs baseline (n = 32): 4.0%, P < .0001.
Post-aHCL vs pre-aHCL (n = 25): 15.3%, P < .0001.
Post-aHCL vs baseline (n = 162): 14.0%, P < .0001.
Post-aHCL vs baseline (n = 32): 6.0%, P < .0001.
Post-aHCL vs pre-aHCL (n = 25): 16.8%, P < .0001.
Longitudinal Cohort
Glucose management indicator values were available for 334 participants in each month and showed stable values, also when stratified for age (Figure 3). Both TIR and TITR decreased slightly over the 12 months (Figure 3). Participants >22 years old showed higher TIR and TITR and lower TAR and TBR than younger participants. Figure 4 presents the number of boluses per day, which is stable during 12 months. HbA1c, height, weight and BMI could not be compared between months, because each month comprised different and much smaller groups of participants.
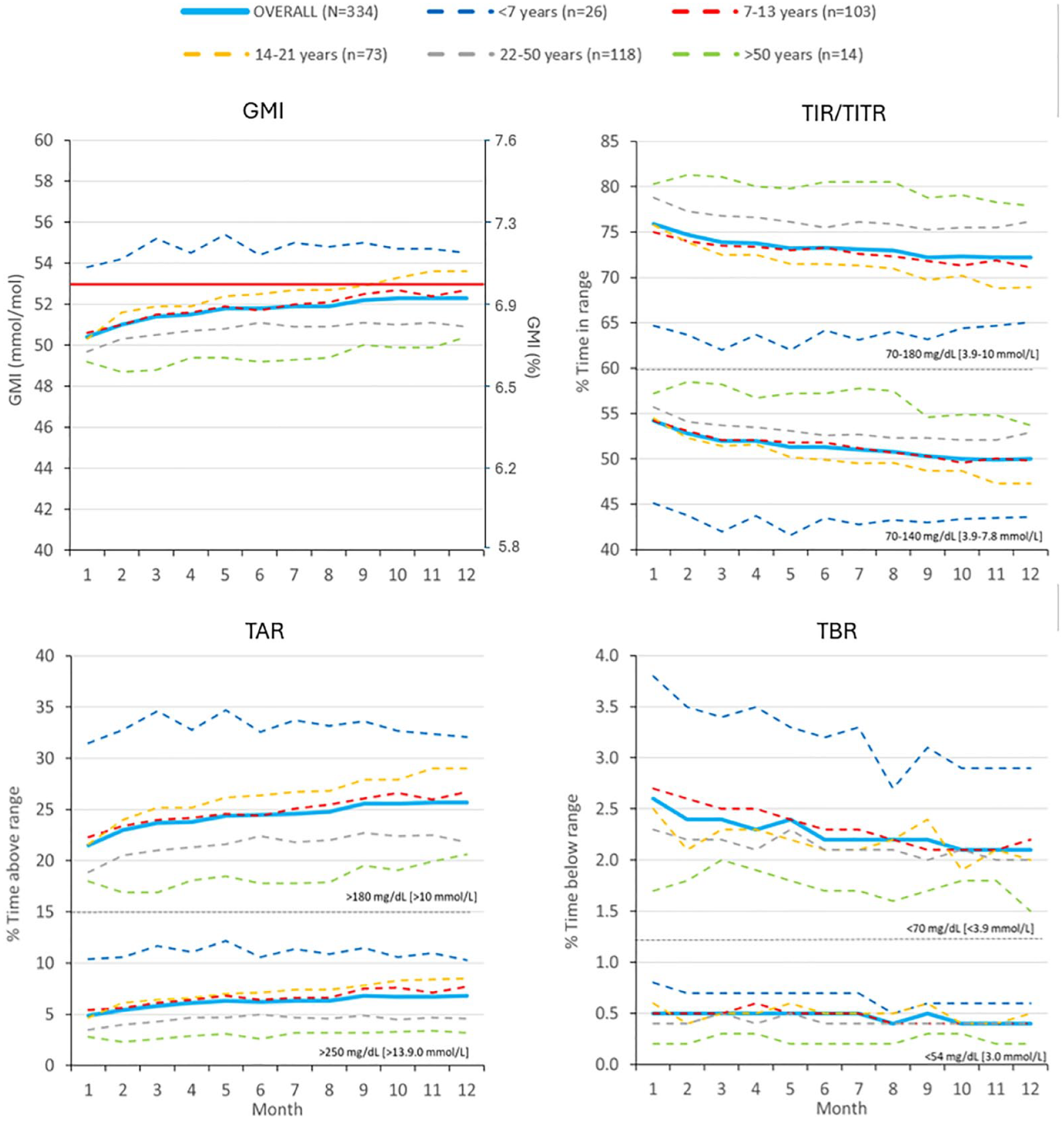
GMI, TIR, TITR, TAR, and TBR of longitudinal cohort.
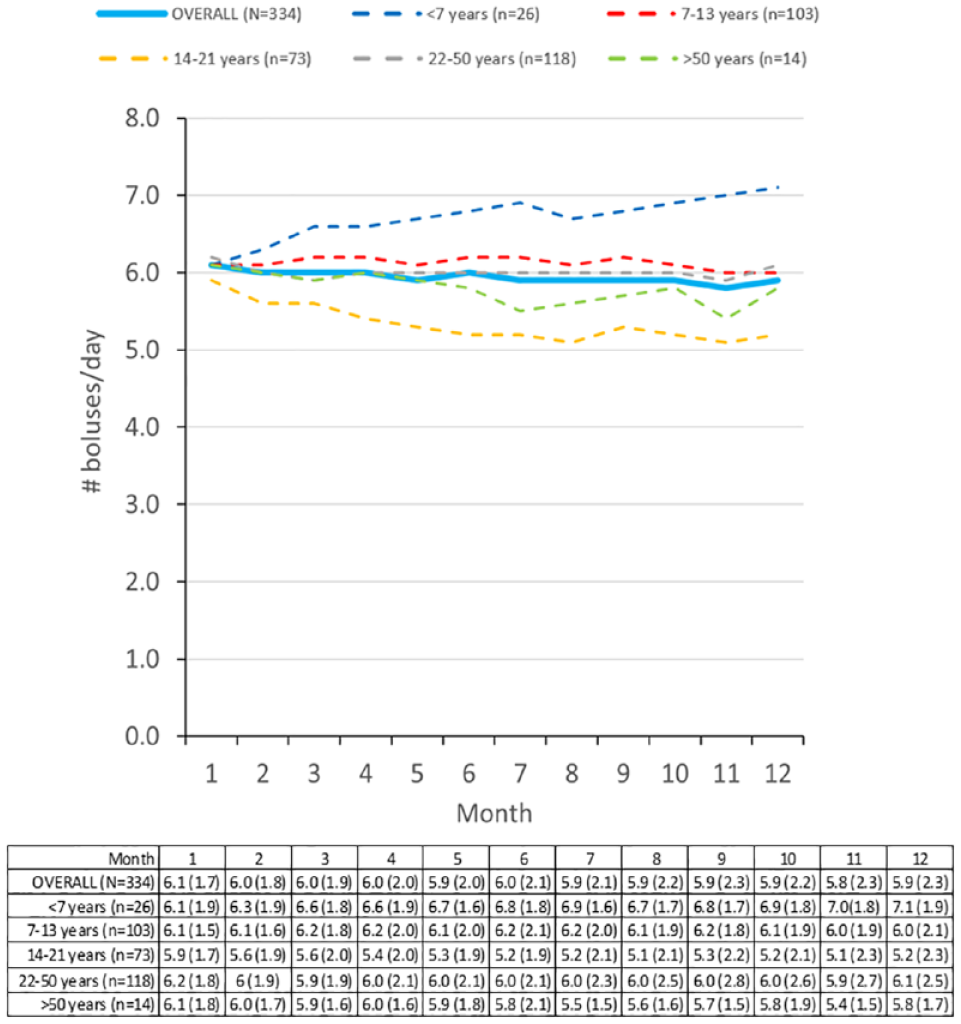
Number of boluses per day for the longitudinal cohort.
Discussion
In this real-world study, rapid glycemic improvements across all age groups (even including the very young of <7 years) were observed after start of aHCL AID use, which were sustained up to 1 year. With 7.1% (54 mmol/mol), post-aHCL HbA1c was significantly lower than baseline levels (7.6% [60 mmol/mol]) and similar decreases were observed after stratifying for age and previous AID system. Despite a slight trend of decrease, TIR remained >70% at least up to one year after aHCL start. Older participants showed higher TIR/TITR and lower TAR and TBR, but even the younger age groups showed sustained good glucometric values within international targets. Participants who previously used earlier HCL systems, such as the MM640G and MM670G sytems, also showed glycemic improvements after switching to an AID system. The small subgroup using optimal system settings showed best glycemic outcomes, with a sustained average TIR reaching up to 77%.
Rapid and sustained improvements after start of automode of AID systems have been reported in other real-world studies.43,50,51,55 -58 Our results suggest that even better improvements in glycemic regulation may be achieved when using the MM780G system with optimal settings. As also shown in this study, optimal system settings (GT and AIT) have been shown to improve TIR (especially TITR), 59 highlighting the need to check settings regularly.
Even in the very young age group (<7 years, currently off-label), we observed a decrease in HbA1c after start of MM780G automode and sustained TIR of almost 70%, as also shown by Tornese et al39,60 RCT studies have also shown that aHCL AID use in this age group improves glycemic control and is safe,37,40,60 -62 and reduces parental diabetes distress.37,40,60 However, younger children were shown to be less able to maintain technology adherence over time compared with teenagers. 63
We observed that PWDs who were on previous systems (MM640G andMM670G) also showed improved glycemic regulation, as also reported by others.8,12,53,62,64,65 From the literature, it is known that PWDs switching from MDI to AID systems also show improved glycemic regulation and unchanged or decreased risk of hypoglycemia.8,42,66 -68 Furthermore, Beato-Víbora et al 8 found that TIR increases were not different between PWDs switching from MDI or from previous pump treatment. The much improved changes in glycemic control and the ability to remotely monitor PWDs with AID systems has the potential to reduce the number of consultations (both face-to-face and virtual) and thus decrease use of HCP (team) resources. In this context, Diabeter has developed an automated population health management system (CloudCare), which translates glucose data from multiple PWDs into internationally defined glycemic parameters to identify those at risk based on a triaging protocol. This allows HCPs to discuss self-management adaptations when necessary, while saving resources by avoiding detailed analysis when glucose values are stable (manuscript submitted).
Despite the increased TIR and TITR after switching from older pump/sensor systems, or going from manual mode to automode, longitudinal analysis showed a slight decrease in TIR/TITR over the 12 months of follow-up. When stratified for age, this decrease was mainly attributable to the 7- to 13-year and 14- to 21-year groups. A slight decrease in TIR in children using the 780G system has been described earlier. 7 This decrease may be partially explained by lower treatment adherence of PWDs in these age groups, for example, due to parental involvement which may be considered as invasive, leading to decreased adherence. 69 Other factors may include alarm fatigue, PWDs getting used to the system thus getting more lax as time goes on, and delayed adjustment of carbohydrate ratios in this rapidly growing/changing age group.
The main strength of this study is the real-world nature of the data. There are also a number of possible limitations. Being an observational study (i.e. without a control group), no causal links can be established. The data are from one center, introducing bias toward methods used at this center. Many participants in this study have initiated aHCL AID treatment remotely due to COVID-19. It may be argued that these participants have not been educated as effectively when compared with face-to-face education, possibly leading to suboptimal results. However, others have reported the absence of notable post-COVID changes. 70 Also due to COVID-19, there was a lack of HbA1c measurements, length, and weight values for many participants. Furthermore, the AID system studied is only one brand. Different brands have been shown to yield different results.71,72 Finally, severe adverse events (SAEs) were not extracted from the EHRs, although the incidence of SAEs is very low in the clinic. Even so, AID systems have been shown to decrease the risk of severe hypoglycemia and diabetic ketoacidosis.41,49
Future analyses will look more in depth at system settings, including assessment of timely attainment of GTs and identification of potential factors which can be improved upon (i.e. education, follow-up, settings). Also, as not much is known yet about effects of AID use on weight, we are currently analyzing this possible association in a larger cohort. We also plan to identify factors predicting success of AID treatment, like previous treatment (i.e. MDI without or with sensor, pump without or with sensor) and HbA1c before initiation of AID treatment.
In conclusion, after initiation of AID systems, PWDs showed significant and sustained glycemic improvements in a real-world setting. Rapid reductions in HbA1c and GMI and maintenance of TIR >70%, sustained for at least 12 months, were observed across various age groups and in participants previously using other HCL systems, especially when using optimal settings. These results underscore the important role of optimal system settings to maximize outcomes. Future research should focus on predictors of aHCL AID treatment success, like system settings, weight, prior treatment modalities, and socioeconomic factors, to further optimize patient outcomes and develop tailored educational initiatives.
Supplemental Material
sj-docx-1-dst-10.1177_19322968241290259 – Supplemental material for Twelve-Month Real-World Use of an Advanced Hybrid Closed-Loop System Versus Previous Therapy in a Dutch Center For Specialized Type 1 Diabetes Care
Supplemental material, sj-docx-1-dst-10.1177_19322968241290259 for Twelve-Month Real-World Use of an Advanced Hybrid Closed-Loop System Versus Previous Therapy in a Dutch Center For Specialized Type 1 Diabetes Care by Pim Dekker, Tim van den Heuvel, Arcelia Arrieta, Javier Castañeda, Dick Mul, Henk Veeze, Ohad Cohen and Henk-Jan Aanstoot in Journal of Diabetes Science and Technology
Footnotes
Abbreviations
aHCL, advanced hybrid closed-loop; AID, automated insulin delivery; AIT, active insulin time; BMI, body mass index; GMI, glucose management index; GT, glucose target; HbA1c, glycated hemoglobin A1c; HCL, hybrid closed-loop; HCP, health care professional; MDI, multiple daily insulin injections; SG, sensor glucose; TAR, Time Above targeted glucose Range; TBR, Time Below targeted glucose Range; TIR, Time In targeted glucose Range; TITR, Time In Tight glucose Range.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TVDH, AA, JC, and OC are full time employees of Medtronic. Diabeter Netherlands is an independent clinic, which was acquired by Medtronic.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
